Microwave Assisted Suzuki-Miyaura and Ullmann Type Homocoupling Reactions of 2- and 3-Halopyridines Using a Pd(OAc)2/Benzimidazolium Salt and Base Catalyst System
Abstract
:1. Introduction
2. Results and Discussion
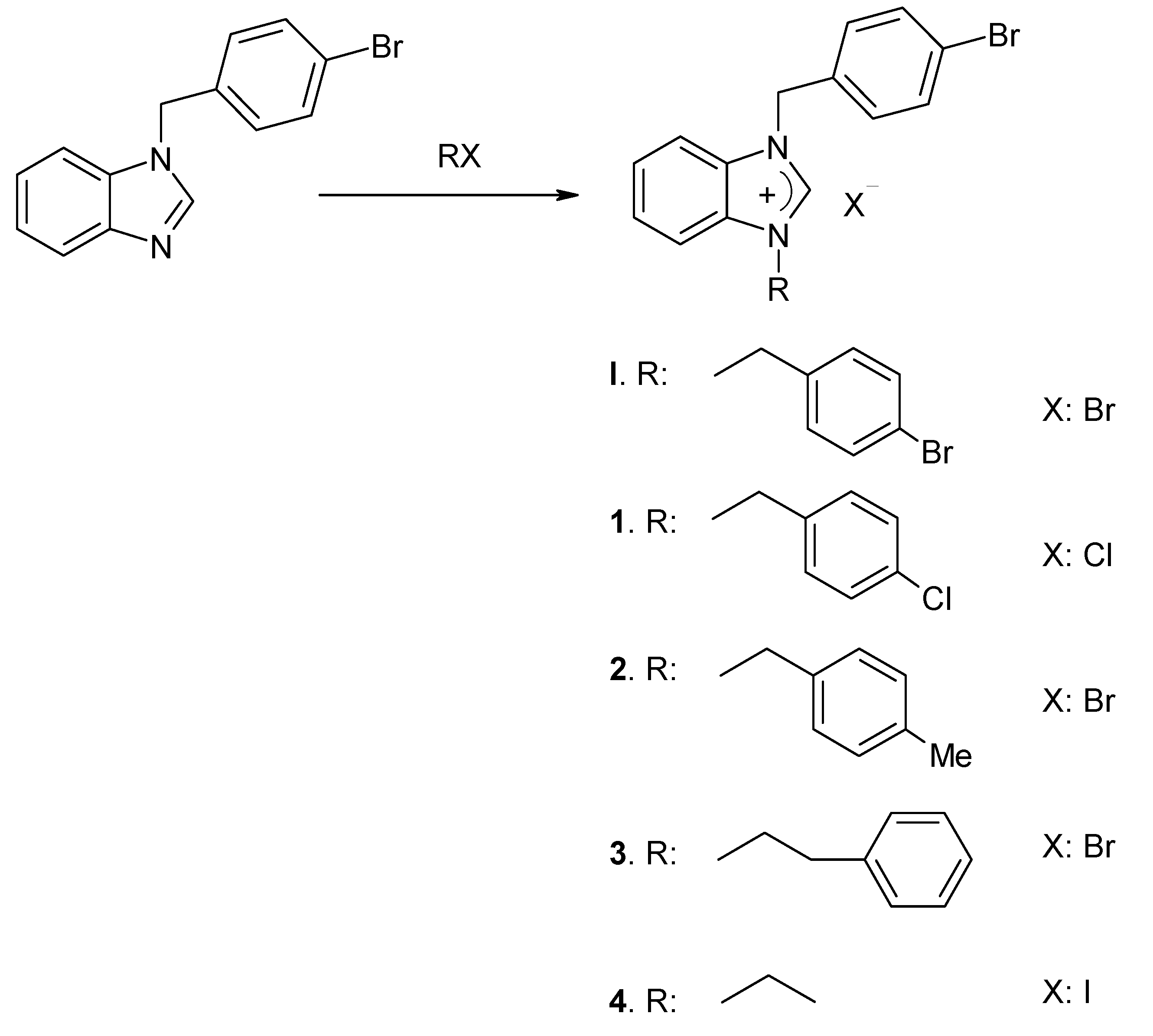
2.1. The Suzuki-Miyaura Type Coupling Reaction
| Entry | X | Y | Z | Salt | % Yields | ||||
|---|---|---|---|---|---|---|---|---|---|
 |  |  |  |  | |||||
| 1 a | Br | N | CH | - | 40.3 | 13.8 | 1.1 | 0.6 | 17.2 |
| 2 b | Br | N | CH | 2 | 2.0 | - | - | - | - |
| 3 c | Br | N | CH | 2 | 57.6 | 5.3 | - | - | 15.6 |
| 4 d | Br | N | CH | 2 | 63.4 | 4.7 | 0.8 | - | 7.1 |
| 5 e | Br | N | CH | 2 | 70.0 | 22.0 | 0.3 | 0.7 | 7.0 |
| 6 | Br | N | CH | I | 65.0 | 23.7 | 0.7 | 0.7 | 9.9 |
| 7 | Br | N | CH | 1 | 67.0 | 27.2 | - | 1.6 | 4.2 |
| 8 | Br | N | CH | 3 | 72.5 | 17.8 | 0.6 | 1.3 | 7.8 |
| 9 | Br | N | CH | 4 | 66.8 | 18.8 | - | - | 4.4 |
| 10 | Cl | N | CH | I | 61.9 | 12.0 | 0.4 | 3.1 | 12.6 |
| 11 | Cl | N | CH | 1 | 64.1 | 14.5 | - | 9.1 | 6.1 |
| 12 | Cl | N | CH | 2 | 65.4 | 12.4 | 0.2 | 5.4 | 12.8 |
| 13 | Cl | N | CH | 3 | 66.1 | 12.5 | - | 4.4 | 9.0 |
| 14 | Cl | N | CH | 4 | 51.8 | 2.5 | - | 6.8 | 4.3 |
| 15 | Br | CH | N | I | 90.0 | - | - | 2.8 | 7.2 |
| 16 | Br | CH | N | 1 | 89.3 | - | - | 2.1 | 8.6 |
| 17 | Br | CH | N | 2 | 91.2 | - | - | 2.9 | 5.9 |
| 18 | Br | CH | N | 3 | 93.4 | 0.7 | - | 1.9 | 4.0 |
| 19 | Br | CH | N | 4 | 90.0 | - | - | 2.4 | 7.6 |
| 20 | Cl | CH | N | I | 66.6 | - | - | 2.5 | 23.8 |
| 21 | Cl | CH | N | 1 | 76.5 | - | - | 6.7 | 12.5 |
| 22 | Cl | CH | N | 2 | 70.1 | 0.7 | - | 2.0 | 16.4 |
| 23 | Cl | CH | N | 3 | 73.5 | - | - | 1.8 | 18.5 |
| 24 | Cl | CH | N | 4 | 58.0 | - | - | 4.8 | 9.2 |
2.2. The Ullmann Type Homocoupling Reaction
| Entry | X | Y | Z | Salt | % Yields | ||
|---|---|---|---|---|---|---|---|
 |  |  | |||||
| 1 a | Br | N | CH | 2 | 1.3 | - | - |
| 2 b | Br | N | CH | 2 | 12.0 | - | 0.4 |
| 3 c | Br | N | CH | 2 | 43.5 | 9.9 | - |
| 4 d | Br | N | CH | 2 | 48.5 | 2.5 | - |
| 5 | Br | N | CH | I | 84.5 | 2.4 | 12.9 |
| 6 | Br | N | CH | 1 | 75.1 | 8.3 | 16.6 |
| 7 | Br | N | CH | 2 | 69.7 | 12.2 | 1.8 |
| 8 | Br | N | CH | 3 | 74.4 | 14.3 | 3.8 |
| 9 | Br | N | CH | 4 | 33.7 | 9.8 | 3.6 |
| 10 | Cl | N | CH | I | 64.3 | 0.6 | 34.4 |
| 11 | Cl | N | CH | 1 | 31.4 | 3.4 | 47.7 |
| 12 | Cl | N | CH | 2 | 46.5 | 5.1 | 23.2 |
| 13 | Cl | N | CH | 3 | 20.9 | 5.5 | 15.2 |
| 14 | Cl | N | CH | 4 | 8.0 | 5.5 | 3.7 |
| 15 | Br | CH | N | I | 15.0 | - | 85.0 |
| 16 | Br | CH | N | 1 | 5.2 | - | 94.8 |
| 17 | Br | CH | N | 2 | 6.6 | - | 93.4 |
| 18 | Br | CH | N | 3 | 2.3 | - | 97.7 |
| 19 | Br | CH | N | 4 | - | - | 62.2 |
| 20 | Cl | CH | N | I | 1.0 | - | 26.1 |
| 21 | Cl | CH | N | 1 | - | - | 40.6 |
| 22 | Cl | CH | N | 2 | 1.6 | - | 11.4 |
| 23 | Cl | CH | N | 3 | 1.0 | - | 10.6 |
| 24 | Cl | CH | N | 4 | - | - | 21.6 |
3. Experimental
3.1. General Procedures
3.2. General Procedure for the Suzuki-Miyaura and the Ullmann Type Homocoupling Reactions
3.2.1. The Suzuki-Miyaura Reaction
3.2.2. Ullmann Type Homocoupling Reaction
4. Conclusions
Supplementary Materials
Acknowledgments
References
- Negishi, E.; de Meijere, A. Handbook of Organopalladium Chemistry for Organic Synthesis; Wiley-Interscience: New York, NY, USA, 2002; p. 3279. [Google Scholar]
- Miyaura, N.; Suzuki, A. Palladium-catalyzed cross-coupling reactions of organoboron compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar] [CrossRef]
- Suzuki, A. Recent advances in the cross-coupling reactions of organoboron derivatives with organic electrophiles, 1995–1998. J. Organomet. Chem. 1999, 576, 147–168. [Google Scholar]
- Fortman, G.C.; Nolan, S.P. N-Heterocyclic carbene (NHC) ligands and palladium in homogeneous cross-coupling catalysis: a perfect union. Chem. Soc. Rev. 2011, 40, 5151–5169. [Google Scholar] [CrossRef]
- Noël, T.; Buchwald, S.L. Cross-coupling in flow. Chem. Soc. Rev. 2011, 40, 5010–5029. [Google Scholar]
- Inoue, A.; Kitagawa, K.; Shinokubo, H.; Oshima, K. Simple and efficient TiCl4-mediated synthesis of biaryls via arylmagnesium compounds. Tetrahedron 2000, 56, 9601–9605. [Google Scholar] [CrossRef]
- McKillop, A.; Elsom, L.F.; Taylor, E.C. Thallium in organic synthesis III. Coupling of aryl and alkyl Grignard reagents. J. Am. Chem. Soc. 1968, 90, 2423–2424. [Google Scholar] [CrossRef]
- Ishikawa, T.; Ogawa, A.; Hirao, T. Oxovanadium(V)-induced oxidative coupling of organolithium and -magnesium compounds. Organometallics 1998, 17, 5713–5716. [Google Scholar] [CrossRef]
- Kharasch, M.S.; Fields, E.K. Factors determining the course and mechanism of Grignard reactions. IV. The effect of metallic halides on the reaction of aryl Grignard reagents and organic halides. J. Am. Chem. Soc. 1941, 63, 2316–2320. [Google Scholar]
- Nagaki, A.; Uesugi, Y.; Tomida, Y.; Yoshida, J. Homocoupling of aryl halides in flow: Space integration of lithiation and FeCl3 promoted homocoupling. Beilstein J. Org. Chem. 2011, 7, 1064–1069. [Google Scholar] [CrossRef] [Green Version]
- Bergeron-Brlek, M.; Giguère, D.; Shiao, T.C.; Saucier, C.; Roy, R. Palladium-catalyzed Ullmann-type reductive homocoupling of iodoaryl glycosides. J. Org. Chem. 2012, 77, 2971–2977. [Google Scholar] [CrossRef]
- Lei, A.W.; Srivastava, M.; Zhang, X.M. Transmetalation of palladium enolate and its application in palladium-catalyzed homocoupling of alkynes: A room-temperature, highly efficient route to make diynes. J. Org. Chem. 2002, 67, 1969–1971. [Google Scholar] [CrossRef]
- Del Zotto, A.; Amoroso, F.; Baratta, W.; Rigo, P. Very fast Suzuki-Miyaura reaction catalyzed by Pd(OAc)2 under aerobic conditions at room temperature in EGME/H2O. Eur. J. Org. Chem. 2009, 1, 110–116. [Google Scholar]
- Zhang, H.; Kwong, F.Y.; Tian, Y.; Chan, K.S. Base and cation effects on the Suzuki cross-coupling of bulky arylboronicacid with halopyridines: Synthesis of pyridylphenols. J. Org. Chem. 1998, 63, 6886–6890. [Google Scholar]
- Nadri, S.; Azadi, E.; Ataei, A.; Joshaghani, M.; Rafiee, E. Investigation of the catalytic activity of a Pd/biphenyl-based phosphine system in the Ullmann homocoupling of aryl bromides. J. Organomet. Chem. 2011, 696, 2966–2970. [Google Scholar] [CrossRef]
- Joshaghani, M.; Faramarzi, E.; Rafiee, E.; Daryanavard, M.; Xiao, J.; Baillie, C. Highly efficient Suzuki coupling using moderately bulky tolylphosphine ligands. J. Mol. Catal. A Chem. 2007, 273, 310–315. [Google Scholar] [CrossRef]
- Joshaghani, M.; Daryanavard, M.; Rafiee, E.; Xiao, J.; Baillie, C. A highly efficient catalyst for Suzuki coupling of aryl halides and bromoarylphosphine oxides. Tetrahedron Lett. 2007, 48, 2025–2027. [Google Scholar] [CrossRef]
- Adamo, C.; Amatore, C.; Ciofini, I.; Jutand, A.; Lakmini, H. Mechanism of the palladium-catalyzed homocoupling of arylboronic acids: Key involvement of a palladium peroxo complex. J. Am. Chem. Soc. 2006, 128, 6829–6836. [Google Scholar]
- Billingsley, K.L.; Anderson, K.W.; Buchwald, S.L. A highly active catalyst for Suzuki-Miyaura cross-coupling reactions of heteroaryl compounds. Angew. Chem. Ind. Ed. Engl. 2006, 45, 3484–3488. [Google Scholar] [CrossRef]
- Seganish, W.M.; Mowery, M.E.; Riggleman, S.; DeShong, P. Palladium-catalyzed homocoupling of aryl halides in the presence of floride. Tetrahedron 2005, 61, 2117–2121. [Google Scholar] [CrossRef]
- Wolfe, J.P.; Tomori, H.; Sodighi, J.P.; Yin, J.J.; Buchwald, S.L. Simple, efficient catalyst system for the palladium-catalyzed amination of aryl chlorides, bromides, and triflats. J. Org. Chem. 2000, 65, 1158–1174. [Google Scholar] [CrossRef]
- Iranpoor, N.; Firouzabadi, H.; Azadi, R. Imidazolium-based phosphinite ionic liquid (IL-OPPh2) as Pd ligand and solvent for selective dehalogenation or homocoupling of aryl halides. J. Organomet. Chem. 2008, 693, 2469–2472. [Google Scholar] [CrossRef]
- Zembayashi, M.; Tamao, K.; Yoshida, J.; Kumada, M. Nickel-Phosphine complex-catalyzed homo coupling of aryl halides in the presence of zinc powder. Tetrahedron Lett. 1977, 47, 4089–4092. [Google Scholar]
- Iyoda, M.; Otsuka, H.; Sato, K.; Nisato, N.; Oda, M. Homocoupling of aryl halides using nickel (II) complex and zinc in the presence of Et4NI. An efficient method for the synthesis of biaryls and bipyridines. Bull. Chem. Soc. Jpn. 1990, 63, 80–87. [Google Scholar]
- Peris, E.; Loch, J.A.; Mata, J.; Crabtre, R.H. A Pd complex of a tridentate pincer CNC bis-carbene ligand as a robust homogenous Heck catalyst. Chem. Commun. 2001, 2, 201–202. [Google Scholar]
- Dawood, K.M. Microwave-assisted Suzuki-Miyaura and Heck-Mizoroki cross-coupling reactions of aryl chlorides and bromides in water using stable benzothiazole-based palladium(II) precatalysts. Tetrahedron 2007, 63, 9642–9651. [Google Scholar] [CrossRef]
- Dallinger, D.; Kappe, C.O. Microwave-assisted synthesis in water as solvent. Chem. Rev. 2007, 107, 2563–2591. [Google Scholar] [CrossRef]
- Irfan, M.; Fuchs, M.; Glasnov, T.N.; Kappe, C.O. Microwave-assisted Cross-Coupling and hydrogenation chemistry by using heterogeneous transition-metal catalysts: An evaluation of the role of selective catalyst heating. Chem. Eur. J. 2009, 15, 11608–11618. [Google Scholar] [CrossRef]
- Brooker, M.D.; Cooper, S.M., Jr.; Hodges, D.R.; Carter, R.R.; Wyatt, J.K. Studies of microwave-enhanced Suzuki-Miyaura vinylation of electron-rich sterically hindered substrates utilizing potassium vinyltrifluoroborate. Tetrahedron Lett. 2010, 51, 6748–6752. [Google Scholar] [CrossRef]
- Martins, D.L.; Alvarez, H.M.; Aguiar, L.C.S. Microwave-assisted Suzuki reaction catalyzed by Pd(0)-PVP nanoparticles. Tetrahedron Lett. 2010, 51, 6814–6817. [Google Scholar] [CrossRef]
- Hajipour, A.R.; Karami, K.; Tavakoli, G. A comparative homocoupling reaction of aryl halides using monomeric orthopalladated complex of 4-methoxybenzoylmethylenetriphenylphosphorane under conventional and microwave irradiation conditions. Appl. Organometal. Chem. 2011, 25, 567–576. [Google Scholar] [CrossRef]
- Gädda, T.M.; Kawanishi, Y.; Miyazawa, A. Microwave-assisted Ullmann-type coupling reactions in alkaline water. Synth. Commun. 2012, 42, 1259–1267. [Google Scholar] [CrossRef]
- Liu, L.-J.; Wang, F.; Shi, M. Elimination of an alkyl group from imidazolium salts: Imidazole-coordinated dinuclear monodentate NHC-palladium complexes driven by self-assembly and their application in the Heck reaction. Eur. J. Inorg. Chem. 2009, 13, 1723–1728. [Google Scholar]
- Yılmaz, Ü.; Şireci, N.; Deniz, S.; Küçükbay, H. Synthesis and microwave-assisted catalytic activity of novel bis-benzimidazole salts bearing furfuryl and thenyl moieties in Heck and Suzuki cross-coupling reactions. Appl. Organometal. Chem. 2010, 24, 414–420. [Google Scholar]
- Yılmaz, Ü.; Küçükbay, H.; Şireci, N.; Akkurt, M.; Günal, S.; Durmaz, R.; Tahir, M.N. Synthesis, microwave-promoted catalytic activity in Suzuki-Miyaura cross-coupling reactions and antimicrobial properties of novel benzimidazole salts bearing trimethylsilyl group. Appl. Organometal. Chem. 2011, 25, 366–373. [Google Scholar]
- Küçükbay, H.; Şireci, N.; Yılmaz, Ü.; Akkurt, M.; Yalçın, Ş.P.; Tahir, M.N.; Ott, H. Synthesis, characterization and microwave-assisted catalytic activity of novel benzimidazole salts bearing piperidine and morpholine moieties in Heck cross-coupling reactions. Appl. Organometal. Chem. 2011, 25, 255–261. [Google Scholar]
- Küçükbay, H.; Şireci, N.; Yılmaz, Ü.; Deniz, S.; Akkurt, M.; Baktır, Z.; Büyükgüngör, O. Synthesis, characterization, and microwave-promoted catalytic activity of novel benzimidazole reactions under aerobic conditions. Turk. J. Chem. 2012, 36, 201–217. [Google Scholar]
- Şireci, N.; Yılmaz, Ü.; Küçükbay, H. Microwave assisted catalytic activity of some bis-5(6)-nitrobenzimidazole salts for Heck and Suzuki cross-coupling reactions. Asian J. Chem. 2010, 22, 7153–7158. [Google Scholar]
- Yılmaz, Ü.; Küçükbay, H.; Deniz, S.; Şireci, N. Synthesis, characterization and microwave-promoted catalytic activity of novel N-phenylbenzimidazolium salts in Heck-Mizoroki and Suzuki-Miyaura cross-coupling reactions under mild condtiyions. Molecules 2013, 18, 2501–2517. [Google Scholar] [CrossRef]
- Bensaid, S.; Doucet, H. Palladium-catalysed direct arylation of heteroaromatics with functionalised bromopyridines. Tetrahedron 2012, 68, 7655–7662. [Google Scholar] [CrossRef]
- Liu, C.; Yang, W. A fast and oxygen-promoted protocol for the ligand-free Suzuki reaction of 2-halogenated pyridines in aqueous media. Chem. Commun. 2009, 41, 6267–6269. [Google Scholar]
- Liu, Y.; Ye, K.; Fan, Y.; Song, W.; Wang, Y.; Hou, Z. Amidinate-ligated iridium(III) bis(2-pyridyl)phenyl complex as an excellent phosphorescent material for electroluminescence devices. Chem. Commun. 2009, 3699–3701. [Google Scholar]
- Mukhopadhyay, S.; Rothenberg, G.; Gitis, D.; Baidossi, M.; Ponde, D.E.; Sasson, Y. Regiospecific cross-coupling of haloaryls and pyridine to 2-phenylpyridine using water, zinc, and catalytic palladium on carbon. J. Chem. Soc. Perkin Trans. 2 2000, 9, 1809–1812. [Google Scholar]
- Tagata, T.; Nishida, M. Palladium charcoal-catalyzed Suzuki-Miyaura coupling to obtain arylpyridines and arylquinolines. J. Org. Chem. 2003, 68, 9412–9415. [Google Scholar] [CrossRef]
- Kudo, N.; Perseghini, M.; Fu, G.C. A versatile method for Suzuki cross-coupling reactions of nitrogen heterocycles. Angew. Chem. Ind. Ed. Engl. 2006, 45, 1282–1284. [Google Scholar] [CrossRef]
- Wen, J.; Qin, S.; Ma, L.-F.; Dong, L.; Zhang, J.; Liu, S.-S.; Duan, Y.-S.; Chen, S.-Y.; Hu, C.-W.; Yu, X.-Q. Iron-Mediated Direct Suzuki-Miyaura Reaction: A New Method for the ortho-Arylation of Pyrrole and Pyridine. Org. Lett. 2010, 12, 2694–2697. [Google Scholar] [CrossRef]
- Hennings, D.D.; Iwama, T.; Rawal, V.H. Palladium-catalyzed (Ullmann-Type) homocoupling of aryl halides: A convenient and general synthesis of symmetrical biaryls via inter- and intramolecular coupling reactions. Org. Lett. 1999, 1, 1205–1208. [Google Scholar] [CrossRef]
- França, K.W.R.; Navarro, M.; Leonel, E.; Durandetti, M.; Nedelec, J.-Y. Electrochemical homocoupling of 2-bromomethylpyridines catalyzed by nickel complexes. J. Org. Chem. 2002, 67, 1838–1842. [Google Scholar] [CrossRef]
- Cravotta, G.; Beggiato, M.; Penoni, A.; Palmisano, G.; Tollari, S.; Lévêque, J.-M.; Bonrath, W. High-intensity ultrasound and microwave, alone or combined, promote Pd/C-catalyzed aryl-aryl couplings. Tetrahedron Lett. 2005, 46, 2267–2271. [Google Scholar]
- Tao, X.; Zhou, W.; Zhang, Y.; Dai, C.; Shen, D.; Huang, M. Homocoupling of aryl bromides catalyzed by nickel chloride in pyridine. Chin. J. Chem. 2006, 24, 939–942. [Google Scholar] [CrossRef]
- Park, B.R.; Kim, K.H.; Kim, T.H.; Kim, J.N. Palladium-catalyzed benzoin-mediated redox process leading to biaryls from aryl halides. Tetrahedron Lett. 2011, 52, 4405–4407. [Google Scholar] [CrossRef]
- Moore, L.R.; Vicic, D.A. A heterogeneous-catalyst-based, microwave-assisted protocol for the synthesis of 2,2'-bipyridines. Chem. Asian. J. 2008, 3, 1046–1049. [Google Scholar] [CrossRef]
- Mo, J.-M.; Ma, Y.-G.; Cheng, Y. Synthesis of novel synthetic intermediates from the reaction of benzimidazole and triazole carbenes with ketenimines and their application in the construction of spiro-pyrroles. Org. Biomol. Chem. 2009, 7, 5010–5019. [Google Scholar] [CrossRef]
- Wan, Y.; Wallinder, C.; Plouffe, B.; Beaudry, H.; Mahalingam, A.K.; Wu, X.; Johansson, B.; Holm, M.; Botoros, M.; Karlén, A.; et al. Design, synthesis, and biological evaluation of the first selective nonpeptide AT2 receptor agonist. J. Med. Chem. 2004, 47, 5995–6008. [Google Scholar] [CrossRef]
- Chanthavong, F.; Leadbeater, N.E. The application of organic bases in microwave-promoted Suzuki coupling reactions in water. Tetrahedron Lett. 2006, 47, 1909–1912. [Google Scholar] [CrossRef]
- Zhao, J.K.; Wang, Y.G. A facile and efficient synthesis of N,N-dimethylarylamines from aryl bromides. Chin. Chem. Lett. 2002, 13, 1149–1151. [Google Scholar]
- Chen, W.-X.; Shao, L.-X. N-Heterocyclic Carbene-Palladium(II)-1-Methylimidazole Complex Catalyzed Amination between Aryl Chlorides and Amides. J. Org. Chem. 2012, 77, 9236–9239. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are all available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yılmaz, Ü.; Deniz, S.; Küçükbay, H.; Şireci, N. Microwave Assisted Suzuki-Miyaura and Ullmann Type Homocoupling Reactions of 2- and 3-Halopyridines Using a Pd(OAc)2/Benzimidazolium Salt and Base Catalyst System. Molecules 2013, 18, 3712-3724. https://doi.org/10.3390/molecules18043712
Yılmaz Ü, Deniz S, Küçükbay H, Şireci N. Microwave Assisted Suzuki-Miyaura and Ullmann Type Homocoupling Reactions of 2- and 3-Halopyridines Using a Pd(OAc)2/Benzimidazolium Salt and Base Catalyst System. Molecules. 2013; 18(4):3712-3724. https://doi.org/10.3390/molecules18043712
Chicago/Turabian StyleYılmaz, Ülkü, Selma Deniz, Hasan Küçükbay, and Nihat Şireci. 2013. "Microwave Assisted Suzuki-Miyaura and Ullmann Type Homocoupling Reactions of 2- and 3-Halopyridines Using a Pd(OAc)2/Benzimidazolium Salt and Base Catalyst System" Molecules 18, no. 4: 3712-3724. https://doi.org/10.3390/molecules18043712






