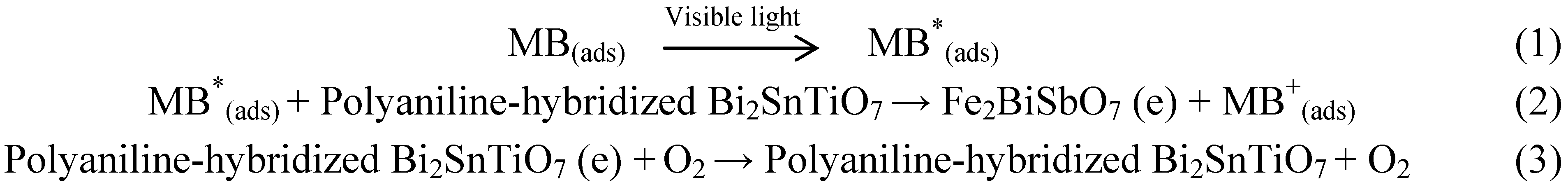2.1. Crystal Structure of Bi2SnTiO7
Figure 1 presents a TEM image, the selected area electron diffraction pattern and the SEM-EDS spectrum of Bi
2SnTiO
7. The TEM image of Bi
2SnTiO
7 showed that the morphology of the Bi
2SnTiO
7 particle was roundish and the Bi
2SnTiO
7 particle size was uniform. It could be seen that the Bi
2SnTiO
7 particles crystallized well and the average particle size of Bi
2SnTiO
7 was about 180 nm. The SEM-EDS spectrum of Bi
2SnTiO
7 revealed that Bi
2SnTiO
7 was pure phase without any other impurities and Bi
2SnTiO
7 displayed the presence of bismuth, tin, titanium and oxygen. It could be seen from
Figure 1 that Bi
2SnTiO
7 crystallized with the pyrochlore-type structure, cubic crystal system and space group
Fd3m.
Figure 1.
TEM image of Bi2SnTiO7 (a) and the selected area electron diffraction pattern of Bi2SnTiO7 (b) and SEM-EDS spectrum of Bi2SnTiO7 (c).
Figure 1.
TEM image of Bi2SnTiO7 (a) and the selected area electron diffraction pattern of Bi2SnTiO7 (b) and SEM-EDS spectrum of Bi2SnTiO7 (c).
The lattice parameter for Bi
2SnTiO
7 was proved to be a = 10.52582(8) Å. According to the calculation results from
Figure 1, the (h k l) value for the main peaks of Bi
2SnTiO
7 could be found and indexed.
Full-profile structure refinements of the collected X-ray diffraction data of Bi
2SnTiO
7 were obtained by the RIETAN
TM [
33] program, which was based on Pawley analysis. The refinement results of Bi
2SnTiO
7 are shown in
Figure 2. The atomic coordinates and structural parameters of Bi
2SnTiO
7 are listed in
Table 1. The results of the final refinement for Bi
2SnTiO
7 indicated a good agreement between the observed and calculated intensities in a pyrochlore-type structure and cubic crystal system with space group
Fd3m. Our XRD results also showed that Bi
2SnTiO
7 and Bi
2InTaO
7 crystallized in the same structure, and 2 theta angles of each reflection of Bi
2SnTiO
7 changed with Sn
4+ and Ti
4+ being replaced by In
3+ and Ta
5+. Bi
2InTaO
7 also crystallized with a cubic structure by space group Fd3m and the lattice parameter of Bi
2InTaO
7 was a = 10.74641(0) Å. The lattice parameter of Bi
2SnTiO
7 was a = 10.52582(8) Å, which indicated that the lattice parameter of Bi
2SnTiO
7 decreased compared with the lattice parameter of Bi
2InTaO
7 because the In
3+ ionic radii (0.92 Å) was larger than the Sn
4+ ionic radii (0.71Å) and the Ta
5+ ionic radii (0.68 Å) was equal to the Ti
4+ ionic radii (0.68Å). The outcome of refinement for Bi
2SnTiO
7 generated the unweighted
R factor,
RP = 10.72% with space group
Fd3m. Zou
et al. [
34] refined the crystal structure of Bi
2InNbO
7 and obtained a large
R factor for Bi
2InNbO
7, which was ascribed to a slightly modified structure model for Bi
2InNbO
7. According to the high purity of the precursors which were utilized in this study and the fact which the EDS results did not trace any other elements, it was unlikely that the observed space groups originated from the presence of impurities. Therefore, it was suggested that the slightly high
R factor for Bi
2SnTiO
7 was owing to a slightly modified structure model for Bi
2SnTiO
7. It should be emphasized that the defects or the disorder/order of a fraction of the atoms could cause the change of structures, including different bond-distance distributions, thermal displacement parameters and/or occupation factors for some of the atoms.
Figure 2.
X-ray powder diffraction pattern and Rietveld refinements of Bi2SnTiO7 prepared by a solid-state reaction method at 1,100 °C.
Figure 2.
X-ray powder diffraction pattern and Rietveld refinements of Bi2SnTiO7 prepared by a solid-state reaction method at 1,100 °C.
Table 1.
Atomic coordinates and structural parameters of Bi2SnTiO7 prepared by the solid state reaction method.
Table 1.
Atomic coordinates and structural parameters of Bi2SnTiO7 prepared by the solid state reaction method.
| Atom | x | y | z | Occupation factor |
|---|
| Bi | 0.0000 | 0 | 0 | 1.0 |
| Sn | 0.5000 | 0.5000 | 0.5000 | 0.5 |
| Ti | 0.5000 | 0.5000 | 0.5000 | 0.5 |
| O(1) | −0.0947 | 0.1250 | 0.1250 | 1.0 |
| O(2) | 0.1250 | 0.1250 | 0.1250 | 1.0 |
In order to reveal the surface chemical compositions and the valence states of various elements of Bi
2SnTiO
7, the X-ray photoelectron spectrum of Bi
2SnTiO
7 for detecting Bi, Sn, Ti and O was performed. The full XPS spectrum confirmed that the prepared Bi
2SnTiO
7 contained the elements Bi, Sn, Ti and O, which was consistent with the SEM-EDS results. The different elemental peaks for Bi
2SnTiO
7 which are corresponding to definite bind energies are given in
Table 2. The results illustrated that the oxidation states of Bi, Sn, Ti and O ions from Bi
2SnTiO
7 were +3, +4, +4 and −2, respectively. Moreover, the average atomic ratio of Bi:Sn:Ti:O for Bi
2SnTiO
7 was 2.00:0.98:1.02:6.97 accoring to our XPS, SEM-EDS and XFS results. Accordingly, it could be deduced that the resulting material was highly pure under our preparation conditions. It was remarkable that there were not any shoulders and widening in the XPS peaks of Bi
2SnTiO
7, which suggested the absence of any other phases.
Table 2.
Binding energies (BE) for key elements from Bi2SnTiO7.
Table 2.
Binding energies (BE) for key elements from Bi2SnTiO7.
| Compound | Bi4f7/2 | Sn3d5/2 | Ti3p | O1s |
|---|
| BE (eV) | BE (eV) | BE (eV) | BE (eV) |
|---|
| Bi2SnTiO7 | 159.50 | 486.55 | 37.50 | 530.15 |
2.2. Photocatalytic Properties
Generally, the direct absorption of band-gap photons would result in the generation of electron–hole pairs within the polyaniline-hybridized Bi
2SnTiO
7, subsequently, the charge carriers began to diffuse to the surface of the polyaniline-hybridized Bi
2SnTiO
7. As a result, the photocatalytic activity for decomposing organic compounds with the polyaniline-hybridized Bi
2SnTiO
7 might be enhanced. Changes in the UV-Vis spectrum of MB upon exposure to visible light (
λ > 400 nm) irradiation with the presence of the polyaniline-hybridized Bi
2SnTiO
7, Bi
2InTaO
7 or N-doped TiO
2 indicated that the polyaniline-hybridized Bi
2SnTiO
7, Bi
2InTaO
7 or N-doped TiO
2 could photodegrade MB effectively under visible light irradiation.
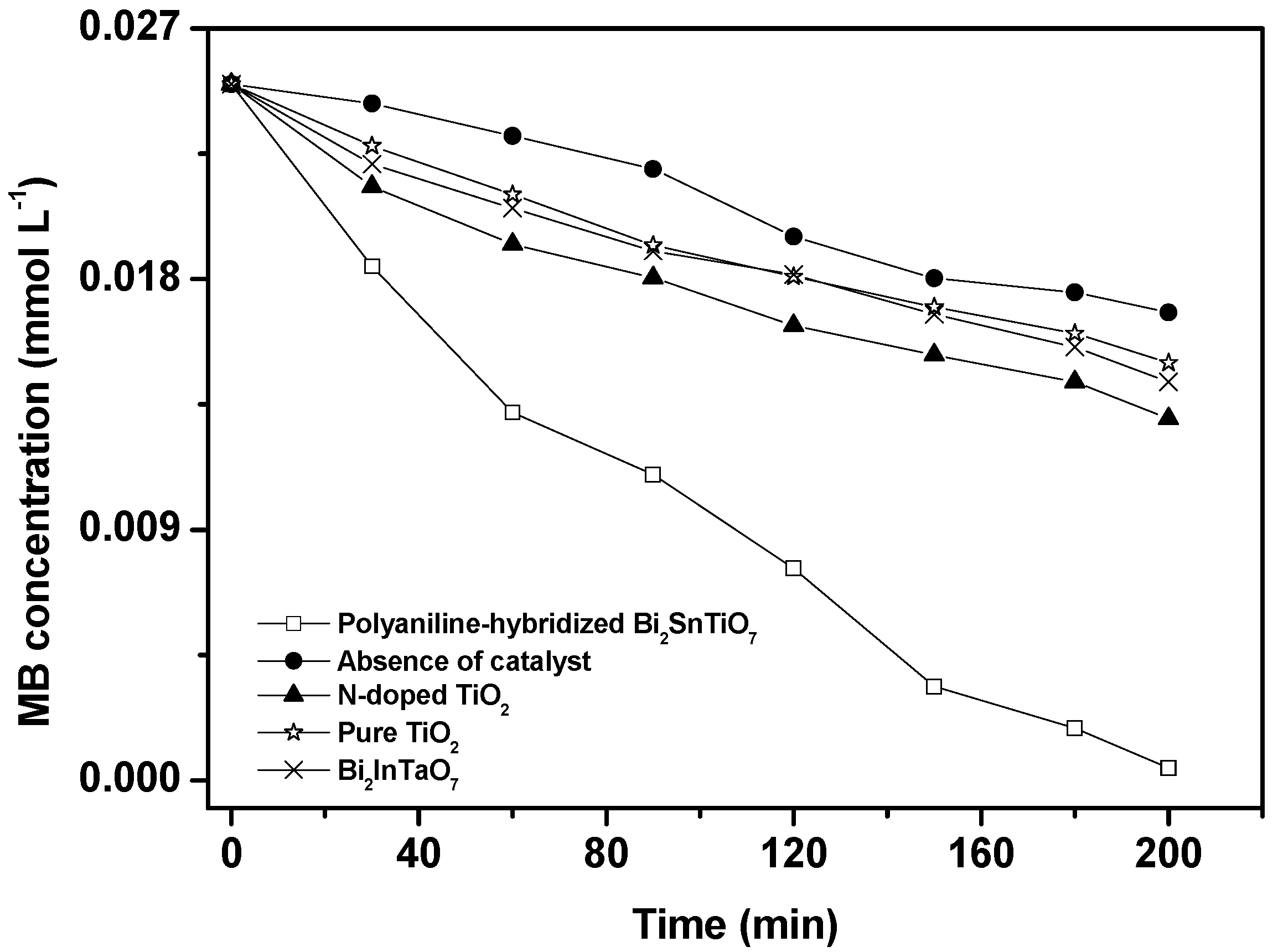
Figure 3 shows the photocatalytic degradation of methylene blue under visible light irradiation in the presence of the polyaniline-hybridized Bi
2SnTiO
7, Bi
2InTaO
7, pure TiO
2, N-doped TiO
2 as well as in the absence of a photocatalyst.
Figure 3.
Photocatalytic degradation of methylene blue under visible light irradiation in the presence of the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7, pure TiO2, N-doped TiO2 as well as in the absence of a photocatalyst.
Figure 3.
Photocatalytic degradation of methylene blue under visible light irradiation in the presence of the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7, pure TiO2, N-doped TiO2 as well as in the absence of a photocatalyst.
The results showed that a reduction in the typical MB peaks at 665 nm and 614 nm was clearly noticed and the photodegradation rate of MB was about 2.046 × 10
−9 mol L
−1 s
−1 and the photonic efficiency was estimated to be 0.0430% (
λ = 420 nm) with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst. Similarly, the photodegradation rate of MB was about 1.001 × 10
−9 mol L
−1 s
−1 and the photonic efficiency was estimated to be 0.0210% (
λ = 420 nm) with N-doped TiO
2 as catalyst. Furthermore, the photodegradation rate of MB was about 0.891 × 10
−9 mol L
−1 s
−1 and the photonic efficiency was estimated to be 0.0187% (
λ = 420 nm) with Bi
2InTaO
7 as catalyst. As a contradistinction, the photodegradation rate of MB within 200 minutes of visible light irradiation was only 0.8338 × 10
−9 mol L
−1 s
−1 and the photonic efficiency was estimated to be 0.0175% (
λ = 420 nm) with pure TiO
2 as catalyst. The photodegradation rate of MB was about 0.6830 × 10
−9 mol L
−1 s
−1 and the photonic efficiency was estimated to be 0.0143% (
λ = 420 nm) in the absence of a photocatalyst. The results showed that the photodegradation rate of MB and the photonic efficiency with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst were both higher than those with N-doped TiO
2 or Bi
2InTaO
7 or pure TiO
2 as catalyst. The photodegradation rate of MB and the photonic efficiency with N-doped TiO
2 as catalyst were both higher than those with Bi
2InTaO
7 or pure TiO
2 as catalyst. The photodegradation rate of MB and the photonic efficiency with Bi
2InTaO
7 as catalyst were both higher than those with pure TiO
2 or the absence of a photocatalyst. The photodegradation rate of MB and the photonic efficiency with pure TiO
2 as catalyst were both higher than those with the absence of a photocatalyst. When the polyaniline-hybridized Bi
2SnTiO
7, N-doped TiO
2, Bi
2InTaO
7 or pure TiO
2 was utilized as catalyst, the photodegradation conversion rate of MB was 98.20%, 48.05%, 42.76% and 40.02% after visible light irradiation for 200 minutes, respectively. Furthermore, the photodegradation conversion rate of MB was 32.78% after visible light irradiation for 200 minutes with the absence of a photocatalyst because of the MB dye photo-sensitization effect [
35]. After visible light irradiation for 220 minutes with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst, complete removal of MB was observed and the complete disappearance of the absorption peaks which presented the absolute color change from deep blue into colorless solution occurred.
Bi
2SbVO
7, Fe
2BiSbO
7 and SrFeO
3 were also prepared by using the preparation methods from the literature reports [
36,
37,
38]. Polyaniline-hybridized Bi
2SnTiO
7, Bi
2SbVO
7, Fe
2BiSbO
7 and SrFeO
3 were utilized as catalysts to degrade MB under the same experimental conditions which were shown in our paper. The results are shown in
Table 3. It could be seen from
Table 3 that the removal rate of MB was 98.2%, 96.5%, 95.0% or 59.7% with polyaniline-hybridized Bi
2SnTiO
7, Bi
2SbVO
7, Fe
2BiSbO
7 or SrFeO
3 as catalyst within 200 minutes under visible light irradiation. The results showed that the polyaniline-hybridized Bi
2SnTiO
7 showed the best photocatalytic activity for photodegradation of MB compared with Bi
2SbVO
7, Fe
2BiSbO
7 or SrFeO
3. Furthermore, our N-doped TiO
2 showed low photocatalytic activity for photodegradation of MB compared with the N-doped TiO
2 which was produced by Yang
et al. [
39] under visible light irradiation. The reason was that N:TiO
2 molar ratio was different between our N-doped TiO
2 and Yang’s N-doped TiO
2 [
39], which indicated that our N-doped TiO
2 contained 62% anatase phase and 38% rutile phase, at the same time, Yang’s N-doped TiO
2 [
39] contained 100% anatase phase. We prepared N-doped TiO
2 by using the preparation method from Yang
et al. and we found that N-doped TiO
2 showed similar results for degradation of MB.
Table 3.
The removal rate of MB by using different catalysts within 200 minutes under visible light irradiation.
Table 3.
The removal rate of MB by using different catalysts within 200 minutes under visible light irradiation.
| Catalyst | Polyaniline-hybridized Bi2SnTiO7 | Bi2SbVO7 | Fe2BiSbO7 | SrFeO3 |
|---|
| Removal rate of MB (%) | 98.2 | 96.5 | 95.0 | 59.7 |
According to above results, the photocatalytic degradation activity of the polyaniline-hybridized Bi2SnTiO7 was much higher than that of N-doped TiO2, Bi2InTaO7 or pure TiO2. Meanwhile, N-doped TiO2 showed higher photocatalytic degradation activity for MB photodegradation compared with Bi2InTaO7 or pure TiO2. Bi2InTaO7 showed higher photocatalytic degradation activity for MB photodegradation compared with pure TiO2. Pure TiO2 was more suitable for MB photodegradation than the absence of a photocatalyst. The photocatalytic property of novel polyaniline-hybridized Bi2SnTiO7 under visible light irradiation was amazing compared with that of N-doped TiO2 or pure TiO2, and the main reason was that the specific surface area of the polyaniline-hybridized Bi2SnTiO7 was much smaller than that of N-doped TiO2 or pure TiO2. BET isotherm measurements of the polyaniline-hybridized Bi2SnTiO7, N-doped TiO2 and pure TiO2 provided a specific surface area of 4.29 m2 g−1, 45.53 m2 g−1 and 46.24 m2 g−1 respectively, which indicated that the photocatalytic degradation activity of the polyaniline-hybridized Bi2SnTiO7 could be improved consumedly by enhancing the specific surface area of the polyaniline-hybridized Bi2SnTiO7.
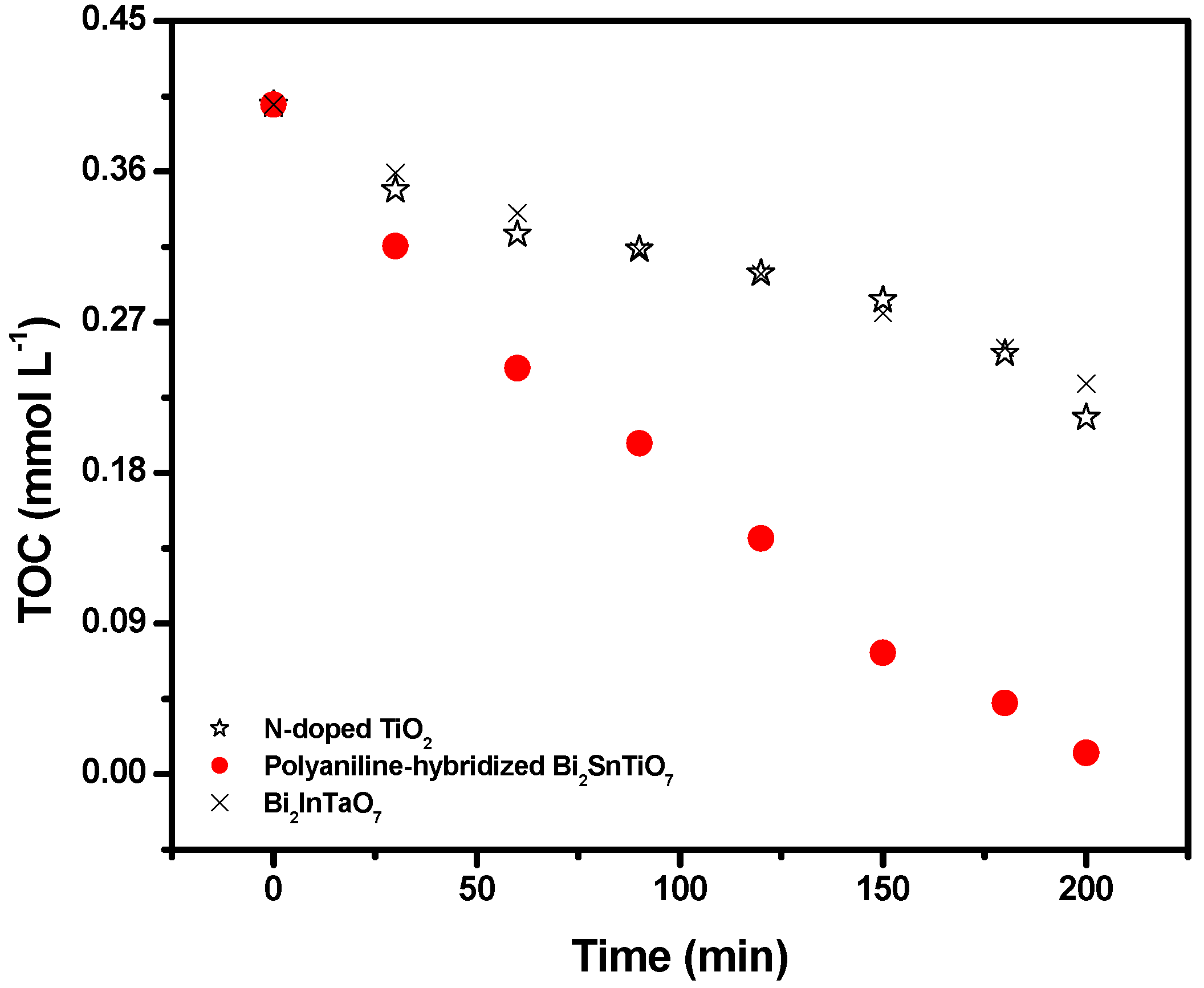
Figure 4 shows the change of TOC during photocatalytic degradation of MB with the polyaniline-hybridized Bi
2SnTiO
7, Bi
2InTaO
7 or N-doped TiO
2 as catalyst under visible light irradiation.
Figure 4.
Disappearance of total organic carbon (TOC) during photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
Figure 4.
Disappearance of total organic carbon (TOC) during photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
The TOC measurements revealed the disappearance of organic carbon when the MB solution which contained the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 was exposed under visible light irradiation. The results showed that 96.77% or 46.77% or 41.71% of TOC decrease was obtained after visible light irradiation for 200 minutes when the polyaniline-hybridized Bi2SnTiO7 or N-doped TiO2 or Bi2InTaO7 was utilized as photocatalyst. Consequently, after visible light irradiation for 220 minutes with the polyaniline-hybridized Bi2SnTiO7 as catalyst, the entire mineralization of MB was observed because of 100% TOC removal. The turnover number which represented the ratio between the total amount of evolved gas and dissipative catalyst was calculated to be more than 0.258 for the polyaniline-hybridized Bi2SnTiO7 after 200 minutes of reaction time under visible light irradiation and this turnover number was evident to prove that this reaction occurred catalytically. Similarly, when the light was turned off in this experiment, the stop of this reaction showed the obvious light response.
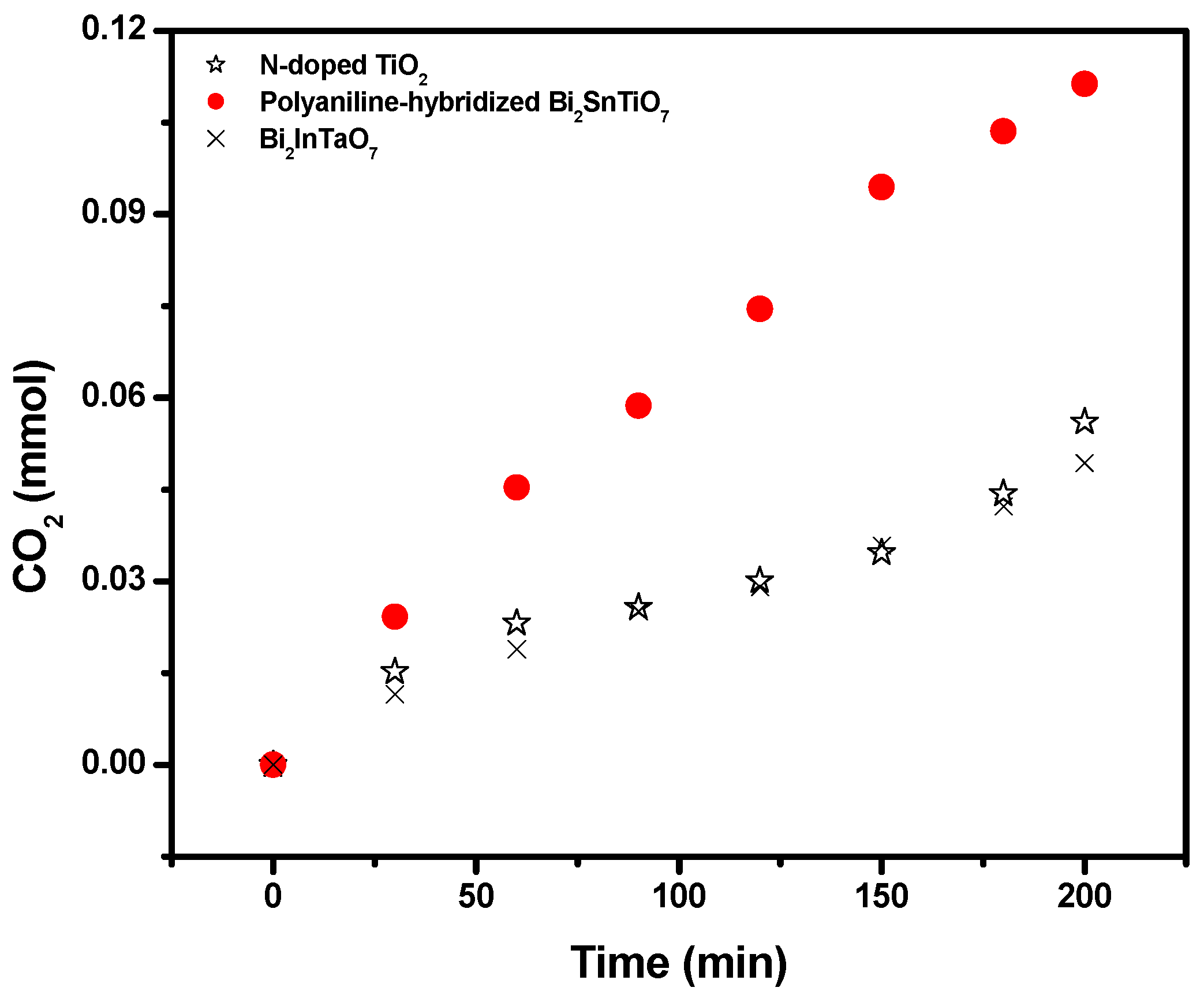
Figure 5 shows the amount of CO
2 which was released during the photodegradation of MB with the polyaniline-hybridized Bi
2SnTiO
7, Bi
2InTaO
7 or N-doped TiO
2 as catalyst under visible light irradiation. The amount of CO
2 increased gradually with increasing reaction time when MB was photodegraded by the polyaniline-hybridized Bi
2SnTiO
7, Bi
2InTaO
7 or N-doped TiO
2. At the same time, after visible light irradiation of 200 minutes, the CO
2 production of 0.11129 mmol with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst was higher than the CO
2 production of 0.05600 mmol with N-doped TiO
2 as catalyst. Meanwhile after visible light irradiation of 200 minutes, the CO
2 production of 0.05600 mmol with N-doped TiO
2 as catalyst was higher than the CO
2 production of 0.04934 mmol with Bi
2InTaO
7 as catalyst.
Figure 5.
CO2 production kinetics during the photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
Figure 5.
CO2 production kinetics during the photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
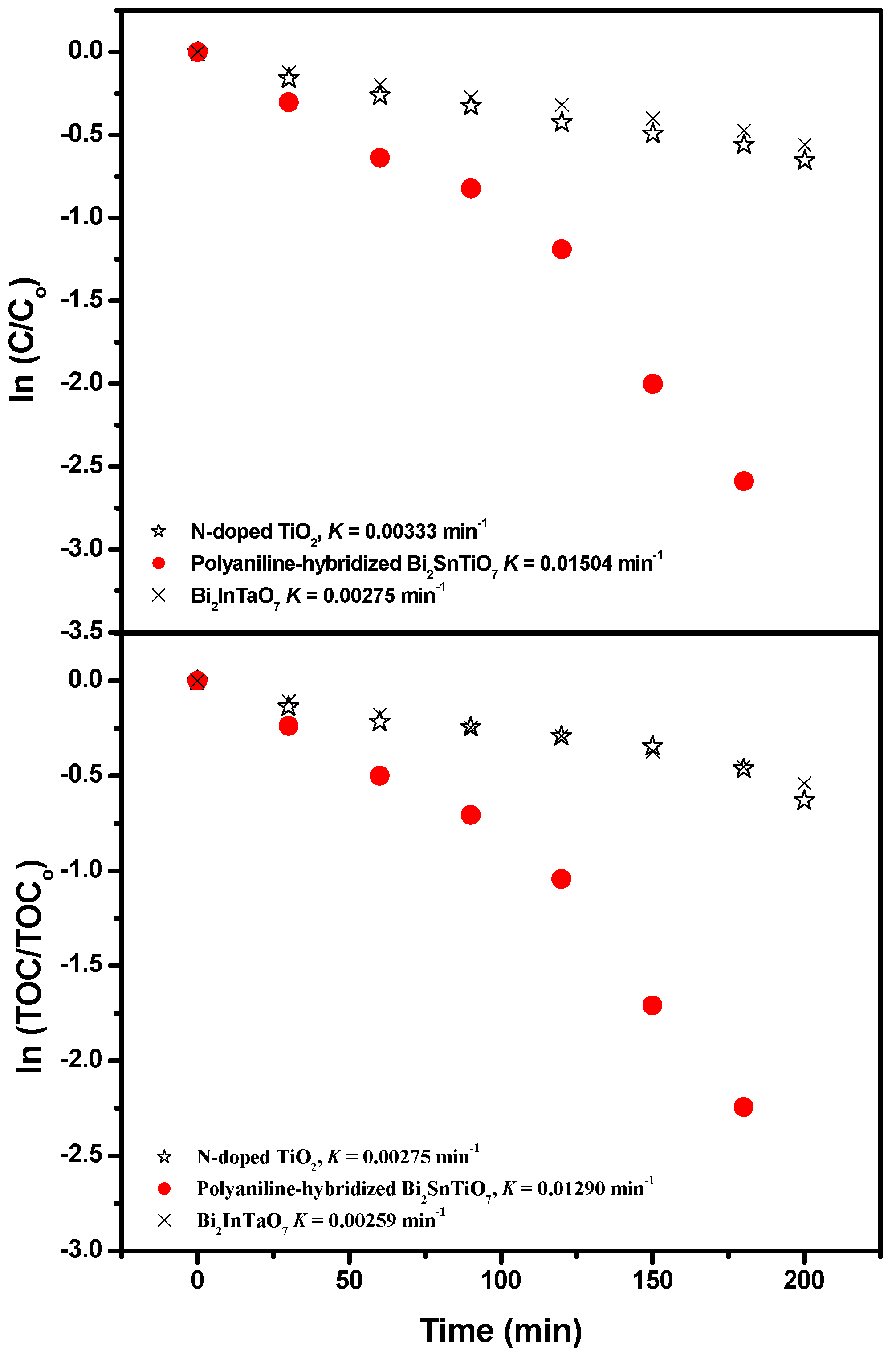
The first-order nature of the photocatalytic degradation kinetics with the polyaniline-hybridized Bi
2SnTiO
7 or Bi
2InTaO
7 or N-doped TiO
2 as catalyst is clearly demonstrated in
Figure 6. The results showed a linear correlation between ln (
C/Co) (or ln (
TOC/TOCo)) and the irradiation time for the photocatalytic degradation of MB under visible light irradiation with the presence of the polyaniline-hybridized Bi
2SnTiO
7 or Bi
2InTaO
7 or N-doped TiO
2. Here,
C represented the MB concentration at time t, and
Co represented the initial MB concentration, and
TOC represented the total organic carbon concentration at time t, and
TOCo represented the initial total organic carbon concentration. According to
Figure 6, the first-order rate constant
kC of MB concentration was estimated to be 0.01504 min
−1 with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst, 0.00275 min
−1 with Bi
2InTaO
7 as catalyst and 0.00333 min
−1 with N-doped TiO
2 as catalyst. The different value of
kC indicated that the polyaniline-hybridized Bi
2SnTiO
7 was more suitable for the photocatalytic degradation of MB under visible light irradiation than N-doped TiO
2 or Bi
2InTaO
7. Meanwhile N-doped TiO
2 was more suitable for the photocatalytic degradation of MB under visible light irradiation than Bi
2InTaO
7.
Figure 6 also showed that the first-order rate constant
KTOC of TOC was estimated to be 0.01290 min
−1 with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst, 0.00275 min
−1 with N-doped TiO
2 as catalyst and 0.00259 min
−1 with Bi
2InTaO
7 as catalyst, which indicated that the photodegradation intermediate products of MB probably appeared during the photocatalytic degradation of MB under visible light irradiation because of the different value between
kC and
KTOC. It could also be seen from
Figure 6 that the polyaniline-hybridized Bi
2SnTiO
7 showed higher mineralization efficiency for MB degradation compared with N-doped TiO
2 or Bi
2InTaO
7. At the same time, N-doped TiO
2 showed higher mineralization efficiency for MB degradation compared with Bi
2InTaO
7.
Figure 6.
Observed first-order kinetic plots for the photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
Figure 6.
Observed first-order kinetic plots for the photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
Some inorganic ions such as NH
4+, NO
3− and SO
42− were formed in parallel as the end products of nitrogen and sulfur atoms which existed in MB.
Figure 7 and
Figure 8 show the concentration variation of SO
42− and NO
3− during photocatalytic degradation of MB with the polyaniline-hybridized Bi
2SnTiO
7 or Bi
2InTaO
7 or N-doped TiO
2 as catalyst under visible light irradiation. The results showed that the concentration of NO
3− or SO
42− increased gradually with increasing reaction time when MB was photodegraded by the polyaniline-hybridized Bi
2SnTiO
7 or Bi
2InTaO
7 or N-doped TiO
2. Monitoring the presence of ions in the solution revealed that the SO
42– ion concentration was 0.01714 mM or 0.00924 mM or 0.00757 mM with the polyaniline-hybridized Bi
2SnTiO
7 or N-doped TiO
2 or Bi
2InTaO
7 as catalyst after visible light irradiation for 200 minutes, indicating that 68.56% or 36.94% or 30.28% of sulfur from MB was converted into sulfate ions with the polyaniline-hybridized Bi
2SnTiO
7 or N-doped TiO
2 or Bi
2InTaO
7 as catalyst after visible light irradiation for 200 minutes.
Figure 7.
The concentration variation of SO42− during photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
Figure 7.
The concentration variation of SO42− during photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
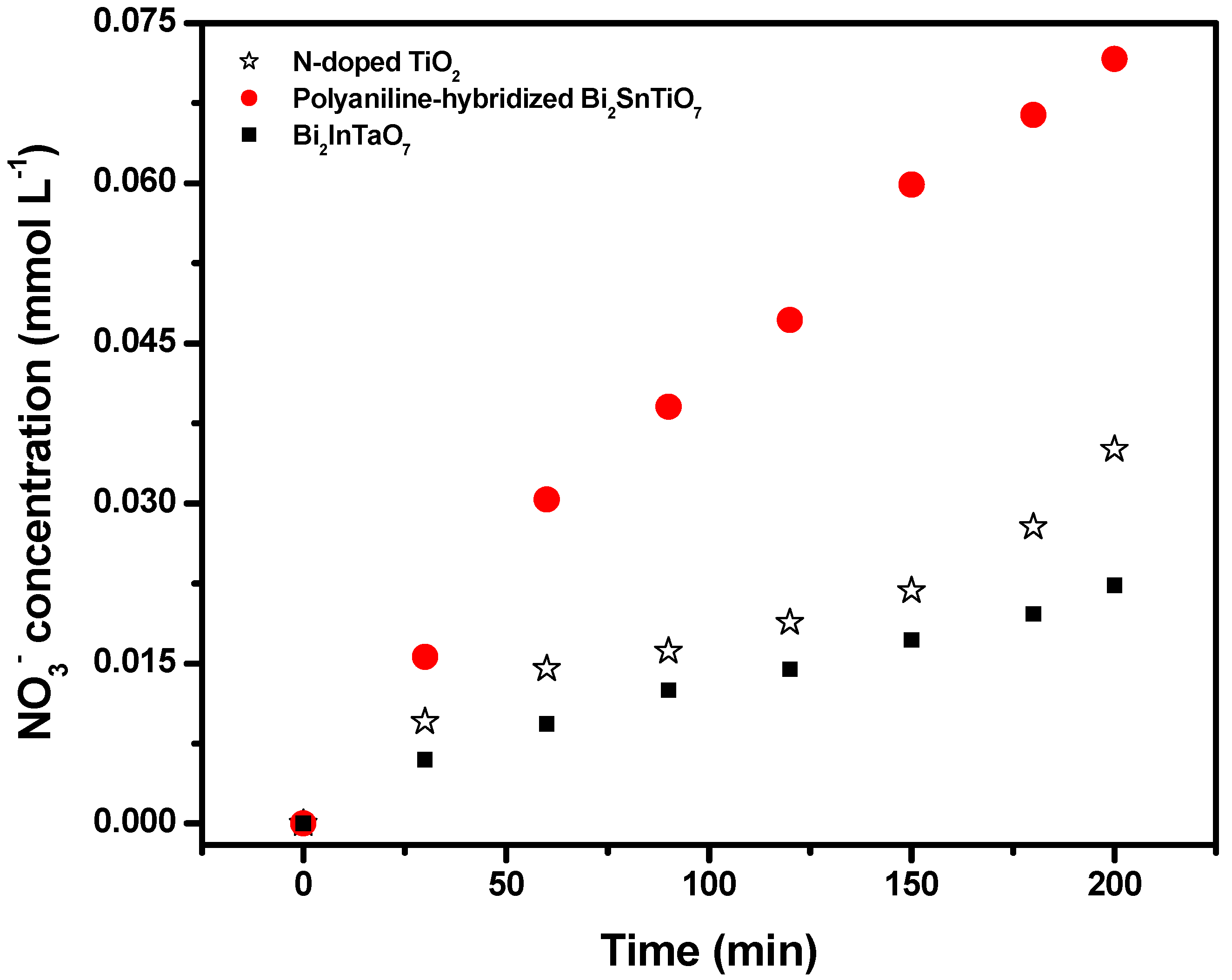
It could be seen from
Figure 8 that the NO
3− ion concentration was 0.07165 mM or 0.0351 mM or 0.02232 mM with the polyaniline-hybridized Bi
2SnTiO
7 or N-doped TiO
2 or Bi
2InTaO
7 as catalyst after visible light irradiation for 200 minutes, indicating that 95.53% or 46.80% or 29.76% of nitrogen from MB was converted into nitrate ions with the polyaniline-hybridized Bi
2SnTiO
7 or N-doped TiO
2 or Bi
2InTaO
7 as catalyst after visible light irradiation for 200 minutes. The sulfur was first hydrolytically removed, and subsequently was oxidized and transformed into SO
42−. At the same time, nitrogen atoms in the −3 oxidation state prduced NH
4+ cations that subsequently were oxidized into NO
3− ions. As expected, the formation kinetics with the polyaniline-hybridized Bi
2SnTiO
7 was significantly faster than that of N-doped TiO
2 or Bi
2InTaO
7 by using the same amount of photocatalyst. Moreover, the formation kinetics with N-doped TiO
2 was faster than that of Bi
2InTaO
7 when using the same amount of photocatalyst. It was noteworthy that the amount of SO
42− ions which was released into the solution was lower than the amount of SO
42− which should come from stoichiometry. One possible reason could be a loss of sulfur-containing volatile compounds such as SO
2. The second possible reason was a partially irreversible adsorption of some SO
42− ions on the surface of the photocatalyst which had been observed by Lachheb
et al. by titanium dioxide [
40]. Regardless whether the sulfate ions were adsorbed irreversibly on the surface or not, it was important to stress that the evidence for restrained photocatalytic activity was not noticed.
Figure 8.
The concentration variation of NO3− during photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
Figure 8.
The concentration variation of NO3− during photocatalytic degradation of methylene blue with the polyaniline-hybridized Bi2SnTiO7, Bi2InTaO7 or N-doped TiO2 as catalyst under visible light irradiation.
The photodegradation intermediate products of MB (
m/z = 284.1) in our experiment were identified as azure B (
m/z = 270.1), azure C (
m/z = 242.1), thionine (
m/z = 228.0), leucomethylene blue (
m/z = 285.0) and aniline (
m/z = 93.0). According to the intermediate products which were found in this work and the observed appearance time of other intermediate products, a possible photocatalytic degradation pathway for MB was proposed.
Scheme 1 shows the suggested photocatalytic degradation pathway scheme for methylene blue under visible light irradiation in the presence of the polyaniline-hybridized Bi
2SnTiO
7. The molecule of MB was converted to small organic species, which were subsequently mineralized into inorganic products such as SO
42− ions, NO
3− ions, CO
2 and water ultimately.
2.3. Photocatalytic Degradation Mechanism
The action spectra of MB degradation with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst was observed under visible light irradiation. A clear photonic efficiency (0.0112% at its maximal point) at wavelengths which corresponded to sub-Eg energies of the photocatalysts (
λ from 490 nm to 800 nm) was observed for the polyaniline-hybridized Bi
2SnTiO
7. The existence of photonic efficiency at this region revealed that photons were not absorbed by the photocatalysts. In particular, the correlation between the low-energy action spectrum and the absorption spectrum of MB clearly demonstrated that any photodegradation results at wavelengths above 490 nm should be attributed to photosensitization effect by the dye MB itself (
Scheme 2).
Scheme 1.
Suggested photocatalytic degradation pathway scheme for methylene blue under visible light irradiation in the presence of the polyaniline-hybridized Bi2SnTiO7.
Scheme 1.
Suggested photocatalytic degradation pathway scheme for methylene blue under visible light irradiation in the presence of the polyaniline-hybridized Bi2SnTiO7.
Scheme 2.
Photosensitization effect by the dye MB.
Scheme 2.
Photosensitization effect by the dye MB.
According to the mechanism which was shown in
Scheme 2, MB which was adsorbed on the polyaniline-hybridized Bi
2SnTiO
7 was excited by visible light irradiation. Subsequently, an electron was injected from the excited MB to the conduction band of the polyaniline-hybridized Bi
2SnTiO
7 where the electron was scavenged by molecular oxygen. Scheme I explained the results which were obtained with the polyaniline-hybridized Bi
2SnTiO
7 as catalyst under visible light irradiation, where the polyaniline-hybridized Bi
2SnTiO
7 could serve to reduce recombination of photogenerated electrons and holes by scavenging of electrons [
41].
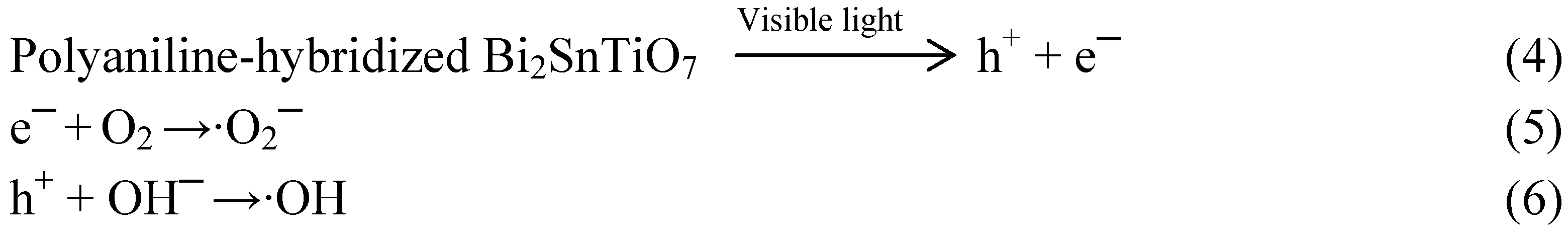
Below 490 nm, the situation was different. The results of photonic efficiency correlated well with the absorption spectra of the polyaniline-hybridized Bi
2SnTiO
7. These results evidently showed that the mechanism which was responsible for the photodegradation of MB went through band gap excitation of the polyaniline-hybridized Bi
2SnTiO
7. Despite the detailed experiments about the effect of oxygen and water were not performed, it was logical to presume that the mechanism in the first step was similar to the observed mechanism for the polyaniline-hybridized Bi
2SnTiO
7 under supra-bandgap irradiation, namely
Scheme 3:
Scheme 3.
The observed mechanism for the polyaniline-hybridized Bi2SnTiO7 under supra-bandgap irradiation.
Scheme 3.
The observed mechanism for the polyaniline-hybridized Bi2SnTiO7 under supra-bandgap irradiation.
According to first principles calculations, we deduced that the conduction band of the polyaniline-hybridized Bi2SnTiO7 was composed of Ti 3d and Sn 5p orbital component, and the valence band of the polyaniline-hybridized Bi2SnTiO7 was composed of O 2p and Bi 6s orbital component. The polyaniline-hybridized Bi2SnTiO7 could produce electron–hole pairs by absorption of photons directly, and it indicated that enough energy which was larger than the band gap energy of the polyaniline-hybridized Bi2SnTiO7 was necessary for the photocatalytic degradation process of MB.
Former luminescent studies had shown that the closer the M–O–M bond angle was to 180°, the more delocalized was the excited state [
42]. As a result, the charge carriers could move more easily in the matrix. The mobility of the photoinduced electrons and holes influenced the photocatalytic activity because high diffusivity indicated the enhancement of probability that the photogenerated electrons and holes would reach the reactive sites of the catalyst surface. According to above results, the lattice parameter a = 10.52582(8) Å for Bi
2SnTiO
7 was smaller than the lattice parameter α = 10.70352(7) Å for Bi
2InTaO
7, thus the photoinduced electrons and holes inside Bi
2SnTiO
7 was easier and faster to reach the reactive sites on the catalyst surface compared with those of Bi
2InTaO
7, which showed the photocatalytic degradation activity of Bi
2SnTiO
7 was higher than that of Bi
2InTaO
7. For the polyaniline-hybridized Bi
2SnTiO
7 in this experiment, the Sn–O–Ti bond angle was 140.196° and the In–O–Ta bond angle was 118.764°, indicating that the Sn–O–Ti or In–O–Ta bond angle was close to 180°. Thus the photocatalytic activity of the polyaniline-hybridized Bi
2SnTiO
7 was correspondingly higher. The crystal structures of Bi
2SnTiO
7 and Bi
2InTaO
7 were the same, but their electronic structures were considered to be somewhat different. For Bi
2SnTiO
7, Ti was 3
d-block metal element, and Bi was 6
p-block rare earth metal element, and Sn was 5
p-block metal element, but for Bi
2InTaO
7, Ta was 5
d-block metal element, and In was 5
p-block metal element, indicating that the crystal structure and the electronic structure of the photocatalysts affect the photocatalytic activity. Based on above analysis, the difference of photocatalytic MB degradation between Bi
2SnTiO
7 and Bi
2InTaO
7 can be attributed mainly to the difference in their crystalline structure and electronic structure. The crystal structure and the electronic structure of the polyaniline-hybridized Bi
2SnTiO
7 and N-doped TiO
2 were totally different. For N-doped TiO
2, Ti was 3
d-block metal element, indicating that the different photodegradation effect of MB between the polyaniline-hybridized Bi
2SnTiO
7 and N-doped TiO
2 could be attributed mainly to the difference of their crystalline structure and electronic structure.
The present results indicated that the polyaniline-hybridized Bi2SnTiO7-visible light photocatalysis system might be regarded as a practical method for treatment of diluted colored waste water. This system could be utilized for decolorization, purification and detoxification of textile, printing and dyeing industries in the long-day countries. Meanwhile, this system did not need high pressure of oxygen, heating or any chemical reagents. Much decolorized and detoxified water were flowed from our new system for treatment, and the results showed that the polyaniline-hybridized Bi2SnTiO7-visible light photocatalysis system might provide a valuable treatment for purifying and reusing colored aqueous effluents.