An Efficient One-Pot Synthesis of Pyrano[3,2-c]quinolin-2,5-dione Derivatives Catalyzed by L-Proline
Abstract
:1. Introduction
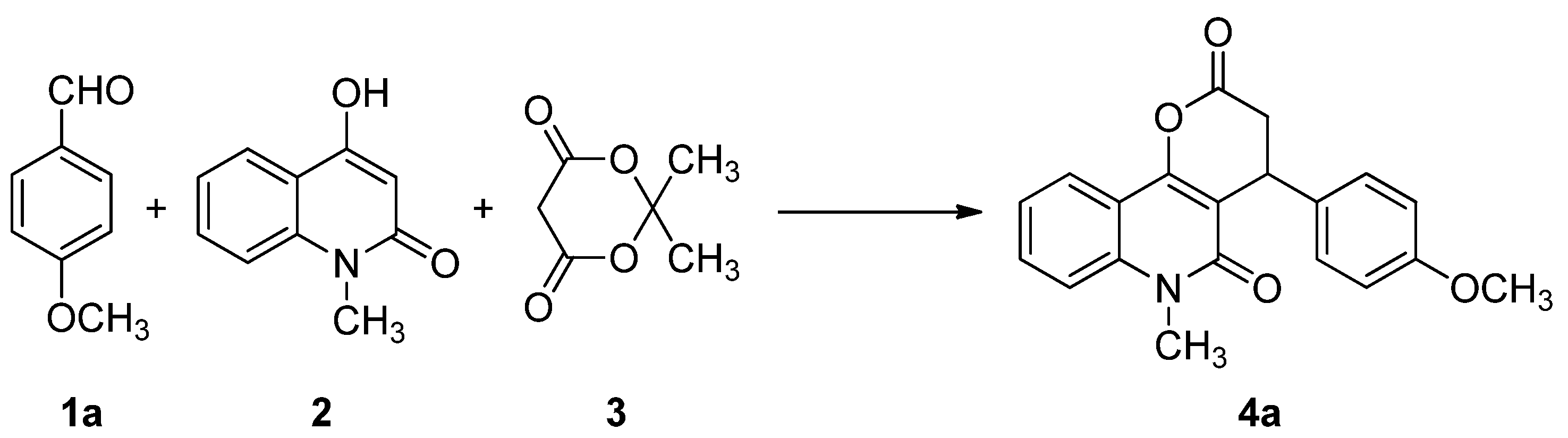
2. Results and Discussion
| Entry | Solvents | Catalyst (mol%) | Temperature (°C) | Time (h) | Yield (%) |
|---|---|---|---|---|---|
| 1 | EtOH | No | reflux | 5 | 45 |
| 2 | EtOH | L-proline (10%) | reflux | 1 | 91 |
| 3 | CH3CN | L-proline (10%) | reflux | 2 | 70 |
| 4 | CHCl3 | L-proline (10%) | reflux | 2 | 63 |
| 5 | HOAc | L-proline (10%) | 100 | 2 | 80 |
| 6 | DMF | L-proline (10%) | 100 | 2 | 52 |
| 7 | H2O | L-proline (10%) | reflux | 4 | 45 |
| 8 | EtOH | L-proline (5%) | reflux | 2 | 75 |
| 9 | EtOH | L-proline (15%) | reflux | 1 | 90 |
| 10 | EtOH | L-proline (20%) | reflux | 1 | 91 |

| Entry | Ar | Product | Time (h) | Yield (%) |
|---|---|---|---|---|
| 1 | 4-CH3OC6H4 | 4a | 2 | 91 |
| 2 | 4-BrC6H4 | 4b | 1 | 94 |
| 3 | 4-HOC6H4 | 4c | 1 | 95 |
| 4 | 4-(CH3)2NC6H4 | 4d | 1.5 | 93 |
| 5 | Thiophen-2-yl | 4e | 2 | 92 |
| 6 | 3-ClC6H4 | 4f | 1.5 | 95 |
| 7 | 4-ClC6H4 | 4g | 1 | 92 |
| 8 | 4-FC6H4 | 4h | 2 | 90 |
| 9 | 3,4-(CH3)2C6H3 | 4i | 2.5 | 91 |

3. Experimental
General
4. Conclusions
Acknowledgments
- Sample Availability: Samples of the compounds 4 are available from the authors.
References
- Nair, V.; Rajesh, C.; Vinod, A.U.; Bindu, S.; Sreekanth, A.R.; Mathen, J.S.; Balagopal, L. Strategies for heterocyclic construction via novel multicomponent reactions based on isocyanides and nucleophilic carbenes. Acc. Chem. Res. 2003, 36, 899–907. [Google Scholar] [CrossRef]
- Domling, A.; Ugi, I. Multicomponent reactions with isocyanides. Angew. Chem. Int. Ed. Engl. 2000, 39, 3168–3210. [Google Scholar] [CrossRef]
- Domling, A. Recent Developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev. 2006, 106, 17–89. [Google Scholar]
- Zhu, J.; Bienaymé, H. Multicomponent Reactions; Wiley-VCH: Weinheim, Germany, 2005; pp. 33–75. [Google Scholar]
- Schnur, R.C. Spiro-quinolylhydantoins. U.S. Patent 4,176,185, 27 November 1979. [Google Scholar]
- Atwal, K.S.; McCullough, J.R.; Hedberg, A.; Conder, M.L.; Ahmed, S.Z.; Cucinotta, G.; Normandin, D.E. The discovery of a novel calcium channel blocker related to the structure of potassium channel opener cromakalim. Bioorg. Med. Chem. Lett. 1992, 2, 1475–1478. [Google Scholar] [CrossRef]
- Magdy, A.I.; Hany, M.H.; Yassin, A.A.G.; Youssef, A.S.A. Novel heterocyclic derivatives of pyrano[3,2-c]quinolinone from 3-(1-ethy1-4-hydroxy-2-oxo-2(1H)-quinolin-3-yl)-3-oxopropanoic acid. Eur. J. Chem. 2010, 1, 195–199. [Google Scholar] [CrossRef]
- Wang, X.S.; Zeng, Z.S.; Shi, D.Q.; Wei, X.Y.; Zong, Z.M. One-pot synthesis of 2-amino-3-cyano-4-aryl-1,4,5,6-tetrahydropyrano[3,2-c]quinolin-5-one derivatives using KF-Al2O3 as catalyst. Synth. Commun. 2004, 34, 3021–3027. [Google Scholar] [CrossRef]
- Wang, X.S.; Shi, D.Q.; Tu, S.J. Synthesis of 2-aminochromene derivatives catalyzed by KF/Al2O3. Chin. J. Chem. 2003, 21, 1114–1117. [Google Scholar]
- Wang, X.S.; Zeng, Z.S.; Shi, D.Q.; Tu, S.J.; Wei, X.Y.; Zong, Z.M. One-pot synthesis of 2-amino-4-aryl-5,6-dihydro-4H-pyrano[3,2-c]quinolin-5-one-3-carboxylate derivatives catalyzed by KF-alumina. Chin. J. Org. Chem. 2005, 25, 579–582. [Google Scholar]
- Vetrivel, N.; Senniappan, T.S.; Helen, P.B.; Sellappan, M.; Thangaian, D.T. Microwave solvent-free condition synthesis and pharmacological evaluation of pyrano[3,2-c]quinolines. Med. Chem. Res. 2012, 21, 2902–2910. [Google Scholar] [CrossRef]
- Igor, V.M.; Madhuri, M.; Marcia, A.O.; Adriana, S.D.; Snezna, R.; Severine, V.S.; Wim, F.A.S.; Nikolai, M.E.; Pavel, Y.U.; Eerik, M.E.; et al. Structural simplification of bioactive natural products with multicomponent synthesis. 2. Antiproliferative and antitubulin activities of pyrano[3,2-c]pyridones and pyrano[3,2-c] quinolones. J. Med. Chem. 2008, 51, 2561–2570. [Google Scholar] [CrossRef]
- Shi, C.L.; Shi, D.Q.; Kim, S.H.; Huang, Z.B.; Ji, S.J.; Ji, M. A novel and efficient one-pot synthesis of furo[3',4':5,6]pyrido[2,3-c]pyrazole derivatives using organocatalysts. Tetrahedron 2008, 64, 2425–2432. [Google Scholar] [CrossRef]
- Shi, C.L.; Shi, D.Q.; Kim, S.H.; Huang, Z.B.; Ji, M. A novel and efficient synthesis of 3,3'-benzylidenebis(4-hydroxy-6-methylpyridin-2(1H)-one) derivatives through a multi-component reaction catalyzed by L-proline. Aust. J. Chem. 2008, 61, 547–551. [Google Scholar] [CrossRef]
- Shi, C.L.; Wang, J.X.; Chen, H.; Shi, D.Q. Regioselective synthesis and in vitro anticancer activity of 4-aza-podophyllotoxin derivatives catalyzed by L-proline. J. Comb. Chem. 2010, 12, 430–434. [Google Scholar] [CrossRef]
- Li, Y.L.; Chen, H.; Shi, C.L.; Shi, D.Q.; Ji, S.J. Efficient one-pot synthesis of spirooxindole derivatives catalyzed by L-proline in aqueous medium. J. Comb. Chem. 2010, 12, 231–237. [Google Scholar] [CrossRef]
- Wang, H.Y.; Li, L.L.; Lin, W.; Xu, P.; Huang, Z.B.; Shi, D.Q. An efficient synthesis of pyrrolo[2,3,4-kl]acridin-1-one derivatives catalyzed by L-proline. Org. Lett. 2012, 14, 4598–4601. [Google Scholar] [CrossRef]
- Alcaide, B.; Almendros, P.; Luna, A.; Torres, M.S. Proline-catalyzed diastereoselective direct Aldol reaction between 4-oxoazoetidine-2-carbaldehydes and ketones. J. Org. Chem. 2006, 71, 4818–4822. [Google Scholar] [CrossRef]
- Zotova, N.; Franzke, A.; Armstrong, A.; Blackmond, D.G. Clarification of the role of water in proline-mediated Aldol reactions. J. Am. Chem. Soc. 2007, 129, 15100–15101. [Google Scholar]
- Janey, J.M.; Hsiao, Y.; Armstrong, J.D., III. Proline-catalyzed, Asymmetric Mannich reactions in the synthesis of a DPP-IV inhibitor. J. Org. Chem. 2006, 71, 390–392. [Google Scholar] [CrossRef]
- Kantam, M.L.; Rajasekhar, C.V.; Gopikrishna, G.; Reddy, K.R.; Choudary, B.M. Proline catalyzed two-component, three-component and self-asymmetric Mannich reactions promoted by ultrasonic conditions. Tetrahedron Lett. 2006, 47, 5965–5967. [Google Scholar] [CrossRef]
- Ramachary, D.B.; Chowdari, N.S.; Barbas, C.F., III. Organocatalytic asymmetric Domino Knoevenagel/Diels-Alder reactions: A bioorganic approach to the diastereospecific and enantioselective construction of highly substituted spiro[5,5]undecane-1,5,9-triones. Angew. Chem. Int. Ed. Engl. 2003, 115, 4365–4369. [Google Scholar] [CrossRef]
- Rasalkar, M.S.; Potdar, M.K.; Mohile, S.S.; Salunkhe, M.M. An ionic liquide influenced L-proline catalyzed asymmetric Michael addition of ketones to nitrostyrene. J. Mol. Catal. A Chem. 2005, 235, 267–270. [Google Scholar] [CrossRef]
- Ramachary, D.B.; Reddy, G.B. Towards organo-click reactions: Development of pharmaceutical ingredients by using direct organo catalytic bio-mimetic reductions. Org. Biomol. Chem. 2006, 4, 4463–4468. [Google Scholar] [CrossRef]
- Ramachary, D.B.; Kishore, M. Organocatalytic sequential one-pot double cascade asymmetric synthesis of Wieland-Miescher ketone analogues from a Knoevenagel/hydrogenation/Robinson Annulation sequence: Scope and applications of organocatalytic biomimetic reductions. J. Org. Chem. 2007, 72, 5056–5068. [Google Scholar] [CrossRef]
- Ramachary, D.B.; Ramakumar, K.; Narayana, V.V. Organocatlytic cascade reactions based on Push-Pull dienamine platform: Synthesis of highly substituted anilines. J. Org. Chem. 2007, 72, 1458–1463. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhu, S.; Wang, J.; Xu, Z.; Li, J. An Efficient One-Pot Synthesis of Pyrano[3,2-c]quinolin-2,5-dione Derivatives Catalyzed by L-Proline. Molecules 2012, 17, 13856-13863. https://doi.org/10.3390/molecules171213856
Zhu S, Wang J, Xu Z, Li J. An Efficient One-Pot Synthesis of Pyrano[3,2-c]quinolin-2,5-dione Derivatives Catalyzed by L-Proline. Molecules. 2012; 17(12):13856-13863. https://doi.org/10.3390/molecules171213856
Chicago/Turabian StyleZhu, Songlei, Jing Wang, Zhou Xu, and Jie Li. 2012. "An Efficient One-Pot Synthesis of Pyrano[3,2-c]quinolin-2,5-dione Derivatives Catalyzed by L-Proline" Molecules 17, no. 12: 13856-13863. https://doi.org/10.3390/molecules171213856




