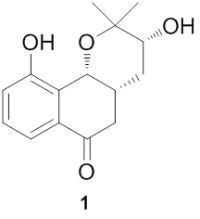
A New Prenylated Naphthoquinoid from the Aerial Parts of Clinopodium chinense (Benth.) O. Kuntze
Abstract
:1. Introduction
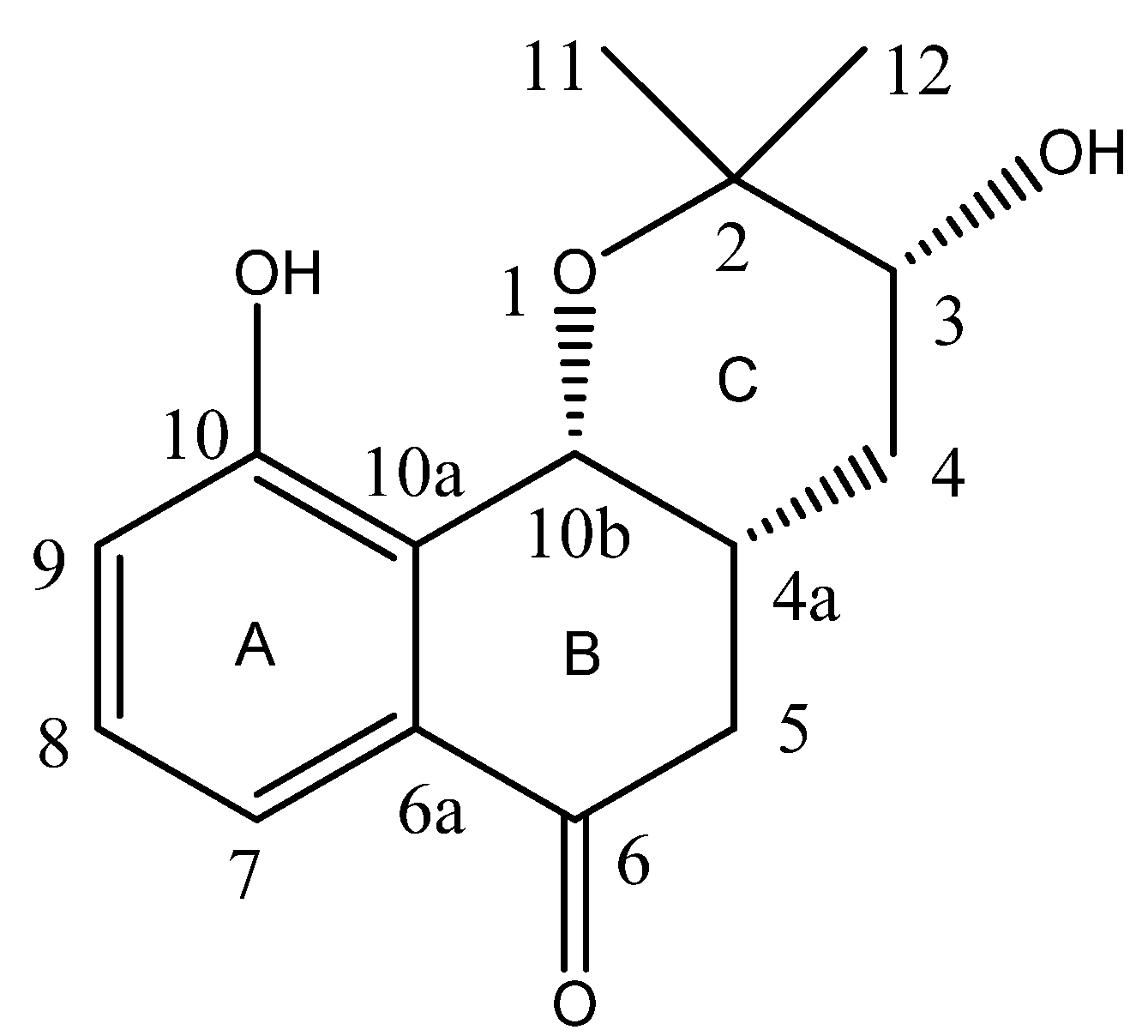
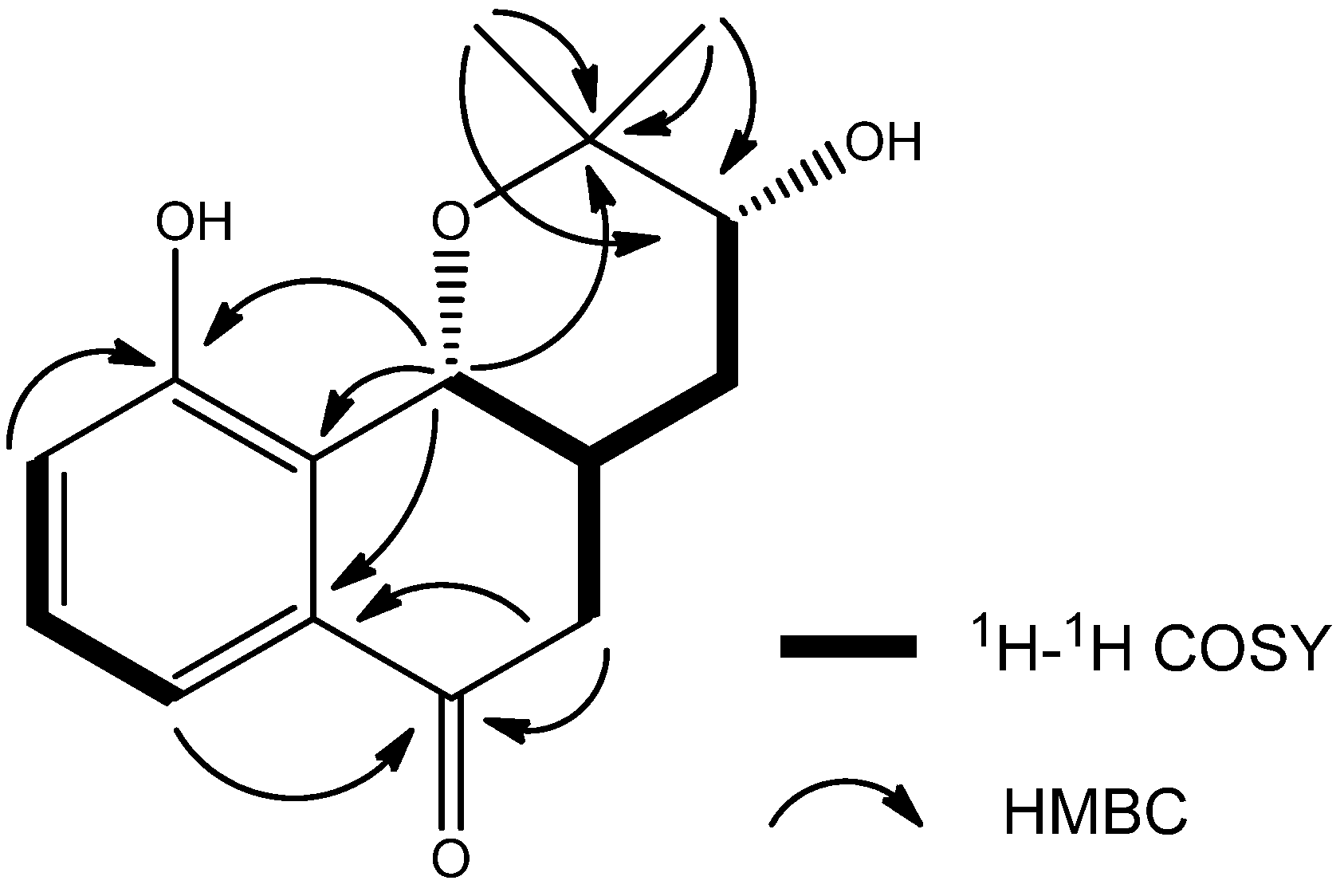
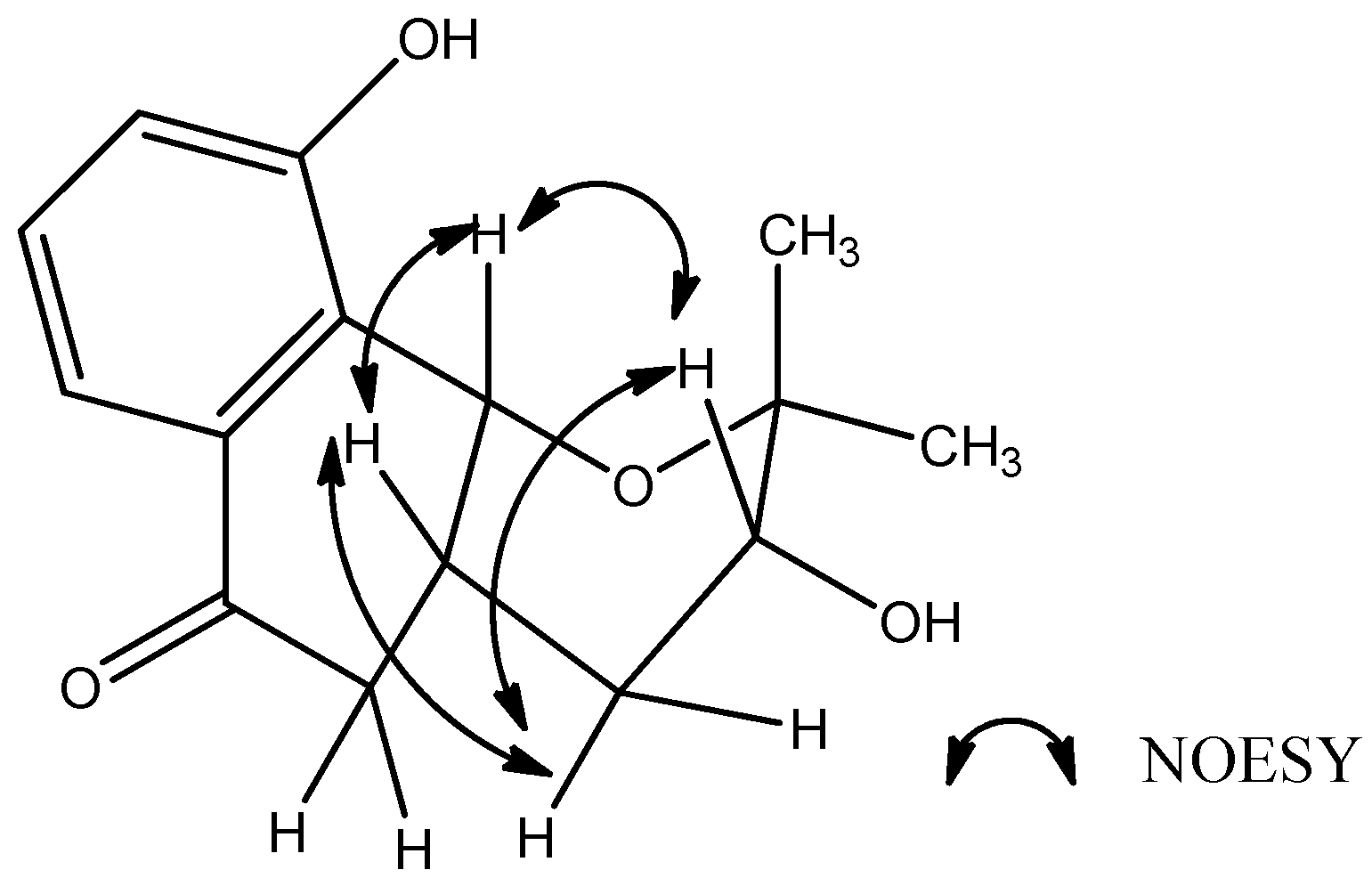
2. Results and Discussion
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectral Data
| Position | 1H(δ) | 13C(δ) | HMBC |
|---|---|---|---|
| 2 | 72.1 | ||
| 3 | 3.99 (1H, dd, J = 7.8 Hz) | 88.2 | 26.6(C-2-Me) |
| 4 | 1.63 (1H, ddd, J = 12.6, 7.8, 5.4 Hz, 4α) | 34.2 | 38.6(C-4a), 41.6(C-5), 74.3(C-10b), 88.2(C-3) |
| 2.24 (1H, dt, J = 12.6, 7.8 Hz, 4β) | 38.6(C-4a), 72.1(C-2), 74.3(C-10b), 88.2(C-3) | ||
| 4a | 2.94 (1H, m) | 38.6 | 88.2(C-3) |
| 5 | 2.59 (1H, dd, J = 15.6, 5.4 Hz, 5α) | 41.6 | 200.6(C-6), 34.2(C-4), 38.6(C-4a), 74.3(C-10b) |
| 2.86 (1H, dd, J = 15.6, 9.6 Hz, 5β) | 134.4(C-6a), 200.6(C-6), 34.2(C-4), 38.6(C-4a), 74.3(C-10b) | ||
| 6 | 200.6 | ||
| 6a | 134.4 | ||
| 7 | 7.41 (1H, d, J = 7.8 Hz) | 118.4 | 128.1(C-10a), 130.7(C-8), 200.6(C-6) |
| 8 | 7.29 (1H, ps t, J = 7.8 Hz) | 130.7 | 118.4(C-7), 122.4(C-9), 128.1(C-10a) |
| 9 | 7.11 (1H, d, J = 7.8 Hz) | 122.4 | 128.1(C-10a), 158.5(C-10) |
| 10 | 158.5 | ||
| 10a | 128.1 | ||
| 10b | 5.14 (1H, d, J = 5.4 Hz) | 74.3 | 128.1(C-10a), 134.4(C-6a), 158.5(C-10), 41.6(C-5), 34.2(C-4) |
| 11-Me | 1.17 (3H, s) | 26.6 | 72.1(C-2), 88.2(C-3) |
| 12-Me | 1.09 (3H, s) | 25.7 | 72.1(C-2), 88.2(C-3) |
4. Conclusions
Acknowledgments
References
- Chi, H.D.; Lu, J.C. Advances in studies on medicinal plants in Clinopodium. J. Shenyang Pharm. Univ. 2007, 6, 100–110. [Google Scholar]
- Zhong, M.L.; Xu, X.D.; Yu, S.C.; Sun, G.L. Advances in studies on medicinal plants in Clinopodium Linn. Chin. Tradit. Herbal Drugs 2012, 43, 820–828. [Google Scholar]
- Miyase, T.; Matsushima, Y. Saikosaponin homologs from Clinopodium spp. The structures of clinoposaponins XII–XX. Chem. Pharm. Bull. 1997, 45, 1493–1497. [Google Scholar] [CrossRef]
- Liu, Z.M.; Jia, Z.J.; Gates, R.G.; Li, D.; Owen, N. Triterpenoid saponins from Clinopodium.chinensis. J. Nat. Prod. 1995, 58, 184–188. [Google Scholar] [CrossRef]
- Macambira, L.M.A.; Andrade, C.H.S.; Matos, F.J.A.; Craveiro, A.A.; Braz Filho, R. Naphthoquinoids from Lippia Sidoids. J. Nat. Prod. 1986, 49, 310–312. [Google Scholar] [CrossRef]
- Kizu, H.; Habe, S.; Ishida, M.; Tomimori, T. Studies on the nepalese crude drugs. XVII. On the naphthalene related compounds from the root bark of Oroxylum indicum Vent. Yakugaku Zasshi 1994, 114, 492–513. [Google Scholar]
- Bringmann, G.; Messer, K.; Saeb, W.; Peters, E.M.; Peters, K. The absolute configuration of (+)-isoshinaolone and in situ LC-CD analysis of its stereoisomer from crude extracts. Phytochemistry 2001, 56, 387–391. [Google Scholar]
- Fathiazad, F.; Ddlazar, A.; Amiri, R.; Sarker, S. Extraction of flavonoids and quantification of rutin from waste tobacco leaves. Iran. J. Pharm. Res. 2006, 3, 222–227. [Google Scholar]
- Owen, R.W.; Haubner, R.; Mier, W.; Giacosa, A.; Hull, W.E.; Spiegelhalder, B.; Bartsch, H. Isolation, structure elucidation and antioxidant potential of the major phenolic and flavonoid compounds in brined olive drupes. Food Chem. Toxicol. 2003, 41, 703–717. [Google Scholar] [CrossRef]
- Li, F.; Meng, F.; Xiong, Z.; Li, Y.; Liu, R.; Liu, H. Stimulative activity of Drynaria fortunei (Kunze) J. Sm. extracts and two of its flavonoids on the proliferation of osteoblastic like cells. Pharmazie 2006, 61, 962–965. [Google Scholar]
- Yamauchi, K.; Mitsunaga, T.; Batubara, I. Isolation, identification and tyrosinase inhibitory avtivities of the extractives from Allamanda cathartica. Nat. Resour. 2011, 2, 167–172. [Google Scholar]
- Shimoda, K.; Kubota, N.; Taniuchi, K.; Sato, D.; Nakajima, N.; Hamada, H.; Hamada, H. Biotransformation of naringin and naringenin by cultured Eucalyptus perriniana cells. Phytochemistry 2010, 71, 201–205. [Google Scholar] [CrossRef]
- Liu, R.M.; Kong, L.Y.; Li, A.F.; Sun, A.L. Preparative Isolation and Purification of Saponin and Flavone Glycoside Compounds from Clinopodium chinense (Benth) O. Kuntze by High Speed Countercurrent Chromatography. J. Liq. Chromatogr. Relat. Technol. 2007, 30, 521–532. [Google Scholar] [CrossRef]
- Sample Availability: Samples of compounds 1–7are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhong, M.; Sun, G.; Zhang, X.; Sun, G.; Xu, X.; Yu, S. A New Prenylated Naphthoquinoid from the Aerial Parts of Clinopodium chinense (Benth.) O. Kuntze. Molecules 2012, 17, 13910-13916. https://doi.org/10.3390/molecules171213910
Zhong M, Sun G, Zhang X, Sun G, Xu X, Yu S. A New Prenylated Naphthoquinoid from the Aerial Parts of Clinopodium chinense (Benth.) O. Kuntze. Molecules. 2012; 17(12):13910-13916. https://doi.org/10.3390/molecules171213910
Chicago/Turabian StyleZhong, Mingliang, Guibo Sun, Xiaopo Zhang, Guangli Sun, Xudong Xu, and Shichun Yu. 2012. "A New Prenylated Naphthoquinoid from the Aerial Parts of Clinopodium chinense (Benth.) O. Kuntze" Molecules 17, no. 12: 13910-13916. https://doi.org/10.3390/molecules171213910