Synthesis and Biological Properties of Caffeic Acid-PNA Dimers Containing Guanine
Abstract
:1. Introduction
| Compounds | Crude yield (%) | Theoretical molecular mass | Experimental molecular mass [M+H]+ | Δ (Da) |
|---|---|---|---|---|
| CA-ag-NH2 (1) | 45 | 745.27 | 746.28 | 0.01 |
| CA-ga-NH2 (2) | 50 | 745.27 | 746.28 | 0.01 |
| CA-cg-NH2 (3) | 55 | 721.27 | 722.27 | 0.00 |
| CA-gc-NH2 (4) | 50 | 721.27 | 722.27 | 0.00 |
| CA-tg-NH2 (5) | 45 | 736.27 | 737.27 | 0.00 |
| CA-gt-NH2 (6) | 55 | 736.27 | 737.27 | 0.00 |
| CA-gg-NH2 (7) | 40 | 761.27 | 762.27 | 0.00 |
2. Results and Discussion
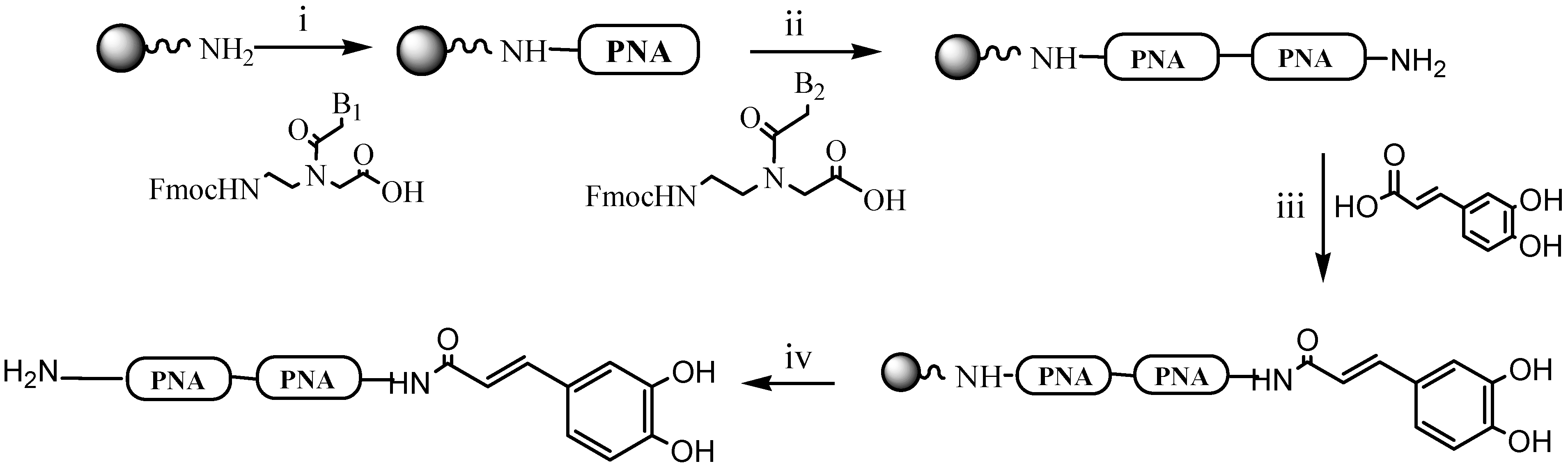
2.1. Synthesis of Dimers 1–7
| Compounds | CLog P a | CLog S b |
|---|---|---|
| Caffeic acid (CA) | 0.97 | −1.41 |
| CA-ag-NH2 (1) | −2.24 | −1.73 |
| CA-ga-NH2 (2) | −2.24 | −1.73 |
| CA-cg-NH2 (3) | −2.52 | −1.12 |
| CA-gc-NH2 (4) | −2.52 | −1.12 |
| CA-tg-NH2 (5) | −2.0 | −1.10 |
| CA-gt-NH2 (6) | −2.0 | −1.10 |
| CA-gg-NH2 (7) | −3.23 | −1.12 |
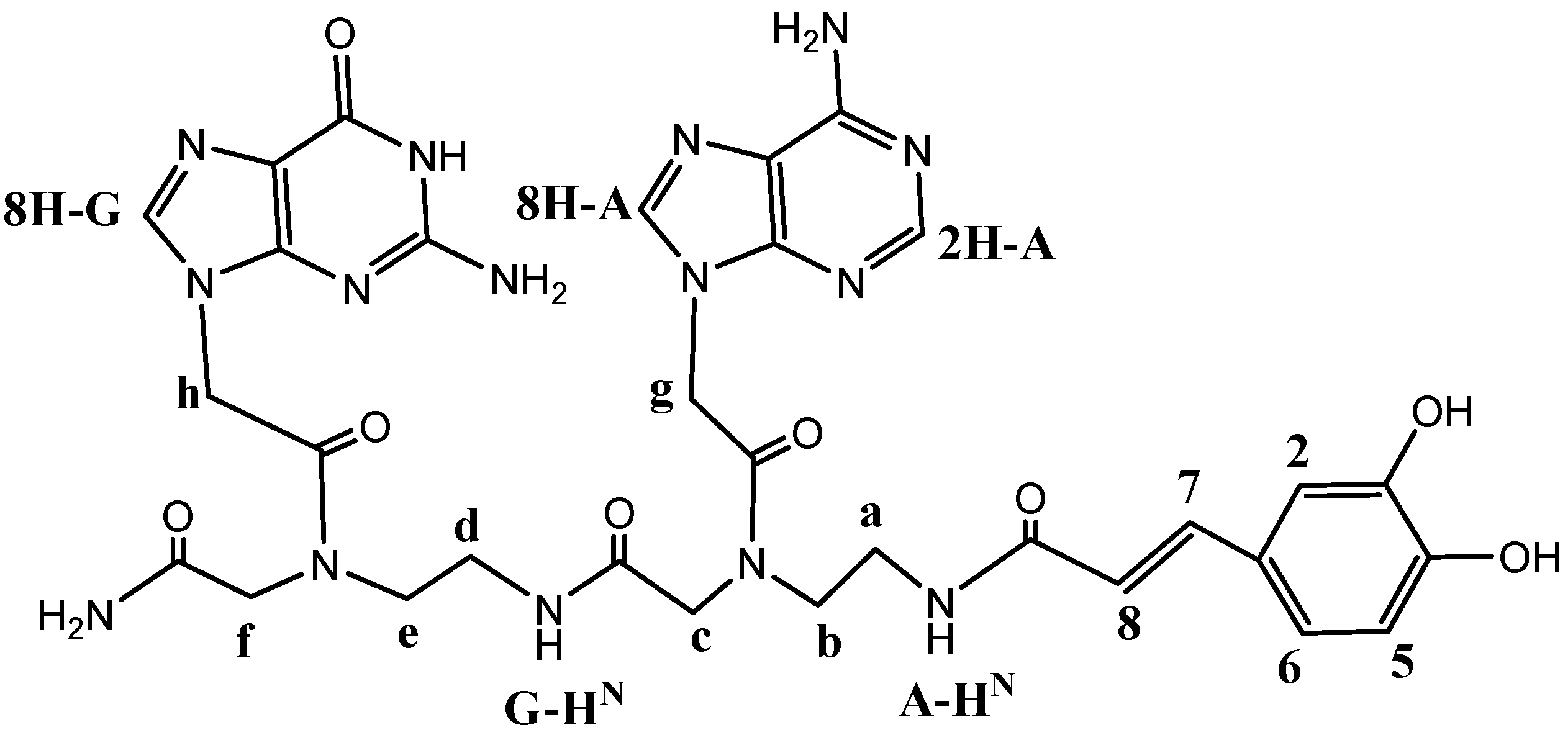
2.2. Chemical Characterization of Dimer 1

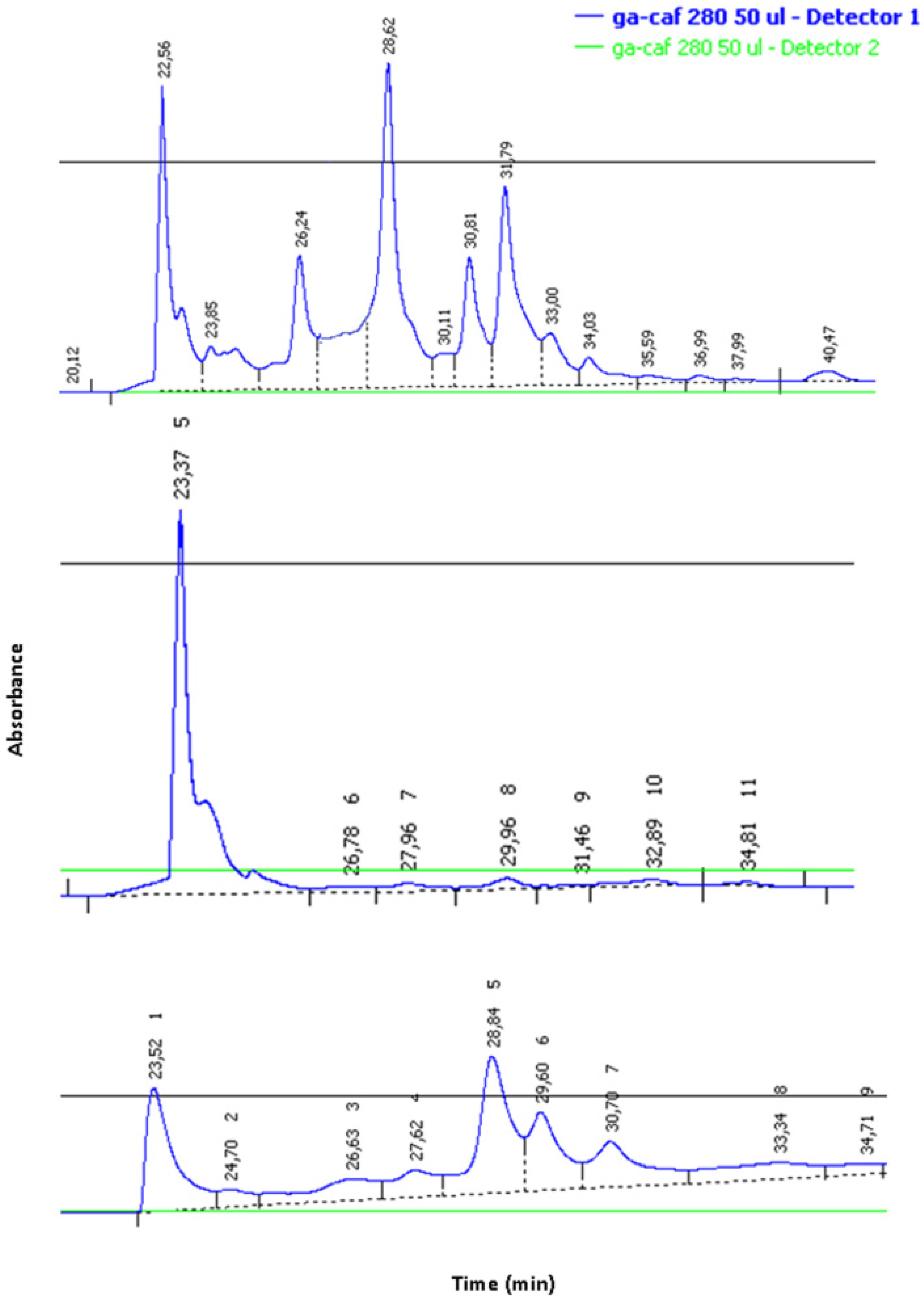
2.3. Biological Characterization

| Compounds | DPPH IC50 | ABTS IC50 | TBARS IC50 |
|---|---|---|---|
| CA-ag-NH2 (1) | >100 | >100 | 85.34 |
| CA-ga-NH2 (2) | 51.36 | 13.91 | 41.38 |


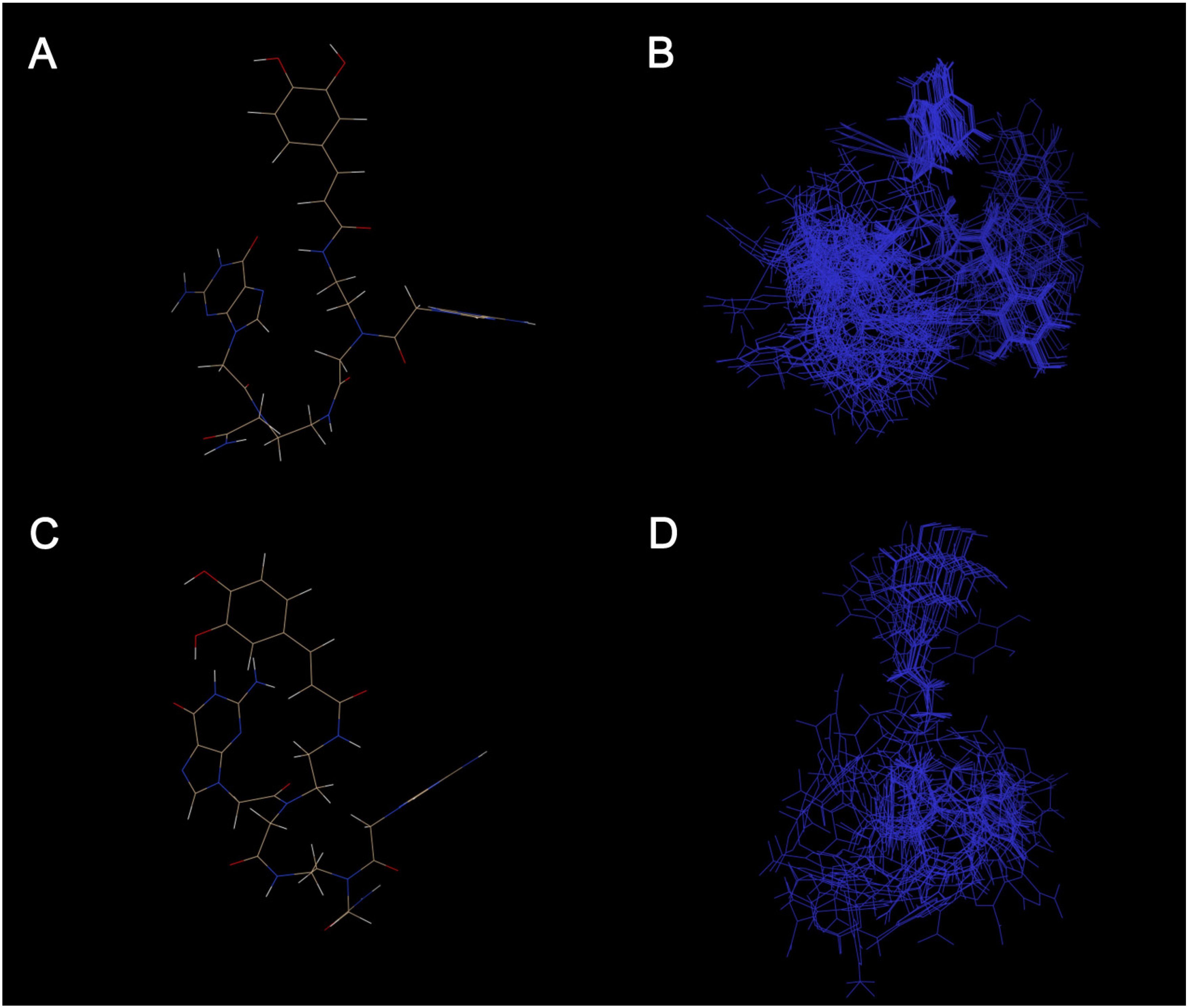
2.4. In Silico Conformational Study
3. Experimental
3.1. Synthetic Procedure for CA-PNA Dimers
3.2. NMR Characterization of CA-PNA Dimer 1
3.3. Conformation Sampling Method
3.4. Cytoprotection Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Fiuza, S.M.; Gomes, C.; Teixeira, L.J.; Girao da Cruz, M.T.; Cordeiro, M.N.; Milhazes, N.; Borges, F.; Marques, M.P. Phenolic acid derivatives with potential anticancer properties—A structure-activity relationship study. Part 1: Methyl, propyl and octyl esters of caffeic and gallic acids. Bioorg. Med. Chem. 2004, 12, 3581–3589. [Google Scholar] [CrossRef]
- Gomes, C.A.; da Cruz, T.G.; Andrade, J.L.; Milhazes, N.; Borges, F.; Marques, M.P. Anticancer Activity of Phenolic Acids of Natural or Synthetic Origin: A Structure−Activity Study. J. Med. Chem. 2003, 46, 5395–5401. [Google Scholar] [CrossRef]
- Son, S.; Lewis, B.A. Free Radical Scavenging and Antioxidative Activity of Caffeic Acid Amide and Ester Analogues: Structure−Activity Relationship. J. Agric. Food Chem. 2002, 50, 468–472. [Google Scholar] [CrossRef]
- Chung, H.S.; Shin, J.C. Characterization of antioxidant alkaloids and phenolic acids from anthocyanin-pigmented rice (Oryza sativa cv. Heugjinjubyeo). Food Chem. 2007, 104, 1670–1677. [Google Scholar] [CrossRef]
- Bezerra, D.P.; Castro, F.O.; Alves, A.P.N.N.; Pessoa, C.; Moraes, M.O.; Silveira, E.R.; Lima, M.A.S.; Elmiro, F.J.M.; Costa-Lotufo, L.V. In vivo growth-inhibition of Sarcoma 180 by piplartine and piperine, two alkaloid amides from Piper. J. Med. Biol. Res. 2006, 39, 801–807. [Google Scholar] [CrossRef]
- Naz, S.; Ahmed, S.; Rasool, S.A.; Sayeed, S.A.; Siddiqi, R. Antibacterial activity directed isolation of compounds from Onosma hispidum. Microb. Res. 2006, 161, 43–48. [Google Scholar] [CrossRef]
- Carvalho, S.A.; da Silva, E.F.; de Souza, M.V.N.; Lourenço, M.C.S.; Vicente, F.R. Synthesis and antimycobacterial evaluation of new trans-cinnamic acid hydrazide derivatives. Bioorg. Med. Chem. Lett. 2008, 18, 538–541. [Google Scholar] [CrossRef]
- Lima, C.F.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Phenolic compounds protect HepG2 cells from oxidative damage: Relevance of glutathione levels. Life Sci. 2006, 79, 2056–2068. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Stanley, R.A.; Adaim, A.; Melton, L.D.; Skinner, M.A. Free radical scavenging and cytoprotective activities of phenolic antioxidants. Mol. Nutr. Food Res. 2006, 50, 996–1005. [Google Scholar] [CrossRef]
- Pérez-Alvarez, V.; Bobadilla, R.A.; Muriel, P. Structure-Hepatoprotective Activity Relationship of 3,4-Dihydroxycinnamic Acid (Caffeic Acid) Derivatives. J. Appl. Toxicol. 2001, 21, 527–531. [Google Scholar] [CrossRef]
- Wang, X.; Stavchansky, S.; Bowman, P.D.; Kerwin, S.M. Cytoprotective effect of caffeic acid phenethyl ester (CAPE) and catechol ring-fluorinated CAPE derivatives against menadione-induced oxidative stress in human endothelial cells. Bioorg. Med. Chem. 2006, 14, 4879–4887. [Google Scholar] [CrossRef]
- Ilhan, A.; Iraz, M.; Gurel, A.; Armutcu, F.; Akyol, O. Caffeic Acid Phenethyl Ester Exerts a Neuroprotective Effect on CNS Against Pentylenetetrazol-Induced Seizures in Mice. Neurochem. Res. 2004, 29, 2287–2292. [Google Scholar] [CrossRef]
- Wang, X.; Bowman, P.D.; Kerwin, S.M.; Stavchansky, S. Stability of caffeic acid phenethyl ester and its fluorinated derivative in rat plasma. Biomed. Chromatogr. 2007, 21, 343–350. [Google Scholar] [CrossRef]
- Rajan, P.; Vedernikova, I.; Cos, P.; Berghe, D.V.; Augustyns, K.; Haemers, A. Synthesis and evaluation of caffeic acid amides as antioxidants. Bioorg. Med. Chem. Lett. 2001, 11, 215–217. [Google Scholar] [CrossRef]
- Yang, J.; Marriner, G.A.; Wang, X.; Bowman, P.D.; Kerwin, S.M.; Stavchansky, S. Synthesis of a series of caffeic acid phenethyl amide (CAPA) fluorinated derivatives: Comparison of cytoprotective effects to caffeic acid phenethyl ester (CAPE). Bioorg. Med. Chem. 2010, 18, 5032–5038. [Google Scholar] [CrossRef]
- Spasova, M.; Kortenska-Kancheva, V.; Totseva, I.; Ivanova, G.; Gerogriev, L.; Milkova, T. Synthesis of cinnamoyl and hydroxycinnamoyl amino acid conjugates and evaluation of their antioxidant activity. Pept. Sci. 2006, 12, 369–375. [Google Scholar] [CrossRef]
- Spasova, M.; Ivanova, G.; Weber, H.; Ranz, A.; Lankmayr, E.; Milkova, T. Amides of substituted cinnamic acids with aliphatic monoamines and their antioxidative potential. Oxid. Commun. 2007, 30, 803–813. [Google Scholar]
- Kwak, S.Y.; Seo, H.S.; Lee, Y.S. Synergistic antioxidative activities of hydroxycinnamoyl-peptides. J. Pept. Sci. 2009, 15, 634–641. [Google Scholar] [CrossRef]
- Seo, H.S.; Kwak, S.Y.; Lee, Y.S. Antioxidative activities of histidine containing caffeic acid-dipeptides. Bioorg. Med. Chem. Lett. 2010, 20, 4266–4272. [Google Scholar] [CrossRef]
- Kwak, S.Y.; Lee, S.; Choi, H.R.; Park, K.C.; Lee, Y.S. Dual effects of caffeoyl-amino acidyl-hydroxamic acid as an antioxidant and depigmenting agent. Bioorg. Med. Chem. Lett. 2011, 21, 5155–5158. [Google Scholar] [CrossRef]
- Koch, T. An Introduction to PNA. In Peptide Nucleic Acids: Protocols and Applications; Nielsen, P.E., Egholm, M., Eds.; Horizon Scientific Press: Norfolk, UK, 1999; pp. 21–37. [Google Scholar]
- Corey, D.R. Peptide nucleic acids: Expanding the scope of nucleic acid recognition. Trends Biotechnol. 1997, 15, 224–229. [Google Scholar] [CrossRef]
- Koppelhus, U.; Nielsen, P.E. Cellular delivery of peptide nucleic acid (PNA). Adv. Drug Deliv. Rev. 2003, 55, 267–280. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Grozema, F.C.; Fonseca Guerra, C.; Bickelhaupt, F.M.; Siebbeles, L.D.A. Mapping the Sites for Selective Oxidation of Guanines in DNA. J. Am. Chem. Soc. 2003, 125, 13658–13659. [Google Scholar]
- Gaglione, M.; Milano, G.; Chambery, A.; Moggio, L.; Romanelli, A.; Messere, A. PNA-based artificial nucleases as antisense and anti-miRNA oligonucleotide agents. Mol. Biosyst. 2011, 7, 2490–2499. [Google Scholar] [CrossRef]
- CS ChemOffice Ultra ver. 10.0. CambridgeSoft: Cambridge, MA, USA, 2006.
- VCCLAB. Virtual Computational Chemistry Laboratory. 2005. Available online: http://www.vcclab.org/lab/alogps/ (accessed on 11 March 2011).
- Broto, P.; Moreau, G.; Vandyke, C. Molecular structures: Perception, autocorrelation descriptor and SAR studies. Eur. J. Med. Chem. 1984, 19, 71–78. [Google Scholar]
- Tetko, I.V.; Bruneau, P. Application of ALOGPS to Predict 1-Octanol/Water Distribution Coefficients, logP, and logD of AstraZeneca In-House Database. J. Pharm. Sci. 2004, 93, 3103–3110. [Google Scholar] [CrossRef]
- Avitabile, C.; Moggio, L.; Malgieri, G.; Capasso, D.; Di Gaetano, S.; Saviano, M.; Pedone, C.; Romanelli, A. γ-sulphate PNA (PNA S): Highly Selective DNA Binding Molecule Showing Promising Antigene Activity. PLoS One 2012, 7, e35774. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Pacifico, S.; Gallicchio, M.; Fiorentino, A.; Fischer, A.; Meyer, U.; Stintzing, F. C. Antioxidant properties and cytotoxic effects on human cancer cell lines of aqueous fermented and lipophilic quince (Cydonia oblonga Mill.) preparations. Food Chem. Toxicol. 2012, 50, 4130–4135. [Google Scholar]
- Steeken, S. Purine bases, nucleosides, and nucleotides: Aqueous solution redox chemistry and transformation reactions of their radical cations and e- and OH adducts. Chem. Rev. 1989, 89, 503–520. [Google Scholar] [CrossRef]
- Mello, L.D.; Hernandez, S.; Marrazza, G.; Marco Mascini, M.; Kubota, L.T. Investigations of the antioxidant properties of plant extracts using a DNA-electrochemical biosensor. Biosens. Bioelectron. 2006, 21, 1374–1382. [Google Scholar] [CrossRef]
- Mersch-Sundermann, V.; Knasmüller, S.; Wu, X.-J.; Darroudi, F.; Kassie, F. Use of a human-derived liver cell line for the detection of cytoprotective, antigenotoxic and cogenotoxic agents. Toxicology 2004, 198, 329–340. [Google Scholar] [CrossRef]
- Martín, M.A.; Ramos, S.; Mateos, R.; Izquierdo-Pulido, M.; Bravo, L.; Goya, L. Protection of human HepG2 cells against oxidative stress by the flavonoid epicatechin. Phytother. Res. 2010, 24, 503–509. [Google Scholar]
- Dhuna, K.; Dhuna, V.; Bhatia, G.; Singh, J.; Kamboj, S.S. Cytoprotective effect of methanolic extract of Nardostachys jatamansi against hydrogen peroxide induced oxidative damage in C6 glioma cells. Acta Biochim. Pol. 2013, 60, 21–31. [Google Scholar]
- Seibert, H.; Maser, E.; Schweda, K.; Seibert, S.; Gulden, M. Cytoprotective activity against peroxide-induced oxidative damage and cytotoxicity of flavonoids in C6 rat glioma cells. Food Chem. Toxicol. 2011, 49, 2398–2407. [Google Scholar] [CrossRef]
- Iwata-Ichikawa, E.; Kondo, Y.; Miyazaki, I.; Asanuma, M.; Ogawa, N. Glial cells protect neurons against oxidative stress via transcriptional up-regulation of the glutathione synthesis. J. Neurochem. 1999, 72, 2334–2344. [Google Scholar]
- Liu, X.; Bai, F.; Ouyang, S.; Wang, X.; Li, H.; Jiang, H. Cyndi: A multi-objective evolution algorithm based method for bioactive molecular conformational generation. BMC Bioinform. 2009, 10, 101–115. [Google Scholar] [CrossRef]
- Hwang, T.L.; Shaka, A.J. Water Suppression That Works. Excitation Sculpting Using Arbitrary Wave-Forms and Pulsed-Field Gradients. J. Magn. Reson. A 1995, 112, 275–279. [Google Scholar] [CrossRef]
- Stejskal, E.O.; Tanner, J.E. Spin Diffusion Measurements: Spin Echoes in the Presence of a Time-Dependent Field Gradient. J. Chem. Phys. 1965, 42, 288–293. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–7 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gaglione, M.; Malgieri, G.; Pacifico, S.; Severino, V.; D'Abrosca, B.; Russo, L.; Fiorentino, A.; Messere, A. Synthesis and Biological Properties of Caffeic Acid-PNA Dimers Containing Guanine. Molecules 2013, 18, 9147-9162. https://doi.org/10.3390/molecules18089147
Gaglione M, Malgieri G, Pacifico S, Severino V, D'Abrosca B, Russo L, Fiorentino A, Messere A. Synthesis and Biological Properties of Caffeic Acid-PNA Dimers Containing Guanine. Molecules. 2013; 18(8):9147-9162. https://doi.org/10.3390/molecules18089147
Chicago/Turabian StyleGaglione, Maria, Gaetano Malgieri, Severina Pacifico, Valeria Severino, Brigida D'Abrosca, Luigi Russo, Antonio Fiorentino, and Anna Messere. 2013. "Synthesis and Biological Properties of Caffeic Acid-PNA Dimers Containing Guanine" Molecules 18, no. 8: 9147-9162. https://doi.org/10.3390/molecules18089147
APA StyleGaglione, M., Malgieri, G., Pacifico, S., Severino, V., D'Abrosca, B., Russo, L., Fiorentino, A., & Messere, A. (2013). Synthesis and Biological Properties of Caffeic Acid-PNA Dimers Containing Guanine. Molecules, 18(8), 9147-9162. https://doi.org/10.3390/molecules18089147




