Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems †
Abstract
:1. Introduction
2. Results and Discussion
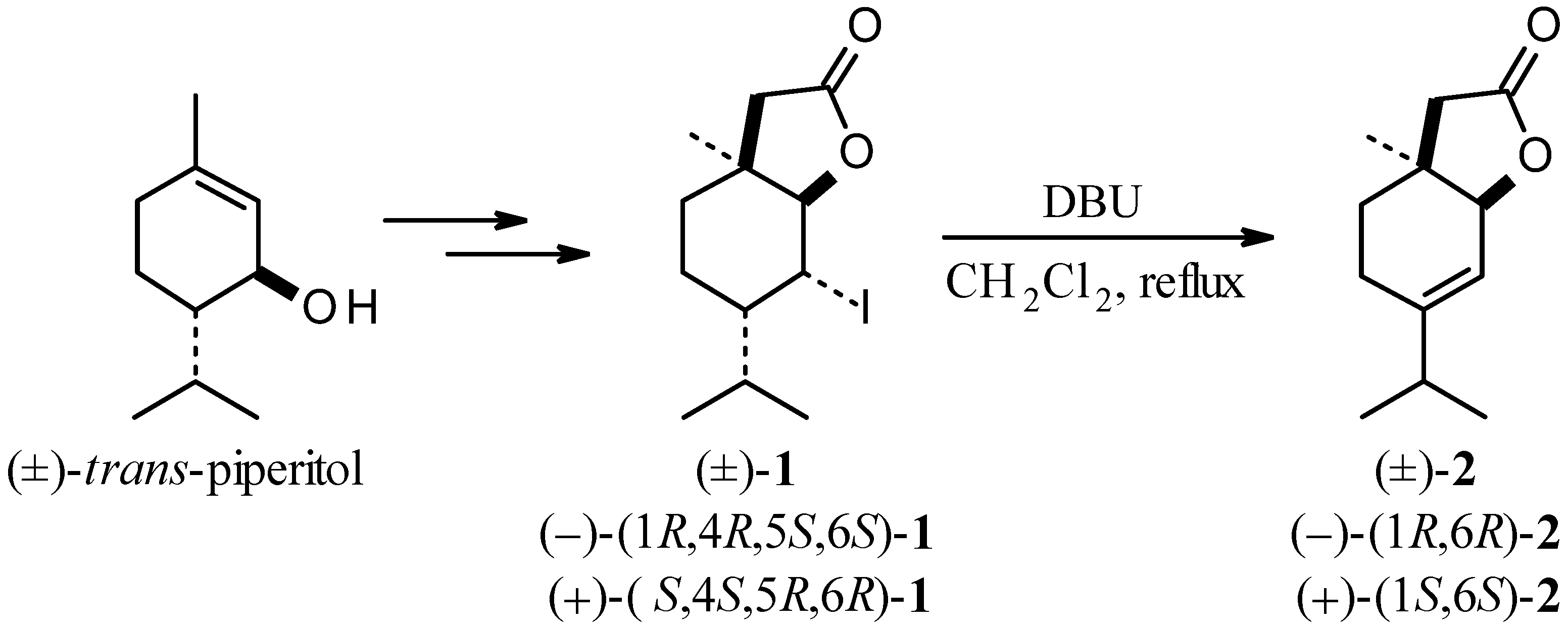
2.1. Synthesis and Odoriferous Properties of Racemic [(±)-2] and Optically Active [(–)-(1R,6R)-2 and (+)-(1S,6S)-2]δ,ε-Unsaturated Lactones
| Compound | Odour description |
|---|---|
| (±)- 2 | Weak, slightly similar to (+)- 2 |
| (–)-(1 R,6R)-2 | Intensive, dill |
| (+)-(1 S,6S)-2 | Medium intensity, coumarin-like |
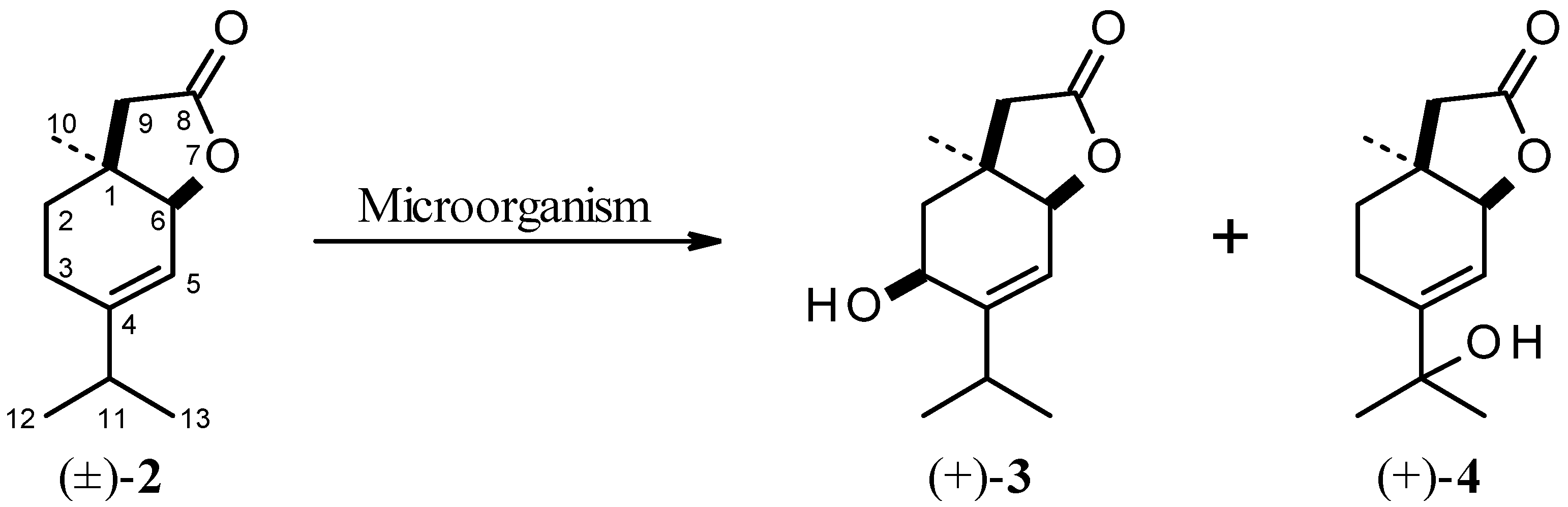
2.2. Biotransformation of Racemic δ,ε-Unsaturated Lactone [(±)-2]
| Microorganism | Time (days) | 2 | 3 | 4 * | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (%) | ee (%) | Config. | (%) | ee (%) |  ** ** | Config. | (%) |  ** ** | ||
| A. cylindrospora AM336 | 1 | 66 | 3 | (1S,6S) | 14 | 14 | (1S,3S,6S) | 20 | ||
| 2 | 41 | 0 | - | 22 | 36 | (1S,3S,6S) | 37 | |||
| 4 | 0 | - | - | 39 | 39 | +8.3 (c 0.38) | (1S,3S,6S) | 61 | +2.2 (c 0.23) | |
| A. glauca AM177 | 2 | 67 | 7 | (1S,6S) | 6 | 0 | - | 27 | ||
| 4 | 32 | 6 | (1S,6S) | 15 | 20 | (1S,3S,6S) | 53 | |||
| 6 | 9 | 9 | (1S,6S) | 23 | 22 | +5.1 (c 0.10) | (1S,3S,6S) | 68 | +0.8 (c 1.28) | |
| S. racemosum AM105 | 2 | 43 | 72 | (1S,6S) | 17 | 18 | (1S,3S,6S) | 40 | ||
| 4 | 11 | 54 | (1S,6S) | 20 | 35 | (1S,3S,6S) | 69 | |||
| 6 | 8 | 50 | (1S,6S) | 21 | 38 | +6.8 (c 0.36) | (1S,3S,6S) | 71 | +1.1 (c 1.19) | |
 = +8.3° (c 0.38, CHCl3), ee = 39%) and 28 mg (22% isolated yield) of (+)-4-(1'hydroxy-1'methylethyl)-1-methyl-7-oxa-cis-bicyclo[4.3.0]non-4-en-8-one (4) (
= +8.3° (c 0.38, CHCl3), ee = 39%) and 28 mg (22% isolated yield) of (+)-4-(1'hydroxy-1'methylethyl)-1-methyl-7-oxa-cis-bicyclo[4.3.0]non-4-en-8-one (4) (  = +2.2° (c 0.23, CHCl3).
= +2.2° (c 0.23, CHCl3). = +5.1 (c 0.10, CHCl3), ee = 22%) and 21 mg (16% isolated yield) of (+)-4-(1'hydroxy-1'methylethyl)-1-methyl-7-oxa-cis-bicyclo[4.3.0]non-4-en-8-one (4) (
= +5.1 (c 0.10, CHCl3), ee = 22%) and 21 mg (16% isolated yield) of (+)-4-(1'hydroxy-1'methylethyl)-1-methyl-7-oxa-cis-bicyclo[4.3.0]non-4-en-8-one (4) (  = +0.8° (c 1.28, CHCl3).
= +0.8° (c 1.28, CHCl3). = +6.8 (c 0.36, CHCl3), ee = 38%) and 4 (29 mg, 22% isolated yield,
= +6.8 (c 0.36, CHCl3), ee = 38%) and 4 (29 mg, 22% isolated yield,  = +1.1° (c 1.19, CHCl3)).
= +1.1° (c 1.19, CHCl3)).3. Experimental
3.1. General
3.2. Chemical Synthesis
 = −1.9° (c 1.15, CHCl3). IR and NMR spectra were identical with those of (±)-2.
= −1.9° (c 1.15, CHCl3). IR and NMR spectra were identical with those of (±)-2. = +2.3° (c 1.24, CHCl3). IR and NMR spectra were identical with those of (±)-2.
= +2.3° (c 1.24, CHCl3). IR and NMR spectra were identical with those of (±)-2.3.3. Microorganisms
3.4. Screening-Scale Biotransformations
3.5. Preparative-Scale Biotransformations
4. Conclusions
Supplementary Materials
Acknowledgments
References
- Dams, I.; Białońska, A.; Ciunik, Z.; Wawrzeńczyk, C. Lactones.21. Synthesis and odoriferous properties of lactones with the p-menthane system. J. Agric. Food Chem. 2004, 52, 1630–1634. [Google Scholar] [CrossRef]
- Frerot, S.; Bagnound, A.; Vuilleumier, C. Menthofurolactone: A new p-menthane lactone in Mentha piperita L.: Analysis, synthesis and olfactory properties. Flavour Fragr. J. 2002, 17, 218–226. [Google Scholar] [CrossRef]
- Bourgeois, M.J.; Montaudon, E. Base-indused reactions of p-menthane and pinane-derived epoxyesters - coconut fragrance. Helv. Chim. Acta 2001, 84, 2430–2438. [Google Scholar] [CrossRef]
- Gaudin, J.M. Synthesis and organoleptic propertiesof p-menthane lactones. Tetrahedron 2000, 56, 4769–4776. [Google Scholar] [CrossRef]
- Naf, R.; Velluz, A. Phenols and lactones in Italo-Mitcham peppermint oil Mentha x piperita L. Flavour Fragr. J. 1998, 13, 203–208. [Google Scholar] [CrossRef]
- Gouth, H. Determination of the configuration of wine lactone. Helv. Chim. Acta 1996, 79, 1559–1570. [Google Scholar] [CrossRef]
- Wawrzeńczyk, C.; Dams, I.; Szumny, A.; Szczepanik, M.; Nawrot, J.; Prądzyńska, A.; Gabryś, B.; Magnucka, E.; Gawdzik, B.; Obara, R.; et al. Synthesis and evaluation of antifeedant, antifungal and antibacterial activity of isoprenoid lactones. Pol. J. Environ. Stud. 2005, 14 (Suppl. II), 69–84. [Google Scholar]
- Szczepanik, M.; Dams, I.; Wawrzeńczyk, C. Terpenoid lactones with the p-menthane system as feeding deterrents to the lesser mealworm, Alphitobius diaperinus. Entomol. Exp. Appl. 2008, 128, 337–345. [Google Scholar] [CrossRef]
- Paruch, E.; Ciunik, Z.; Nawrot, J.; Wawrzeńczyk, C. Lactones. 9. Synthesis of terpenoid lactones—Active insect antifeedants. J. Agric. Food Chem. 2000, 48, 4973–4977. [Google Scholar] [CrossRef]
- Masłowiec, D.; Nawrot, J.; Białońska, A.; Wawrzeńczyk, C. Lactones. Part XL. Synthesis and antifeedant activity of ε-lactones fused to the methoxysubstituted benzene ring. Przem. Chem. 2012, 91, 883–889. [Google Scholar]
- Mori, K. Chirality and insect pheromones. Chirality 1998, 10, 578–586. [Google Scholar] [CrossRef]
- Onken, J.; Berger, R.G. Effects of R-(+)-limonene on submerged cultures of the terpene transforming basidiomycete Pleurotus sapidus. J. Biotechnol. 1999, 69, 163–168. [Google Scholar] [CrossRef]
- Rao, S.C. V.; Rao, R.; Agrawal, R. Enhanced production of verbenol, a highly valued food flavourant, by an intergeneric fusant strain of Aspergillus niger and Penicillium digitatum. Biotechnol. Appl. Biochem. 2003, 37, 145–147. [Google Scholar] [CrossRef]
- Vanek, T.; Halik, J.; Vankowa, R.; Valterova, I. Formation of trans-verbenol and verbenone from α-pinene catalysed by immobilised Picea abies cells. Biosci. Biotechnol. Biochem. 2005, 69, 321–325. [Google Scholar] [CrossRef]
- Berger, R.G. Flavours and Fragrances. Chemistry, Bioprocessing and Sustainability, 1st ed.; Springer: Heidelberg, Germany, 2007; pp. 540–549. [Google Scholar]
- Bailey, P.D.; Higgins, S.D.; Ridyard, C.H.; Roberts, S.M.; Rosair, G.M.; Whittaker, R.A.; Willetts, A.J. Selective biohydroxylation of 1-substituted adamantanes using Absidia cylindrospora (I.M.I. 342950). Chem. Commun. 1996, 1833–1834. [Google Scholar]
- Guo, N.; Zhao, Y.; Fang, W. Biotransformation of 3-oxo-oleanolic acid by Absidia glauca. Planta Med. 2010, 76, 1904–1907. [Google Scholar] [CrossRef]
- Huszcza, E.; Dmochowska-Gładysz, J. Transformations of testosterone and related steroids in Absidia glauca culture. J. Basic Microbiol. 2003, 2, 113–120. [Google Scholar]
- Huang, F.; Yang, W.; Ye, F.; Tian, J.; Hu, H.; Feng, L.; Guo, D.; Ye, M. Microbial transformation of ursolic acid by Syncephalastrum racemosum (Cohn) Schroter AS 3.264. Phytochemistry 2012, 82, 56–60. [Google Scholar]
- McClanahan, R.H.; Robertson, L.W. Biotransformation of olivetol by Syncephalastrum racemosum. J. Nat. Prod. 1984, 47, 828–834. [Google Scholar] [CrossRef]
- Grudniewska, A.; Dancewicz, K.; Białońska, A.; Ciunik, Z.; Gabryś, B.; Wawrzeńczyk, C. Synthesis of piperitone-derived halogenated lactones and their effect on aphid probing, feeding, and settling behavior. RSC Adv. 2011, 1, 498–510. [Google Scholar] [CrossRef]
- Gładkowski, W.; Grabarczyk, M.; Wińska, K.; Ratuś, B.; Białońska, A.; Ciunik, Z.; Wawrzeńczyk, C. Lactones 26: Stereoselective microbial epoxidation of unsaturated bicyclic γ-lactones with the alkylsubstituted cyclohexane system. J. Mol. Catal. B: Enzym. 2007, 49, 79–87. [Google Scholar] [CrossRef]
- Gladkowski, W.; Grabarczyk, M.; Wińska, K.; Bialońska, A.; Ciunik, Z.; Wawrzeńczyk, C. Lactones 27: Transformations of γ-lactones fused to a dimethylcyclohexane ring in Absidia cylindrospora cultures. J. Mol. Catal. B: Enzym. 2006, 39, 31–39. [Google Scholar] [CrossRef]
- Garcia-Granados, A.; Gutierrez, M.C.; Rivas, F. Improved microbiological hydroxylation of sesquiterpenoids: Semisynthesis, structural determination and biotransformation studies of cyclic sulfite eudesmane derivatives. Org. Biomol. Chem. 2003, 1, 2314–2320. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds (±)-2, (–)-2 and (+)-2 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Grudniewska, A.; Wawrzeńczyk, C. Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems. Molecules 2013, 18, 2778-2787. https://doi.org/10.3390/molecules18032778
Grudniewska A, Wawrzeńczyk C. Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems. Molecules. 2013; 18(3):2778-2787. https://doi.org/10.3390/molecules18032778
Chicago/Turabian StyleGrudniewska, Aleksandra, and Czesław Wawrzeńczyk. 2013. "Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems" Molecules 18, no. 3: 2778-2787. https://doi.org/10.3390/molecules18032778
APA StyleGrudniewska, A., & Wawrzeńczyk, C. (2013). Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems. Molecules, 18(3), 2778-2787. https://doi.org/10.3390/molecules18032778




