Recent Developments in the Reformatsky-Claisen Rearrangement
Abstract
:1. Introduction
2. Pioneering Works of the Reformatsky-Claisen Rearrangement
| Substrate | R1 | R2 | R3 | R4 | Solvent | Temp (°C) | Yield of 2a–d |
|---|---|---|---|---|---|---|---|
| 1a | Me | Me | H | H | PhH | 80 | 100% |
| 1b | Me | H | H | Me | PhMe | 110 | 96% |
| 1c | H | H | H | H | Xylene | 140 | <15% |
| 1d | Me | H | Ph | H | PhMe | 110 | 16% |
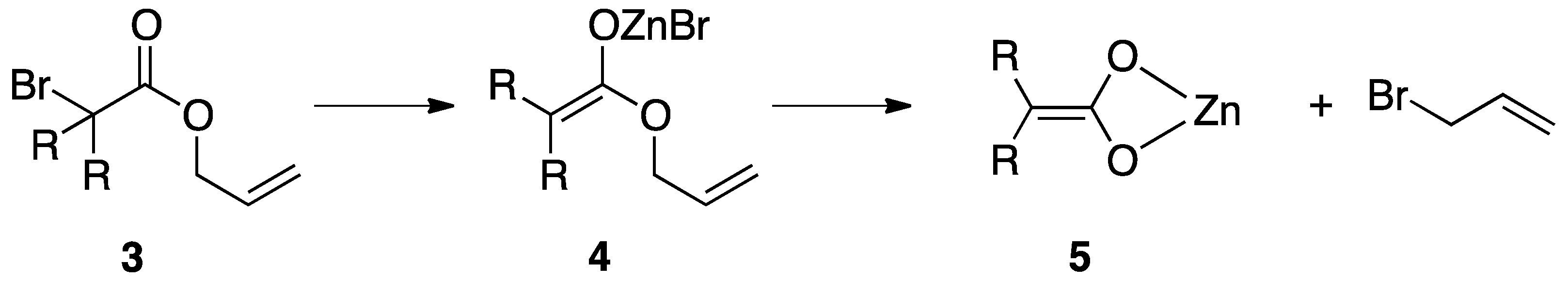
2.1. Reformatsky-Claisen Rearrangement in the Presence of Zinc and a Silylating Reagent
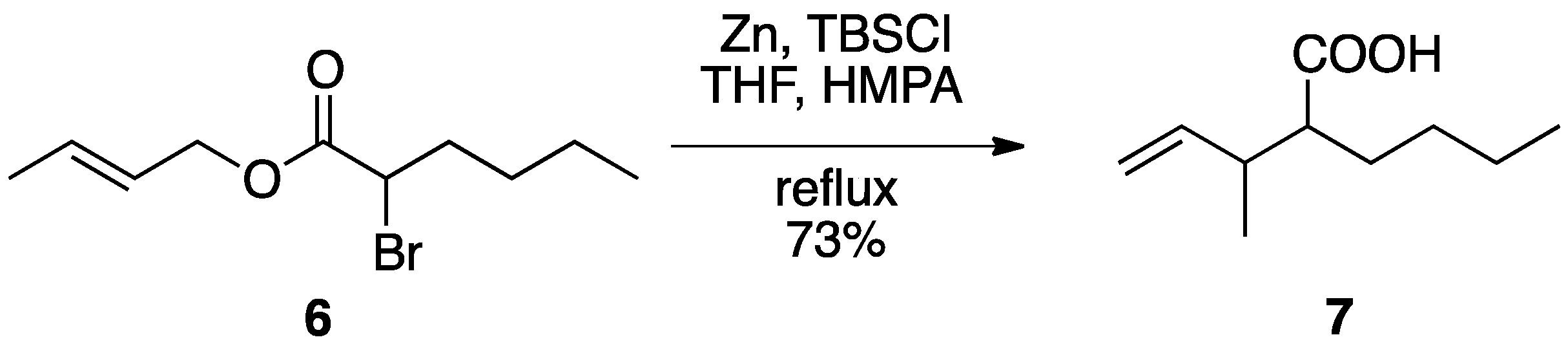

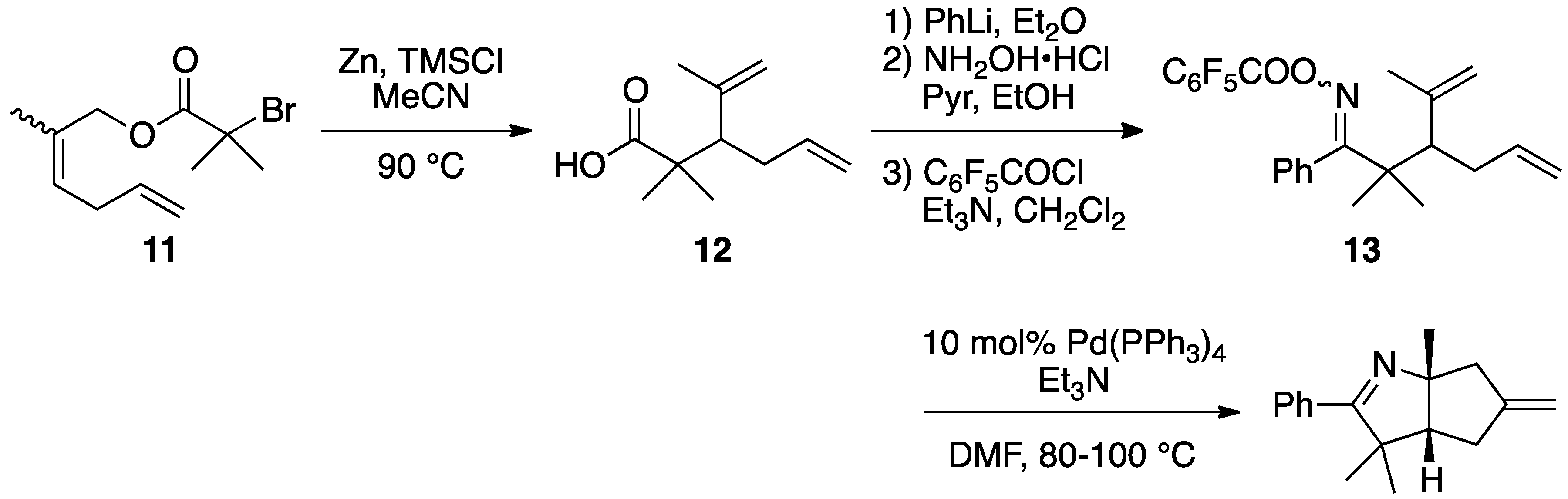
2.2. Application of Reformatsky-Claisen Rearrangement with Zinc and a Silylating Reagent
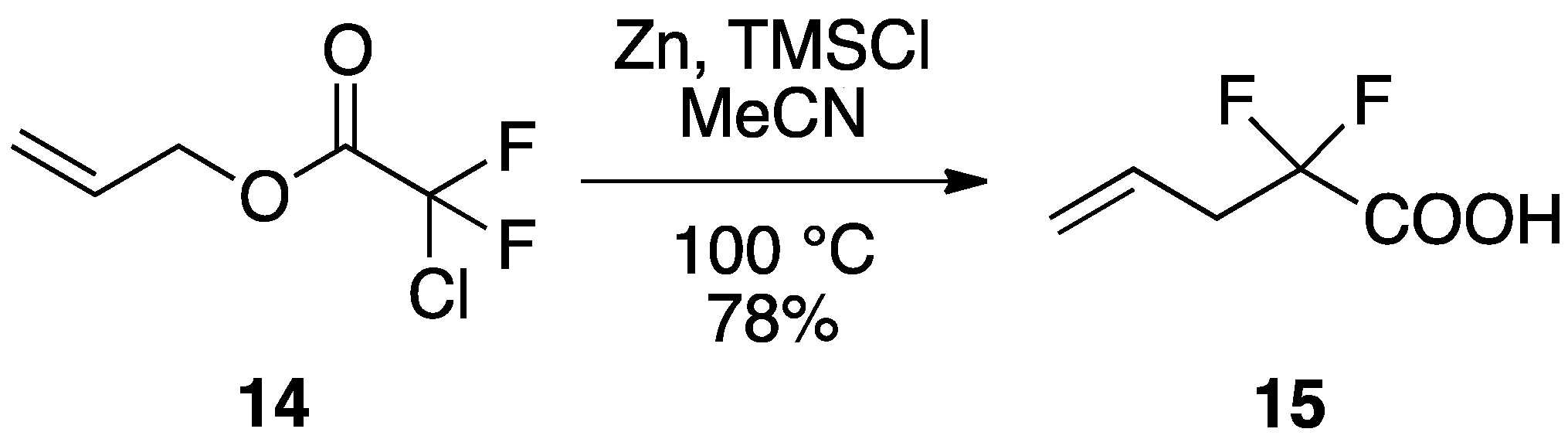
2.2.1. Preparation of α-Fluorocarboxylic Acid by Reformatsky-Claisen Rearrangement
2.2.2. Application of α-Fluorocarboxylic Acid Induced by Reformatsky-Claisen Rearrangement to Biological Active Compounds

3. Indium-Mediated Reformatsky-Claisen Rearrangement
3.1. Indium-Mediated Reformatsky-Claisen Rearrangement of α-Bromopropionates

3.2. Indium-Mediated Reformatsky-Claisen Rearrangement of Various Substrates
| Substrates | R | Method | Products | Yield | |
|---|---|---|---|---|---|
 | 39a: Me | a |  | 40a | 96% |
| 39b: H | b | 40b | 84% (1.5:1) | ||
 | 41a: Me | a |  | 42a | 94% |
| 41b: H | b | 42b | 61% (1.6:1) | ||
 | 43a: Me | a |  | 44a | 71% |
| 43b: H | b | 44b | 54% (1.4:1) | ||
 | 45a: Me | a |  | 46a | 63% |
| 45b: H | b | 46b | 62% | ||
 | 47a: Me | a |  | 48a | 91% |
| 47b: H | b | 48b | 34% |
3.3. The Reaction of Base-Sensitive Compounds

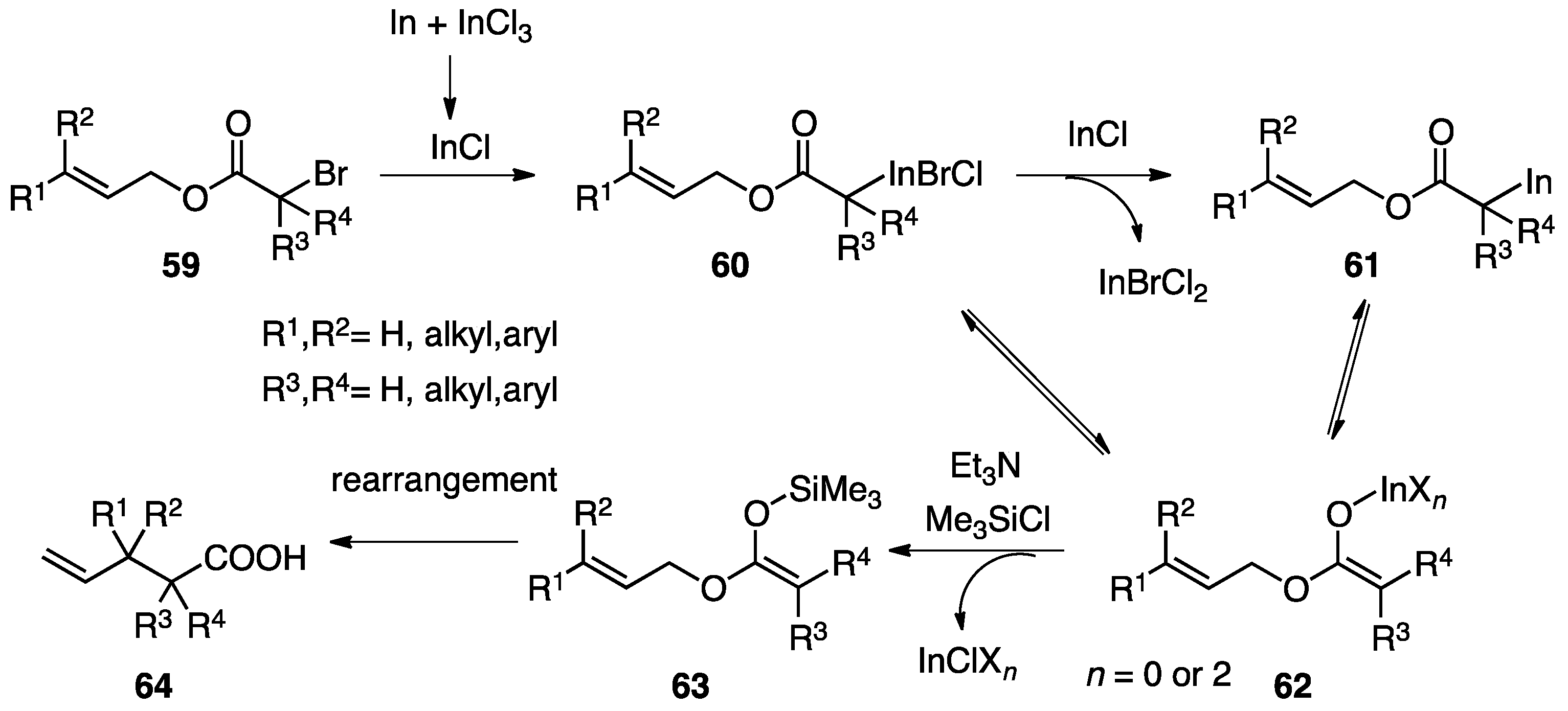
3.4. Proposed Mechanism of the Indium-Mediated Reformatsky-Claisen Rearrangement
4. Conclusions
Acknowledgments
References
- Ziegler, F.E. Stereo- and regiochemistry of the Claisen rearrangement: Applications to natural products synthesis. Acc. Chem. Res. 1977, 10, 227–232. [Google Scholar] [CrossRef]
- Tadano, K.-I. Natural products synthesis starting with carbohydrates based on the Claisen rearrangement protocol. In Studies in Natural Products Chemistry: Stereoselective Synthesis (Part F); Atta-Ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 1992; Volume 10, pp. 405–455. [Google Scholar]
- Ilardi, E.A.; Stivala, C.E.; Zakarian, A. [3,3]-Sigmatropic rearrangements: Recent applications in the total synthesis of natural products. Chem. Soc. Rev. 2009, 38, 3133–3148. [Google Scholar] [CrossRef]
- Kotha, S.; Krishna, N.G.; Halder, S.; Misra, S. A synergistic approach to polycyclics via a strategic utilization of Claisen rearrangement and olefin metathesis. Org. Biomol. Chem. 2011, 9, 5597–5624. [Google Scholar]
- The Claisen Rearrangement: Methods and Applications; Hiersemann, M.; Nubbemeyer, U. (Eds.) Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2007.
- Bennett, G.B. The Claisen rearrangement in organic synthesis: 1967 to January 1977. Synthesis 1977, 589–606. [Google Scholar] [CrossRef]
- Blechert, S. The hetero-Cope rearrangement in organic synthesis. Synthesis 1989, 71–82. [Google Scholar] [CrossRef]
- Wipf, P. Claisen rearrangements. In Comprehensive Organic synthesis; Trost, B.M., Ed.; Pergamon Press: Oxford, UK, 1991; Volume 5, pp. 827–873. [Google Scholar]
- Martin Castro, A.M. Claisen rearrangement over the past nine decades. Chem. Rev. 2004, 104, 2939. [Google Scholar] [CrossRef]
- Ireland, R.E.; Mueller, R.H. Claisen rearrangement of allyl esters. J. Am. Chem. Soc. 1972, 94, 5897–5898. [Google Scholar]
- Ireland, R.E.; Mueller, R.H.; Willard, A.K. The ester enolate Claisen rearrangement. Stereochemical control through stereoselective enolate formation. J. Am. Chem. Soc. 1976, 98, 2868–2877. [Google Scholar]
- Ireland, R.E.; Wipf, P.; Armstrong, J.D. Stereochemical control in the ester enolate Claisen rearrangement. 1. Stereoselectivity in silyl ketene acetal formation. J. Org. Chem. 1991, 56, 650–657. [Google Scholar] [CrossRef]
- Ireland, R.E.; Wipf, P.; Xiang, J.-N. Stereochemical control in the ester enolate Claisen rearrangement. 2. Chairlike vs boatlike transition-state selection. J. Org. Chem. 1991, 56, 3572–3582. [Google Scholar]
- Pereira, S.; Srebnik, M. The Ireland-Claisen rearrangement. Aldrichim. Acta 1993, 26, 17–29. [Google Scholar]
- Baldwin, J.E.; Walker, J.A. The Reformatsky-Claisen reaction, A new synthetically useful sigmatropic process. J. Chem. Soc. Chem. Commun. 1973, 117–118. [Google Scholar] [CrossRef]
- Wada, M.; Shigehisa, T.; Akiba, K. A new synthesis of medium-membered lactones via intramolecular condensation of α-chlorosulfides containing an ester group and allylsilanyl moiety. Tetrahedron Lett. 1985, 26, 5191–5194. [Google Scholar]
- Zaman, S.; Kitamura, M.; Narasaka, K. Synthesis of polycyclic imines by palladium-catalyzed domino cyclization of di- and trienylketone O-pentafluorobenzoyloximes. Bull. Chem. Soc. Jpn. 2003, 76, 1055–1062. [Google Scholar] [CrossRef]
- Greuter, H.; Lang, R.W.; Romann, A.J. Fluorine-containing organozinc reagents. V.1: The Reformatskii-claisen reaction of chlorodifluoroacetic acid derivatives. Tetrahedron Lett. 1988, 29, 3291–3294. [Google Scholar]
- Chen, L.; Kim, Y.M.; Kucera, D.J.; Harrison, K.E.; Bahmanyar, S.; Scott, J.M.; Yazbeck, D. Fluorination-free synthesis of a 4,4-difluoro-3,3-dimethylproline derivative. J. Org. Chem. 2006, 71, 5468–5473. [Google Scholar] [CrossRef]
- Zheng, F.; Zhang, X.; Qing, F.-L. Stereoselective Reformatskii–Claisen rearrangement: Synthesis of 2’,3’-dideoxy-6’,6’-difluoro-2’-thionucleosides. Chem. Commun. 2009, 1505–1507. [Google Scholar]
- Yang, Y.-Y.; Xu, J.; You, Z.-W.; Xu, X.-H.; Qiu, X.-L.; Qing, F.-L. Synthesis of 3',3'-difluoro-2'-hydroxymethyl-4',5'-unsaturated carbocyclic nucleosides. Org. Lett. 2007, 9, 5437–5440. [Google Scholar]
- Yang, Y.; Zheng, F.; Qing, F.-L. Synthesis of 2',3'-dideoxy-6'-fluorocarbocyclic nucleosides via Reformatskii–Claisen rearrangement. Tetrahedron 2011, 67, 3388–3394. [Google Scholar] [CrossRef]
- Ishihara, J.; Koyama, N.; Nishino, Y.; Takahashi, K.; Hatakeyama, S. A new variant of Reformatsky-Claisen rearrangement mediated by indium chloride. Synlett 2009, 2351–2355. [Google Scholar]
- Ishihara, J.; Watanabe, Y.; Koyama, N.; Nishino, Y.; Takahashi, K.; Hatakeyama, S. Indium-mediated Reformatsky-Claisen rearrangement. Tetrahedron 2011, 67, 3659–3667. [Google Scholar] [CrossRef]
- Chao, L.-C.; Rieke, R.D. Activated metals. IX. New reformatsky reagent involving activated indium for the preparation of β-hydroxy esters. J. Org. Chem. 1975, 40, 2253–2255. [Google Scholar]
- Babu, S.A.; Yasuda, M.; Shibata, I.; Baba, A. In- or In(I)-Employed diastereoselective Reformatsky-type reactions with ketones:1H-NMR investigations on the active species. Org. Lett. 2004, 6, 4475–4478. [Google Scholar] [CrossRef]
- Babu, S.A.; Yasuda, M.; Shibata, I.; Baba, A. In- or In(I)-employed tailoring of the stereogenic centers in the Reformatsky-type reactions of simple ketones, α-alkoxy ketones, and β-keto esters. J. Org. Chem. 2005, 70, 10408–10419. [Google Scholar] [CrossRef]
- Babu, S.A.; Yasuda, M.; Okabe, Y.; Shibata, I.; Baba, A. High chelation control of three contiguous stereogenic centers in the Reformatsky reactions of indium enolates with α-hydroxyketones: Unexpected stereochemistry of lactone formation. Org. Lett. 2006, 8, 3029–3032. [Google Scholar] [CrossRef]
- Inomata, K.; Hirata, T.; Sasada, Y.; Asada, T.; Senda, H.; Kinoshita, H. Crystallographic approach to the origin of “syn-effect”. Chem. Lett. 1990, 2153–2156. [Google Scholar]
- Hirata, T.; Sasada, Y.; Ohtani, T.; Asada, T.; Kinoshita, H.; Senda, H.; Inomata, K. “Syn-effect” in the conversion of (E)-vinylic sulfones to the corresponding allylic sulfones. Bull. Chem. Soc. Jpn. 1992, 65, 75–96. [Google Scholar] [CrossRef]
- Inomata, K. “Syn–effect” in the base–induced isomerization of vinylic sulfones to allylic sulfones and the related various reactions. J. Synth. Org. Chem. Jpn. 2009, 67, 1172–1182. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ishihara, J.; Hatakeyama, S. Recent Developments in the Reformatsky-Claisen Rearrangement. Molecules 2012, 17, 14249-14259. https://doi.org/10.3390/molecules171214249
Ishihara J, Hatakeyama S. Recent Developments in the Reformatsky-Claisen Rearrangement. Molecules. 2012; 17(12):14249-14259. https://doi.org/10.3390/molecules171214249
Chicago/Turabian StyleIshihara, Jun, and Susumi Hatakeyama. 2012. "Recent Developments in the Reformatsky-Claisen Rearrangement" Molecules 17, no. 12: 14249-14259. https://doi.org/10.3390/molecules171214249





