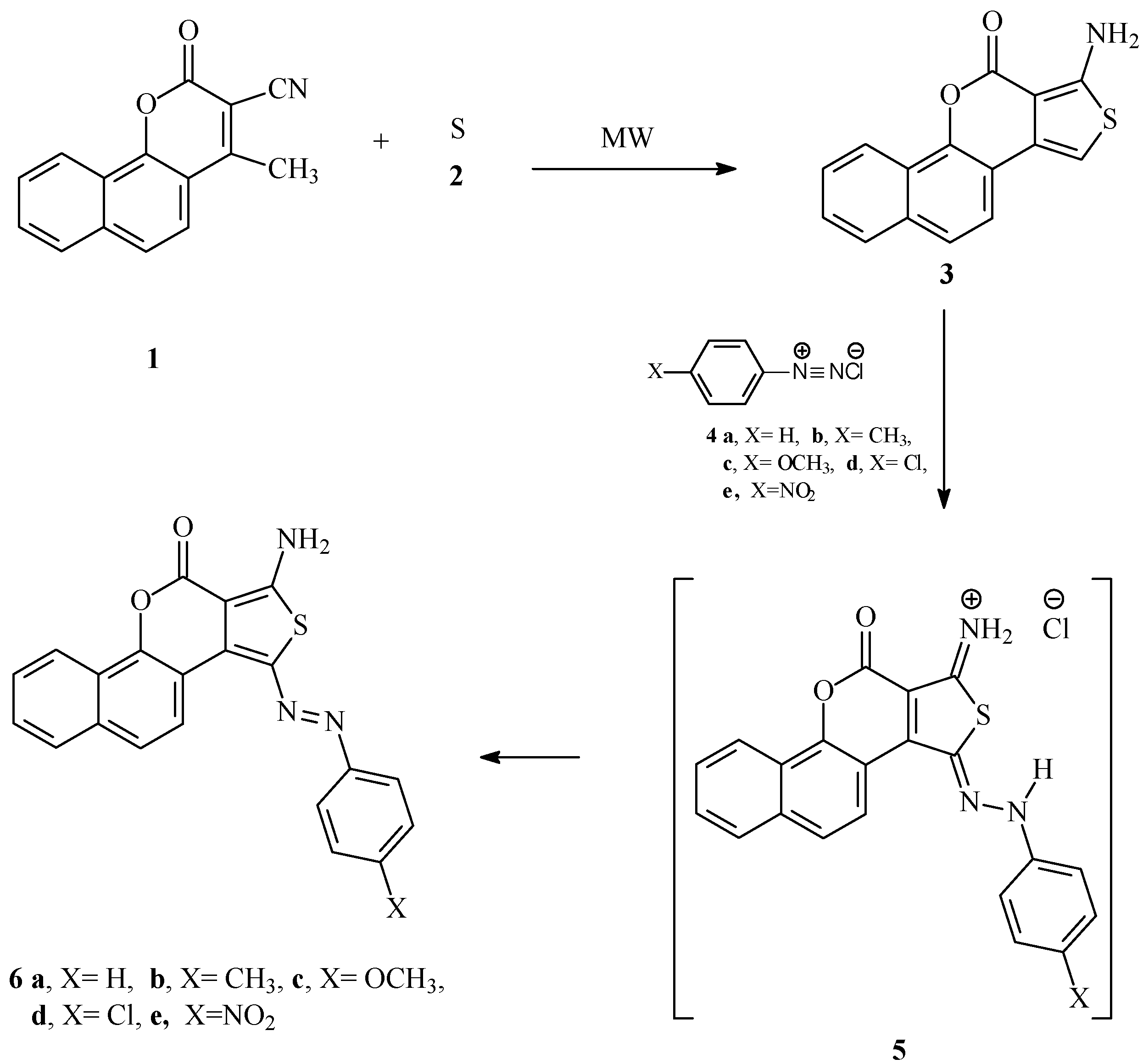
3.2. 17-Amino-11-oxa-16-thia-cyclopenta[a]phenanthren-12-one (3)
A mixture of compound 1 (2.35 g, 10 mmol), sulfur (0.32 g, 10 mmol) and dioxane (2 mL) in the presence of few drops of morpholine, was irradiated by focused microwave at 130 °C for 5 min. Completion of the reactions was monitored by TLC. The build-up of pressure in the closed reaction vessel was carefully monitored. After the irradiation, the reaction tube was cooled with high-pressure air through an inbuilt system in the instrument until the temperature had fallen below 50 °C. The mixtures were cooled and then poured into ice-water. The solids that formed were collected by using filtration and crystallized from dioxane to give compound 3 as a green powder (93%), mp 262–264 °C, IR (KBr): 3410, 3296 (NH2), 1679 (CO) cm−1. 1H-NMR (DMSO-d6): δ = 6.97 (1H, s, 1-H); 7.57–7.65 (m, 2H, arom-H), 7.75 (d, 1H, J = 8.4 Hz, arom-H), 7.84 (s. 2H, NH2, D2O-exchangeable), 7.93 (t, 2H, J = 8.0 Hz, arom-H), 8.24 (d, 1H, J = 8.0 Hz, arom-H). 13C-NMR (DMSO-d6): δ = 166.6 (CO), 158.5, 145.6, 139.2, 131.3, 127.9, 127.1, 127.0, 123.9, 123.2, 121.3, 121.0, 113.1, 97.9, 97.6. MS (EI) m/z = 267 (M]+, 100), HRMS: m/z (EI) for C15H9NO2S; calcd. 267.0384; found: 267.0384.
3.3. Reaction of Compounds 3 with Aromatic Diazonium Salts
A solution of each aryldiazonium chloride (10 mmol. prepared as described earlier [
18]) was added at 0 °C to a solution of
3 (10 mmol) in acetic acid (50 mL) containing sodium acetate (0.60 g). The reaction mixture was stirred at room temperature for 1 hr and the solid product was collected by filtration and crystallized from DMF/ethanol (3:1).
3-Amino-1-phenylazo)thieno[3,4-c][1]benzopyran-4-one (
6a). Brown crystals (81%), mp >300 °C (lit. [
18], mp >300 °C). IR: ν
max/cm
−1 3391 and 3274 (NH
2), 1699 (CO);
1H-NMR (DMSO-
d6): δ
H 7.07 (br s, 2H, NH
2), 7.40 (t, 1H,
J = 7.4 Hz, phenyl-H), 7.55 (t, 2H,
J = 7.6 Hz, phenyl-H), 7.71 (t, 2H,
J = 7.2 Hz, arom-H), 7.76 (d, 2H,
J = 8.8 Hz, phenyl-H). 7.95 (d, 1H,
J = 8.9 Hz, arom-H), 8.04 (t, 1H,
J = 7.2 Hz, arom-H). 8.37 (t, 1H,
J = 7.2 Hz, arom-H), 8.93 (d, 1H,
J = 8.8 Hz, arom-H).
13C-NMR (DMSO-
d6): δ
C 169.55 (CO), 157.4, 153.2, 149.8, 147.4, 137.6, 135.1, 131.6, 130.5, 129.9, 129.0, 128.4, 125.3, 123.8, 123.0, 122.7, 113.7, 104.8, 102.3. MS:
m/z (100%) 371 [M
+]. Anal. Calcd for C
21H
13N
3O
2S: C, 67.91; H, 3.52, N, 11.31; S, 8.63. Found C, 67.96; H, 3.23; N, 11.06; S, 8.50%.
3-Amino-1-(p-tolylazo)-thieno[3,4-c][1]benzopyran-4-one (6b). Orange powder (75%), IR (KBr): 3397 and 3275 (NH2), 1702 (CO) cm−1; 1H-NMR (DMSO-d6): δ = 2.39 (s, 3H, CH3), 7.34 (d, 2H, J = 7.8 Hz, arom-H), 7.65-7.71 (m, 6H, arom-H and NH2), 7.91 (d, 1H, J = 9.0 Hz, arom-H), 8.01 (t, 1H, J = 7.2 Hz, arom-H), 8.34 (t, 1H, J = 5.4 Hz, arom-H), 8.88 (d, 1H, J = 9.0 Hz, arom-H). MS (EI) m/z = 385 (M]+, 100), HRMS: m/z (EI) for C22H15N3O2S; calcd. 385.0871; found: 385.0871.
3-Amino-1-(4-methoxyphenyl)-thieno[3,4-c][1]benzopyran-4-one (6c). Deep orange powder (70%), mp 283–284 °C. IR (KBr): 3392 and 3277 (NH2), 1708 (CO) cm−1. 1H-NMR (DMSO-d6): δ = 3.56 (s, 3H, OCH3), 7.02 (d, 2H, J = 9.0 Hz, arom-H), 7.62–7.80 (m, 7H, arom-H and NH2), 7.92 (d, 1H, J = 7.2 Hz, arom-H), 8.25 (d, 1H, J = 9.0 Hz, arom-H), 8.74 (d, 1H, J = 9.0 Hz, arom-H). MS (EI) m/z = 401 (M]+, 17). HRMS: m/z (EI) for C22H15N3O3S; calcd. 401.0821; found: 401.0821.
3-Amino-1-(4-chlorophenylazo)thieno[3,4-c][1]benzopyran-4-one (
6d). Orange crystals (78%), mp. >300 °C (lit. [
18], mp >300 °C). IR: ν
max/cm
−1 3395 and 3280 (NH
2), 1699 (CO).
1H-NMR (CDCl
3): δH 7.58 (d, 2H,
J = 8.4 Hz, 4-chlorophenyl-H), 7.70–7.76 (m, 4H, arom-H and 4-chlorophenyl-H), 7.93 (d, 1H,
J = 8.8 Hz, arom-H), 8.03–8.05 (m, 1H, arom-H), 8.32 (br s, 2H, NH
2, D
2O exchangeable), 8.36–8.38 (m, 1H, arom-H). 8.88 (d, 1H,
J = 8.8 Hz, arom-H). MS (EI)
m/z = 405 (M]
+, 100), Anal. Calcd for C
21H
12ClN
3O
2S: C, 62.14; H, 2.98; N, 10.35; S, 7.90. Found C, 62.21; H, 3.17; N, 10.46; S, 7.67%. HRMS: calcd. 405.0336; found: 405.0336.
3-Amino-1-(4-nitrophenylazo)thieno[3,4-c][1]benzopyran-4-one (
6e). Violet crystals (74%), mp. > 300 °C (lit. [
18], mp >300 °C). IR: ν
max/cm
−1 3387 and 3270 (NH
2), 1703 (CO);
1H-NMR (DMSO-
d6): δ
H 7.12 (br s, 2H, NH
2, D
2O exchangeable), 7.72–7.73 (m, 2H, arom-H), 7.84 (d, 2H,
J = 9 Hz, 4-nitrophenyl-H), 7.94 (d, 1H,
J = 8.8 Hz, arom-H), 8.06 (m, 1H, arom-H), 8.32 (d, 2H,
J = 9 Hz, 4-nitrophenyl-H), 8.38 (d, 1H,
J = 7 Hz arom-H), 8.90 (d, 1H,
J = 8.8 Hz, arom-H). MS (EI)
m/z = 416 (M]
+, 100). Anal. Calcd for C
21H
12N
4O
4S: C, 60.57; H, 2.90; N, 13.15; S, 7.70. Found C, 60.22; H, 3.14; N, 13.18; S, 7.27%. HRMS: calcd. 416.0573; found: 416.0573.