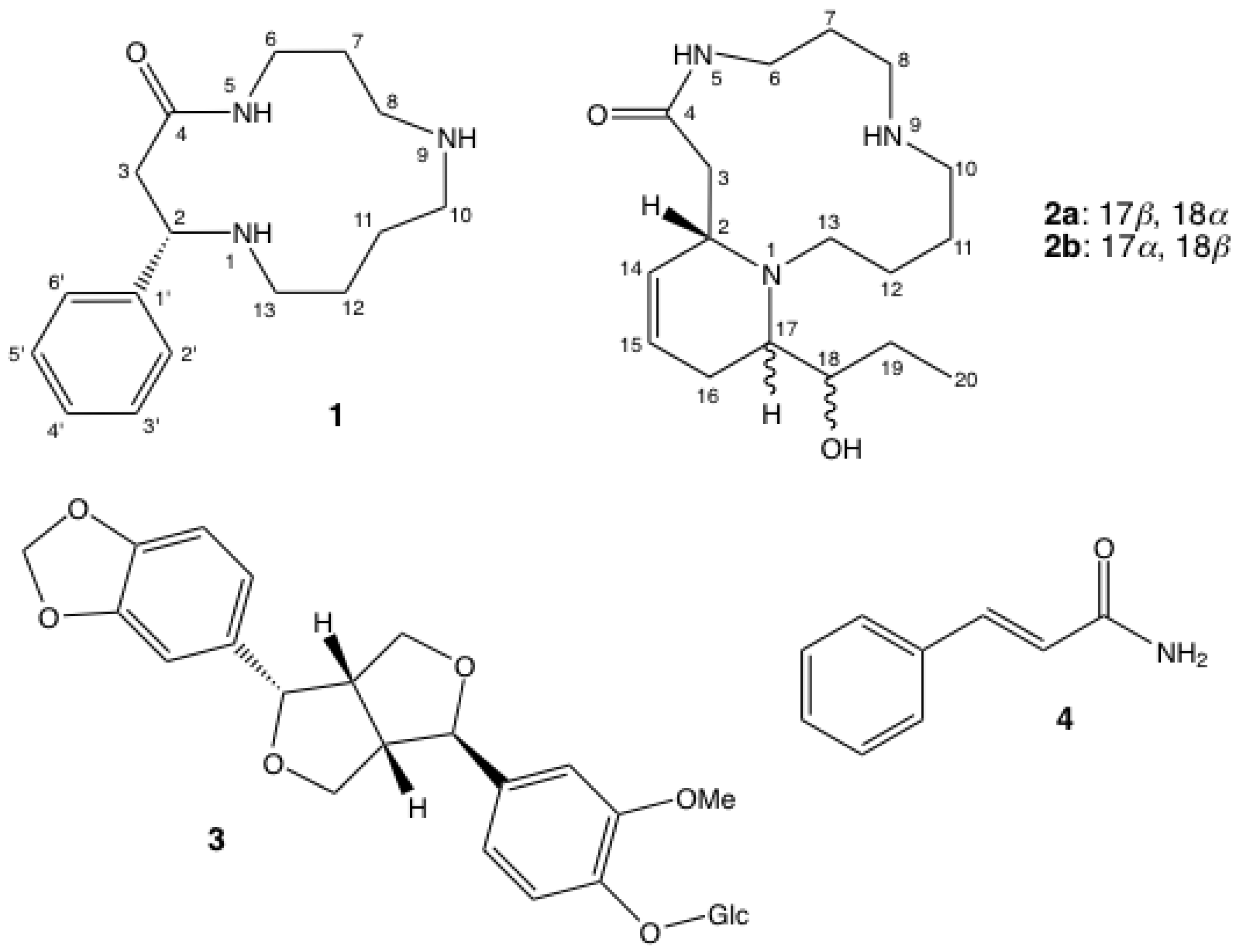
Macrocyclic Spermidine Alkaloids from Androya decaryi L. Perrier
Abstract
:1. Introduction
2. Results and Discussion
| δH (J in Hz) a | δC | |
|---|---|---|
| 2 | 4.01 dd (11.5, 2.9) | 60.0 |
| 3 | 2.48 dd (15.0, 2.9); 2.56 dd (15.0, 11.5) | 45.0 |
| 4 | - | 171.7 |
| 5 | 8.57 bs | - |
| 6 | 3.19 dt (13.7, 5.5); 3.70 dt (13.7, 5.2) | 39.6 |
| 7 | 1.80–1.85 m | 27.8 |
| 8 | 2.86 dt (12.1, 5.0) 2.96 dt (12.1, 5.6) | 49.7 |
| 10 | 2.74–2.75 m | 49.1 |
| 11 | 1.55–1.61 m; 1.74–1.80 m | 27.9 |
| 12 | 1.40–1.49 m; 1.63–1.69 m | 27.6 |
| 13 | 2.33 ddd (12.5, 8.8, 1.9); 2.52–2.56 m | 45.8 |
| 1’ | - | 142.7 |
| 2'/6' | 7.23–7.27 m | 126.4 |
| 3'/5' | 7.30–7.36 m | 128.7 |
| 4' | 7.23–7.27 m | 127.3 |
| 2a | 2b | |||
|---|---|---|---|---|
| δH (J in Hz) a | δC | δH | δCa | |
| 2 | 3.56–3.62 m | 56.5 | 3.71–3.73, m | 57.05 |
| 3 | 2.22 dd (13.7, 2.3); 2.52–2.62 m | 41.9 | 2.40 dd (14.4, 3.8); 2.56–2.61 m | 40.4 |
| 4 | - | 173.2 | - | 173.9 |
| 5 | 7.45–7.53 m | - | 8.10 br s | - |
| 6 | 2.97–3.03 m; 4.12–4.21 m | 37.4 | 3.05–3.11 m; 3.76–3.81 m | 38.8 |
| 7 | 2.02–2.06 m; 2.42–2.52 m | 26.9 | 1.98–2.03 m; 2.20–2.26 m | 25.8 |
| 8 | 2.87–2.95 m; 3.27–3.36 m | 46.3 | 3.05–3.11 m; 3.16–3.22 m | 49.5 |
| 10 | 2.87–2.95 m; 3.21–3.27 m | 45.8 | 2.88 ddd (13.2, 11.5, 2.4); 2.97–3.01 m | 50.1 |
| 11 | 1.70–1.77 m; 1.94–2.02 m | 22.5 | 1.67–1.77 m; 1.98–2.09 m | 26.4 |
| 12 | 1.73–1.82 m | 22.1 | 1.57–1.78 m | 24.7 |
| 13 | 2.28–2.37 m; 2.87–2.93 m | 54.4 | 2.46–2.53 m; 2.56–2.61 m | 46.8 |
| 14 | 5.59 d (10.4) | 126.6 | 5.55–5.58 m | 126.95 |
| 15 | 5.79–5.84 m | 123.7 | 5.83 ddd (10.0, 4.3, 3.7) | 127.0 |
| 16 | 1.85–1.92 m; 2.28–2.37 m | 20.5 | 1.83–1.85 m | 22.2 |
| 17 | 2.73–2.79 m | 63.5 | 3.16–3.22 m | 57.0 |
| 18 | 3.53 dt (11.0, 1.8) | 73.0 | 3.54, ddd (9.5, 6.4, 5.2) | 72.2 |
| 19 | 1.61–1.77 m | 28.9 | 1.50–1.56 m | 26.7 |
| 20 | 1.13 t (7.4) | 10.7 | 1.06 t (7.3) | 10.4 |

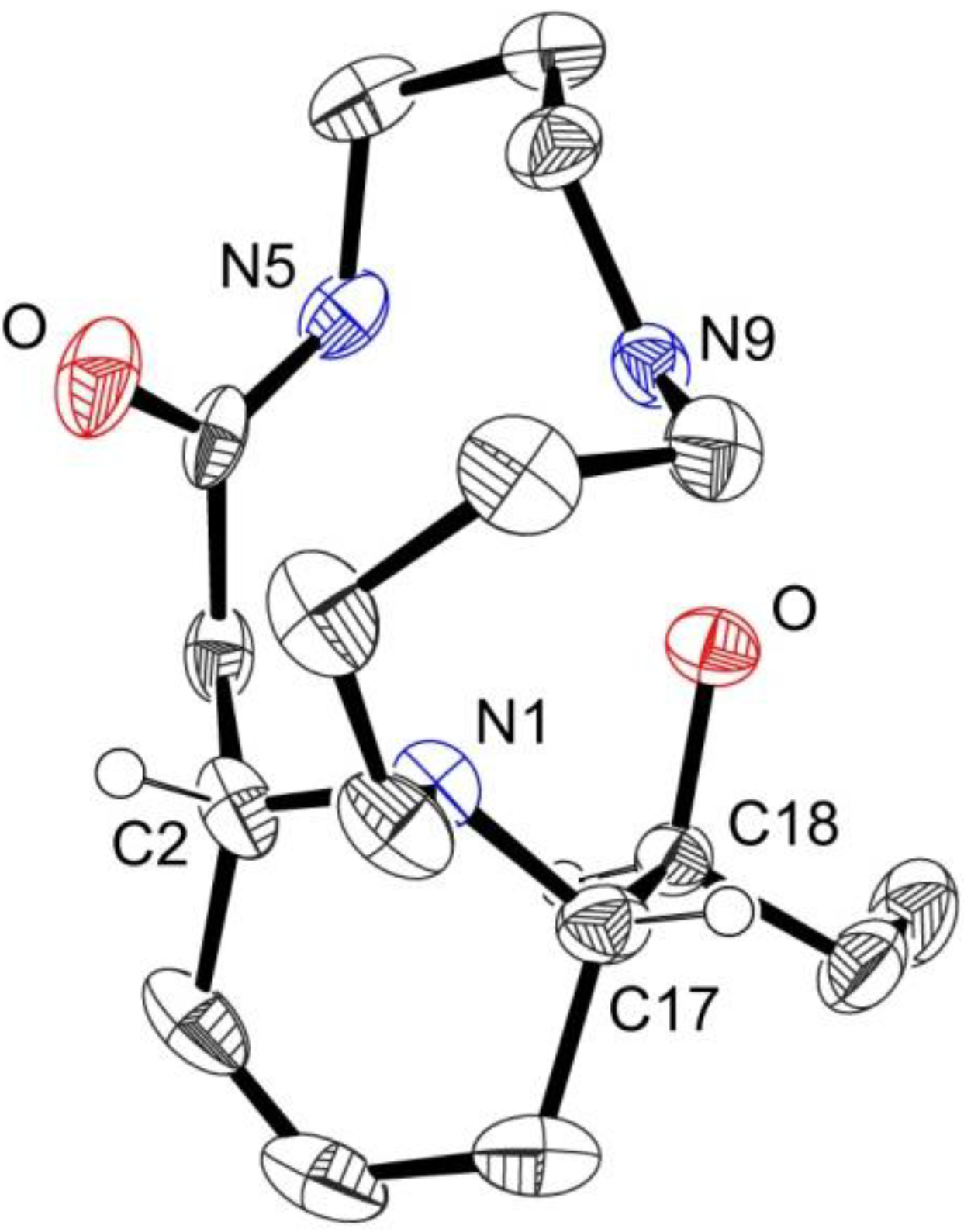

3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Antiplasmodial Bioassay
4. Conclusions
Supplementary Materials
Acknowledgments
References
- Oxelman, B.; Kornhall, P.; Olmstead, R.G.; Bremer, B. Further disintregration of Scrophulariaceae. Taxon 2005, 54, 411–425. [Google Scholar] [CrossRef]
- Petersen, I.; Eastman, R.; Lanzer, M. Drug-resistant malaria: Molecular mechanisms and implications for public health. FEBS Lett. 2011, 585, 1551–1562. [Google Scholar] [CrossRef]
- Bero, J.; Frédérich, M.; Quetin-Leclercq, J. Antimalarial compounds isolated from plants used in traditional medicine. J. Pharm. Pharmacol. 2009, 61, 1401–1433. [Google Scholar] [CrossRef]
- Bero, J.; Quetin-Leclercq, J. Natural products published in 2009 from plants traditionally used to treat malaria. Planta Med. 2011, 77, 631–640. [Google Scholar] [CrossRef] [Green Version]
- Nogueira, C.R.; Lopes, L.M.X. Antiplasmodial natural products. Molecules 2011, 16, 2146–2190. [Google Scholar] [CrossRef]
- Kuehne, P.; Linden, A.; Hesse, M. Asymmetric synthesis of the alkaloids mayfoline and N(1)-acetyl-N(1)-deoxymayfoline. Helv. Chim. Acta 1996, 79, 1085–1094. [Google Scholar] [CrossRef]
- Kuehne, P.; Guggisberg, A.; Hesse, M. Synthesis and chiroptical properties of 13-membered spermidine alkaloids (–)-(S)-Celacininne. (0)-(S)-celabenzine, (–)-(S)-celafurine, and (+)-(S)-viburnine. Helv. Chim. Acta 1997, 80, 1802–1808. [Google Scholar] [CrossRef]
- Wassermann, H.H.; Matsuyama, H.; Robinson, R. β-lactams as building blocks in the synthesis of macrocyclic spermine and spermidine alkaloids. Tetrahedron 2002, 58, 7177–7190. [Google Scholar] [CrossRef]
- Flack, H.D. On enantiomorph-polarity estimation. Acta Cryst. 1983, A39, 876–881. [Google Scholar]
- Kerrer, P.; Eugster, C.H. An alkaloid from Equisetum palustre. Helv. Chim. Acta 1948, 31, 1062–1066. [Google Scholar] [CrossRef]
- Natsume, M.; Ogawa, M. Total synthesis of (±)-palustrine and structure revision. Chem. Pharm. Bull. 1984, 32, 3789–3791. [Google Scholar] [CrossRef]
- Schultz, K.; Kuehne, P.; Häusermann, U.A.; Hesse, M. Absolute configuration of macrocyclic spermidine alkaloids. Chirality 1997, 9, 3789–3791. [Google Scholar]
- Guggisberg, A.; Hesse, M. Natural polyamine derivatives—New aspects of their isolation, structure elucidation and synthesis. In The Alkaloids, Chemistry and Biology; Academic Press: San Diego, CA, USA, 1998; Volume 50, pp. 219–256. [Google Scholar]
- Murata, T.; Miyase, T.; Yoshizaki, F. Meehanines A–K, Spermidine alkaloidal glycosides from Meehania urticifolia. J. Nat. Prod. 2009, 72, 1049–1056. [Google Scholar] [CrossRef]
- Murata, T.; Miyase, T.; Yoshizaki, F. Meehanines L–W, Spermidine alkaloidal glycosides from Meehania urticifolia. J. Nat. Prod. 2009, 72, 1937–1943. [Google Scholar] [CrossRef]
- Murata, T.; Miyase, T.; Yoshizaki, F. Cyclic spermidine alkaloids and flavone glycosides from Meehania fargesii. Chem. Pharm. Bull. 2010, 58, 696–702. [Google Scholar] [CrossRef]
- Wang, S.Q.; Ren, D.M.; Xiang, F.; Wang, X.N.; Zhu, C.J.; Yuan, H.Q.; Sun, L.M.; Ly, B.B.; Sun, X.J.; Lou, H.X. Dracotanosides A–D, Spermidine glycosides from Dracocephalum tanguticum: Structure and amide rotational barrier. J. Nat. Prod. 2009, 72, 1006–1010. [Google Scholar] [CrossRef]
- Khanjin, N.A.; Hesse, M. Synthesis of the spermidine alkaloids (–)-(2R,3R)- and (–)-(2R,3S)-3-hydrocelacinnine: Macrocyclisation with oxirane-ring opening and inversion via cyclic sulfamidates. Helv. Chim. Acta 2003, 86, 2028–2057. [Google Scholar] [CrossRef]
- Häusermann, U.A.; Linden, A.; Song, J.; Hesse, M. Total synthesis of (–)-(2R)-dihydromyricoidine. Helv. Chim. Acta 1996, 79, 1995–2003. [Google Scholar] [CrossRef]
- Li, T.; Hao, X.J.; Gu, Q.Q.; Zhu, W.M. Minor furofurano lignans from the Tibetan herb. Lancea tibetica. Planta Med. 2008, 74, 1391–1396. [Google Scholar] [CrossRef]
- Cai, M.Z.; Zhao, H.; Hu, W.Y.; Hale, P.D. Silica-supported Arsine Palladium(0) complex: A highly active and stereoselective catalyst for arylation of butyl acrylate and acrylamide. Chin. J. Chem. 2005, 23, 443–447. [Google Scholar] [CrossRef]
- Trager, W.; Jensen, J.B. Human malaria parasites in continuous culture. Science 1976, 193, 673–675. [Google Scholar]
- Benoit, F.; Valentin, A.; Pelissier, Y.; Marion, C.; Dakuyo, Z.; Mallie, M.; Bastide, J.M. Antimalarial activity in vitro of Cochlospermum tinctorium tubercle extracts. Trans. Roy. Soc. Trop. Med. Hyg. 1995, 89, 217–218. [Google Scholar] [CrossRef]
- Lambros, C.; Vanderberg, J.P. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J. Parasitol. 1979, 65, 418–420. [Google Scholar] [CrossRef]
- Desjardins, R.E.; Canfield, C.J.; Haynes, J.D.; Chulay, J.D. Quantitative assessment of antimalarial activity in vitro by a semiautomated microdilution technique. Antimicrob. Agent Chemother. 1979, 16, 710–718. [Google Scholar] [CrossRef]
- Valentin, A.; Benoit-Vical, F.; Moulis, C.; Stanislas, E.; Mallie, M.; Fouraste, I.; Bastide, J.-M. In vitro antimalarial activity of penduline, A bisbenzylisoquinoline from Isopyrum thalictroides. Antimicrob. Agent Chemother. 1997, 41, 2305–2307. [Google Scholar]
- Häusermann, U.A.; Hesse, M. Total syntheses of (+)-(2S)-dihydromyricoidine and (+)-(2S)-myricoidine. Tetrahedron Lett. 1998, 39, 257–260. [Google Scholar] [CrossRef]
- Bienz, S.; Bisegger, P.; Guggisberg, A.; Hesse, M. Polyamine alkaloids. Nat. Prod. Rep. 2005, 22, 647–658. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1 and 2a are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Le Lamer, A.-C.; Ibrahim, N.; Manjary, F.; Mallet-Ladeira, S.; Girardi, C.; Valentin, A.; Fabre, N.; Moulis, C. Macrocyclic Spermidine Alkaloids from Androya decaryi L. Perrier. Molecules 2013, 18, 3962-3971. https://doi.org/10.3390/molecules18043962
Le Lamer A-C, Ibrahim N, Manjary F, Mallet-Ladeira S, Girardi C, Valentin A, Fabre N, Moulis C. Macrocyclic Spermidine Alkaloids from Androya decaryi L. Perrier. Molecules. 2013; 18(4):3962-3971. https://doi.org/10.3390/molecules18043962
Chicago/Turabian StyleLe Lamer, Anne-Cécile, Nehal Ibrahim, Frédéric Manjary, Sonia Mallet-Ladeira, Cynthia Girardi, Alexis Valentin, Nicolas Fabre, and Claude Moulis. 2013. "Macrocyclic Spermidine Alkaloids from Androya decaryi L. Perrier" Molecules 18, no. 4: 3962-3971. https://doi.org/10.3390/molecules18043962




