Application of Hydrogen Peroxide Encapsulated in Silica Xerogels to Oxidation Reactions
Abstract
:1. Introduction
2. Results and Discussion
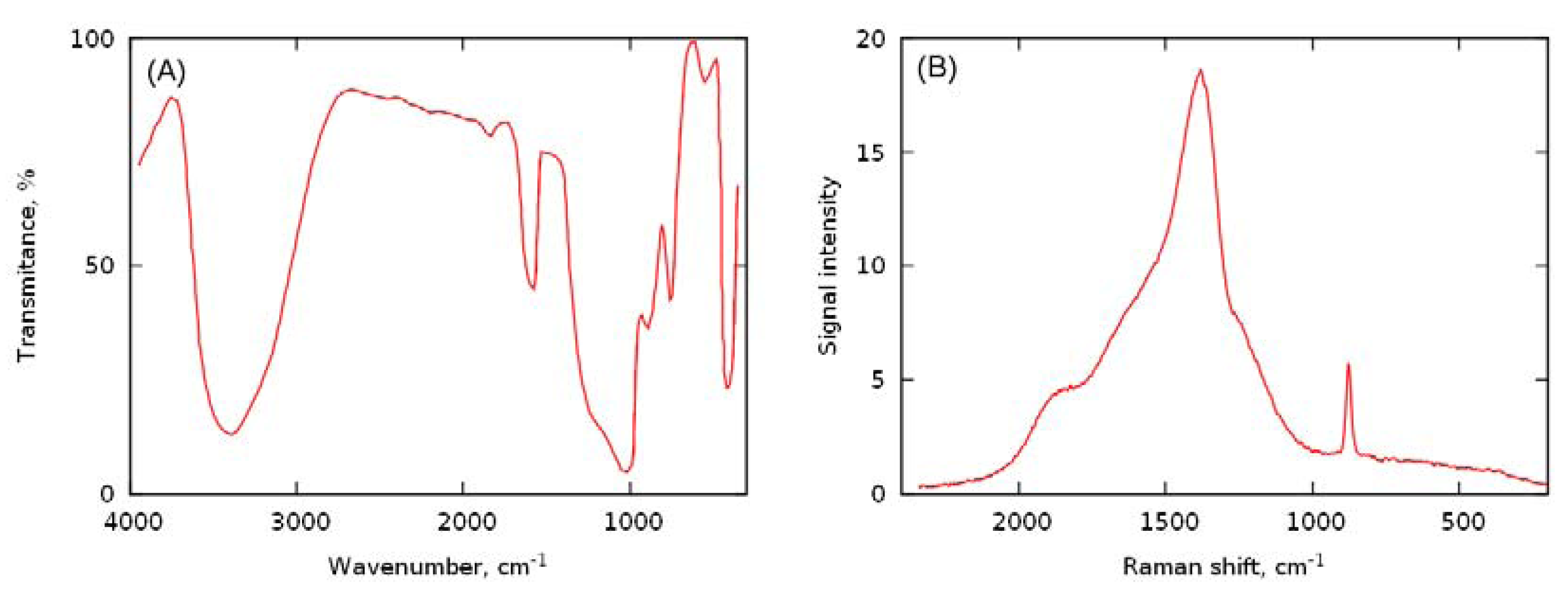
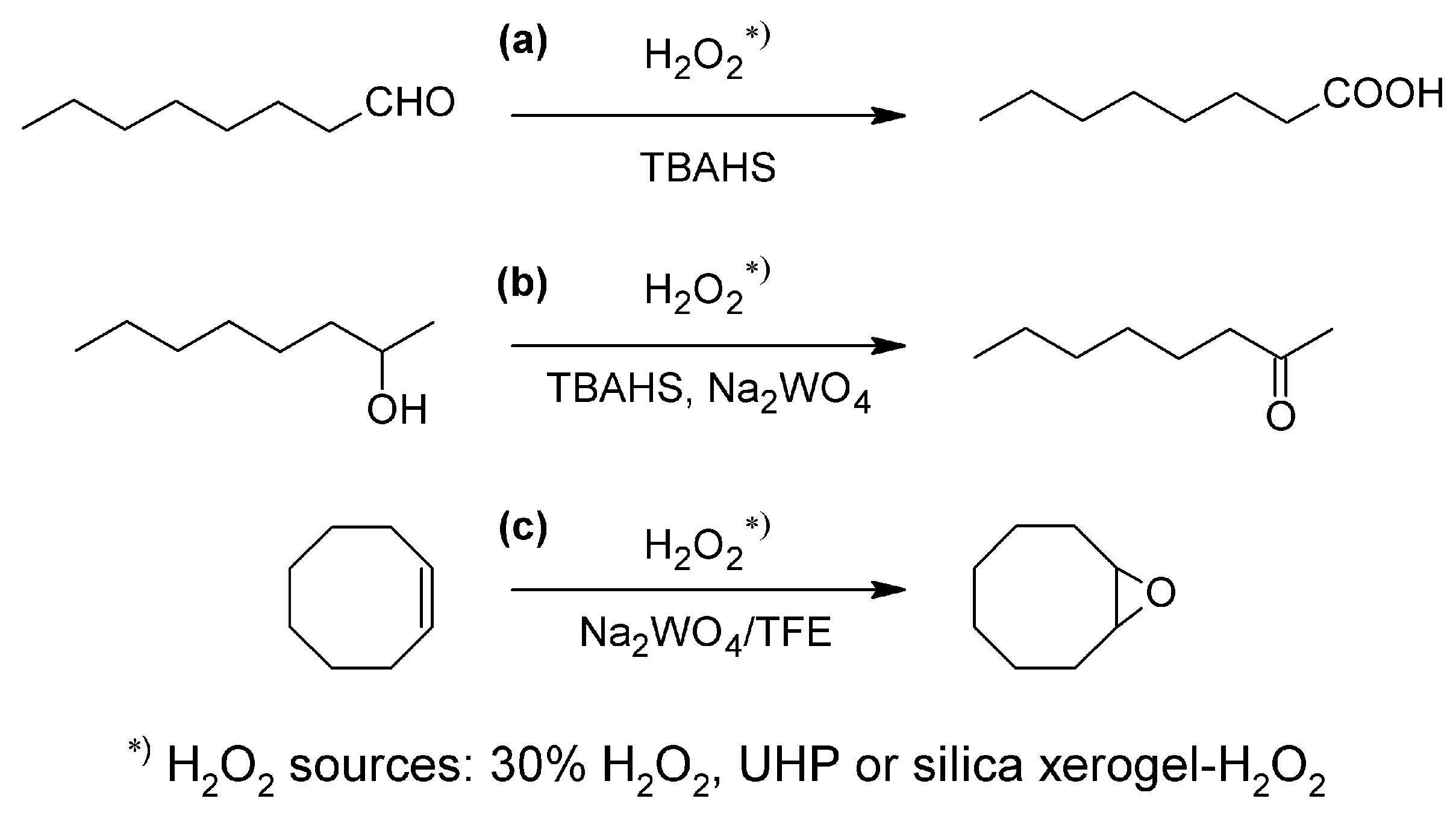
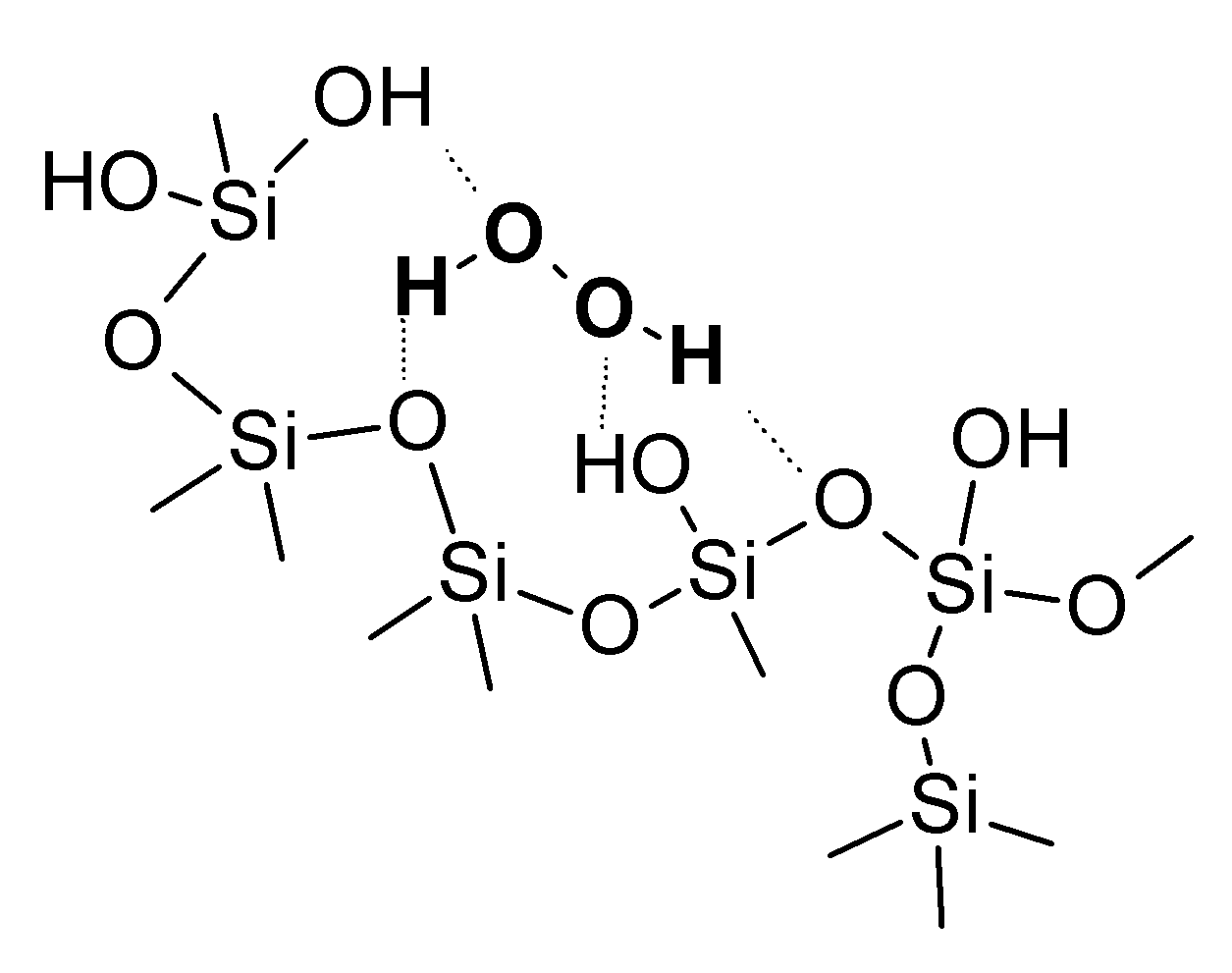
| H2O2 source | Yield of octanoic acid, % | |
|---|---|---|
| Conventional conditions, | Microwave irradiation, | |
| 90 °C, 120 min | 90-95 °C, 45 min | |
| 30% H2O2 | 86 | 81 |
| UHP | 49 | 0 a |
| The xerogel | 83 | 73 |
| H2O2 source | Yield of 2-octanone, % | |
|---|---|---|
| Conventional conditions, 80 °C, 30 min | Microwave irradiation, 80-90 °C, 15 min | |
| 30% H2O2 | 92 (64 a) | 99 |
| UHP | 69 (6 a) | <1 |
| The xerogel | 98 (56 a) | 46 |
| H2O2 source | Yield of cyclooctene oxide, % | |
|---|---|---|
| Conventional conditions, 3 h 60 °C | Microwave irradiation, 1 h 60–70 °C | |
| 30% H2O2 | 88 | 90 |
| UHP | 94 | 95 |
| The xerogel | 58 (69 a, 74 b) | 71 |
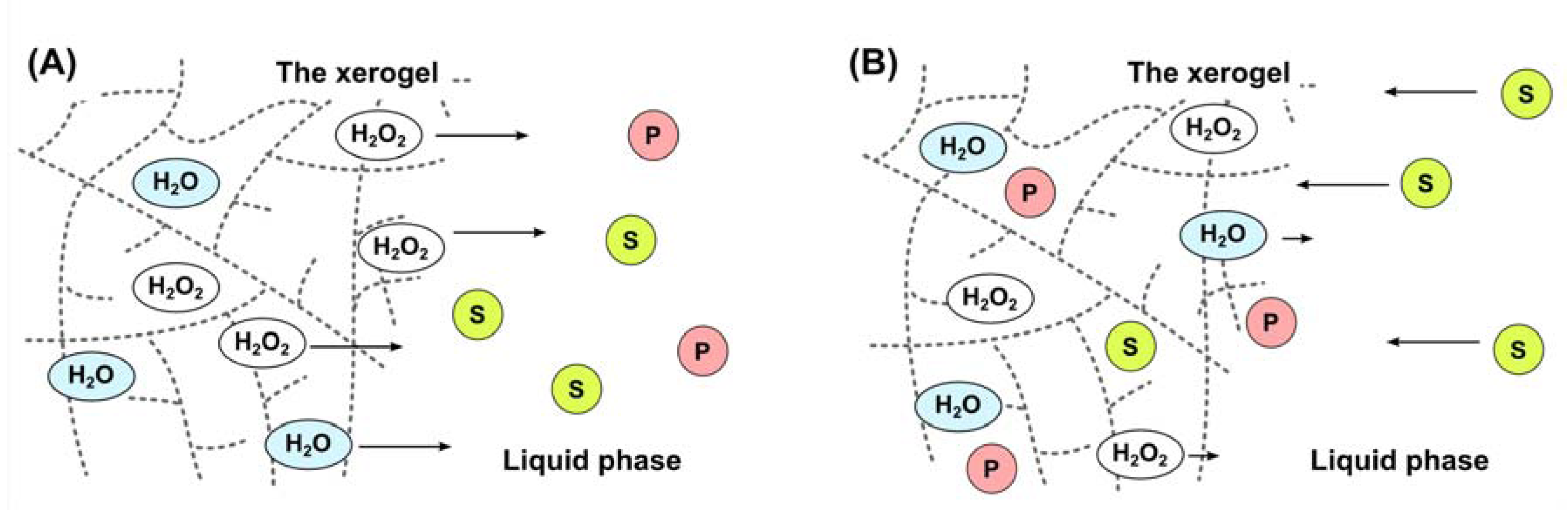
3. Experimental Section
3.1. Equipment
3.2. Materials
3.3. Preparation of Silica Xerogel-Hydrogen Peroxide Composites by the Sol-Gel Method
3.4. Model Reactions
3.4.1. Octanal Oxidation
3.4.2. 2-Octanol Oxidation
3.4.3. Cyclooctene Epoxidation
3.5. Microwave Assisted Reactions
4. Conclusions
Acknowledgments
References
- de Jong, K.P. Synthesis of Solid Catalysts; Wiley-VCH: Weinheim, Germany, 2009. [Google Scholar]
- Kochkar, H.; Figueras, F. Synthesis of hydrophobic TiO2-SiO2 mixed oxides for the epoxidation of cyclohexene. J. Catal. 1997, 171, 420–430. [Google Scholar]
- Lukasiewicz, M.; Bogdal, D.; Pielichowski, J. Microwave assisted oxidation of some aromatics by hydrogen peroxide at supported tungsten catalyst. Mol. Divers. 2006, 10, 491–493. [Google Scholar] [CrossRef]
- Barbosa, G.N.; Mac Leod, T.C.O.; Guedes, D.F.C.; Assis, M.D.; Oliveira, H.P. Preparation, characterization and catalytic studies of V2O5-SiO2 xerogel composite. J. Sol-Gel Sci. Technol. 2008, 46, 99–105. [Google Scholar]
- Neumann, R.; Levin-Elad, M. Metal oxide (TiO2, MoO3, WO3) substituted silicate xerogels as catalysts for the oxidation of hydrocarbons with hydrogen peroxide. J. Catal. 1997, 166, 206–217. [Google Scholar]
- Quintanar-Guerrero, D.; Ganem-Quintanar, A.; Nava-Arzaluz, M.G.; Piñón-Segundo, E. Silica xerogels as pharmaceutical drug carriers. Expert Opin. Drug Del. 2009, 6, 485–498. [Google Scholar] [CrossRef]
- Żegliński, J.; Cabaj, A.; Strankowski, M.; Czerniak, J.; Haponiuk, J.T. Silica xerogel-hydrogen peroxide composites: Their morphology, stability, and antimicrobial activity. Colloids Surf. B 2007, 54, 165–172. [Google Scholar] [CrossRef]
- Żegliński, J.; Piotrkowski, G.P.; Piękoś, R.J. A study of interaction between hydrogen peroxide and silica gel by FTIR spectroscopy and quantum chemistry. Mol. Struct. 2006, 794, 83–91. [Google Scholar] [CrossRef]
- Sato, K.; Hyodo, M.; Takagi, J.; Aoki, M.; Noyori, R. Hydrogen peroxide oxidation of aldehydes to carboxylic acids: An organic solvent-, halide- and metal-free procedure. Tetrahedron Lett. 2000, 41, 1439–1442. [Google Scholar] [CrossRef]
- Noyori, R.; Aoki, M.; Sato, K. Green oxidation with aqueous hydrogen peroxide. Chem. Commun. 2003, 16, 1977–1986. [Google Scholar]
- Bogdal, D.; Lukasiewicz, M. Microwave-assisted oxidation of alcohols using aqueous hydrogen peroxide. Synlett 2000, 1, 143–145. [Google Scholar]
- Bogdal, D.; Bednarz, S.; Plezia, M. Efficient epoxidation of olefins catalyzed by tungstate (VI) in fluoroalcohols. Tetrahedron 2012, in press.. [Google Scholar]
- Bogdal, D.; Lukasiewicz, M.; Pielichowski, J.; Bednarz, S. Microwave-assisted epoxidation of simple alkenes in the presence of hydrogen peroxide. Synth. Commun. 2005, 35, 2973–2983. [Google Scholar]
- Ogata, Y.; Kawasaki, A.; Okumura, N. Kinetics of the condensation of urea with some aliphatic aldehydes. Tetrahedron 1966, 22, 1731–1739. [Google Scholar] [CrossRef]
- Heaney, H. Oxidation reactions using magnesium monoperphthalate and urea hydrogen peroxide. Aldrichim. Acta 1993, 26, 35–45. [Google Scholar]
- Legros, J.; Crousse, B.; Bonnet-Delpon, D.; Bégué, J.P. Urea-Hydrogen peroxide/hexafluoro-2-propanol: An efficient system for a catalytic epoxidation reaction without a metal. Eur. J. Org. Chem. 2002, 19, 3290–3293. [Google Scholar]
- Meredith, R.J. Engineers’ Handbook of Industrial Microwave Heating, 1st ed; Institution of Electrical Engineers: London, UK, 1998. [Google Scholar]
- Lukasiewicz, M.; Bogdal, D.; Pielichowski, J. Microwave-Assisted oxidation of side chain arenes by magtrieve. J. Adv. Synth. Catal. 2003, 345, 1269–1272. [Google Scholar] [CrossRef]
- Lukasiewicz, M.; Bogdal, D.; Pielichowski, J. 8th International electronic conference on synthetic organic chemistry. Available online: http://www.usc.es/congresos/ecsoc/8/MAS/003/index.htm (accessed on 2 May 2012).
- dos Santos, A.A.; Wendler, E.P.; de Marques, F.A.; Simonelli, F. Microwave-accelerated epoxidation of α,β-unsaturated ketones with urea-hydrogen peroxide. Lett. Org. Chem. 2004, 1, 47–49. [Google Scholar] [CrossRef]
- Ravikumar, K.S.; Zhang, Y.M.; Bégué, J.P.; Bonnet-Delpon, D. Role of Hexafluoro-2-propanol in selective oxidation of sulfide to sulfoxide: Efficient preparation of glycosyl sulfoxides. Eur. J. Org. Chem. 1998, 2937–2940. [Google Scholar]
- Neimann, K.; Neumann, R. Electrophilic activation of hydrogen peroxide: Selective oxidation reactions in perfluorinated alcohol solvents. Org. Lett. 2000, 2, 2861–2863. [Google Scholar] [CrossRef]
- de Visser, S.P.; Kaneti, J.; Neumann, R.; Shaik, S. Fluorinated alcohols enable olefin epoxidation by h2o2: Template catalysis. J. Org. Chem. 2003, 68, 2903–2912. [Google Scholar]
- Berkessel, A.; Adrio, J.A. Kinetic studies of olefin epoxidation with hydrogen peroxide in 1,1,1,3,3,3-Hexafluoro-2-propanol reveal a crucial catalytic role for solvent clusters. Adv. Synth. Catal. 2004, 346, 275–280. [Google Scholar] [CrossRef]
- Berkessel, A.; Adrio, J.A. Dramatic acceleration of olefin epoxidation in fluorinated alcohols: Activation of hydrogen peroxide by multiple h-bond networks. J. Am. Chem. Soc. 2006, 128, 13412–13420. [Google Scholar] [CrossRef]
- Yoshida, A. Silica nucleation, polymerization, and growth preparation of monodispersed sols. In Colloidal Silica: Fundamentals and Applications, 1st; Bergna, H.E., Roberts, W.O., Eds.; Taylor & Francis Group: Boca Raton, FL, USA, 2006. [Google Scholar]
- Alexander, G.B. Aquasols of positively-charged coated silica particles and their production. U.S. Patent 3007878, 1961. [Google Scholar]
- Panarin, E.F.; Kalninsh, K.K.; Pestov, D.V. Molecular complexes between hydrogen peroxide and polyvinylpyrrolidone. Doklady Akademii Nauk 1998, 363, 208–210. [Google Scholar]
- Panarin, E.F.; Kalninsh, K.K.; Pestov, D.V. Complexation of hydrogen peroxide with polyvinylpyrrolidone: Ab initio calculations. Eur. Pol. J. 2001, 37, 375–379. [Google Scholar]
- Shiraeff, D.A. Solid stabilized hydrogen peroxide compositions. U.S. Patent 3480557, 1969. [Google Scholar]
- Panarin, E.F.; Kalninsh, K.K.; Azanova, V.V. IR spectra and structure of poly(vinylamide) complexes with hydrogen peroxide. Polym. Sci. Ser. A 2007, 49, 275–283. [Google Scholar]
- Breitenbach, J.; Sanner, A. Hydrogen peroxide complexes in powder form are prepared by spray drying aqueous or alcoholic solutions. U.S. Patent 5674436, 1997. [Google Scholar]
- Sample Availability: Samples of the compounds xerogels are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bednarz, S.; Ryś, B.; Bogdał, D. Application of Hydrogen Peroxide Encapsulated in Silica Xerogels to Oxidation Reactions. Molecules 2012, 17, 8068-8078. https://doi.org/10.3390/molecules17078068
Bednarz S, Ryś B, Bogdał D. Application of Hydrogen Peroxide Encapsulated in Silica Xerogels to Oxidation Reactions. Molecules. 2012; 17(7):8068-8078. https://doi.org/10.3390/molecules17078068
Chicago/Turabian StyleBednarz, Szczepan, Barbara Ryś, and Dariusz Bogdał. 2012. "Application of Hydrogen Peroxide Encapsulated in Silica Xerogels to Oxidation Reactions" Molecules 17, no. 7: 8068-8078. https://doi.org/10.3390/molecules17078068