Anti-Platelet Aggregation and Vasorelaxing Effects of the Constituents of the Rhizomes of Zingiber officinale
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Isolated Compounds
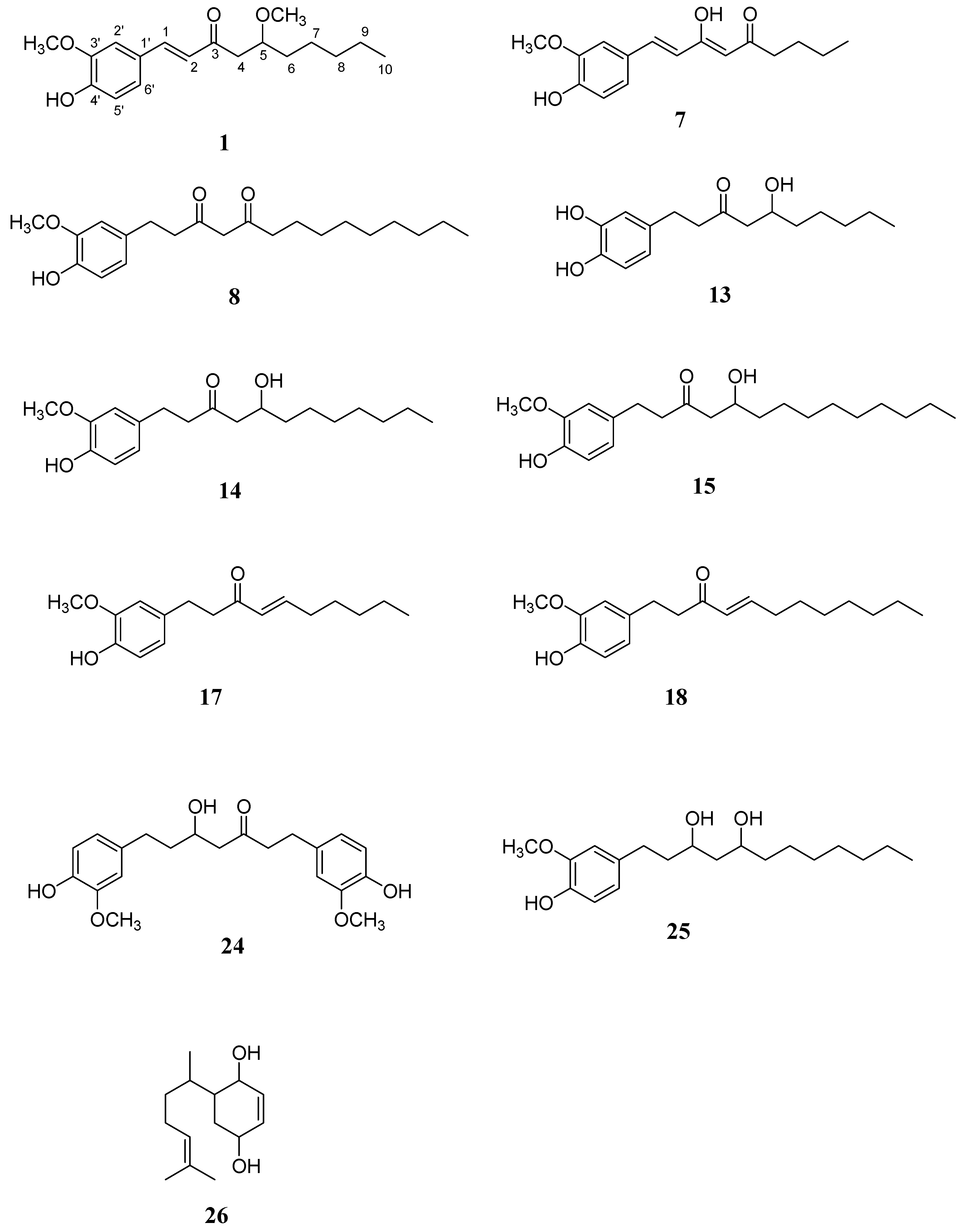
2.2. Anti-Platelet Aggregation Evaluation Bioassay
2.3. Vasorelaxing Effect Evaluation Bioassay
| Aggregation (%) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Inducer | Control | Conc. (μg/mL) | 7 | 8 | 13 | 14 | 15 | 17 | 18 | 24 | 25 | 26 |
| AA | 85.9 ± 0.6 | 0.1 | - | - | 83.3 ± 2.0 | - | - | - | ||||
| (100 μM) | 0.2 | 85.6 ± 0.2 | - | 33.7 ± 19.1 † | 80.6 ± 3.6 | 80.9 ± 1.7 † | - | - | ||||
| 0.5 | 17.6 ± 12.5 ‡ | 82.9 ± 1.4 | 16.3 ± 10.1 ‡ | 82.1 ± 7.3 | 79.8 ± 3.8 † | 16.6 ± 14.4 ‡ | 1.4 ± 1.2 ‡ | - | 85.6 ± 1.7 | |||
| 1.0 | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0‡ | 18.1 ± 3.8 ‡ | 2.7 ± 2.3 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | - | 44.4 ± 21.1 * | |||
| 2.0 | - | - | - | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | - | 86.1 ± 0.7 | 34.9 ± 18.3 ‡ | ||||
| 5.0 | - | - | - | - | 41.5 ± 20.8 * | 0.0 ± 0.0 ‡ | ||||||
| 10.0 | - | - | - | - | 27.9±16.4 ‡ | - | ||||||
| 20.0 | - | - | - | - | 0.0 ± 0.0 ‡ | - | ||||||
| 50.0 | - | - | - | - | - | - | 84.8 ± 0.5 | |||||
| 100.0 | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 32.1 ± 16.7 † | ||
| Col | 87.1 ± 1.0 | 0.5 | 83.3 ± 0.7 * | - | - | |||||||
| (10 μg/mL) | 1.0 | - | - | 84.4 ± 2.9 | 71.9 ± 4.7 ‡ | - | - | |||||
| 2.0 | 78.5 ± 2.7 | - | 69.6 ± 9.9 † | 84.2 ± 3.7 | 30.6 ± 10.8 ‡ | 82.5 ± 4.0 | ||||||
| 5.0 | 37.4 ± 15.6 ‡ | 86.3 ± 1.3 | 40.6 ± 17.9 ‡ | 76.8 ± 4.8 † | 77.7 ± 1.9 ‡ | 0.0 ± 0.0 ‡ | 25.4 ± 14.0 ‡ | |||||
| 10.0 | 4.6 ± 4.0 ‡ | 54.8 ± 18.0 * | 12.6 ± 10.3 ‡ | 29.0 ± 16.9 ‡ | 20.6 ± 10.6 ‡ | 0.0 ± 0.0 ‡ | 80.1 ± 5.1 | |||||
| 20.0 | 0.0 ± 0.0 ‡ | 4.6 ± 4.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 77.6 ± 4.0 † | 54.7 ± 15.6 * | |||||
| 50.0 | - | 0.0 ± 0.0 ‡ | - | 33.0 ± 17.2 ‡ | 47.2 ± 13.7 ‡ | |||||||
| 100.0 | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 0.0 ± 0.0 ‡ | 3.8 ± 3.3 ‡ | 15.5 ± 8.9 ‡ | 89.5 ± 1.8 | ||
| PAF | 88.6 ± 1.8 | 20.0 | 84.3 ± 3.6 | |||||||||
| (2 nM) | 50.0 | 83.9 ± 1.0 | 83.0 ± 1.0 * | 83.2 ± 1.4 | 83.4 ± 1.6 | 59.9 ± 8.5 † | 84.9 ± 4.0 | |||||
| 100.0 | 55.8 ± 4.7 ‡ | 55.3 ± 11.6 † | 24.4 ± 8.6 ‡ | 10.5 ± 8.6 ‡ | 0.0 ± 0.0 ‡ | 55.8 ± 9.2 † | 84.5 ± 2.4 | 84.9 ± 1.5 | 85.6 ± 1.3 | |||
| Thr (0.1 μg/mL) | 92.3 ± 1.0 | 100.0 | 87.4 ± 1.1 | 84.5 ± 0.6 ‡ | 85.0 ± 0.5 ‡ | 84.3 ± 2.5 * | 70.5 ± 2.7 ‡ | 80.2 ± 0.9 ‡ | 85.1 ± 4.7 | 88.5 ± 1.1 * | 88.2 ± 3.6 | |
| Compound | Conc. (μM) | NE (3 μM)contraction (%) | K+ (80 mM) + Ca2+ (1.9 mM)contraction (%) | |
|---|---|---|---|---|
| Phasic | Tonic | Tonic | ||
| Control | 100.0 ± 5.1 | 100.0 ± 4.1 | 100.0 ± 4.9 | |
| 7 | 362 | 86.6 ± 1.6 | 84.7 ± 1.5 | 75.4 ± 3.2 |
| 8 | 287.4 | 100.0 ± 0.0 | 85.3 ± 2.8 | 46.9 ± 4.2 |
| 86.2 | – | – | 72.3 ± 3.7 | |
| 13 | 357.1 | 49.2 ± 2.3 | 40.3 ± 3.8 | 13.4 ± 1.1 |
| 107.1 | 88.7 ± 1.9 | 93.3 ± 0.0 | 26.4 ± 2.4 | |
| 35.7 | – | – | 96.7 ± 1.9 | |
| 14 | 310.6 | 63.3 ± 3.1 | 48.1 ± 3.9 | 12.5 ± 0.0 |
| 93.2 | 89.9 ± 9.2 | 73.2 ± 2.8 | 20.9 ± 2.1 | |
| 31.1 | – | – | 83.2 ± 4.9 | |
| 15 | 285.7 | 104.2 ± 1.9 | 65.1 ± 7.2 | 23.8 ± 3.1 |
| 85.7 | – | – | 48.1 ± 0.9 | |
| 28.6 | – | – | 68.6 ± 4.9 | |
| 17 | 362.3 | 60.0 ± 14.1 | 36.7 ± 2.4 | 11.0 ± 0.4 |
| 108.7 | 90.3 ± 0.9 | 89.9 ± 1.7 | 29.7 ± 3.8 | |
| 36.2 | – | – | 83.0 ± 1.1 | |
| 18 | 328.9 | 109.7 ± 3.7 | 78.6 ± 2.8 | 24.3 ± 4.3 |
| 98.7 | – | – | 58.0 ± 1.9 | |
| 32.9 | – | – | 93.2 ± 1.9 | |
| 24 | 267.4 | 76.7 ± 10.0 | 72.2 ± 3.2 | 50.9 ± 7.8 |
| 80.2 | – | – | 95.0 ± 4.9 | |
| 25 | 308.6 | 78.3 ± 2.3 | 52.7 ± 2.9 | 12.0 ± 1.0 |
| 92.6 | 83.9 ± 1.8 | 83.5 ± 4.1 | 71.7 ± 1.8 | |
| 26 | 446.4 | 72.5 ± 1.8 | 53.0 ± 1.3 | 45.1 ± 5.1 |
| 133.9 | 80.0 ± 0.0 | 75.3 ± 3.8 | 88.6 ± 7.8 | |
2.4. Structure-Bioactivity Relationships
3. Experimental
3.1. General Procedures
3.2. Isolation
3.3. Antiplatelet Aggregatory and Vasorelaxing Activity Bioassays
3.4. Spectral Data
4. Conclusion
Acknowledgments
References
- Tang, W.; Eisenbrand, G. Chinese Drugs of Plant Origin; Springer-Verlag: Berlin, Germany, 1992; pp. 1011–1052.
- Nurtjahja-Tjendraputra, E.; Ammit, A.J.; Roufogalis, B.D.; Tran, V.H.; Duke, C.C. Effective anti-platelet and COX-1 enzyme inhibitors from pungent constituents of ginger. Thromb. Res. 2003, 111, 259–265. [Google Scholar] [CrossRef]
- Wu, T.S.; Wu, Y.C.; Wu, P.L.; Chern, C.H.; Leu, Y.L.; Chan, Y.Y. Structure and synthesis of [n]-dehydroshogaols from Zingiber officinale. Phytochemistry 1998, 48, 889–891. [Google Scholar]
- Tao, Q.F.; Xu, Y.; Lam, R.Y.Y.; Schneider, B.; Dou, H.; Leung, P.S.; Shi, S.Y.; Zhou, C.X.; Yang, L.X.; Zhang, R.P.; et al. Diarylheptanoids and a monoterpenoid from the rhizomes of Zingiber officinale: Antioxidant and cytoprotective properties. J. Nat. Prod. 2008, 71, 12–15. [Google Scholar] [CrossRef]
- Shukla, Y.; Singh, M. Cancer preventive properties of ginger: A brief review. Food Chem. Toxicol. 2007, 45, 683–687. [Google Scholar] [CrossRef]
- Jolad, S.D.; Lantz, R.C.; Chen, G.J.; Bates, R.B. Commercially processed dry ginger (Zingiber officinale): Composition and effects on LPS-stimulated PGE2 production. Phytochemistry 2005, 6, 1614–1617. [Google Scholar]
- Weyerstahl, P.; Schlicht, V. Synthesis of rac-ar-curcumen-15-al. Liebigs Ann. 1997, 1, 175–178. [Google Scholar] [CrossRef]
- Hsieh, P.W.; Chang, F.R.; Wu, C.C.; Li, C.M.; Wu, K.Y.; Chen, S.L.; Yen, H.F.; Wu, Y.C. Longicalycinin A, a new cytotoxic cyclic peptide from Dianthus superbus var. longicalycinus (Maxim.) Will. Chem. Pharm. Bull. 2005, 53, 336–339. [Google Scholar] [CrossRef]
- Uehara, S.I.; Yasuda, I.; Akiyama, K.; Morita, H.; Takeya, K.; Itokawa, H. Diarylheptanoids from the rhizomes of Curcuma xanthorrhiza and Alpinia officinarum. Chem.Pharm.Bull. 1987, 35, 3298–3302. [Google Scholar] [CrossRef]
- Castro, V.; Tamayo-Castillo, G.; Jakupovic, J. Sesquiterpene lactones and other constituents from Calea prunifolia and C. Peckii.Phytochemistry 1989, 28, 2415–2418. [Google Scholar]
- Kiuchi, F.; Iwakami, S.; Shibuya, M.; Hanaoka, F.; Sankawa, U. Inhibition of prostaglandin and leukotriene biosynthesis by gingerols and diarylheptanoids. Chem. Pharm. Bul. 1992, 40, 387–391. [Google Scholar] [CrossRef]
- Shoji, N.; Iwasa, A.; Takemoto, T.; Ishida, Y.; Ohizumi, Y. Cardiotonic principles of ginger (Zingiber officinale Roscoe). J. Pharm.Sci. 1982, 71, 1174–1179. [Google Scholar] [CrossRef]
- Hatanaka, M.; Himeda, Y.; Imashiro, R.; Tanaka, Y.; Ueda, I. Allylidenetriphenylphosphorane as a bifunctional reagent: Synthesis of cyclopentenones and α,β-unsaturated ketones with (3-(alkoxycarbonyl)-2-ethoxy-2-propenylidene)triphenylphosphorane. J. Org. Chem. 1994, 59, 111–115. [Google Scholar] [CrossRef]
- Wu, T.S.; Tsang, Z.J.; Wu, P.L.; Lin, F.W.; Li, C.Y.; Teng, C.M.; Lee, K.H. New constituents and antiplatelet aggregation and anti-HIV principles of Artemisia capillaris. Bioorg. Med. Chem. 2001, 9, 77–83. [Google Scholar] [CrossRef]
- Denniff, P.; Macleod, I.; Whiting, D.A. Studies in the biosynthesis of [6]-gingerol, pungent principle of ginger (Zingiber officinale). J. Chem. Soc. Perkin Trans. 1 1980, 2637–2641. [Google Scholar]
- Hagiwara, H.; Okabe, T.; Ono, H.; Kamat, V.P.; Hoshi, T.; Suzuki, T.; Ando, M. Total synthesis of bisabolane sesquiterpenoids, α-bisabol-1-one, curcumene, curcuphenol and elvirol: Utility of catalytic enamine reaction in cyclohexenone synthesis. J. Chem. Soc. Perkin Trans. 1 2002, 895–899. [Google Scholar]
- Wu, P.L.; Su, G.C.; Wu, T.S. Bioactive constituents from the stems of Aristolochia manshuriensis. J. Nat. Prod. 2003, 66, 996–998. [Google Scholar] [CrossRef]
- Ohtsu, H.; Xiao, Z.; Ishida, J.; Nagai, M.; Wang, H.K.; Itokawa, H.; Su, C.Y.; Shin, C.; Chiang, T.; Chang, E.; et al. Antitumor agents. 217. Curcumin analogues as novel androgen receptor antagonists with potential as anti-prostate cancer agents. J. Med. Chem. 2002, 45, 5037–5041. [Google Scholar]
- Kikuzaki, H.; Tsai, S.M.; Nakatani, N. Gingerdiol related compounds from the rhizomes of Zingiber officinale. Phytochemistry 1992, 31, 1783–1785. [Google Scholar] [CrossRef]
- Ohshiro, M.; Kuroyanagi, M.; Ueno, A. Structures of sesquiterpenes from Curcuma longa. Phytochemistry 1990, 29, 2201–2204. [Google Scholar] [CrossRef]
- Rejzek, M.; Vacek, M.; Wimmer, Z. A mild approach to the synthesis of sn-Glycerol 1,2-Di-γ-linolenate 3-palmitate. Helv. Chim. Acta 2000, 83, 2756–2759. [Google Scholar] [CrossRef]
- Kikuzaki, H.; Usuguchi, J.; Nakatani, N. Constitutents of Zingiberaceae. I. Diarylheptanoids from the rhizomes of ginger (Zingiber officinale Roscoe). Chem. Pharm. Bull. 1991, 39, 120–125. [Google Scholar] [CrossRef]
- Wu, S.L.; Li, W.S. Chemical constituents from the roots of Neolitsea hiiranensis. J. Chin. Chem. Soc. 1997, 42, 555–559. [Google Scholar]
- Wu, T.S.; Chern, H.J.; Damu, A.G.; Kuo, P.C.; Su, C.R.; Lee, E.J.; Teng, C.M. Flavonoids and andrographolides from Andrographis paniculata and their antiplatelet aggregatory and vasorelaxing effects. J. Asian Nat. Prod. Res. 2008, 10, 17–24. [Google Scholar] [CrossRef]
- Guh, J.H.; Ko, F.N.; Yu, S.M.; Wu, Y.C.; Teng, C.M. Pharmacological evaluation of N-methyl-actinodaphnine, a new vascular α-adrenoceptor antagonist, isolated from Illigera luzonensis. Eur.J.Pharm. 1995, 279, 33–41. [Google Scholar] [CrossRef]
- Teng, C.M.; Chen, W.C.; Ko, F.N.; Lee, L.G.; Huang, T.F.; Chen, Y.P.; Hsu, H.Y. Two antiplatelet agents from Magnolia officinalis. Thromb. Res. 1988, 50, 757–765. [Google Scholar] [CrossRef]
- Born, G.V.R.; Cross, M.J. Effects of inorganic ions and of plasma proteins on the aggregation of blood platelets by adenosine diphosphate. J. Physiol. 1963, 168, 178–195. [Google Scholar]
- Sample Availability: Samples of the Z. officinale are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liao, Y.-R.; Leu, Y.-L.; Chan, Y.-Y.; Kuo, P.-C.; Wu, T.-S. Anti-Platelet Aggregation and Vasorelaxing Effects of the Constituents of the Rhizomes of Zingiber officinale. Molecules 2012, 17, 8928-8937. https://doi.org/10.3390/molecules17088928
Liao Y-R, Leu Y-L, Chan Y-Y, Kuo P-C, Wu T-S. Anti-Platelet Aggregation and Vasorelaxing Effects of the Constituents of the Rhizomes of Zingiber officinale. Molecules. 2012; 17(8):8928-8937. https://doi.org/10.3390/molecules17088928
Chicago/Turabian StyleLiao, Yu-Ren, Yann-Lii Leu, Yu-Yi Chan, Ping-Chung Kuo, and Tian-Shung Wu. 2012. "Anti-Platelet Aggregation and Vasorelaxing Effects of the Constituents of the Rhizomes of Zingiber officinale" Molecules 17, no. 8: 8928-8937. https://doi.org/10.3390/molecules17088928




