In Situ Formation of Steroidal Supramolecular Gels Designed for Drug Release
Abstract
:1. Introduction
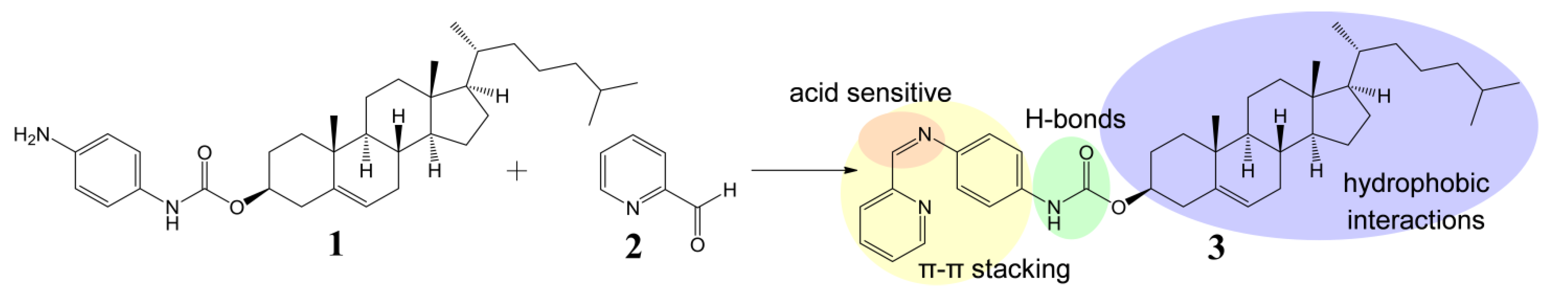
2. Results and Discussion
2.1. Preparation and Gelation Behaviour of Imine 3
| Solvents | 1 | 2 | 3 (“ in situ”) | 3 | Solvents | 1 | 2 | 3 (“ in situ”) | 3 |
|---|---|---|---|---|---|---|---|---|---|
| toluene | S | S | P | P | hexan-1-ol | S | S | pG | pG |
| CH2Cl2 | S | S | P | P | heptan-1-ol | S | S | pG | pG |
| CHCl3 | S | S | S | S | octan-1-ol | S | S | pG | pG |
| CCl4 | S | S | P | P | water | I | S | - | I |
| methanol | P | S | I | I | DMF | S | S | pG | pG |
| ethanol | P | S | I | I | DMSO | G | S | G | G |
| propan-1-ol | S | S | G | G | cyclohexane | G | I | - | I |
| butan-1-ol | S | S | G | G | hexane | I | I | - | I |
| pentan-1-ol | S | S | G | G |

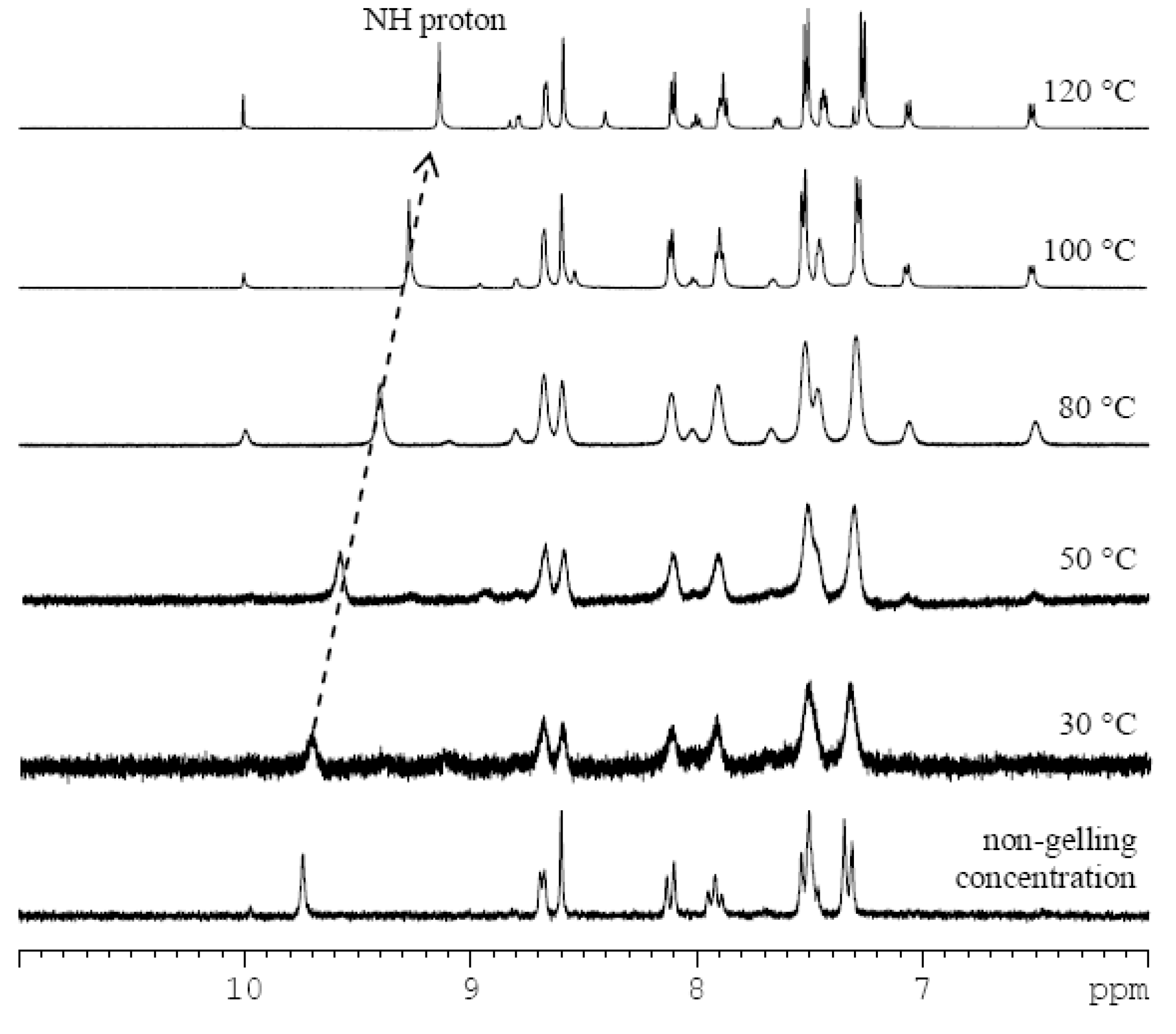
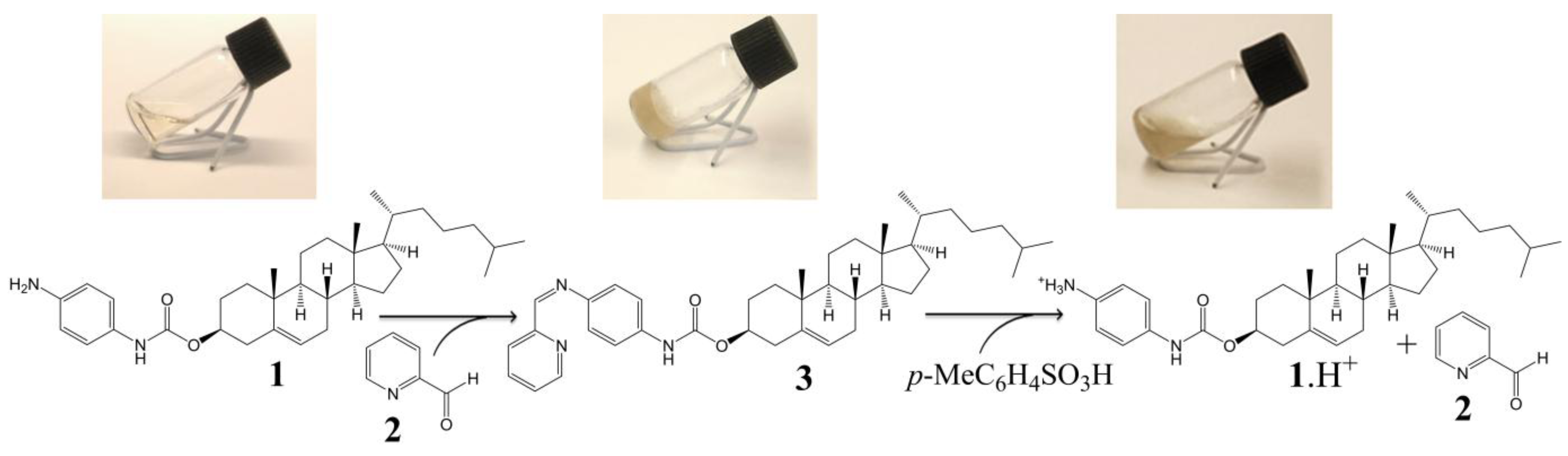
2.2. Acid-Responsive Gels
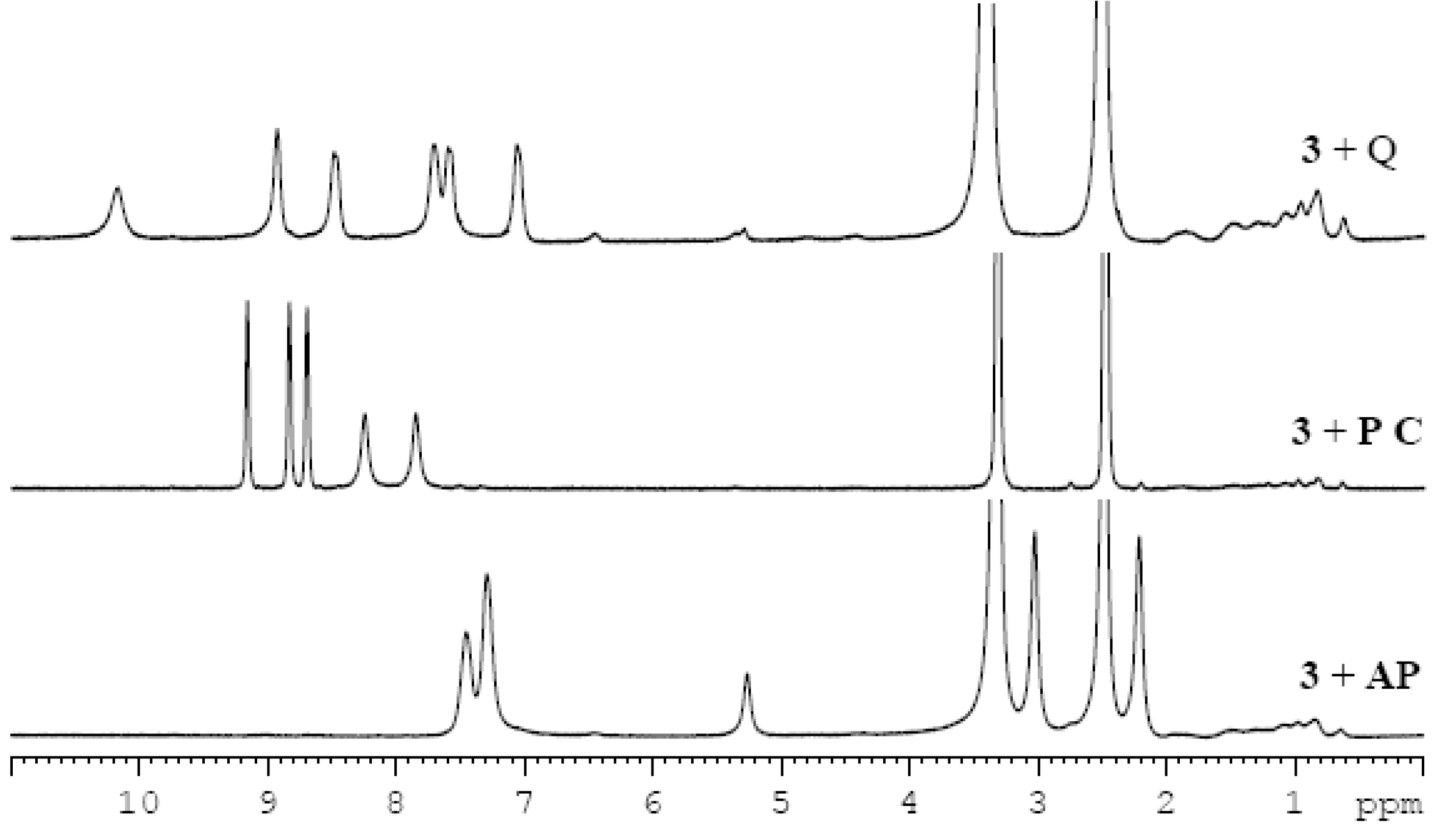
2.3. Two Component Gel System

| Solvents | 3 | Q | 3 + Q | PC | 3 + PC | AP | 3 + AP |
|---|---|---|---|---|---|---|---|
| ethanol | I | R | P | I | S | I | |
| propan-1-ol | G | G | G | P | G | S | G |
| butan-1-ol | G | R | G | P | G | S | G |
| pentan-1-ol | G | R | pG | P | G | S | G |
| hexan-1-ol | pG | R | pG | P | G | S | G |
| octan-1-ol | pG | R | pG | P | pG | S | pG |
| DMF | pG | S | pG | S | pG | S | pG |
| DMSO | G | S | G | S | G | S | G |

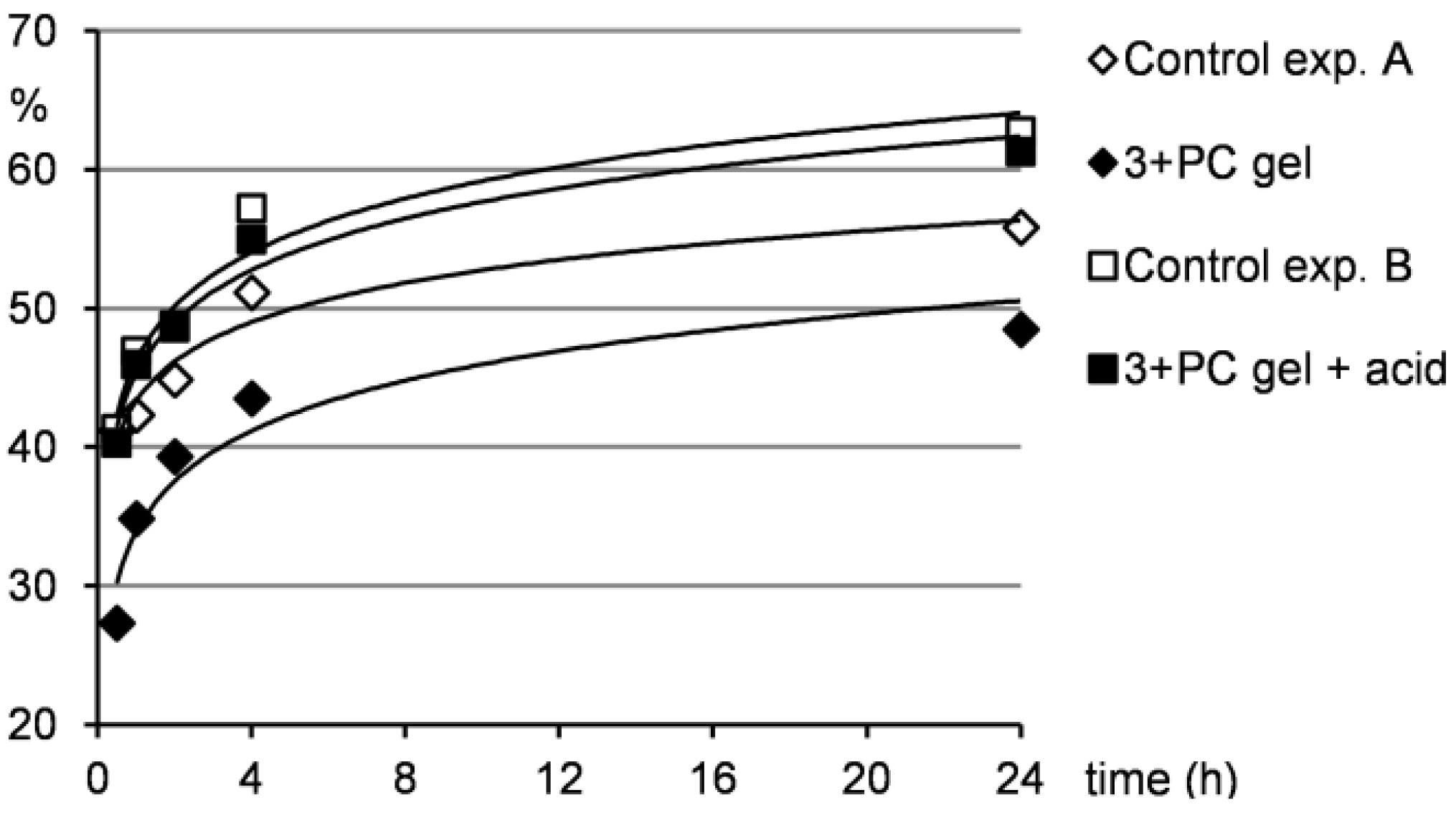
2.4. Controlled Drug Release

3. Experimental
3.1. General
3.2. Preparation of compound 1
3.3. Preparation of Compound 3
3.4. Gelation Tests
3.5. NMR Studies
3.6. SEM Measurments
3.7. Drug Release Experiments
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Smith, D.K. Self-Assembling Fibrillar Networks—Supramolecular Gels. In Supramolecular Chemistry: From Molecules to Nanomaterials, 1st ed.; Steed, J.W., Gale, A.P., Eds.; Wiley & Sons Ltd.: Chichester, UK, 2012; Volume 7, pp. 3355–3376. [Google Scholar]
- Steed, J.W. Supramolecular Gel Chemistry: Developments Over the Last Decade. Chem. Commun. 2011, 47, 1379–1383. [Google Scholar] [CrossRef]
- Johnson, E.K.; Adams, D.J.; Cameron, P.J. Peptide Based Low Molecular Weight Gelators. J. Mater. Chem. 2011, 21, 2024–2027. [Google Scholar] [CrossRef]
- Dastidar, P. Supramolecular Gelling Agents: Can they be Designed? Chem. Soc. Rev. 2008, 37, 2699–2715. [Google Scholar] [CrossRef]
- Weiss, R.G.; Terech, P. Molecular Gels: Materials with Self-Assembled Fibrillar Networks; Springer: Dordrecht, The Netherlands, 2006. [Google Scholar]
- Estroff, L.A.; Hamilton, A.D. Water Gelation by Small Organic Molecules. Chem. Rev. 2004, 104, 1201–1218. [Google Scholar] [CrossRef]
- Dawn, A.; Shiraki, T.; Haraguchi, S.; Tamaru, S.; Shinkai, S. What Kind of “Soft Materials” can we Design from Molecular Gels? Chem. Asian J. 2011, 6, 266–282. [Google Scholar] [CrossRef]
- Escuder, B.; Rodriguez-Llansola, F.; Miravet, J.F. Supramolecular Gels as Active Media for Organic Reactions and Catalysis. New J. Chem. 2010, 34, 1044–1054. [Google Scholar] [CrossRef]
- Banerjee, S.; Das, R.K.; Maitra, U. Supramolecular Gels “in Action”. J. Mater. Chem. 2009, 19, 6649–6687. [Google Scholar] [CrossRef]
- Hirst, A.; Escuder, B.; Miravet, J.; Smith, D. High-Tech Applications of Self-Assembling Supramolecular Nanostructured Gel-Phase Materials: From Regenerative Medicine to Electronic Devices. Angew. Chem. Int. Ed. Engl. 2008, 47, 8002–8018. [Google Scholar] [CrossRef]
- Sangeetha, N.M.; Maitra, U. Supramolecular Gels: Functions and Uses. Chem. Soc. Rev. 2005, 34, 821–836. [Google Scholar]
- Truong, W.T.; Su, Y.; Meijer, J.T.; Thordarson, P.; Braet, F. Self-Assembled Gels for Biomedical Applications. Chem. Asian J. 2011, 6, 30–42. [Google Scholar] [CrossRef]
- Vintiloiu, A.; Leroux, J. Organogels and their use in Drug Delivery—A Review. J. Controll. Release 2008, 125, 179–192. [Google Scholar] [CrossRef]
- Murdan, S. Organogels in Drug Delivery. Expert Opin. Drug Deliv. 2005, 2, 489–505. [Google Scholar] [CrossRef]
- Ajayaghosh, A.; Praveen, V.K.; Vijayakumar, C. Organogels as Scaffolds for Excitation Energy Transfer and Light Harvesting. Chem. Soc. Rev. 2008, 37, 109–122. [Google Scholar]
- Liu, X. Gelation with Small Molecules: From Formation Mechanism to Nanostructure Architecture. Top. Curr. Chem. 2005, 256, 1–37. [Google Scholar] [CrossRef]
- Svobodova, H.; Noponen, V.; Kolehmainen, E.; Sievänen, E. Recent Advances in Steroidal Supramolecular Gels. RSC Adv. 2012, 2, 4985–5007. [Google Scholar]
- Žinić, M.; Vögtle, F.; Fages, F. Cholesterol-Based Gelators. Top. Curr. Chem. 2005, 256, 39–76. [Google Scholar] [CrossRef]
- Shinkai, S.; Murata, K. Cholesterol-Based Functional Tectons as Versatile Building-Blocks for Liquid Crystals, Organic Gels and Monolayers. J. Mater. Chem. 1998, 8, 485–495. [Google Scholar] [CrossRef]
- Babu, P.; Sangeetha, N.M.; Maitra, U. Supramolecular Chemistry of Bile Acid Derivatives: Formation of Gels. Macromol. Symp. 2006, 241, 60–67. [Google Scholar]
- Buerkle, L.E.; von Recum, H.A.; Rowan, S.J. Toward Potential Supramolecular Tissue Engineering Scaffolds Based on Guanosine Derivatives. Chem. Sci. 2012, 3, 564–572. [Google Scholar] [CrossRef]
- Sukul, P.K.; Malik, S. Supramolecular Hydrogels of Adenine: Morphological, Structural and Rheological Investigations. Soft Matter 2011, 7, 4234–4241. [Google Scholar] [CrossRef]
- Buerkle, L.E.; Li, Z.; Jamieson, A.M.; Rowan, S.J. Tailoring the Properties of Guanosine-Based Supramolecular Hydrogels. Langmuir 2009, 25, 8833–8840. [Google Scholar] [CrossRef]
- Tomasini, C.; Castellucci, N. Peptides and Peptidomimetics that Behave as Low Molecular Weight Gelators. Chem. Soc. Rev. 2013, 42, 156–172. [Google Scholar] [CrossRef]
- Ryan, D.M.; Nilsson, B.L. Self-Assembled Amino Acids and Dipeptides as Noncovalent Hydrogels for Tissue Engineering. Polym. Chem. 2012, 3, 18–33. [Google Scholar] [CrossRef]
- Adams, D.J. Dipeptide and Tripeptide Conjugates as Low-Molecular-Weight Hydrogelators. Macromol. Biosci. 2011, 11, 160–173. [Google Scholar] [CrossRef]
- Frkanec, L.; Žinić, M. Chiral Bis(Amino Acid)- and Bis(Amino Alcohol)-Oxalamide Gelators. Gelation Properties, Self-Assembly Motifs and Chirality Effects. Chem. Commun. 2010, 46, 522–537. [Google Scholar] [CrossRef]
- Suzuki, M.; Hanabusa, K. L-Lysine-Based Low-Molecular-Weight Gelators. Chem. Soc. Rev. 2009, 38, 967–975. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, G.; Zhang, D. Stimuli Responsive Gels Based on Low Molecular Weight Gelators. J. Mater. Chem. 2012, 22, 38–50. [Google Scholar] [CrossRef]
- De Jong, J.; Feringa, B.L.; van Esch, J. Responsive Molecular Gels. In Molecular Switches; Feringa, B.L., Browne, W.R., Eds.; Willey-VCH Vertag&Co.: Weinheim, Germany, 2011; pp. 517–562. [Google Scholar]
- Liu, K.; He, P.; Fang, Y. Progress in the Studies of Low-Molecular Mass Gelators with Unusual Properties. Sci. China Chem. 2011, 54, 575. [Google Scholar] [CrossRef]
- Piepenbrock, M.M.; Lloyd, G.O.; Clarke, N.; Steed, J.W. Metal- and Anion-Binding Supramolecular Gels. Chem. Rev. 2010, 110, 1960–2004. [Google Scholar] [CrossRef]
- Cravotto, G.; Cintas, P. Molecular Self-Assembly and Patterning Induced by Sound Waves. The Case of Gelation. Chem. Soc. Rev. 2009, 38, 2684–2697. [Google Scholar] [CrossRef]
- Maeda, H. Anion-Responsive Supramolecular Gels. Chem. Eur. J. 2008, 14, 11274–11282. [Google Scholar] [CrossRef]
- Svobodova, H.; Nonappa; Wimmer, Z.; Kolehmainen, E. Design, Synthesis and Stimuli Responsive Gelation of Novel stigmasterol—Amino Acid Conjugates. J. Colloid Interface Sci. 2011, 361, 587–593. [Google Scholar] [CrossRef]
- Li, Y.; Liu, K.; Liu, J.; Peng, J.; Feng, X.; Fang, Y. Amino Acid Derivatives of Cholesterol as “Latent” Organogelators with Hydrogen Chloride as a Protonation Reagent. Langmuir 2006, 22, 7016–7020. [Google Scholar] [CrossRef]
- Maitra, U.; Chakrabarty, A. Protonation and Deprotonation Induced organo/hydrogelation: Bile Acid Derived Gelators Containing a Basic Side Chain. Beilstein J. Org. Chem. 2011, 7, 304–309. [Google Scholar] [CrossRef]
- Smith, M.M.; Edwards, W.; Smith, D.K. Self-Organisation Effects in Dynamic Nanoscale Gels Self-Assembled from Simple Mixtures of Commercially Available Molecular-Scale Components. Chem. Sci. 2013, 4, 671–676. [Google Scholar]
- Suzuki, M.; Nakajima, Y.; Yumoto, M.; Kimura, M.; Shirai, H.; Hanabusa, K. In Situ Organogelation at Room Temperature: Direct Synthesis of Gelators in Organic Solvents. Org. Biomol. Chem. 2004, 2, 1155–1159. [Google Scholar] [CrossRef]
- George, M.; Weiss, R.G. Chemically Reversible Organogels: Aliphatic Amines as “Latent” Gelators with Carbon Dioxide. J. Am. Chem. Soc. 2001, 123, 10393–10394. [Google Scholar] [CrossRef]
- Wu, H.; Ni, B.-B.; Wang, C.; Zhai, F.; Ma, Y. Acid-Responsive Organogel Mediated by Arene–Perfluoroarene and Hydrogen Bonding Interactions. Soft Matter 2012, 8, 5486–5492. [Google Scholar] [CrossRef]
- Buerkle, L.E.; Rowan, S.J. Supramolecular Gels Formed from Multi-Component Low Molecular Weight Species. Chem. Soc. Rev. 2012, 41, 6089–6102. [Google Scholar] [CrossRef]
- Hirst, A.R.; Smith, D.K. Two-Component Gel-Phase Materials—Highly Tunable Self-Assembling Systems. Chem. Eur. J. 2005, 11, 5496–5508. [Google Scholar] [CrossRef]
- Hongmanee, P.; Rukseree, K.; Buabut, B.; Somsri, B.; Palittapongarnpim, P. In Vitro Activities of Cloxyquin (5-Chloroquinolin-8-Ol) Against Mycobacterium Tuberculosis. Antimicrob. Agents Chemother. 2007, 51, 1105–1106. [Google Scholar] [CrossRef]
- Kalinda, A.S.; Aldrich, C.C. Pyrazinamide: A Frontline Drug used for Tuberculosis. Molecular Mechanism of Action Resolved After 50 Years? ChemMedChem 2012, 7, 558–560. [Google Scholar] [CrossRef]
- Kolehmainen, E.; Salo, H.; Korpela, J. Gelation Behavior of 5-Chloro-8-Hydroxyquinoline, an Antituberculosis Agent in Aqueous Alcohol Solutions. Antibiotics 2012, 1, 17–24. [Google Scholar]
- Escuder, B.; LLusar, M.; Miravet, J.F. Insight on the NMR Study of Supramolecular Gels and its Application to Monitor Molecular Recognition on Self-Assembled Fibers. J. Org. Chem. 2006, 71, 7747–7752. [Google Scholar] [CrossRef]
- Noponen, V.; Nonappa; Lahtinen, M.; Valkonen, A.; Salo, H.; Kolehmainen, E.; Sievänen, E. Bile Acid-Amino Acid Ester Conjugates: Gelation, Structural Properties, and Thermoreversible Solid to Solid Phase Transition. Soft Matter 2010, 6, 3789–3796. [Google Scholar] [CrossRef]
- Hou, X.; Gao, D.; Yan, J.; Ma, Y.; Liu, K.; Fang, Y. Novel Dimeric Cholesteryl Derivatives and their Smart Thixotropic Gels. Langmuir 2011, 27, 12156–12163. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bunzen, H.; Kolehmainen, E. In Situ Formation of Steroidal Supramolecular Gels Designed for Drug Release. Molecules 2013, 18, 3745-3759. https://doi.org/10.3390/molecules18043745
Bunzen H, Kolehmainen E. In Situ Formation of Steroidal Supramolecular Gels Designed for Drug Release. Molecules. 2013; 18(4):3745-3759. https://doi.org/10.3390/molecules18043745
Chicago/Turabian StyleBunzen, Hana, and Erkki Kolehmainen. 2013. "In Situ Formation of Steroidal Supramolecular Gels Designed for Drug Release" Molecules 18, no. 4: 3745-3759. https://doi.org/10.3390/molecules18043745




