Aporphine Alkaloids from the Leaves of Phoebe grandis (Nees) Mer. (Lauraceae) and Their Cytotoxic and Antibacterial Activities
Abstract
:1. Introduction
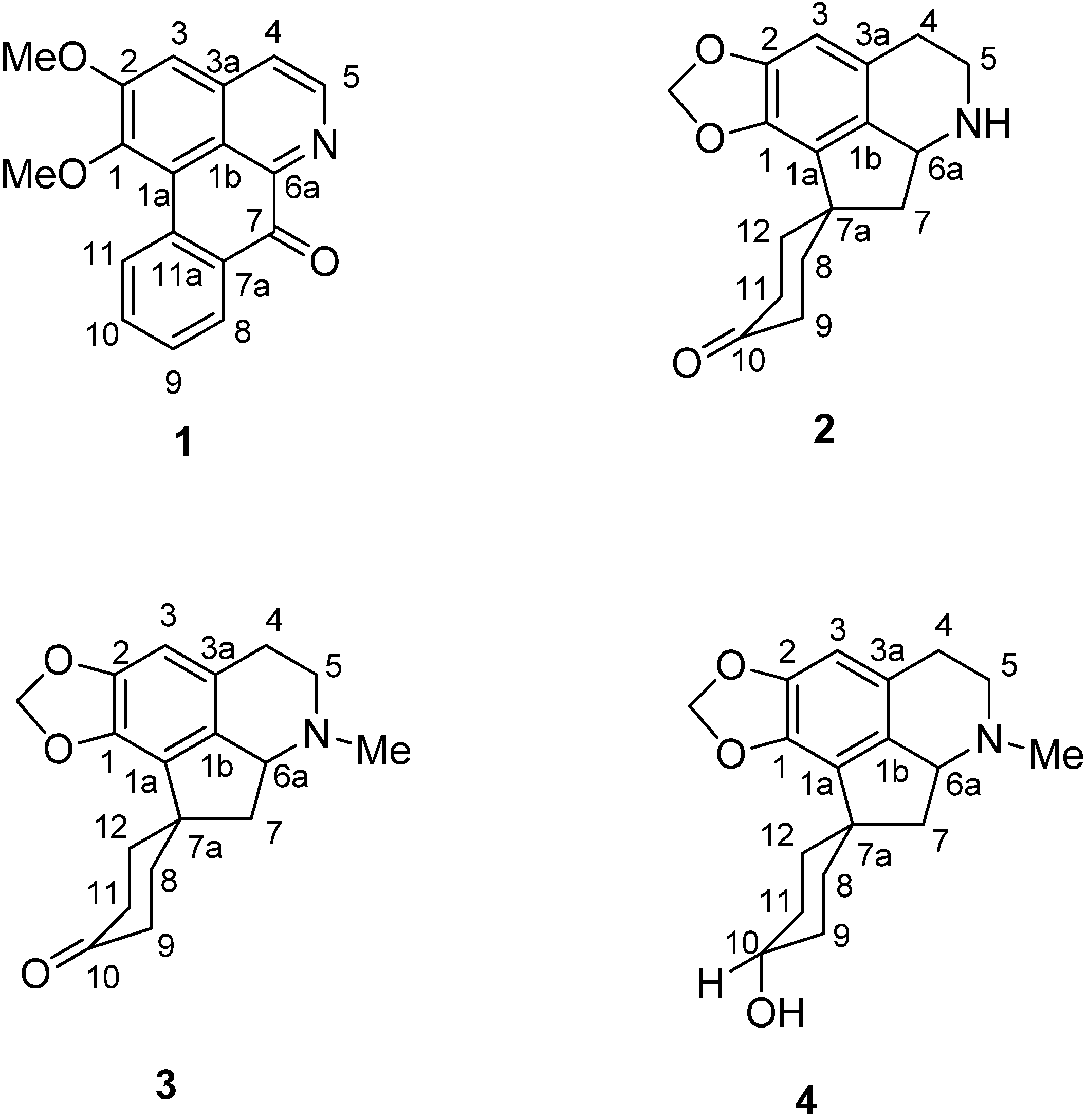
2. Results and Discussion
2.1. Compound characterization
 = −25 (c = 0.00008, CHCl3). The mass spectrum showed the [M+H]+ peak at m/z = 292.0963, which corresponds to a molecular formula of C18H13NO3. (calcd. for C18H14NO3, 292.0929). Other significant fragmentations observed were at m/z 277, which may be attributed to the loss of a CH3 molecule, [M−15]+. The UV spectrum showed absorption maxima at 236, 267, 360 and 396 nm, indicating the existence of a highly unsaturated oxoaporphine chromophore [36,37]. The IR spectrum showed a conjugated ketone peak at 1665 cm−1 [37,38]. The 1H-NMR spectrum showed two distinct methoxyl peaks at δ 4.00 and δ 4.08, which were probably situated at C-1 and C-2. H-3 appeared as a singlet at δ 7.21. Two doublets (J = 5.2 Hz) typical of the H-4 and H-5 signals of an oxoaporphine were observed at δ 7.78 and δ 8.88, respectively. The H-5 proton was deshielded by the neighbouring N atom. A very downfield signal at δ 9.16 appeared as doublet of doublet with J1 = 8.4 and J2 = 0.7 belongs to H-11. In addition, a doublet of doublet peak was observed at δ 8.57 (1H, dd, J1 = 7.9 Hz, J2 = 1.4 Hz; H-8) which experienced a deshielding effect from the neighbouring C-7 carbonyl group. The peak appeared as doublet-triplet at δ 7.75 with J1 = 8.52 Hz and J2 = 1.64Hz was assigned for H-9 whereas H-10 resonated at δ 7.56 as doublet-triplet with J1 = 8.52 Hz and J2 = 1.64 Hz. The 13C-NMR spectrum gave a total of eighteen carbons which validated the molecular formula of C18H13NO3. Analysis of the 13C-NMR spectrum gave nine quaternary carbons. Hence, compound 1 is an oxoaporphine alkaloid and in fact it was identified as lysicamine by the full agreement of the 1H- and 13C-NMR data of 1 with the literature values for that compound [39,40].
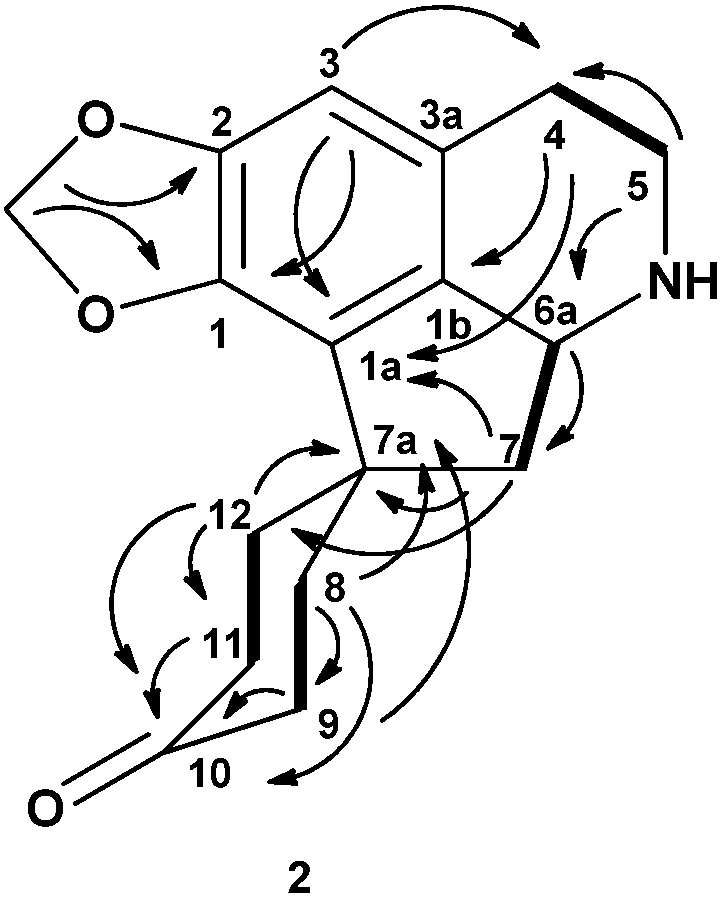
= −25 (c = 0.00008, CHCl3). The mass spectrum showed the [M+H]+ peak at m/z = 292.0963, which corresponds to a molecular formula of C18H13NO3. (calcd. for C18H14NO3, 292.0929). Other significant fragmentations observed were at m/z 277, which may be attributed to the loss of a CH3 molecule, [M−15]+. The UV spectrum showed absorption maxima at 236, 267, 360 and 396 nm, indicating the existence of a highly unsaturated oxoaporphine chromophore [36,37]. The IR spectrum showed a conjugated ketone peak at 1665 cm−1 [37,38]. The 1H-NMR spectrum showed two distinct methoxyl peaks at δ 4.00 and δ 4.08, which were probably situated at C-1 and C-2. H-3 appeared as a singlet at δ 7.21. Two doublets (J = 5.2 Hz) typical of the H-4 and H-5 signals of an oxoaporphine were observed at δ 7.78 and δ 8.88, respectively. The H-5 proton was deshielded by the neighbouring N atom. A very downfield signal at δ 9.16 appeared as doublet of doublet with J1 = 8.4 and J2 = 0.7 belongs to H-11. In addition, a doublet of doublet peak was observed at δ 8.57 (1H, dd, J1 = 7.9 Hz, J2 = 1.4 Hz; H-8) which experienced a deshielding effect from the neighbouring C-7 carbonyl group. The peak appeared as doublet-triplet at δ 7.75 with J1 = 8.52 Hz and J2 = 1.64Hz was assigned for H-9 whereas H-10 resonated at δ 7.56 as doublet-triplet with J1 = 8.52 Hz and J2 = 1.64 Hz. The 13C-NMR spectrum gave a total of eighteen carbons which validated the molecular formula of C18H13NO3. Analysis of the 13C-NMR spectrum gave nine quaternary carbons. Hence, compound 1 is an oxoaporphine alkaloid and in fact it was identified as lysicamine by the full agreement of the 1H- and 13C-NMR data of 1 with the literature values for that compound [39,40]. = −25 (c = 0.00004, CHCl3). This proaporphine alkaloid exhibited an [M+H]+ peak in the LCMS-IT-TOF ESI (positive mode) mass spectrum at m/z 286.1421 which correlated to the molecular formula of C17H19NO3 (calcd. for C17H20NO3, 286.1432). The UV spectrum revealed three peaks at 300, 236 and 207 nm, which indicated the existence of a conjugated system [37]. The IR spectrum revealed a very significant carbonyl absorption at 1712.52 cm−1. In addition, the presence of a methylenedioxyl group was proven by its characteristic absorption peaks at 1248.37 cm−1 and 934.90 cm−1, which indicate asymmetric C-O-C stretching.
= −25 (c = 0.00004, CHCl3). This proaporphine alkaloid exhibited an [M+H]+ peak in the LCMS-IT-TOF ESI (positive mode) mass spectrum at m/z 286.1421 which correlated to the molecular formula of C17H19NO3 (calcd. for C17H20NO3, 286.1432). The UV spectrum revealed three peaks at 300, 236 and 207 nm, which indicated the existence of a conjugated system [37]. The IR spectrum revealed a very significant carbonyl absorption at 1712.52 cm−1. In addition, the presence of a methylenedioxyl group was proven by its characteristic absorption peaks at 1248.37 cm−1 and 934.90 cm−1, which indicate asymmetric C-O-C stretching.| Position | 13C (δ, CDCl3) | Type | 1H (J, Hz) | HMBC (2J, 3J) |
|---|---|---|---|---|
| 1 | 148.9 | Cq | - | |
| 1a | 141.1 | Cq | - | |
| 1b | 123.9 | Cq | - | |
| 2 | 141.1 | Cq | - | |
| 3 | 106.9 | CH | 6.51 s | C1, C1a, C2, C4 |
| 3a | 126.7 | Cq | - | |
| 4 | 25.1 | CH2 | 2.87 m (ax) | C3, C1b, C1a |
| 2.75 m (eq) | C3, C1b, C1a | |||
| 5 | 43.9 | CH2 | 3.55 m (ax) | C4, C6a, C1b |
| 3.16 m (eq) | C4, C6a | |||
| 6a | 56.8 | CH | 4.26 dd | C5, C7 |
| (J = 6.9 Hz, | ||||
| J = 9.8 Hz) | ||||
| 7 | 44.1 | CH2 | 2.73 m (ax) | C3, C3a, C1a |
| 1.87 m (eq) | C12, C7a, C6a | |||
| 7a | 46.2 | Cq | - | - |
| 8 | 38.5 | CH2 | 2.68 m (ax) | C10 |
| 2.41 m (eq) | C9, C7a, C10 | |||
| 9 | 36.2 | CH2 | 2.50 m (ax) | C8, C7a, C10 |
| 1.91 m (eq) | C11, C7a, C10 | |||
| 10 | 211.1 | C=O | - | - |
| 11 | 38.9 | CH2 | 2.46 m (ax) | C10 |
| 2.45 m (eq) | C12, C7a, C10 | |||
| 12 | 34.3 | CH2 | 2.10 m (ax) | C9, C7, C7a, C3a, C10 |
| 2.00 m (eq) | C9, C7a, C3a, C10 | |||
| Methylenedioxy (O-CH2-O) | 100.9 | CH2 | 5.89 d (J = 1.3 Hz) | C1,C2 |
| 5.86 d (J = 1.3 Hz) | C1,C2 |
 = −25 (c = 0.00004, CHCl3). The LCMS-IT-TOF mass spectrum of this proaporphine type of alkaloid showed a [M+H]+ peak at m/z = 300.1596 which correlated to the molecular formula C18H21NO3 (calcd. for C18H22NO3, 300.1521). The UV spectrum showed an absorption peak at 203 nm. The IR spectrum showed a very significant carbonyl absorption peak at 1712.80 cm−1 due to C=O stretching vibrations. The presence of the methylenedioxyl group was proven by its characteristic absorption peaks at 1254.45 and 944.84 cm−1, which indicate the asymmetric C-O-C stretching.
= −25 (c = 0.00004, CHCl3). The LCMS-IT-TOF mass spectrum of this proaporphine type of alkaloid showed a [M+H]+ peak at m/z = 300.1596 which correlated to the molecular formula C18H21NO3 (calcd. for C18H22NO3, 300.1521). The UV spectrum showed an absorption peak at 203 nm. The IR spectrum showed a very significant carbonyl absorption peak at 1712.80 cm−1 due to C=O stretching vibrations. The presence of the methylenedioxyl group was proven by its characteristic absorption peaks at 1254.45 and 944.84 cm−1, which indicate the asymmetric C-O-C stretching.| Position | 13C (δ, CDCl3) | Type | 1H (J, Hz) | HMBC (2J, 3J) |
|---|---|---|---|---|
| 1 | 140.7 | Cq | - | - |
| 1a | 134.3 | Cq | - | - |
| 1b | 124.5 | Cq | - | - |
| 2 | 148.2 | Cq | - | - |
| 3 | 106.5 | CH | 6.49 s | C1a, C1, C2,C4 |
| 3a | 126.9 | Cq | - | |
| 4 | 27.4 | CH2 | 2.92 (m) ax | C1b, C5 |
| 2.72 (m) eq | C1b, C1a, C3 | |||
| 5 | 55.0 | CH2 | 3.09 (m) ax | C1b, C4, NCH3, C6a |
| 2.45 (m) eq | ||||
| 6a | 65.7 | CH | 3.30 br s | - |
| 7 | 44.5 | CH2 | 2.59 (m) ax | C3a, C1a, C8, C7a, C6a |
| 1.75 (m) eq | C8, C12, C7a, C6a | |||
| 7a | 46.0 | Cq | - | - |
| 8 | 34.6 | CH2 | 2.14 (m) ax | C3a, C12, C7 |
| 2.02 (m) eq | C3a, C12, C7a | |||
| 9 | 39.0 | CH2 | 2.47 (m) | C8, C11, C7a |
| C8, C11, C7a | ||||
| 10 | 211.7 | C=O | - | - |
| 11 | 38.6 | CH2 | 2.70 (m) ax | C12, C7a |
| 2.43 (m) eq | C12, C7a | |||
| 12 | 36.5 | CH2 | 2.50 (m) ax | C8, C11, C7 |
| 1.93 (m) eq | C3a, C8, C11, C7a | |||
| N-CH3 | 43.5 | CH3 | 2.39 s | C5, C6a |
| Methlenedioxy | 100.6 | CH2 | 5.88 d (J = 1.2) | C1, C2 |
| (O-CH2-O) | 5.83 d (J = 1.2) | C1, C2 |

 = +100 (c = 0.00003, CHCl3). The mass spectrum showed a molecular ion peak at m/z = 302.9177 [M+H]+, thus suggesting a molecular formula of C18H23NO3 (calcd. for C18H24NO3, 302.9167). The UV spectrum showed three peaks at 300, 245 and 265 nm, which indicate the existence of a conjugated system [37]. The IR spectrum showed a broad absorption band at 3391.78 cm−1, indicating the presence of a hydroxyl group. The methylenedioxyl group absorbed at 1254.36 and 929.07 cm−1. There was no carbonyl group present in the IR spectrum.
= +100 (c = 0.00003, CHCl3). The mass spectrum showed a molecular ion peak at m/z = 302.9177 [M+H]+, thus suggesting a molecular formula of C18H23NO3 (calcd. for C18H24NO3, 302.9167). The UV spectrum showed three peaks at 300, 245 and 265 nm, which indicate the existence of a conjugated system [37]. The IR spectrum showed a broad absorption band at 3391.78 cm−1, indicating the presence of a hydroxyl group. The methylenedioxyl group absorbed at 1254.36 and 929.07 cm−1. There was no carbonyl group present in the IR spectrum.| Position | 13C (δ, CDCl3) | Type | 1H (J, Hz) | HMBC (2J, 3J) |
|---|---|---|---|---|
| 1 | 148.1 | Cq | - | - |
| 1a | 129.0 | Cq | - | - |
| 1b | 131.0 | Cq | - | - |
| 2 | 140.8 | Cq | - | - |
| 3 | 105.9 | CH | 6.46 (s) | C1, C2,C4 |
| 3a | 124.0 | Cq | - | - |
| 4 | 27.3 | CH2 | 2.93 (m) ax | C5 |
| 2.71 (m) eq | C3a | |||
| 5 | 54.9 | CH2 | 3.11 (m) ax | C3a, C6a |
| 2.46 (m) eq | ||||
| 6a | 65.7 | CH | 3.26 (m) | - |
| 7 | 44.3 | CH2 | 2.44 (m) ax | C1a |
| 1.58 (m) eq | C12, C9 | |||
| 7a | 46.6 | Cq | - | - |
| 8 | 30.2 | CH2 | 2.03 (m) ax | C7a, C10 |
| 1.54 (m) eq | C12, C7a, C10 | |||
| 9 | 31.7 | CH2 | 2.41 (m) ax | |
| 1.46 (m) eq | C8, C7a, C10 | |||
| 10 | 67.1 | CH | 4.00 br, m | |
| 11 | 31.0 | CH2 | 1.75 (m) | C12, C7a, C10 |
| 12 | 29.7 | CH2 | 1.25 (m) | |
| N-CH3 | 43.2 | CH3 | 2.39 (s) | C5, C6a |
| (OCH2O) | 100.5 | CH2 | 5.90 (d, J = 1.2) | C1, C2 |
| 5.86 (d, J = 1.2) | C1, C2 |

2.2. Cell Culture and MTT Cytotoxicity Activity
| Compounds | IC50 (µg/mL) at 24 h | |
|---|---|---|
| MCF7 | HepG2 | |
| 1 | 26 | 27 |
| 2 | 60 | 14 |
| 3 | >100 | 81 |
| 4 | >100 | 20 |
| Doxorubicin | 0.2 | 1.06 |
2.3. Antibacterial Activity
| Sample | Inhibition diameter (mm ± SD) | ||||
|---|---|---|---|---|---|
| Staphylococcus epidermidis | Staphylococcus aureus | Bacillus subtilis | Pasteurella multocida | Enterobacter cloacae | |
| (Gram +ve) | (Gram +ve) | (Gram +ve) | (Gram −ve) | (Gram −ve) | |
| 1 | 12.00 ± 0.00 | 13.33 ± 0.57 | 15.50 ± 0.57 | NI | NI |
| 2 | NI | NI | NI | NI | NI |
| 3 | NI | NI | NI | NI | NI |
| 4 | nt | nt | nt | nt | nt |
| Streptomycin sulfate a | 20.00 ± 0.00 | 13.66 ± 0.57 | 21.00 ± 0.00 | 21.33 ± 1.15 | NI |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation of the Alkaloids
3.4. Cell Culture and MTT Cytotoxicity Assay
3.5. Bacterial Cultures and Disc Diffusion Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Tayarani-Najaran, Z.; Ahmad Emami, S. Cytotoxic Plants: Potential Uses in Prevention and Treatment of Cancer, Current Cancer Treatment—Novel Beyond Conventional Approaches; Ozdemir, O., Ed.; In Tech: New York, NY, USA, 2011. Available online: http://www.intechopen.com (accessed on 9 December 2011).
- Parkin, D.M.; Fernandez, L.M.G. Use of statistics to assess the global burden of breast cancer. Breast J. 2006, 12, S70–S80. [Google Scholar] [CrossRef]
- Sakpakdeejaroen, I.; Itharat, A. Cytotoxic compounds against breast adenocarcinoma cells (MCF-7) from pikutbenjakul. J. Health Res. 2009, 23, 71–76. [Google Scholar]
- Abu-Dahab, R.; Afifi, F. Antiproliferative activity of selected medicinal plants of Jordan against a breast adenocarcinoma cell line (MCF7). Sci. Pharm. 2007, 75, 121–136. [Google Scholar] [CrossRef]
- Saleem, M.; Nazir, M.; Shaiq Ali, M.; Hussain, H.; Sup Lee, Y.; Riaz, N.; Jabbar, A. Antimicrobial natural products: An update on future antibiotic drug candidates. Nat. Prod. Rep. 2010, 27, 238–254. [Google Scholar] [CrossRef]
- Schleifer, K.H.; Kloos, W.E. Isolation and characterization of staphylococci from human skin I. Amended Descriptions of Staphylococcus epidermidis and Staphylococcus saprophyticus and Descriptions of three new species: Staphylococcus cohnii, Staphylococcus haemolyticus, and Staphylococcus xylosus. Int. J. Syst. Bacteriol. 1975, 25, 50–61. [Google Scholar] [CrossRef]
- Kluytmans, J.; van Belkum, A.; Verbrugh, H. Nasal carriage of Staphylococcus aureus: Epidemiology, underlying mechanisms, and associated risks. Clin. Microbiol. Rev. 1997, 10, 505–520. [Google Scholar]
- Cole, A.M.; Tahk, S.; Oren, A.; Yoshioka, D.; Kim, Y.H.; Park, A.; Ganz, T. Determinants of Staphylococcus aureus nasal carriage. Clin. Diagn. Lab. Immunol. 2001, 8, 1064–1069. [Google Scholar]
- Hong, H.A.; Khaneja, R.; Tam, N.M.K.; Cazzato, A.; Tan, S.; Urdaci, M.; Brisson, A.; Gasbarrini, A.; Barnes, I.; Cutting, S.M. Bacillus subtilis isolated from the human gastrointestinal tract. Res. Microbiol. 2009, 160, 134–143. [Google Scholar] [CrossRef]
- Boyce, J.D.; Adler, B. The capsule is a virulence determinant in the pathogenesis of Pasteurella multocida M1404 (B:2). Infect. Immun. 2000, 68, 3463–3468. [Google Scholar] [CrossRef]
- Harper, M.; Cox, A.D.; St Michael, F.; Wilkie, I.W.; Boyce, J.D.; Adler, B. A heptosyltransferase mutant of Pasteurella multocida produces a truncated lipopolysaccharide structure and is attenuated in virulence. Infect. Immun. 2004, 72, 3436–3443. [Google Scholar] [CrossRef]
- Musil, I.; Jensen, V.; Schilling, J.; Ashdown, B.; Kent, T. Enterobacter cloacae infection of an expanded polytetrafluoroethylene femoral-popliteal bypass graft: A case report. J. Med. Case Rep. 2010, 4, 131. [Google Scholar] [CrossRef]
- Ng, F.S.P. Tree Flora of Malaya; Longman: Kuala Lumpur, Malaysia, 1989; p. 4. [Google Scholar]
- Henry, T.A. The Plant Alkaloids, 4th ed.; J.A. Churchill Ltd.: London, UK, 1949; p. 317. [Google Scholar]
- Perry, L.M.; Metzgr, J. Medicinal Plants East and Southest Asia: Attributed Properties and Uses; MIT Press: Cambridge, MA, USA and London, UK, 1980; pp. 20, 201–202. [Google Scholar]
- Bhakuni, D.S.; Gupta, S. The Alkaloids of Stephania glabra. J. Nat Prod. 1982, 45, 407–411. [Google Scholar] [CrossRef]
- Castro, O.; Lopez, J.; Stermitz, F.R. New aporphine alkaloids from Phoebe valeriana. J. Nat. Prod. 1986, 49, 1036–1040. [Google Scholar] [CrossRef]
- CSIR, The Wealth of India, Raw Materials; Sp-W CSIR: New Delhi, India, 1989; Volume 10, pp. 41–44.
- Mukhtar, M.R.; Martin, M.T.; Domansky, M.; Pais, M.; Hadi, A.H.A.; Awang, K. Phoebegrandines A and B, proaporphine-tryptamine dimers, from Phoebe grandis. Phytochemistry 1997, 45, 1543–1546. [Google Scholar] [CrossRef]
- Mukhtar, M.R.; Hadi, A.H.A.; Sévenet, T.; Martin, M.T.; Awang, K. Phoebegrandines C A novel proaporphine-tryptamine dimer, from Phoebe grandis (Nees) Merr. Nat. Prod. Res. 2004, 18, 163–167. [Google Scholar] [CrossRef]
- Stermitz, F.R.; Castro, C.O. Pentasubstituted aporphine alkaloids from Phoebe molicella. J. Nat. Prod. 1983, 46, 913–916. [Google Scholar] [CrossRef]
- Castro, O.C.; Lopez, J.V.; Vergara, A.G. Aporphine alkaloids from Phoebe pittieri. Phytochemistry 1985, 24, 203–204. [Google Scholar] [CrossRef]
- Chen, C.C.; Huang, Y.L.; Lee, S.S.; Ou, J.C. Laurodionine, a new oxalyl-fused aporphine alkaloid from Phoebe formosana. J. Nat. Prod. 1997, 60, 826–827. [Google Scholar] [CrossRef]
- Semwal, D.K.; Rawat, U.; Singh, G. Further aporphine alkaloids from Phoebe lanceolata. Molbank 2008, 3, 581. [Google Scholar] [CrossRef]
- Mukhtar, M.R.; Aziz, A.N.; Thomas, N.F.; Hadi, A.H.A.; Litaudon, M.; Awang, K. Grandine A, a new proaporphine alkaloid from the bark of Phoebe grandis. Molecules 2009, 14, 1227–1233. [Google Scholar] [CrossRef]
- Hufford, C.; Morgan, J. Synthesis of (+-)-3-methoxy-N-acetylnornantenine. J. Organ. Chem. 1976, 41, 375–376. [Google Scholar] [CrossRef]
- Awang, K.; Mukhtar, M.R.; Mustafa, M.R.; Litaudon, M.; Shaari, K.; Mohamad, K.; Hadi, A.H.A. New alkaloids from Phoebe scortechinii. Nat. Prod. Res. 2007, 21, 704–709. [Google Scholar] [CrossRef]
- Chiou, C.M.; Kang, J.J.; Lee, S.S. Litebamine N-Homologues: Preparation and anti-acetylcholinesterase activity. J. Nat. Prod. 1998, 61, 46–50. [Google Scholar] [CrossRef]
- Semwal, D.K.; Rawat, U.; Bamola, A.; Semwal, R. Antimicrobial activity of Phoebe lanceolata and Stephania Glabra: Preliminary screening studies. J. Sci. Res. 2009, 1, 662–666. [Google Scholar]
- Ridley, H.N. The Flora of Malay Peninsular; L. Reeve, Co. Ltd.: London, UK, 1967; pp. 103–106. [Google Scholar]
- Corner, E.H.J. Wayside Trees of Malaya, 3rd ed.; The Malayan Nature Society: Kuala Lumpur, Malaysia, 1988; pp. 371–381. [Google Scholar]
- Awang, K.; Mukhtar, M.R.; Hadi, A.H.A.; Litaudon, M.; Latip, J.; Abdullah, N.R. New alkaloids from Phoebe grandis (Nees) Merr. Nat. Prod. Res. 2006, 20, 567–572. [Google Scholar] [CrossRef]
- Castro, O.; Lopez, J.; Vergara, A.; Stermitz, F.R. Phenylpropanoids in alkaloid-free species of Phoebe. J. Nat. Prod. 1985, 48, 640–641. [Google Scholar] [CrossRef]
- Mukhtar, M.R.; Hadi, A.H.A.; Rondeau, D.; Richomme, P.; Litaudon, M.; Mustafa, M.R.; Awang, K. New proaporphines from the bark of Phoebe scortechinii. Nat. Prod. Res. 2008, 22, 921–926. [Google Scholar] [CrossRef] [Green Version]
- Nakasato, T.; Asada, S. Structure of litsericine. J. Pharmac. Soc. Jpn. 1966, 86, 1205. [Google Scholar]
- Chen, C.L.; Chang, H.M.; Cowling, E.B. Aporphine alkaloids and lignans in heartwood of Liriodendron tulipifera. Phytochemistry 1976, 15, 547–550. [Google Scholar] [CrossRef]
- Svatava, D.; Ldislav, H.; Vladimir, P.; Frantisek, S. The oxoaporphine alkaloids. Heterocycles 1975, 3, 575–613. [Google Scholar] [CrossRef]
- Duddley, H.W.; Fleming, I. Spectroscopic Methods in Organic Chemistry, 4th ed.; Mc Graw Hill: London, UK, 1989; pp. 29–62. [Google Scholar]
- Guinaudeau, H.; Leboeuf, M.; Cavé, A. Aporphinoid alkaloids, III. J. Nat. Prod. 1983, 46, 761–835. [Google Scholar] [CrossRef]
- Harrigan, G.G.; Gunatilaka, A.L.; Kingston, D.G.I.; Chan, G.W.; Johnson, R.K. Isolation of bioactive and other oxoaporphine alkaloids from two annonaceous plants, Xylopia aethiopica and Miliusa cf. banacea. J. Nat. Prod. 1994, 57, 68–73. [Google Scholar] [CrossRef]
- Casagrande, C.; Canonica, L.; Ricca, G.S. Title. J. Chem. Soc. Perkin 1 1975, 17, 1659. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Likhitwitayawuid, K.; Angerhofer, C.K.; Chai, H.; Pezzuto, J.M.; Cordell, G.A. Cytotoxic and antimalarial alkaloids from the tubers of Stephania pierrei. J. Nat. Prod. 1993, 56, 1468–1478. [Google Scholar] [CrossRef]
- Mohammed, M.M.D.; Ibrahim, N.A.; Awad, N.E.; Matloub, A.A.; Mohamed-ali, A.G.; Barakat, E.E.; Mohamed, E.E.; Colla, P.L. Anti-HIV-1 and cytotoxicity of the alkaloids of Erythrina abyssinica Lam. Growing in Sudan. Nat. Prod. Res. 2012, 26, 1565–1575. [Google Scholar] [CrossRef]
- Makarasen, A.; Sirithana, W.; Mogkhuntod, S.; Khunnawutmanotham, N.; Chimnoi, N.; Techasakul, S. Cytotoxic and antimicrobial activities of aporphine alkaloids isolated from Stephania venosa (blume) spreng. Planta Med. 2011, 77, 1519–1524. [Google Scholar] [CrossRef]
- Vlietinck, A.J.; Hoof, L.V.; Totte, J.; Lasure, A.; Berghe, D.V.; Rwangabo, P.C.; Mvukiyumwami, J. Screening of hundred Rwandese medicinal plants for antimicrobial and antiviral properties. J. Ethnopharmacol. 1995, 46, 31–47. [Google Scholar] [CrossRef]
- Martin, G.J. A Methods Manual; Chapman and Hall: London, UK, 1995; p. 80. [Google Scholar]
- Paz, E.A.; Cerdeiras, M.P.; Fernandez, J.; Ferreira, F.; Moyna, P.; Soubes, M.; Vazquez, A.; Vero, S.; Zunino, L. Screening of Uruguayan medicinal plants for antimicrobial activity. J. Ethnopharmacol. 1995, 45, 67–70. [Google Scholar] [CrossRef]
- Yao, J.; Moellering, R. Antibacterial Agents. In Manual of Clinical Microbiology; Murray, P., Baron, E., Pfaller, M., Tenover, F., Yolken, R., Eds.; ASM Press: Washington, DC, USA, 1995; pp. 1281–1290. [Google Scholar]
- Nikaido, H. Outer Membrane. In Escherichia coli and Salmonella: Cellular and Molecular Biology; Neidhardt, F.C., Ed.; ASM Press: Washington, DC, USA, 1996; Volume 1, pp. 29–47. [Google Scholar]
- Gao, Y.; van Belkum, M.J.; Stiles, M.E. The outer membrane of Gram-negative bacteria inhibits antibacterial activity of brochocin-C. Appl. Environ. Microb. 1999, 65, 4329–4333. [Google Scholar]
- Duffy, C.F.; Power, R.F. Antioxidant and antimicrobial properties of some Chinese plant extracts. Int. J. Antimicrob. Agent. 2001, 17, 527–529. [Google Scholar] [CrossRef]
- Westh, H.; Zinn, C.S.; Rosdahl, V.T.; Sarisa, S. An international multicenter study of antimicrobial consumption and resistance in Staphylococcus aureus isolates from 15 hospitals in 14 countries. Microb. Drug Resis. 2004, 10, 169–176. [Google Scholar] [CrossRef]
- Hashim, N.M.; Rahmani, M.; Ee, G.C.L.; Sukari, M.A.; Yahayu, M.; Amin, M.A.M.; Ali, A.M.; Go, R. Antioxidant, antimicrobial and tyrosinase inhibitory activities of xanthones isolated from Artocarpus obtusus FM Jarrett. Molecules 2012, 17, 6071–6082. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Omar, H.; Hashim, N.M.; Zajmi, A.; Nordin, N.; Abdelwahab, S.I.; Azizan, A.H.S.; Hadi, A.H.A.; Ali, H.M. Aporphine Alkaloids from the Leaves of Phoebe grandis (Nees) Mer. (Lauraceae) and Their Cytotoxic and Antibacterial Activities. Molecules 2013, 18, 8994-9009. https://doi.org/10.3390/molecules18088994
Omar H, Hashim NM, Zajmi A, Nordin N, Abdelwahab SI, Azizan AHS, Hadi AHA, Ali HM. Aporphine Alkaloids from the Leaves of Phoebe grandis (Nees) Mer. (Lauraceae) and Their Cytotoxic and Antibacterial Activities. Molecules. 2013; 18(8):8994-9009. https://doi.org/10.3390/molecules18088994
Chicago/Turabian StyleOmar, Hanita, Najihah Mohd. Hashim, Asdren Zajmi, Noraziah Nordin, Siddiq Ibrahim Abdelwahab, Ainnul Hamidah Syahadah Azizan, A. Hamid A. Hadi, and Hapipah Mohd Ali. 2013. "Aporphine Alkaloids from the Leaves of Phoebe grandis (Nees) Mer. (Lauraceae) and Their Cytotoxic and Antibacterial Activities" Molecules 18, no. 8: 8994-9009. https://doi.org/10.3390/molecules18088994






