Structural Correlation of Some Heterocyclic Chalcone Analogues and Evaluation of Their Antioxidant Potential
Abstract
:1. Introduction
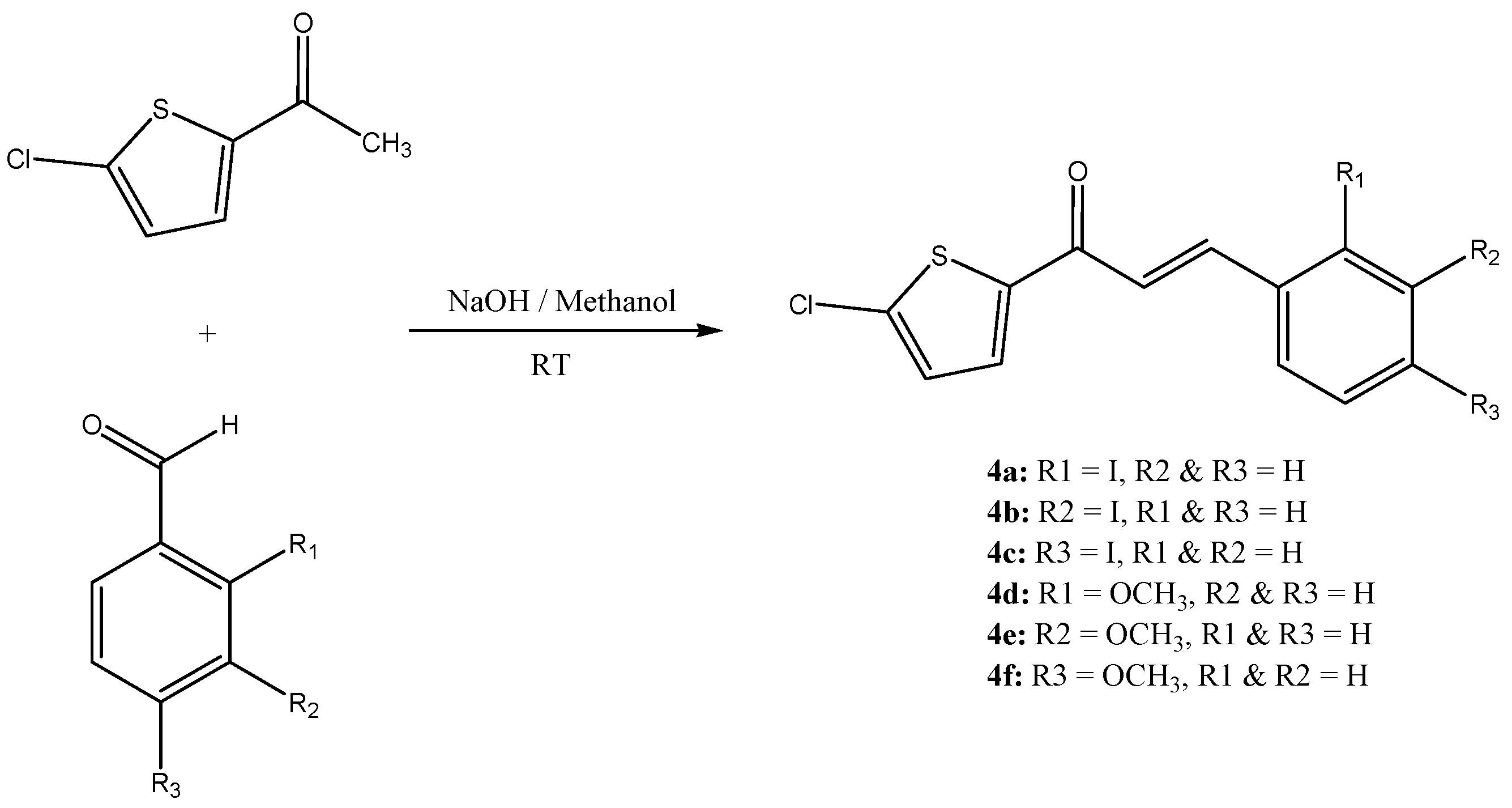
2. Results and Discussion
| Compound | 4a | 4b | 4c | 4d | 4e | 4f |
|---|---|---|---|---|---|---|
| CCDC deposition numbers | 939878 | 942739 | 942740 | 948858 | 944070 | 944071 |
| Molecular formula | C13H8ClIOS | C13H8ClIOS | C13H8ClIOS | C14H11ClO2S | C14H11ClO2S | C14H11ClO2S |
| Molecular weight | 374.60 | 374.60 | 374.60 | 278.74 | 278.74 | 278.74 |
| Crystal system | Monoclinic | Triclinic | Triclinic | Monoclinic | Monoclinic | Monoclinic |
| Space group | Cc | P-1 | P-1 | P21 | P21/c | Pc |
| a (Å) | 34.308(7) | 6.0486(8) | 6.1259(4) | 11.9567(15) | 5.9343(6) | 3.9116(9) |
| b (Å) | 4.1026(9) | 8.4673(15) | 8.6600(6) | 17.986(2) | 30.575(3) | 15.867(4) |
| c (Å) | 23.367(5) | 13.5924(15) | 13.5854(9) | 12.2265(15) | 7.3758(7) | 10.146(2) |
| α (°) | 90 | 88.616(2) | 98.330(1) | 90 | 90 | 90 |
| β (°) | 126.544(10) | 86.511(2) | 98.812(1) | 90.01 | 104.623(3) | 95.115(4) |
| γ (°) | 90 | 70.131(2) | 110.605(1) | 90 | 90 | 90 |
| V (Å3) | 2642.4(9) | 653.48(16) | 651.06(8) | 2629.3(5) | 1294.9(2) | 627.2(3) |
| Z | 8 | 2 | 2 | 8 | 4 | 2 |
| Dcalc (g cm−3) | 1.883 | 1.904 | 1.911 | 1.408 | 1.430 | 1.476 |
| Crystal dimensions (mm) | 0.35 × 0.17 × 0.09 | 0.52 × 0.22 × 0.09 | 0.76 × 0.41 × 0.13 | 0.69 × 0.15 × 0.06 | 0.56 × 0.40 × 0.07 | 0.66 × 0.26 × 0.07 |
| Colour | Colourless | Colourless | Colourless | Yellow | Colourless | Colourless |
| μ (mm−1) | 2.76 | 2.79 | 2.80 | 0.44 | 0.45 | 0.46 |
| Radiation λ (Å) | 0.71073 | 0.71073 | 0.71073 | 0.71073 | 0.71073 | 0.71073 |
| Tmin/Tmax | 0.448/0.779 | 0.322/0.779 | 0.223/0.705 | 0.753/0.972 | 0.790/0.971 | 0.753/0.968 |
| Reflections measured | 19059 | 10400 | 10227 | 19862 | 15699 | 6086 |
| Ranges/indices (h, k, l) | −44, 44; −5, 5; −30, 30 | −7, 7; −10, 10; −17, 17 | −7, 7; −11, 11; −17, 16 | −14, 14; −21, 20; −14, 14 | −8, 8; −44, 42; −9, 10 | −5, 4; −22, 22; −14, 14 |
| θ limit (°) | 2.4–29.1 | 1.5–26.5 | 1.6–27.5 | 2.8–26.1 | 1.3–31.2 | 2.6–39.9 |
| Unique reflections | 5911 | 2662 | 2972 | 8732 | 4170 | 3237 |
| Observed reflections (I > 2σ(I)) | 4676 | 2482 | 2735 | 6388 | 2644 | 2846 |
| Parameters | 308 | 154 | 154 | 650 | 163 | 163 |
| Goodness of fit on F2 | 1.09 | 1.13 | 1.08 | 0.97 | 1.02 | 1.04 |
| R1, wR2 [I ≥ 2σ(I)] | 0.068, 0.186 | 0.039, 0.127 | 0.036, 0.098 | 0.044, 0.113 | 0.047, 0.144 | 0.036, 0.086 |
| D–H···A | d(D–H) (Å) | d(H···A)(Å) | d(D···A)(Å) | Angle (D–H···A)(°) |
|---|---|---|---|---|
| 4d | ||||
| C3A—H3AA···O1D i | 0.93 | 2.45 | 3.319(6) | 156 |
| C3B—H3BA···O1C ii | 0.93 | 2.43 | 3.288(6) | 154 |
| C3C—H3CA···O1B iii | 0.93 | 2.42 | 3.277(6) | 154 |
| C3D—H3DA···O1A iv | 0.93 | 2.43 | 3.275(7) | 151 |
| 4e | 0.96 | 2.87 | 3.620(2) | 136 |
| C14-H14B··· Cg1 | ||||
| 4f | ||||
| C3-H3A···O1 v | 0.93 | 2.54 | 3.426(3) | 160 |
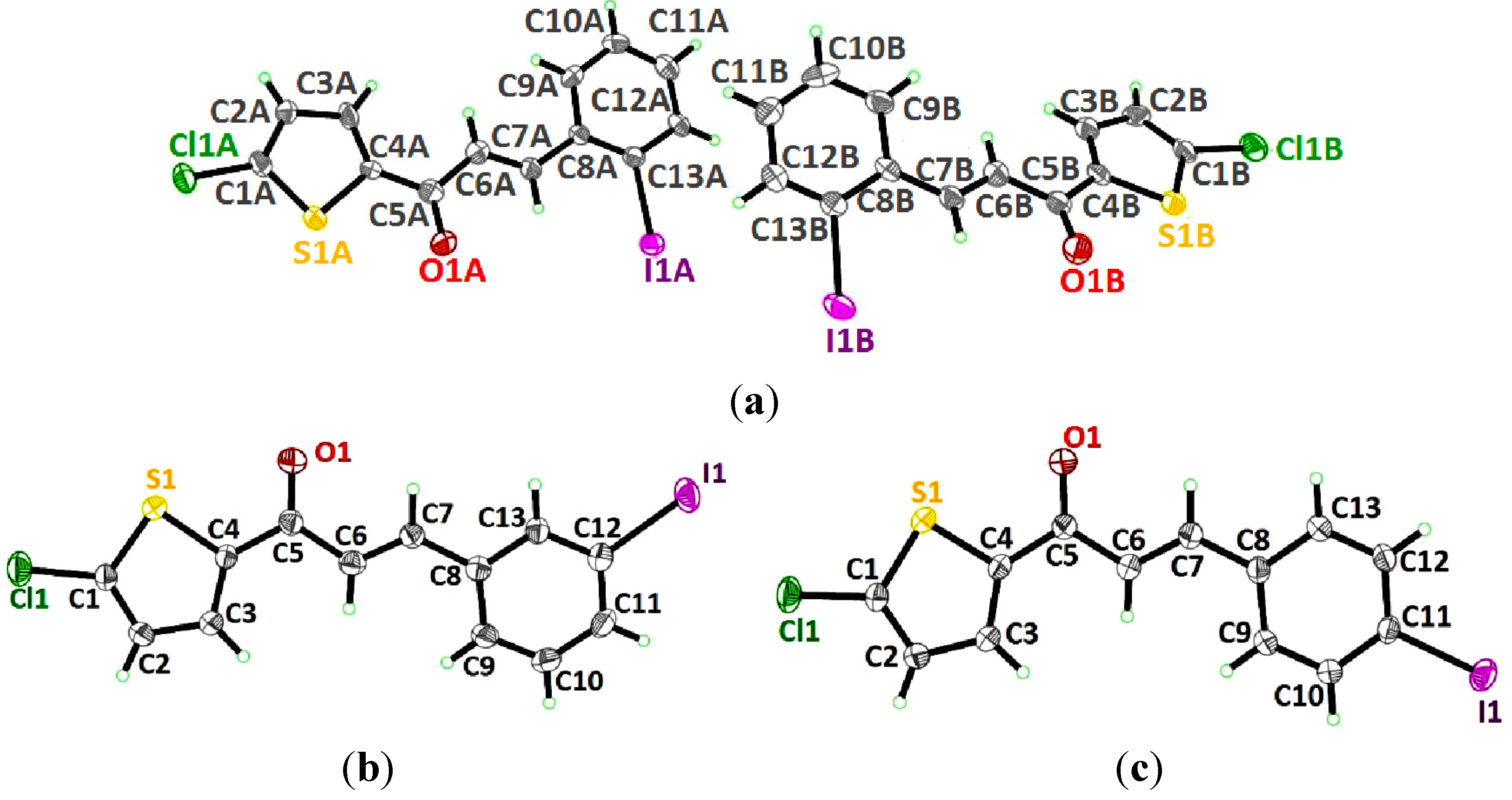
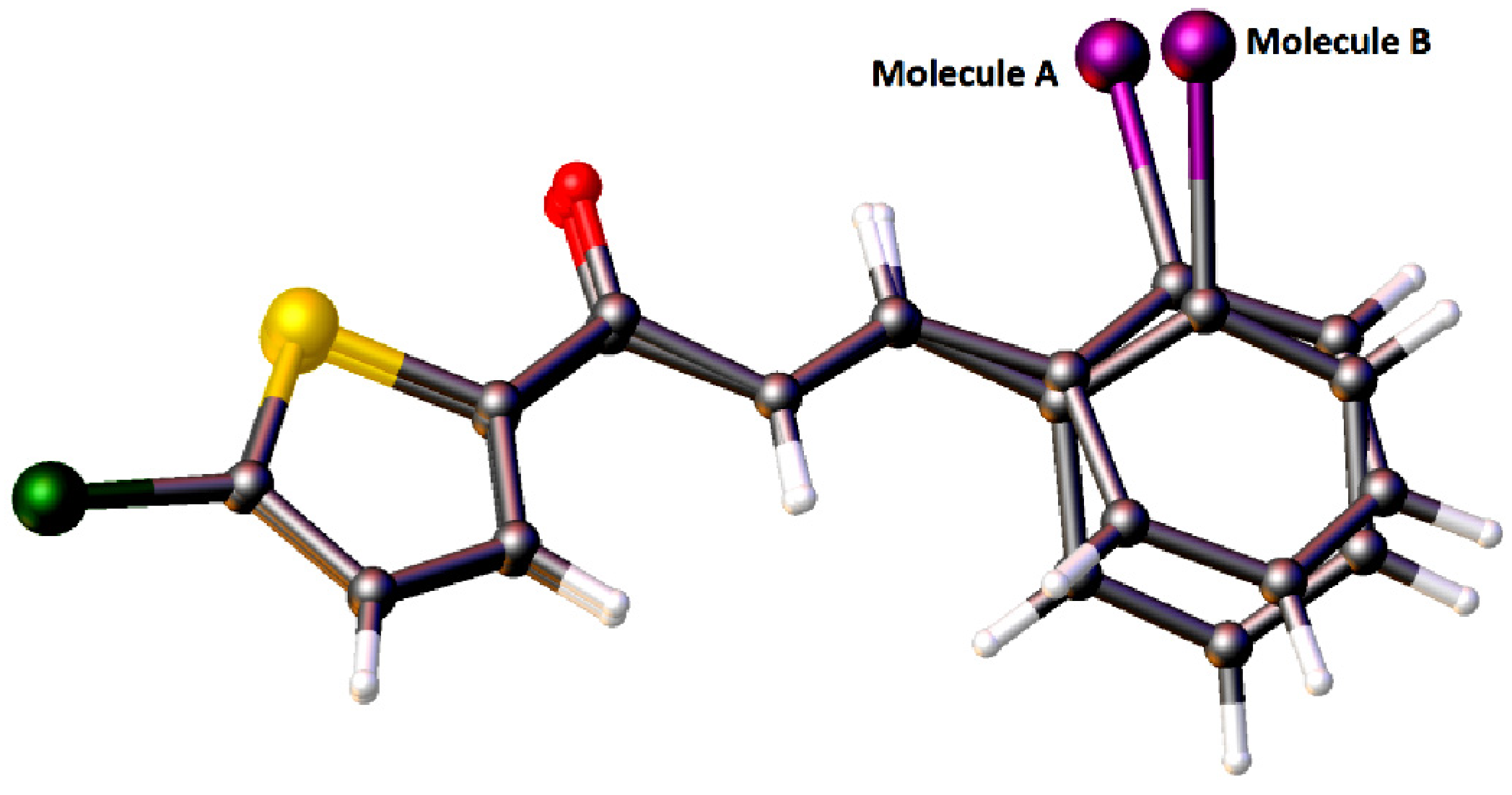
2.1. X-ray Crystal Structure Description for Compound 4a, 4b and 4c
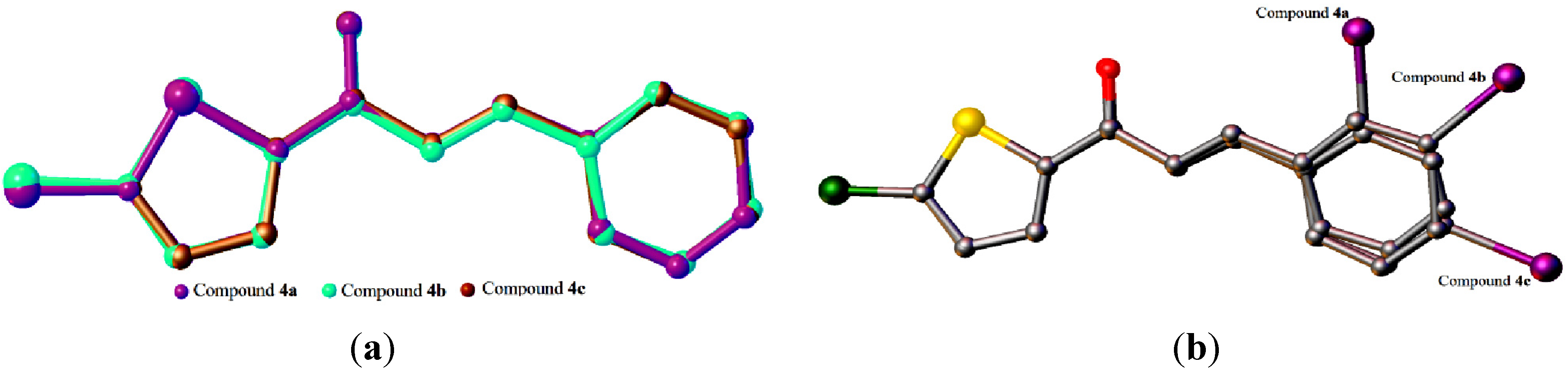

2.2. X-ray Crystal Structure Description for Compounds 4d, 4e and 4f


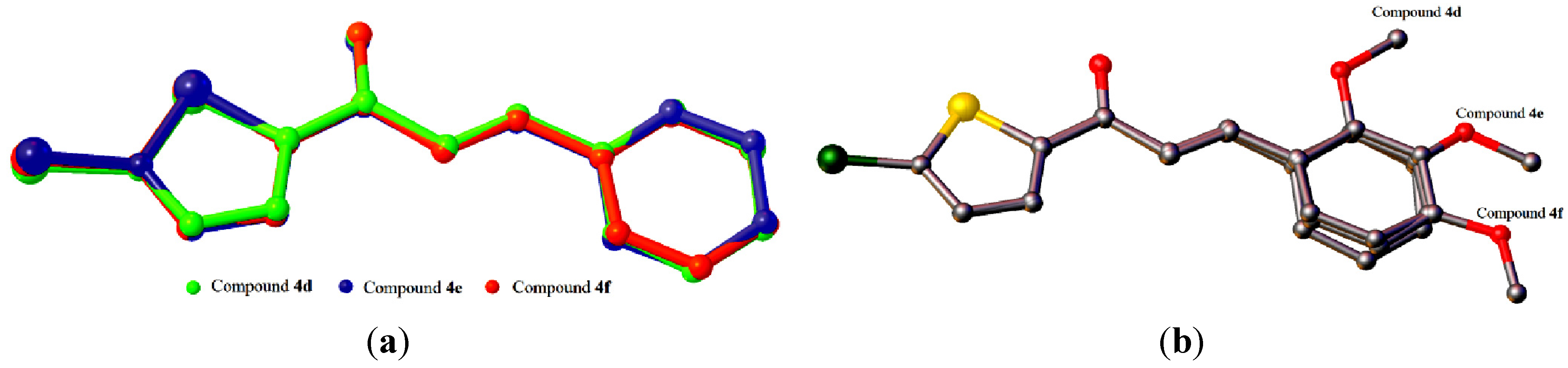
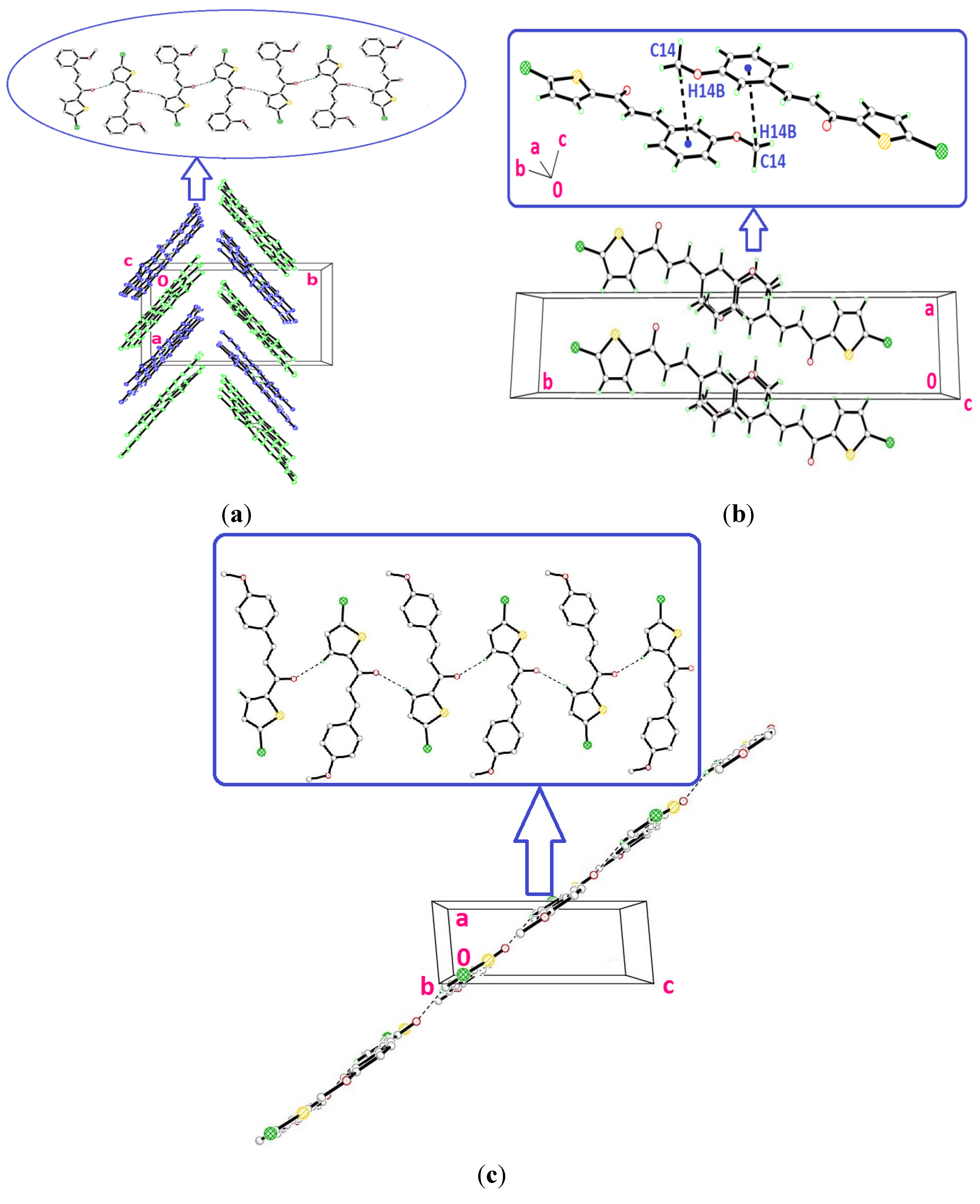
2.3. Antioxidant Activity
2.3.1. DPPH Radical Scavenging Assay
2.3.2. ABTS Radical Scavenging Assay

2.3.3. Ferric Reducing Antioxidant Power (FRAP) Assay
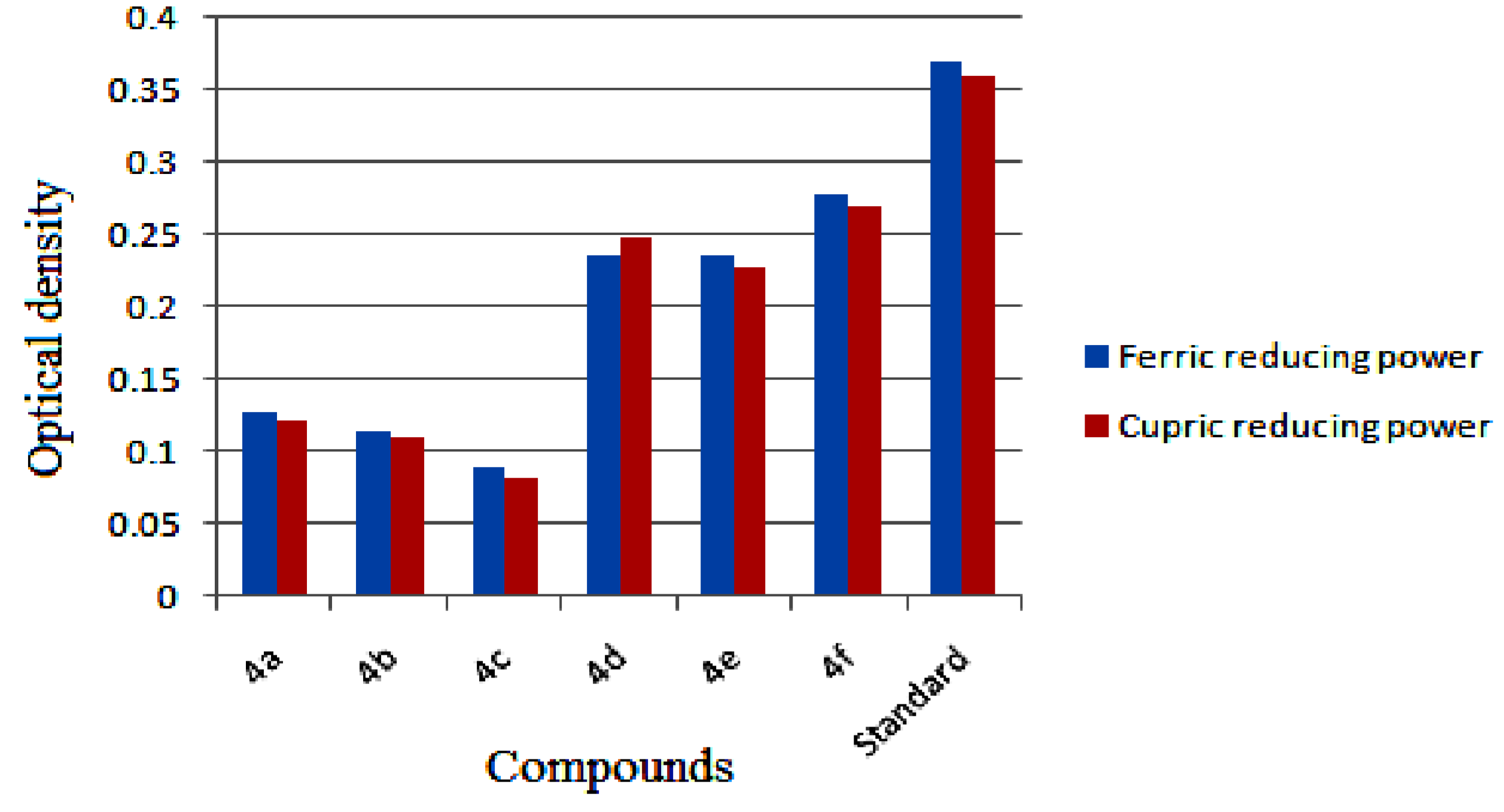
2.3.4. Cupric Ion Reducing Antioxidant Capacity (CUPRAC) Assay
3. Experimental
3.1. General Information
3.2. X-ray Crystallographic Analysis
3.3. General Procedure for the Synthesis of Chalcones 4(a–f)
3.4. In Vitro Antioxidant Activity
3.4.1. DPPH Radical Scavenging Assay
3.4.2. ABTS Radical Scavenging Assay
3.4.3. Ferric Reducing Antioxidant Power (FRAP) Assay
3.4.4. Cupric Ion Reducing Antioxidant Capacity (CUPRAC) Assay
3.4.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Alcaraz, L.E.; Blanco, S.E.; Puig, O.N.; Tomas, F.; Ferretti, F.H. Antibacterial activity of Flavonoids against methicillin- resistant Staphylococcus aureus strains. J. Theor. Biol. 2000, 205, 231–240. [Google Scholar] [CrossRef]
- Baviskar, B.A.; Baviskar, B.; Shiradkar, M.R.; Deokate, U.A.; Khadabadi, S.S. Synthesis and antimicrobial activity of some novel benzimidazolyl chalcones. J. Chem. 2009, 6, 196–200. [Google Scholar]
- Echeverria, C.; Santibañez, J.F.; Donoso-Tauda, O.; Escobar, C.A.; Ramirez-Tagle, R. Structural antitumoral activity relationships of synthetic chalcones. Int. J. Mol. Sci. 2009, 10, 221–231. [Google Scholar] [CrossRef]
- Modzelewska, A.; Pettit, C.; Achanta, G.; Davidson, N.E.; Huang, P.; Khan, S.R. Anticancer activities of novel chalcone and bis-chalcone derivatives. Bioorg. Med. Chem. 2006, 14, 3491–3495. [Google Scholar] [CrossRef]
- Vogel, S.; Heilmann, J. Synthesis, cytotoxicity, and antioxidative activity of minor prenylated chalcones from Humulus lupulus. J. Nat. Prod. 2008, 71, 1237–1241. [Google Scholar] [CrossRef]
- Babasaheb, P.B.; Sachin, A.P.; Rajesh, N.G. Synthesis and biological evaluation of nitrogencontaining chalcones as possible anti-inflammatory and antioxidant agents. Bioorg. Med. Chem. Lett. 2010, 20, 730–733. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, J.; Park, H.; Kim, H.P. Anti-inflammatory activity of the synthetic chalcone derivatives: Inhibition of inducible nitric oxide synthase-catalyzed nitric oxide production from lipopolysaccharide-treated RAW 264.7 cells. Biol. Pharm. Bull. 2007, 30, 1450–1455. [Google Scholar] [CrossRef]
- Ballesteros, J.F.; Sanz, M.J.; Ubeda, A.; Miranda, M.A.; Iborra, S.; Paya, M.; Alcaraz, M.J. Synthesis and pharmacological evaluation of 2’-hydroxychalcones and flavones as inhibitors of inflammatory mediators generation. J. Med. Chem. 1995, 38, 2794–2797. [Google Scholar] [CrossRef]
- Vogel, S.; Barbic, M.; Jürgenliemk, G.; Heilmann, J. Synthesis, cytotoxicity, anti-oxidative and anti-inflammatory activity of chalcones and influence of A-ring modifications on the pharmacological effect. Eur. J. Med. Chem. 2010, 45, 2206–2213. [Google Scholar] [CrossRef]
- Lopez, S.N.; Castelli, M.V.; Zacchino, S.A.; Domnguez, J.N.; Lobo, G.; Charris-Charris, J.; Cortes, J.C.G.; Ribas, J.C.; Devia, C.; Rodrguez, A.M.; et al. In vitro antifungal evaluation and structure–activity relationships of a new series of chalcone derivatives and synthetic analogues, with inhibitory properties against polymers of the fungal cell wall. Bioorg. Med. Chem. 2001, 9, 1999–2013. [Google Scholar] [CrossRef]
- Beom-Tae, K.; Kwang-Joong, O.; Jae-Chul, C.; Ki-Jun, H. Synthesis of dihydroxylated chalcone derivatives with diverse substitution patterns and their radical scavenging ability toward DPPH free radicals. Bull. Korean Chem. Soc. 2008, 29, 1125–1130. [Google Scholar] [CrossRef]
- Doan, T.N.; Tran, T.-D. Synthesis, antioxidant and antimicrobial activities of a novel series of chalcones, pyrazolic chalcones, and allylic chalcones. Pharmacol. Pharm. 2011, 2, 282–288. [Google Scholar] [CrossRef]
- Go, M.L.; Wu, X.; Liu, X.L. Chalcones: An update on cytotoxic and chemoprotective properties. Curr. Med. Chem. 2005, 12, 481–99. [Google Scholar]
- Sivakumar, P.M.; Prabhakar, P.K.; Doble, M. Synthesis, antioxidant evaluation, and quantitative structure–activity relationship studies of chalcones. Med. Chem. Res. 2011, 20, 482–492. [Google Scholar] [CrossRef]
- Vogel, S.; Ohmayer, S.; Brunner, G.; Heilmann, J. Natural and non-natural prenylated chalcones: Synthesis, cytotoxicity and anti-oxidative activity. Bioorg. Med. Chem. 2008, 16, 4286–4293. [Google Scholar] [CrossRef]
- Bruker APEX2, SAINT and SADABS. Bruker AXS Inc.: Madison, WI, USA, 2009.
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Lai, L.S.; Chou, S.T.; Chao, W.W. Studies on the antioxidant activities of Hsian-tsao (Mesona procumbens Hemsl) leaf gum. J. Agric. Food Chem. 2001, 49, 963–968. [Google Scholar] [CrossRef]
- Re, R.; Pellergini, N.; Proteggenete, A.; Pannala, A.; Yang, M.; Rice Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Apak, R.; Guclu, K.; Ozyurek, M.; Celik, S.E. Mechanism of antioxidant capacity assays and the CUPRAC (cupric ion reducing antioxidant capacity) assay. Microchim. Acta 2008, 160, 413–419. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kumar, C.S.C.; Loh, W.-S.; Ooi, C.W.; Quah, C.K.; Fun, H.-K. Structural Correlation of Some Heterocyclic Chalcone Analogues and Evaluation of Their Antioxidant Potential. Molecules 2013, 18, 11996-12011. https://doi.org/10.3390/molecules181011996
Kumar CSC, Loh W-S, Ooi CW, Quah CK, Fun H-K. Structural Correlation of Some Heterocyclic Chalcone Analogues and Evaluation of Their Antioxidant Potential. Molecules. 2013; 18(10):11996-12011. https://doi.org/10.3390/molecules181011996
Chicago/Turabian StyleKumar, C. S. Chidan, Wan-Sin Loh, Chin Wei Ooi, Ching Kheng Quah, and Hoong-Kun Fun. 2013. "Structural Correlation of Some Heterocyclic Chalcone Analogues and Evaluation of Their Antioxidant Potential" Molecules 18, no. 10: 11996-12011. https://doi.org/10.3390/molecules181011996
APA StyleKumar, C. S. C., Loh, W.-S., Ooi, C. W., Quah, C. K., & Fun, H.-K. (2013). Structural Correlation of Some Heterocyclic Chalcone Analogues and Evaluation of Their Antioxidant Potential. Molecules, 18(10), 11996-12011. https://doi.org/10.3390/molecules181011996




