Two New Monoterpenes from the Fruits of Illicium lanceolatum
Abstract
:1. Introduction

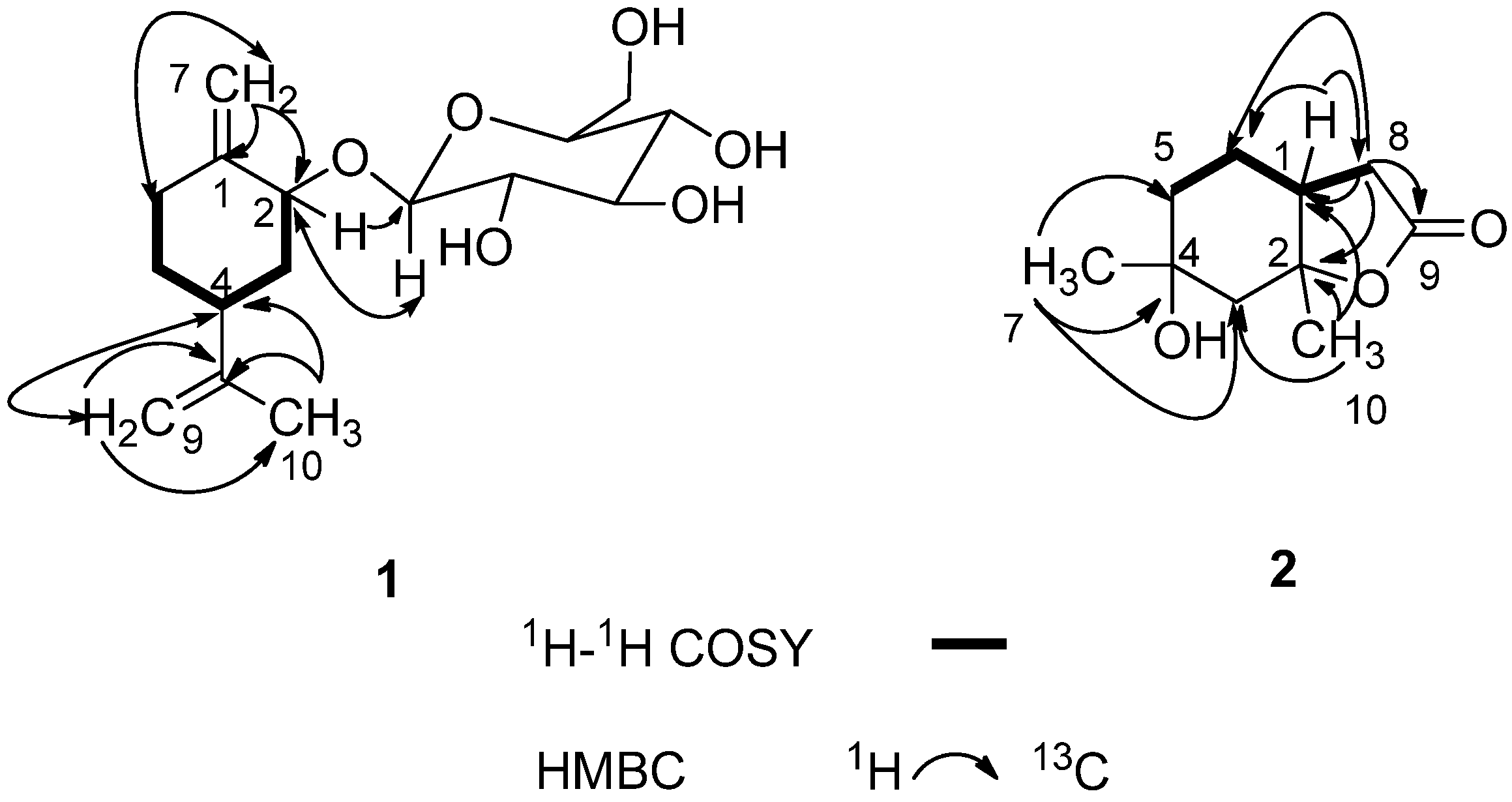
2. Results and Discussion
| Position | δC | δH | Position | δC | δH |
|---|---|---|---|---|---|
| 1 | 147.9 | - | 8 | 148.7 | - |
| 2 | 76.7 | 4.29 (m) | 9 | 108.1 | 4.70 (s) |
| 3 | 40.5 | 2.16–2.19 (m) | 4.69 (d, 1.8) | ||
| 1.29–1.39 (m) | 10 | 19.6 | 1.71 (s) | ||
| 4 | 44.1 | 2.24–2.28 (m) | 1' | 101.8 | 4.40 (d, 7.8) |
| 5 | 33.7 | 2.39–2.44 (m) | 2' | 74.1 | 3.23–3.28 (m) |
| 2.01–2.09 (m) | 3' | 77.5 | 3.27–3.36 (m) | ||
| 6 | 32.9 | 1.78–1.81 (m) | 4' | 70.3 | 3.26–3.34 (m) |
| 1.13–1.18 (m) | 5' | 76.5 | 3.22–3.27 (m) | ||
| 7 | 104.9 | 5.27 (s) | 6' | 61.4 | 3.87 (dd, 12.0, 2.0) |
| 4.77 (s) | 3.65 (dd, 11.9, 5.6) |

| Position | 2 | 3 | ||
|---|---|---|---|---|
| δC | δH | δC | δH | |
| 1 | 47.2 | 2.01–2.07 (dddd, 3.3, 7.8, 11.8, 15.2) | 39.8 | 2.36–2.41 (m) |
| 2 | 86.2 | - | 86.1 | - |
| 3 | 48.9 | 2.12 (d, 13.1.) | 46.3 | 1.89 (d, 14.5) |
| 1.80 (d, 13.1) | 1.73 (d, 14.5) | |||
| 4 | 71.3 | - | 69.5 | - |
| 5 | 40.4 | 1.81–1.86 (m) | 33.2 | 1.56–1.60 (m) |
| 1.53–1.60 (m) | 1.48–1.55 (m) | |||
| 6 | 21.3 | 1.70–1.74 (m) | 21.7 | 2.01–2.07 (m) |
| 1.60–1.67 (m) | 1.49–1.53 (m) | |||
| 7 | 33.4 | 1.32 (s) | 31.1 | 1.30 (s) |
| 8 | 33.3 | 2.43–2.49 (dd, 8.4, 16.4) | 34.0 | 2.53–2.60 (dd, 7.9, 16.8) |
| 2.37–2.42 (dd, 8.4, 16.5) | 2.40–2.48 (dd, 7.9, 16.8) | |||
| 9 | 176.6 | - | 176.3 | - |
| 10 | 19.5 | 1.48 (s) | 27.4 | 1.57 (s) |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
 +4.7 (c 0.102, MeOH); IR (KBr) vmax 3423, 2923, 1643, 1437, 1074, 1020 cm−1; 1H- and 13C-NMR spectral data, see Table 1; HRESIMS: m/z 337.1622 (calcd. for C16H26O6Na, 337.1627).
+4.7 (c 0.102, MeOH); IR (KBr) vmax 3423, 2923, 1643, 1437, 1074, 1020 cm−1; 1H- and 13C-NMR spectral data, see Table 1; HRESIMS: m/z 337.1622 (calcd. for C16H26O6Na, 337.1627). +133.3 (c 0.105, MeOH); IR (KBr) vmax 3446, 2972, 2940, 1746, 1378, 1268, 1105 cm−1; 1H- and 13C-NMR spectral data, see Table 2; HRESIMS: m/z 207.0999 (calcd. for C10H16O3Na, 207.0997).
+133.3 (c 0.105, MeOH); IR (KBr) vmax 3446, 2972, 2940, 1746, 1378, 1268, 1105 cm−1; 1H- and 13C-NMR spectral data, see Table 2; HRESIMS: m/z 207.0999 (calcd. for C10H16O3Na, 207.0997). +54.5 (c 0.101, MeOH); IR (KBr) vmax 3473, 2924, 2852, 1735, 1382, 1269, 1100 cm−1; 1H- and 13C-NMR spectral data, see Table 2; HRESIMS: m/z 207.0994 (calcd. for C10H16O3Na, 207.0997).
+54.5 (c 0.101, MeOH); IR (KBr) vmax 3473, 2924, 2852, 1735, 1382, 1269, 1100 cm−1; 1H- and 13C-NMR spectral data, see Table 2; HRESIMS: m/z 207.0994 (calcd. for C10H16O3Na, 207.0997).4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Liu, Y.N.; Su, X.H.; Huo, C.H.; Zhang, X.P.; Shi, Q.W.; Gu, Y.C. Chemical constituents of plants from the genus Illicium. Chem. Biodivers. 2009, 6, 963–989. [Google Scholar] [CrossRef]
- Yang, C.C.; Sun, J.N.; Huang, J.M. Study and Application of Chinese Materia Medica from Asarum and Illicium; People’s Medical Publishing House: Beijing, China, 2006. [Google Scholar]
- Liang, J.; Huang, B.K.; Wang, G.W. Chemical composition, antinociceptive and anti-inflammatory properties of essential oil from the roots of Illicium lanceolatum. Nat. Prod. Res. 2012, 26, 1712–1714. [Google Scholar] [CrossRef]
- Liang, J. Study on the Chemical Composition and Biological Activities of Illicium lanceolatum A. C. Smith; Fujian University of Traditional Chinese Medicine Press: Fuzhou, China, 2011; pp. 7–10. [Google Scholar]
- Wang, G.W. Chemical Constituents and Anti-inflammatory Activities of the Stems and Leaves of Illicium lanceolatum A.C. Smith; The Second Military Medical University Press: Shanghai, China, 2012; pp. 12–17. [Google Scholar]
- Wolinsky, J.; Chan, D. Carbonyl-olefin reactions. The cyclization of 3-isopropenyl-6-oxoheptanoic acid. J. Org. Chem. 1966, 31, 2471–2474. [Google Scholar] [CrossRef]
- Abraham, W.R.; Stumpf, B; Kieslich, K. Microbial transformations of terpenoids with 1-p-menthene skeleton. Appl. Microbiol. Biotechnol. 1986, 24, 24–30. [Google Scholar]
- Yoshikawa, M.; Morikawa, T.; Oominami, H.; Matsuda, H. Absolute stereostructures of olibanumols A, B, C, H, I, and J from olibanum, gum-resin of Boswellia. carterii, and inhibitors of nitric oxide production in lipopolysaccharide-activated mouse peritoneal macrophages. Chem. Pharm. Bull. 2009, 57, 957–964. [Google Scholar] [CrossRef]
- Kitajima, J.; Ishikawa, T.; Urabe, A.; Satoh, M. Monoterpenoids and their glycosides from the leaf of thyme. Phytochemistry 2004, 65, 3279–3287. [Google Scholar] [CrossRef]
- Matsumura, T.; Ishikawa, T.; Kitajima, J. New p-menthanetriols and their glucosides from the fruit of caraway. Tetrahedron 2001, 57, 8067–8074. [Google Scholar] [CrossRef]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Broch micro-dilution method for determining the susceptibility of Escherichia coli and Staphylococcus aureus to the essential oil of Melaleuca. altermifolia (tea tree oil). Microbios 1995, 82, 181–185. [Google Scholar]
- Sample Availability: Samples of the compounds 1–7 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, J.-F.; Li, H.-J.; Wang, L.-X.; Liu, M.-Q.; Bi, Y.-F.; Zhang, Y.-B. Two New Monoterpenes from the Fruits of Illicium lanceolatum. Molecules 2013, 18, 11866-11872. https://doi.org/10.3390/molecules181011866
Liu J-F, Li H-J, Wang L-X, Liu M-Q, Bi Y-F, Zhang Y-B. Two New Monoterpenes from the Fruits of Illicium lanceolatum. Molecules. 2013; 18(10):11866-11872. https://doi.org/10.3390/molecules181011866
Chicago/Turabian StyleLiu, Ji-Feng, Hui-Juan Li, Li-Xia Wang, Meng-Qi Liu, Yue-Feng Bi, and Yan-Bing Zhang. 2013. "Two New Monoterpenes from the Fruits of Illicium lanceolatum" Molecules 18, no. 10: 11866-11872. https://doi.org/10.3390/molecules181011866




