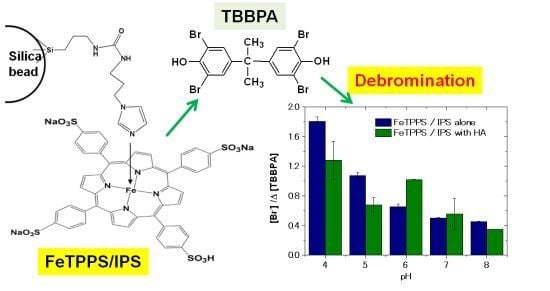
Oxidative Debromination and Degradation of Tetrabromo-bisphenol A by a Functionalized Silica-Supported Iron(III)-tetrakis(p-sulfonatophenyl)porphyrin Catalyst
Abstract
:1. Introduction
2. Results and Discussion
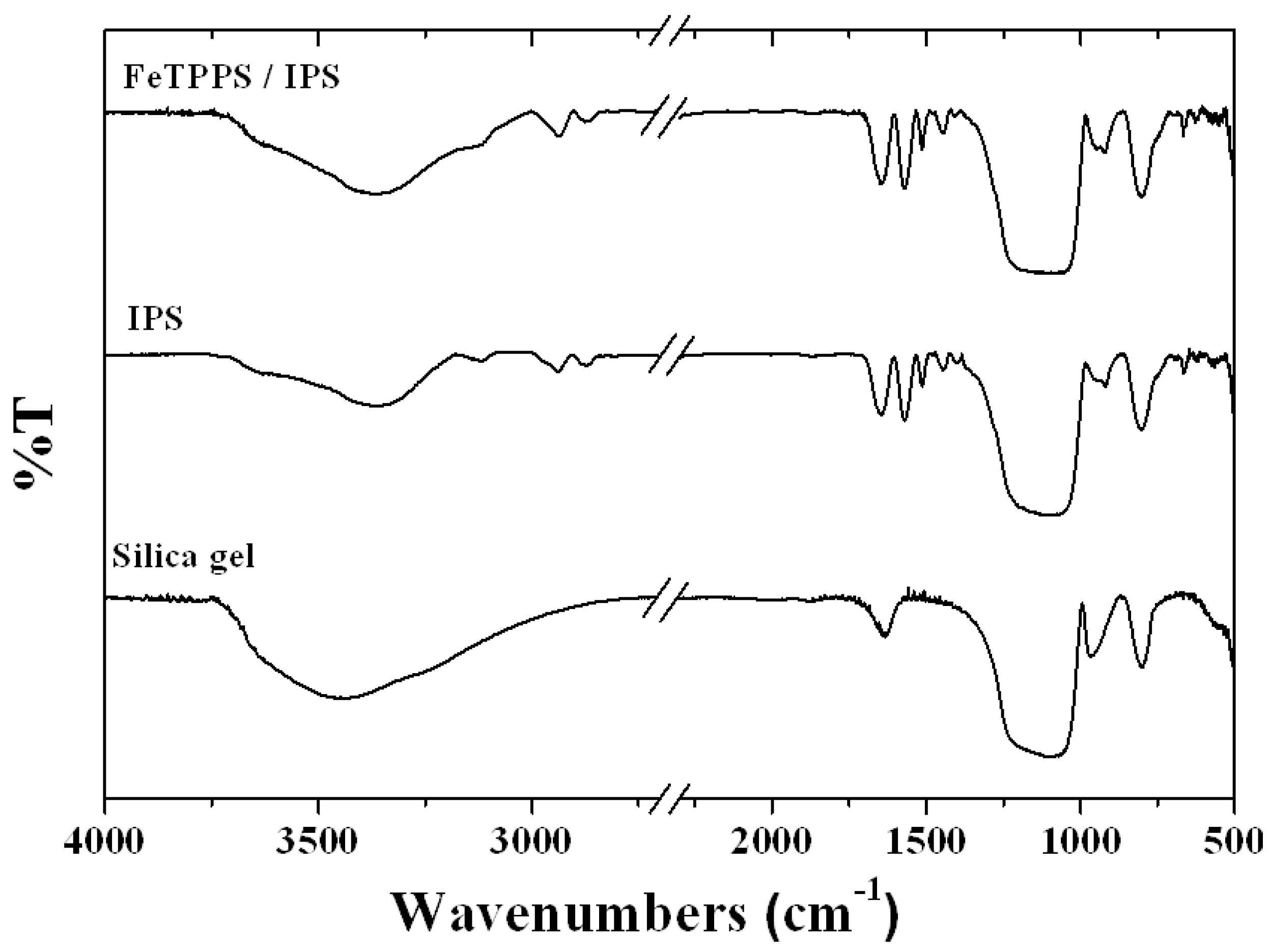
2.1. Characterization of FeTPPS / IPS

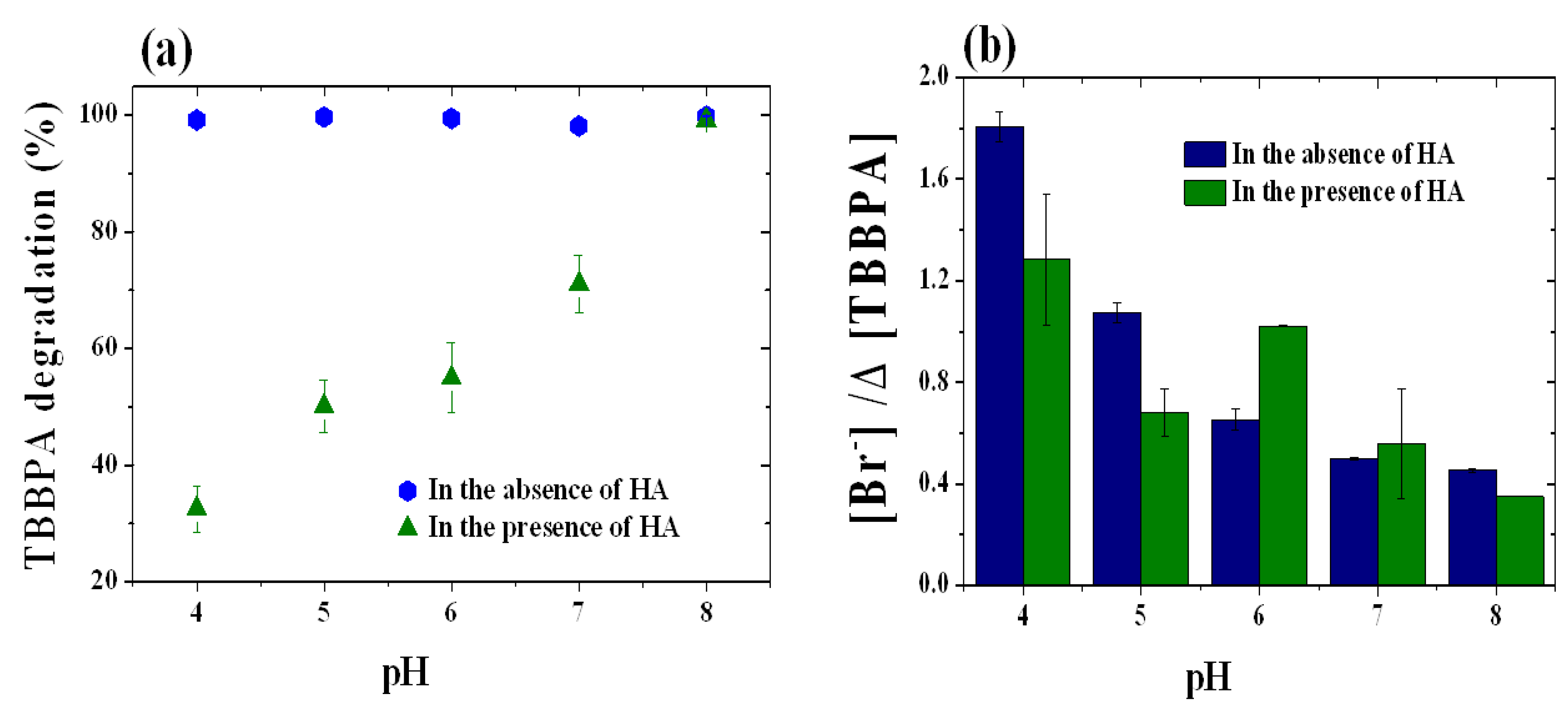
2.2. Influence of pH on the Degradation of TBBPA

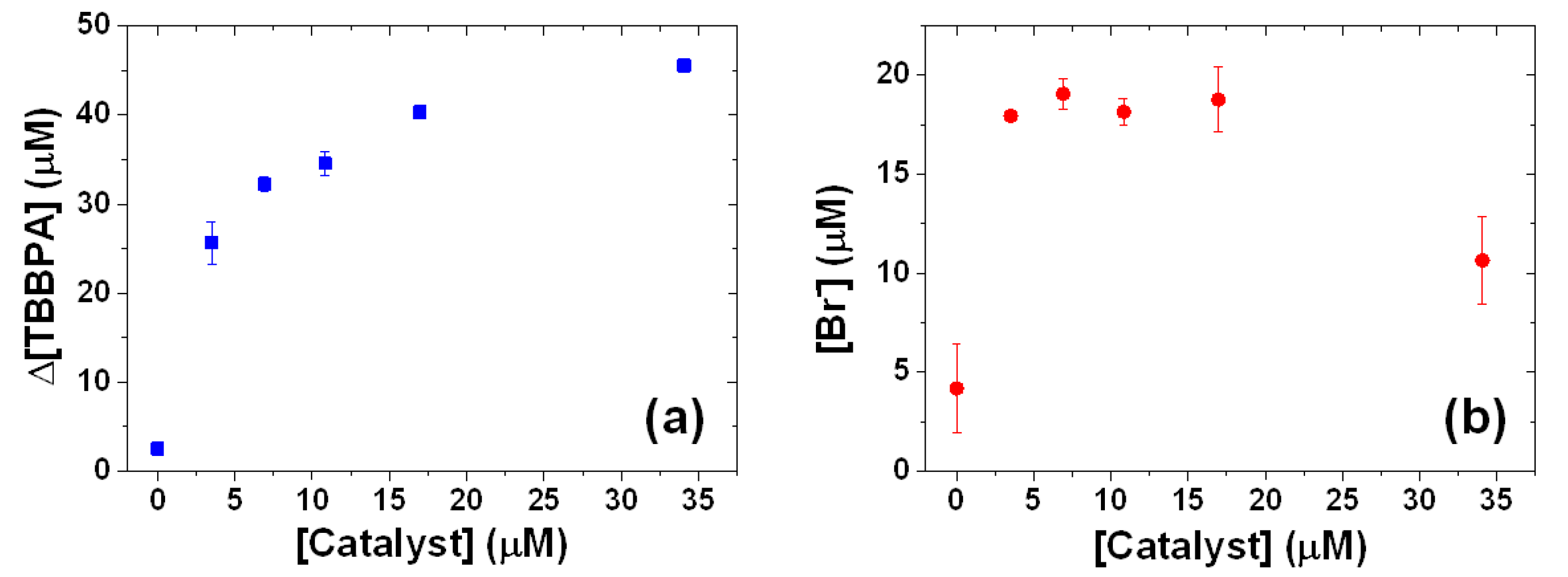
2.3. Influence of Catalyst Concentration on the TBBPA Degradation and Debromination
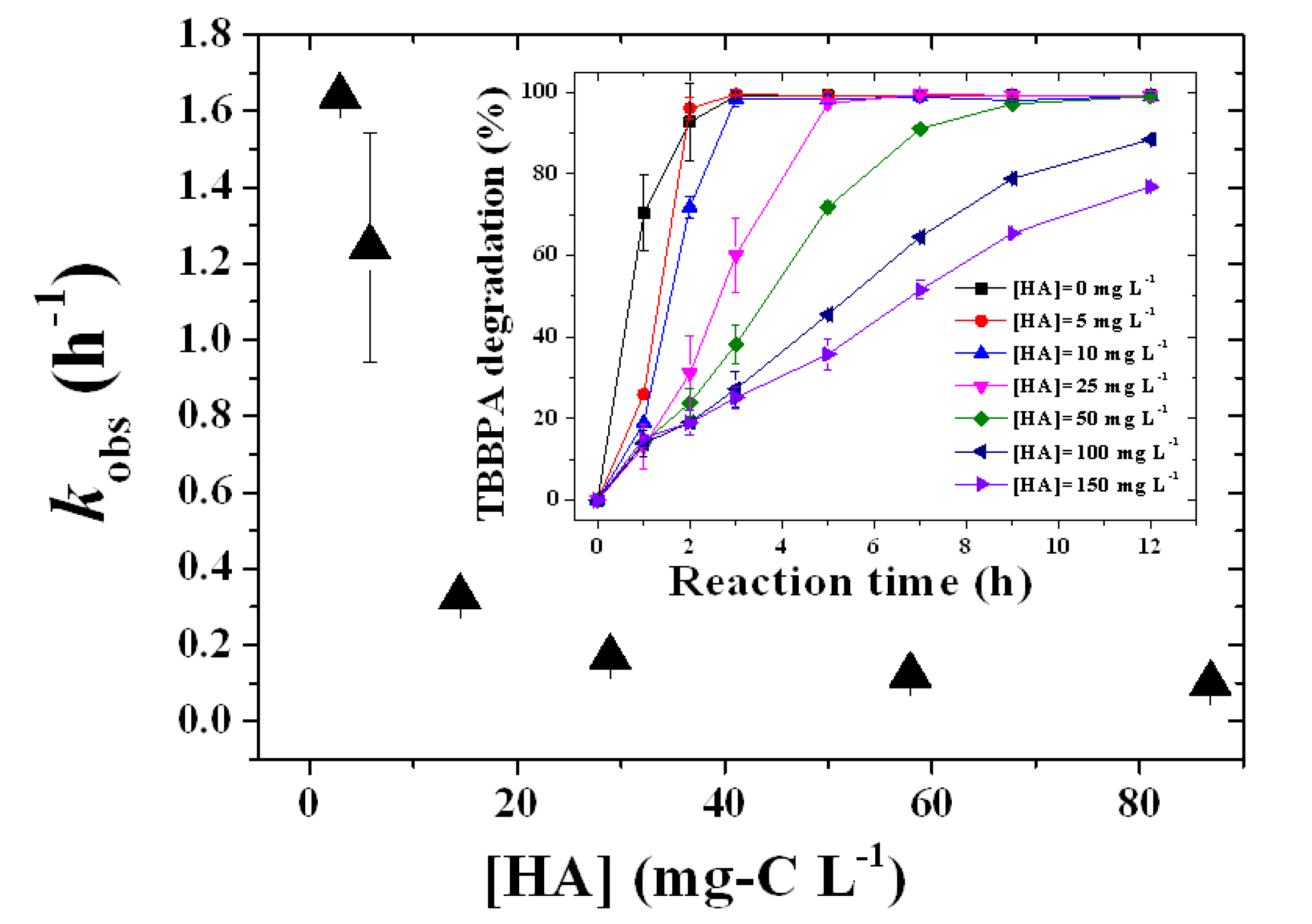
2.4. Influence of HA Concentration
2.5. Reusability of FeTPPS/IPS


3. Experimental
3.1. Materials
3.2. Synthesis of Silica Supported FeTPPS Catalyst

3.3. Characterization of the Synthesized Catalyst
3.4. Assay for TBBPA Degradation


4. Conclusions
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Tetrabromobisphenol A and Derivatives: A General Introduction, Environmental Health Criteria 172; World Health Organization: Geneva, Switzerland, 1997. [Google Scholar]
- Choi, K.-I.; Lee, S.-H.; Osako, M. Leaching of brominated flame retardants from TV housing plastics in the presence of dissolved humic matter. Chemosphere 2009, 74, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Osako, M.; Kim, Y.-J.; Sakai, S. Leaching of brominated flame retardants in leachate from landfills in Japan. Chemosphere 2004, 57, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Strack, S.; Detzel, T.; Wahl, M.; Kuch, B.; Krug, H.F. Cytotoxicity of TBBPA and effects on proliferation, cell cycle and MAPK pathways in mammalian cells. Chemosphere 2007, 67, S405–S411. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Liu, W.; Gan, J. Reaction of tetrabromobisphenol A (TBBPA) with manganese dioxide: Kinetics, products, and pathways. Environ. Sci. Technol. 2009, 43, 4480–4486. [Google Scholar] [CrossRef] [PubMed]
- Han, S.-K.; Bilski, P.; Karriker, B.; Sik, R.H.; Chignell, C.F. Oxidation of flame retardant tetrabromobisphenol A by singlet oxygen. Environ. Sci. Technol. 2008, 42, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Bastos, P.M.; Eriksson, J.; Green, N.; Bergman, Å. A standardized method for assessment of oxidative transformations of brominated phenols in water. Chemosphere 2008, 70, 1196–1202. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Ji, H.; Huang, X. Photocatalytic degradation of methyl orange over metalloporphyrins supported on TiO2 degussa P25. Molecules 2012, 17, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.-C.; She, Y.-B.; Zhou, Y.; Song, X.-F.; Li, K. Synthesis characterization and spectral properties of substituted tetraphenyl-porphyrin iron chloride complexes. Molecules 2011, 16, 2960–2970. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, M.; Ishida, Y.; Shigematsu, S.; Kuramitz, H.; Nagao, S. Pattern of oxidation products derived from tetrabromobisphenol A in a catalytic system comprised of iron(III)-tetrakis(p-sulfophenyl)porphyrin, KHSO5 and humic acids. Chemosphere 2010, 80, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, M.; Mizutani, Y.; Maeno, S.; Zhu, Q.; Kuramitz, H.; Nagao, S. Influence of halogen substituents on the catalytic oxidation of 2,4,6-halogenated phenols by Fe(III)-tetrakis(p-hydroxyphenyl)porphyrins and potassium monopersulfate. Molecules 2012, 17, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Nappa, M.J.; Tolman, C.A. Steric and electronic control of iron porphyrin catalyzed hydrocarbon oxidations. Inorg. Chem. 1985, 24, 4711–4719. [Google Scholar] [CrossRef]
- Fukushima, M.; Tatsumi, K. Effect of hydroxypropyl-β-cyclodextrin on the degradation of pentachlorophenol by potassium monopersulfate catalyzed with iron(III)−porphyrin complex. Environ. Sci. Technol. 2005, 39, 9337–9342. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, M.; Tatsumi, K. Complex formation of water-soluble iron(III)-porphyrin with humic acids and their effects on the catalytic oxidation of pentachlorophenol. J. Mol. Catal. A-Chem. 2006, 245, 178–184. [Google Scholar] [CrossRef]
- Machado, G.S.; Castro, K.A.D.F.; Wypych, F.; Nakagaki, S. Immobilization of metalloporphyrins into nanotubes of natural halloysite toward selective catalysts for oxidation reactions. J. Mol. Catal. A-Chem. 2008, 283, 99–107. [Google Scholar] [CrossRef]
- Liu, J.; Guan, J.; Lu, M.; Kan, Q.; Li, Z. Hemoglobin immobilized with modified “fish-in-net” approach for the catalytic removal of aniline. J. Hazard. Mater. 2012, 217–218, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Nazari, K.; Shokrollahzadeh, S.; Mahmoudi, A.; Mesbahi, F.; Matin, N. S.; Moosavi-Movahedi, A.A. Iron(III)-protoporphyrin/MCM41 catalyst as a peroxidase enzyme model: Preparation and typical test reactions. J. Mol. Catal. A-Chem. 2005, 239, 1–9. [Google Scholar] [CrossRef]
- Nur, H.; Hamid, H.; Endud, S.; Hamdan, H.; Ramli, Z. Iron-porphyrin encapsulated in poly(methacrylic acid) and mesoporpous Al-MCM-41 as catalysts in the oxidation of benzene to phenol. Mater. Chem. Phys. 2006, 96, 337–342. [Google Scholar] [CrossRef]
- Zucca, P.; Mocci, G.; Rescigno, A.; Sanjust, E. 5,10,15,20-Tetrakis(4-sulfonato-phenyl)porphine-Mn(III) immobilized on imidazole-activated silica as a novel lignin-peroxidase-like biomimetic catalyst. J. Mol. Cata. A-Chem. 2007, 278, 220–227. [Google Scholar] [CrossRef]
- Zucca, P.; Sollai, F.; Garau, A.; Rescigno, A.; Sanjust, E. Fe(III)-5,10,15,20-tetrakis-(pentafluorophenyl)porphine supported on pyridyl-functionalized, crosslinked poly(vinyl alcohol) as a biomimetic versatile-peroxidase-like catalyst. J. Mol. Catal. A-Chem. 2009, 306, 89–96. [Google Scholar] [CrossRef]
- Christoforidis, K.C.; Serestatidou, E.; Louloudi, M.; Konstantinou, I.K.; Milaeva, E.R.; Deligiannakis, Y. Mechanism of catalytic degradation of 2,4,6-trichlorophenol by a Fe-porphyrin catalyst. Appl. Catal. B-Environ. 2011, 101, 417–424. [Google Scholar] [CrossRef]
- Christoforidis, K.C.; Louloudi, M.; Deligiannakis, Y. Complete dechlorination of pentachlorophenol by a heterogeneous SiO2-Fe-porphyrin catalyst. Appl. Catal. B-Environ. 2010, 95, 297–302. [Google Scholar] [CrossRef]
- Shigematsu, S.; Fukushima, M.; Nagao, S. Oxidative degradation of 2,6-dibromophenol using an anion-exchange resin supported supramolecular catalysts of iron(III)-5,10,15,20-tetrakis (p-hydroxyphenyl)porphyrin bound to humic acid prepared via formaldehyde and urea-formaldehyde polycondensation. J. Environ. Sci. Heal. A 2010, 45, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, M.; Tanaka, S.; Hasebe, K.; Taga, M.; Nakamura, H. Interpretation of acid-base equilibrium of humic acid by continuous pK distribution model and electrostatic model. Anal. Chim. Acta 1995, 302, 365–373. [Google Scholar] [CrossRef]
- Fukushima, M.; Tanabe, Y.; Morimoto, K.; Tatsumi, K. Role of humic acid fraction with higher aromaticity in enhancing the activity of a biomimetic catalyst, tetra(p-sulfonatophenyl)-porphineiron(III). Biomacromolecules 2007, 8, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, M.; Kuriss, A.; Fukushima, M.; Sawada, A.; Tatsumi, K. Effects of pH and organic co-solvents on the oxidation of naphthalene with peroxosulfate catalyzed by iron(III) tetrakis(p-sulfonatophenyl)porphyrin, J. Porphyr. Phthalocya. 2003, 7, 645–650. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhu, Q.; Mizutani, Y.; Maeno, S.; Fukushima, M. Oxidative Debromination and Degradation of Tetrabromo-bisphenol A by a Functionalized Silica-Supported Iron(III)-tetrakis(p-sulfonatophenyl)porphyrin Catalyst. Molecules 2013, 18, 5360-5372. https://doi.org/10.3390/molecules18055360
Zhu Q, Mizutani Y, Maeno S, Fukushima M. Oxidative Debromination and Degradation of Tetrabromo-bisphenol A by a Functionalized Silica-Supported Iron(III)-tetrakis(p-sulfonatophenyl)porphyrin Catalyst. Molecules. 2013; 18(5):5360-5372. https://doi.org/10.3390/molecules18055360
Chicago/Turabian StyleZhu, Qianqian, Yusuke Mizutani, Shohei Maeno, and Masami Fukushima. 2013. "Oxidative Debromination and Degradation of Tetrabromo-bisphenol A by a Functionalized Silica-Supported Iron(III)-tetrakis(p-sulfonatophenyl)porphyrin Catalyst" Molecules 18, no. 5: 5360-5372. https://doi.org/10.3390/molecules18055360