Improved Quality Control Method for Prescriptions of Polygonum capitatum through Simultaneous Determination of Nine Major Constituents by HPLC Coupled with Triple Quadruple Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of LC-MS/MS Conditions
2.2. Method Validation
2.2.1. Linearity and Detection Limit
| Compounds | Characterization of 11 markers | MRM parameters | ||||
|---|---|---|---|---|---|---|
| RT (min) | M-H/M (m/z) | Lost ions | Quantification transition (m/z) | Frag (V) | CE (V) | |
| Gallic acid | 0.774 | 169 | 125[M-COOH]-; 79[M-COOH-3OH+3H]- | 169→125 | 80 | 10 |
| Protocatechuic acid | 0.793 | 153 | 109[M-COOH-H]-; 91[M-COOH-H20]- | 153→109 | 80 | 10 |
| Vanillic acid | 0.837 | 167 | 152[M-CH3]-; 123[M-COOH]- | 167→152 | 80 | 10 |
| Syringic acid | 0.846 | 197 | 182[M-CH3]-; 153[M-COOH]- | 197→182 | 80 | 20 |
| Catechin | 6.184 | 289 | 245[M-CH3CHO]-; 109[M-2OH-C6H5-CH3CHO-CH3]- | 289→109 | 150 | 20 |
| Schizandriside | 7.634 | 491 | 359[M-xylose]-; 344[M-xylose-CH3]- | 491→359 | 200 | 20 |
| Quercitrin | 8.129 | 447 | 301[M-rhamnose]-; 255[M-rhamnose-COOH]- | 447→301 | 180 | 20 |
| Quercetin | 8.677 | 301 | 179[M-2OH-C6H4-CH2]-; 151[M-2OH-C6H4-CH3CHO] | 301→151 | 150 | 15 |
| Kaempferol | 9.046 | 285 | 117[M-2OH-C6H6-CH2O-2H2O]-; 93[C6H5+OH] | 285→ 93 | 150 | 25 |
| Internal standard1 | 3.021 | 151 | 107[M-COOH]-; 92[M-COOH-CH3]- | 151→107 | 80 | 5 |
| Internal standard2 | 7.749 | 579 | 459[M-glucose+CH3COOH]-; 271[M-glucose-rhamnose]- | 579→271 | 200 | 30 |
| Compounds | Calibration curves | Linear ranges (ng·mL−1) | Correlation coefficient | LOD (ng·mL−1) | LOQ (ng·mL−1) |
|---|---|---|---|---|---|
| Gallic acid | Y = 0.7624X − 1.8747 | 100–10,000 | 0.9937 | 5.0 | 10 |
| Protocatechuic acid | Y = 0.9881X − 0.0169 | 5–500 | 0.9974 | 1.0 | 5.0 |
| Vanillic acid | Y = 0.6889X − 0.6085 | 100–10,000 | 0.9982 | 1.0 | 5.0 |
| Syringic acid | Y = 11.5119X + 68.0960 | 5–1,000 | 0.9992 | 1.0 | 5.0 |
| Catechin | Y = 72.2795X + 637.9497 | 5–1,000 | 0.9965 | 1.0 | 5.0 |
| Schizandriside | Y = 0.0019X − 0.0032 | 50–10,000 | 0.9971 | 5.0 | 10 |
| Quercitrin | Y = 9.6795X − 0.8127 | 5–10,000 | 0.9989 | 1.0 | 5.0 |
| Quercetin | Y = 0.7283X − 0.0865 | 50–10,000 | 0.9991 | 1.0 | 5.0 |
| Kaempferol | Y = 0.3322X − 0.0300 | 10–5,000 | 0.9987 | 1.0 | 5.0 |
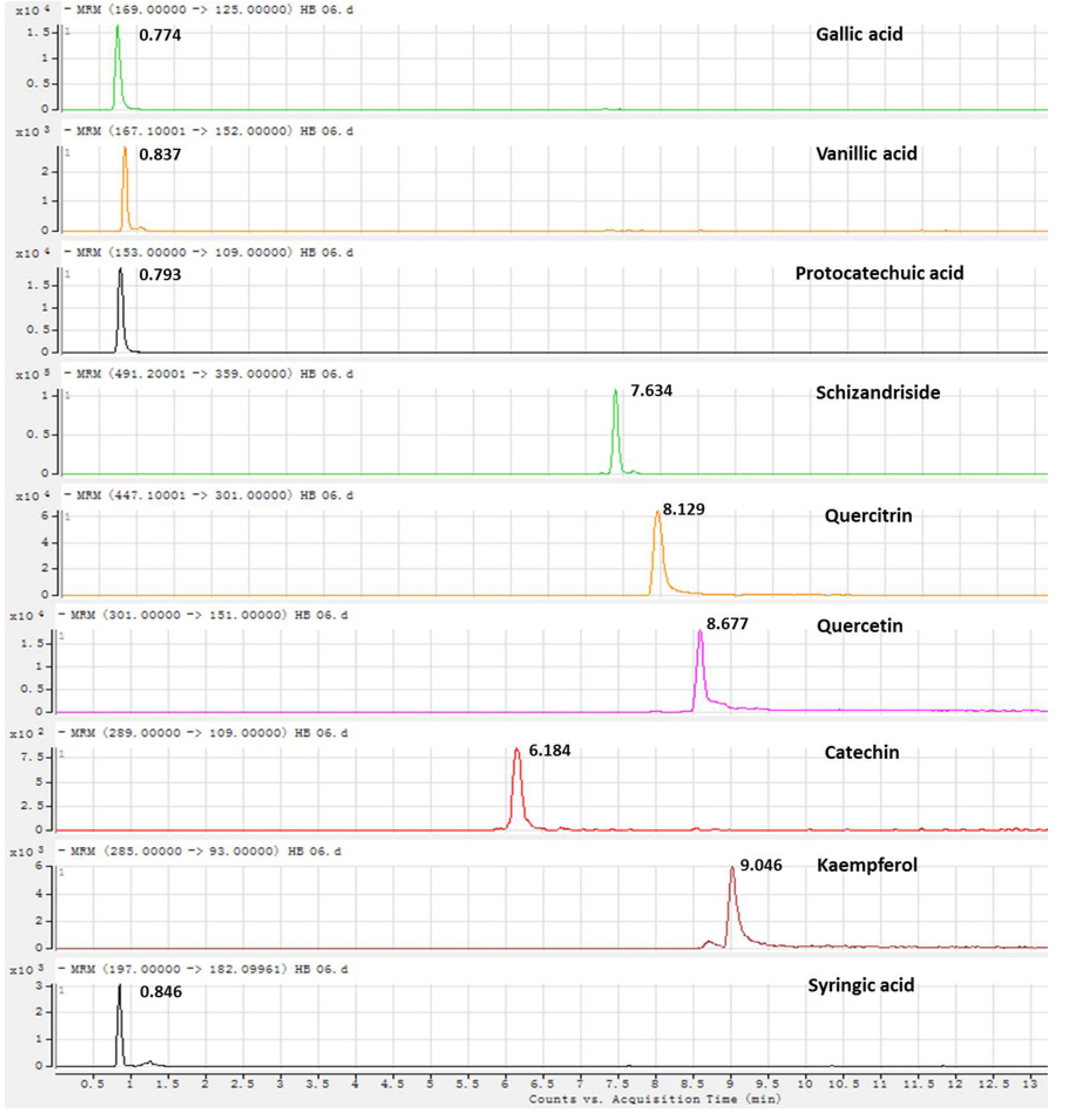
2.2.2. Precision, Repeatability and Stability
2.2.3. Recovery
| Compounds | Precision (RSD, n = 6) % | Repeatability (RSD, n = 6) % | Stability (RSD, n = 6) % | |||
|---|---|---|---|---|---|---|
| Intra-day | Inter-day | G1 C1 T1 | ||||
| Gallic acid | 1.68 | 2.11 | 2.75 | 2.27 | 2.35 | 3.16 |
| Protocatechuic acid | 1.59 | 2.35 | 2.17 | 2.38 | 2.67 | 2.78 |
| Vanillic acid | 2.36 | 2.47 | 3.03 | 2.79 | 2.08 | 2.91 |
| Syringic acid | 2.07 | 2.58 | 2.69 | 2.67 | 3.24 | 3.56 |
| Catechin | 1.73 | 2.72 | 2.78 | 3.29 | 2.49 | 2.44 |
| Schizandriside | 2.32 | 2.78 | 3.47 | 2.88 | 2.37 | 3.72 |
| Quercitrin | 1.25 | 1.47 | 2.05 | 1.84 | 2.16 | 2.38 |
| Quercetin | 1.54 | 1.87 | 2.19 | 2.17 | 3.24 | 2.27 |
| Kaempferol | 1.68 | 2.31 | 2.57 | 2.74 | 2.35 | 3.04 |
| Compounds | Granules | Capsule | Tablet | |||
|---|---|---|---|---|---|---|
| Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | |
| Gallic acid | 98.61% | 2.18 | 99.50% | 3.03 | 101.38% | 2.13 |
| 100.65% | 2.33 | 96.91% | 2.45 | 99.76% | 2.65 | |
| 95.68% | 2.46 | 101.05% | 2.19 | 101.33% | 2.29 | |
| Protocatechuic acid | 97.34% | 2.98 | 96.57% | 3.17 | 96.57% | 3.15 |
| 98.29% | 3.10 | 101.47% | 2.27 | 102.25% | 3.27 | |
| 102.51% | 2.78 | 95.00% | 3.66 | 97.70% | 3.26 | |
| Vanillic acid | 98.91% | 2.59 | 104.50% | 2.49 | 101.38% | 3.55 |
| 101.70% | 2.84 | 97.70% | 2.85 | 98.05% | 2.64 | |
| 103.12% | 3.14 | 108.54% | 3.14 | 103.82% | 2.74 | |
| Syringic acid | 102.07% | 3.19 | 102.72% | 2.19 | 101.84% | 2.15 |
| 102.43% | 3.28 | 98.13% | 3.28 | 97.40% | 3.28 | |
| 95.58% | 2.97 | 95.29% | 2.87 | 95.14% | 2.97 | |
| Catechin | 98.68% | 2.46 | 103.88% | 3.26 | 101.85% | 2.46 |
| 107.80% | 2.79 | 95.79% | 2.89 | 102.40% | 2.79 | |
| 98.23% | 3.13 | 95.93% | 3.13 | 97.63% | 3.03 | |
| Schizandriside | 101.36% | 3.28 | 101.98% | 3.08 | 101.20% | 3.48 |
| 101.53% | 3.24 | 95.01% | 3.24 | 100.57% | 2.14 | |
| 98.30% | 2.85 | 97.94% | 2.85 | 97.59% | 2.15 | |
| Quercitrin | 100.97% | 3.77 | 101.22% | 3.27 | 101.83% | 3.67 |
| 102.73% | 3.54 | 101.53% | 3.54 | 104.04% | 3.34 | |
| 99.53% | 3.42 | 98.27% | 3.62 | 98.27% | 2.62 | |
| Quercetin | 97.13% | 2.79 | 95.26% | 2.79 | 101.89% | 2.79 |
| 104.62% | 2.57 | 95.81% | 2.57 | 97.74% | 2.57 | |
| 97.62% | 2.84 | 104.03% | 2.84 | 96.30% | 2.53 | |
| Kaempferol | 93.60% | 2.13 | 95.25% | 2.23 | 97.15% | 2.13 |
| 105.60% | 2.15 | 104.64% | 2.15 | 102.32% | 2.42 | |
| 95.07% | 3.08 | 96.03% | 2.38 | 97.43% | 3.18 | |
2.3. Sample Determination
| Samples | Contents of each compound in different relinqing samples | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | |
| G1 | 21,751.04 | 568.31 | 463.72 | 68.92 | 56.31 | 852.04 | 699.64 | 84.32 | 46.95 |
| G2 | 19,667.32 | 587.64 | 457.58 | 70.25 | 58.67 | 657.38 | 687.81 | 80.77 | 45.57 |
| G3 | 20,996.64 | 495.94 | 471.46 | 69.67 | 53.71 | 789.68 | 696.79 | 93.94 | 44.82 |
| G4 | 19,589.24 | 488.73 | 482.88 | 75.39 | 59.86 | 856.41 | 663.84 | 95.63 | 46,78 |
| 20,501.06 | 535.15 | 468.91 | 71.05 | 57.14 | 788.87 | 687.02 | 88.66 | 45.78 | |
| RSD | 5.14% | 9.37% | 2.32% | 4.13% | 4.76% | 11.76% | 2.36% | 8.17% | 2.36% |
| C1 | 17,855.36 | 359.67 | 134.37 | 58.98 | 138.68 | 679.78 | 609.64 | 185.37 | 47.81 |
| C2 | 17,015.81 | 468,95 | 147.63 | 72.74 | 156.42 | 516.89 | 626.79 | 168,98 | 46.03 |
| C3 | 19,389.43 | 443.64 | 159.83 | 65.81 | 140.17 | 591.57 | 614.46 | 174.46 | 44.42 |
| C4 | 18,544.16 | 369,97 | 138.52 | 73.57 | 163.59 | 714,62 | 621.69 | 151.63 | 47.83 |
| C | 18,201.19 | 401.65 | 145.08 | 67.77 | 149.71 | 596.08 | 618.14 | 170.48 | 46.52 |
| RSD | 5.54% | 14.78% | 7.76% | 10.05% | 8.18% | 13.67% | 1.23% | 10.09% | 3.51% |
| T1 | 9,439.76 | 277.79 | 426.14 | 59.42 | 51.37 | 839.17 | 403.48 | 220.17 | 48.96 |
| T2 | 12,920.56 | 321.46 | 437.27 | 66.89 | 59.47 | 765.04 | 409.79 | 160.79 | 42.37 |
| T3 | 15,145.27 | 258.42 | 419.49 | 72.47 | 62.89 | 323.66 | 302.42 | 345.53 | 54.89 |
| T4 | 11,075.44 | 306.54 | 403.72 | 58.81 | 60.34 | 691.74 | 423.54 | 193.41 | 46.54 |
| T | 12,145.26 | 291.05 | 421.65 | 64.39 | 58.51 | 654.9 | 564.50 | 229.97 | 48.19 |
| RSD | 20.21% | 9.72% | 3.32% | 10.11% | 8.51% | 34.94% | 14.43% | 35.12% | 10.85% |
| total | 16,919.17 | 409.28 | 345.21 | 67.73 | 88.45 | 679.95 | 395.07 | 163.03 | 46.83 |
2.4. Discussion


3. Experimental
3.1. Materials and Reagents
3.2. Preparation of Sample Solutions
3.3. Preparation of Standard Solutions
3.4. Chromatographic and Mass Spectrometric Conditions
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Li, S.P.; Zhao, J.; Yang, B. Strategies for quality control of Chinese medicines. J. Pharm. Biomed. Anal. 2011, 55, 802–809. [Google Scholar] [CrossRef]
- Liang, X.M.; Jin, Y.; Wang, Y.P.; Jin, G.W.; Fu, Q.; Xiao, Y.S. Qualitative and quantitative analysis in quality control of traditional Chinese medicines. J. Chromatogr. A 2009, 1216, 2033–2044. [Google Scholar] [CrossRef]
- Shekarchi, M.; Hajimehdipoor, H.; Saeidnia, S.; Gohari, A.R. Comparative study of rosmarinic acid content in some plants of Labiatae family. Pharmacogn. Mag. 2012, 8, 37–41. [Google Scholar] [CrossRef]
- Jiang, Y.; David, B.; Tu, P.F.; Barbin, Y. Recent analytical approaches in quality control of traditional Chinese medicines—A review. Anal. Chim. Acta 2010, 657, 9–18. [Google Scholar] [CrossRef]
- Liang, X.; Zhang, L.; Zhang, X.; Dai, W.X.; Li, H.Y.; Hu, L.W.; Liu, H.; Su, J.; Zhang, W.D. Qualitative and quantitative analysis of traditional Chinese medicine Niu Huang Jie Du Pill using ultra performance liquid chromatography coupled with tunable UV detector and rapid resolution liquid chromatography coupled with time-of-flight tandem mass spectrometry. J. Pharm. Biomed. Anal. 2010, 51, 565–571. [Google Scholar] [CrossRef]
- Hu, F.L.; Deng, C.H.; Liu, Y.; Zhang, X.M. Quantitative determination of chlorogenic acid in Honeysuckle using microwave-assisted extraction followed by nano-LC-ESI mass spectrometry. Talanta 2009, 77, 1299–1303. [Google Scholar] [CrossRef]
- Song, L.R.; Ding, X.L.; Zang, Z.Y.; Hong, X. Dictionary of Modern TCD, 1st ed.; People’s Medical Publishing House: Beijing, China, 2001; pp. 580–581. [Google Scholar]
- Jiangsu New Medicinal College. Dictionary of Chinese Herbal Medicine; Shanghai People’s Publishing House: Shanghai, China, 1977; p. 611. [Google Scholar]
- Ren, G.Y.; Chang, F.G.; Lu, S.L.; Zhong, H.L.; Zhang, G.L. Pharmacological studies of Polygonum capitatum Buch-Ham ex D Don. (in Chinese). Zhongguo Zhong Yao Za Zhi 1995, 2, 107–109. [Google Scholar]
- Yan, X.L.; Li, C.Q.; Liu, Y.X.; Chang, X.; Kang, W.Y. Antioxidant activity of Polygonum capitatum. China Pharmacy 2010, 21, 3659–3661. [Google Scholar]
- Li, Y.M.; Gong, Y. The research progress on the chemical component and the pharmacology of Polygotum capitatum Ham ex D. Don. (in Chinese). J. Guizhou Univ. (Nat. Sci.) 2007, 24, 205–207. [Google Scholar]
- Liu, Z.J.; Qi, J.; Zhu, D.N.; Yu, B.Y. Chemical constituents from Polygonum capitatum and their antioxidation activities in vitro. (in Chinese). Zhong Yao Cai 2008, 31, 995–998. [Google Scholar]
- Yang, Y.; Cai, F.; Yang, Q.; Yang, Y.B.; Sun, L.N.; Chen, W.S. Study on chemical constituents of Polygonum capitatum Buch.-Ham. ex D. Don(I). (in Chinese). Acad. J. Second Military Med. Univ. 2009, 30, 937–940. [Google Scholar]
- Zhao, H.X.; Bai, H.; Li, W.; Wang, Y.S.; Liu, Y.J.; Liu, A.Q. Chemical constituents from Polygonum capitatum. Nat. Prod. Res. Dev. 2011, 23, 262–266. [Google Scholar]
- Yu, M.; Li, Z.L.; Li, N.; Li, X. Chemical constituents of the aerial parts of Polygonum capitatum. (in Chinese). J. Shenyang Pharmaceut. Univ. 2008, 25, 633–635. [Google Scholar]
- Liao, S.G.; Zhang, L.J.; Sun, F.; Zhang, J.J.; Chen, A.Y.; Lan, Y.Y.; Li, Y.J.; Wang, A.M.; He, X.; Xiong, Y.; et al. Antibacterial and anti-inflammatory effects of extracts and fractions from Polygonum capitatum. J. Ethnopharmacol. 2011, 134, 1006–1009. [Google Scholar] [CrossRef]
- Watson, D.G.; Oliveira, E.J. Solid-phase extraction and gas chromatography—Mass spectrometry determination of kaempferol and quercetin in human urine after consumption of Ginkgo biloba tablets. J. Chromatogr. B 1999, 723, 203–210. [Google Scholar] [CrossRef]
- Boue, S.M.; Carter-Weintjies, C.H.; Shih, B.Y.; Cleveland, T.E. Identification of flavone aglycones and glycosides in soybean pods by liquid chromatography—Tandem mass spectrometry. J. Chromatogr. A 2003, 991, 61–68. [Google Scholar] [CrossRef]
- Zhao, H.X.; Bai, H.; Li, W.; Wang, Y.S. Study on lignans of Polygonum capitatum. (in Chinese). Zhong Yao Cai 2010, 33, 1409–1411. [Google Scholar]
- Li, Y.S.; Wang, X.P.; Wan, D.G.; Wu, H.M. The thinking of study and development of series of Huoxiang Zhengqi preparations. (in Chinese). Pharmacy Clin. Chin. Materia Medica 2011, 2, 24–26. [Google Scholar]
- Xie, Y.; Zhang, L.Y.; Liang, B.; Li, M.L.; Tang, J.W. Determination of quercitrin in Polygonum capitatum and Relinqing granules by HPLC. (in Chinese). China J. Chin. Materia Medica 2009, 34, 984–986. [Google Scholar]
- Yang, B.B.; Feng, R.; Wang, W.C.; Zhang, L.Y.; Ye, X.M.; Wang, Y.; Wang, M.Z. Quantitative analysis of three active constituents in Miao regional herb Polygonum capitatum by HPLC/DAD/MS. (in Chinese). Chin. J. Pharm. Anal. 2008, 28, 1793–1796. [Google Scholar]
- Steinmann, D.; Ganzera, M. Recent advances on HPLC/MS in medicinal plant analysis. J. Pharm. Biomed. Anal. 2011, 55, 744–757. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.; Chen, X.; Chen, C.; Wang, H.; Meng, F.; Yang, H.; Huang, L. Simultaneous quantification of 17 constituents from Yuanhu Zhitong tablet using rapid resolution liquid chromatography coupled with a triple quadrupole electrospray tandem mass spectrometry. J. Pharm. Biomed. Anal. 2011, 56, 497–504. [Google Scholar] [CrossRef]
- Peng, J.B.; Jia, H.M.; Liu, Y.T.; Zhang, H.W.; Dong, S.; Zou, Z.M. Qualitative and quantitative characterization of chemical constituents in Xin-Ke-Shu preparations by liquid chromatography coupled with a LTQ Orbitrap mass spectrometer. J. Pharm. Biomed. Anal. 2011, 55, 984–995. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds—(1) gallic acid; (2) protocatechuic acid; (3) vanillic acid; (4) syringic acid; (5) catechin; (6) schizandriside; (7) quercitrin; (8) quercetin; (9) kaempferol—are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, K.-X.; Wang, Y.-S.; Jing, W.-G.; Zhang, J.; Liu, A. Improved Quality Control Method for Prescriptions of Polygonum capitatum through Simultaneous Determination of Nine Major Constituents by HPLC Coupled with Triple Quadruple Mass Spectrometry. Molecules 2013, 18, 11824-11835. https://doi.org/10.3390/molecules181011824
Zhang K-X, Wang Y-S, Jing W-G, Zhang J, Liu A. Improved Quality Control Method for Prescriptions of Polygonum capitatum through Simultaneous Determination of Nine Major Constituents by HPLC Coupled with Triple Quadruple Mass Spectrometry. Molecules. 2013; 18(10):11824-11835. https://doi.org/10.3390/molecules181011824
Chicago/Turabian StyleZhang, Kai-Xia, Yue-Sheng Wang, Wen-Guang Jing, Jun Zhang, and An Liu. 2013. "Improved Quality Control Method for Prescriptions of Polygonum capitatum through Simultaneous Determination of Nine Major Constituents by HPLC Coupled with Triple Quadruple Mass Spectrometry" Molecules 18, no. 10: 11824-11835. https://doi.org/10.3390/molecules181011824




