A Novel Antibacterial Compound from Siegesbeckia glabrescens
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of Active Compound from S. glabrescens
2.2. Structure Determination of Isolated Compound
| Position | 13C (δ) | DEPT | 1H (δ) a (multiplicity, J) | 1H-1H COSY | HMBC b (1H→13C) |
|---|---|---|---|---|---|
| 1' | 173.5 | CO | |||
| 2' | 33.9 | CH2 | 2.33 (t, 7.0, 7.5) | H-3' | C-1', C-3', C-4' |
| 3' | 24.9 | CH2 | 1.62 (m) | H-2', H-4' | C-1', C-2', C-4' |
| 4' | 29.0 | CH2 | 1.29 (br) | ||
| 5' | 29.2 | CH2 | 1.29 (br) | ||
| 6' | 29.3 | CH2 | 1.29 (br) | ||
| 7' | 29.4 | CH2 | 1.29 (br) | ||
| 8', 9' | 29.6 | 2(CH2) | 1.29 (br) | ||
| 10' | 31.9 | CH2 | 1.29 (br) | ||
| 11' | 22.6 | CH2 | 1.29 (br) | ||
| 12' | 13.3 | CH3 | 0.90 (t, 7.0) | H-11' | C-11', C-10' |
| 1 | 169.8 | CO | |||
| 2 | 72.0 | CH | 5.20 (d, 4.5) | H-3 | C-1, C-7, C-3 |
| 3 | 76.5 | CH | 5.11 (dd, 7.5, 7.0) | H-2, H-4 | C-1', C-1, C-2, C-4, C-5, C-6 |
| 4 | 29.0 | CH | 2.16 (m) | H-3, H-5, H-6 | C-3, C-2, C-5, C-6 |
| 5 | 17.3 | CH3 | 0.93 (d, 6.5) | H-4 | C-3, C-4, C-6 |
| 6 | 18.4 | CH3 | 1.01 (d, 6.5) | H-4 | C-3, C-4, C-5 |
| 7 | 176.3 | CO | |||
| 8 | 33.9 | CH | 2.63 (m) | H-9, H-10 | C-7, C-9, C-10 |
| 9, 10 | 18.0 | 2(CH3) | 1.19 (dd, 7.0) | H-10, H-8 | C-7, C-8 |

2.3. Antimicrobial Activity of Isolated Compound
| Compounds | Diameter of clear zone (mm) | |||
|---|---|---|---|---|
| 6.25 | 12.50 | 25.00 | 50.00 a | |
| Lauric acid | - b | - | - | 11 c |
| 3-(Dodecanoyloxy)-2(isobutyryloxy)-4-methylpentanoic acid | 11 | 12 | 15 | 19 |
| Erythromycin | 40 | 40 | 42 | 46 |
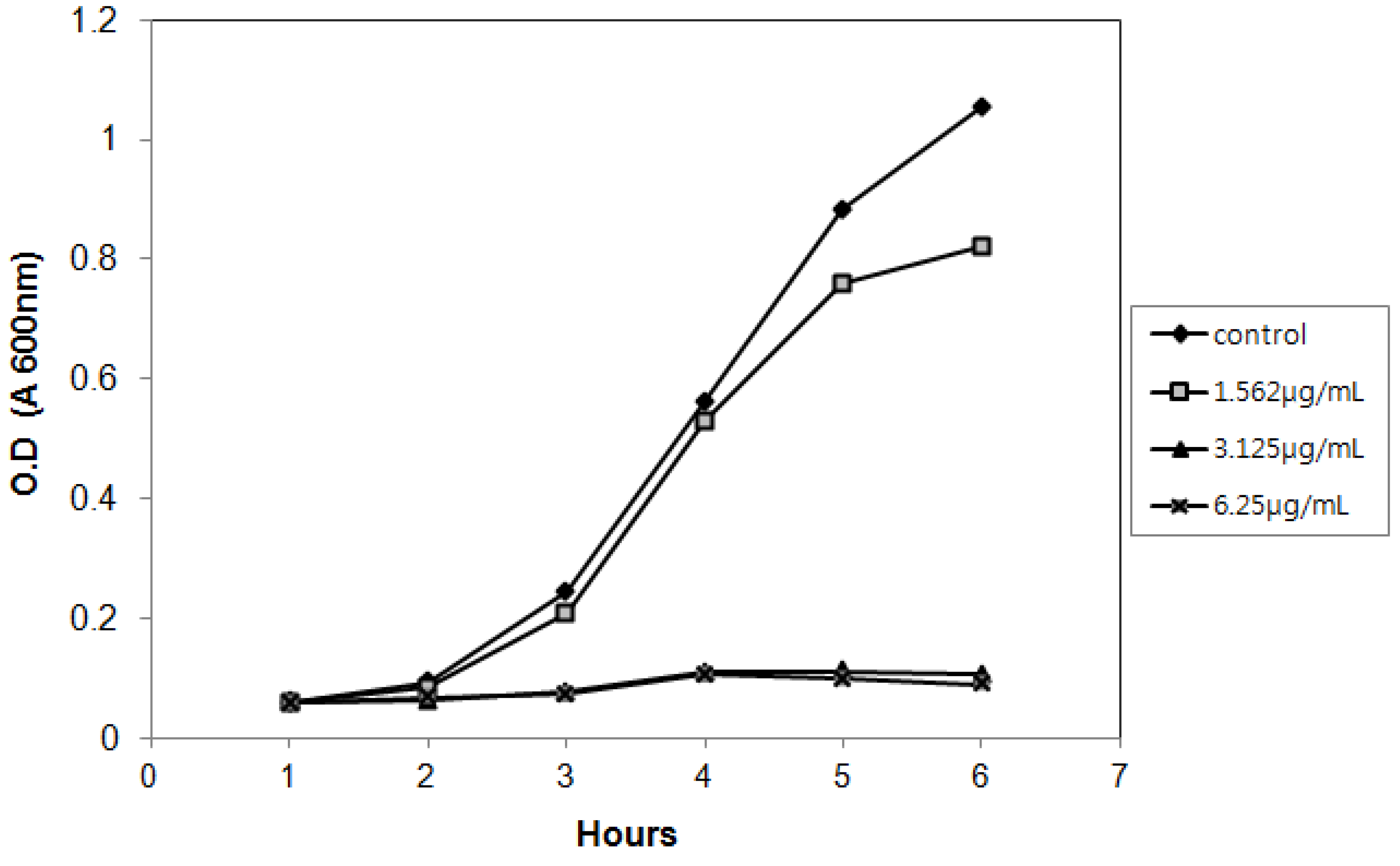
| Organism | MIC (μg/mL) | Organism | MIC (μg/mL) | |
|---|---|---|---|---|
| Gram-positive bacteria | B. subtilis | 6.25 | S. epidermidis | 25.00 |
| E. faecalis | 25.00 | S. schleiferi | 12.50 | |
| P. acnes | 25.00 | S. agalactiae | 6.25 | |
| S. aureus | 3.12 | S. pyrogens | 6.25 | |
| Gram-negative bacteria | E. coli | - a | P. aeruginosa | - |
| yeast | C. alibicans | - | F. neoformans | - |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Antibacterial Activity Assay
4. Conclusions
Conflict of Interest
Acknowledgements
References
- Centers for Disease Control and Prevention. Update: Staphylococcus aureus with reduced susceptibility to vancomycin–United States, 1997. Morb. Mortal. Wkly. Rep. 1997, 46, 813–815.
- Centers for Disease Control and Prevention. Nosocomial enterococci resistant to vancomycin: United States, 1989–1993: National Nosocomial Infection Surveillance. Morb. Mortal. Wkly. Rep. 1993, 42, 597–599.
- Archer, G.L. Staphylococcus aureus: A well-armed pathogen. Clin. Infect. Dis. 1998, 26, 1179–1181. [Google Scholar]
- Richard, M.J.; Edwards, J.R.; Culver, D.H.; Gaynes, R.P. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit. Care Med. 1999, 27, 887–892. [Google Scholar] [CrossRef]
- Nakata, K.; Inoue, Y.; Harada, J.; Maeda, N.; Watanabe, H.; Tano, Y.; Shimomura, Y.; Harino, S.; Sawa, M. A high incidence of Staphylococcus aureus colonization in the externall eyes of patients with atopic dermatitis. Ophthalmology 2000, 107, 2167–2171. [Google Scholar] [CrossRef]
- Shanmuganathan, V.A.; Armstrong, M.; Buller, A.; Tullo, A.B. External ocular infections due to methicillin-resistant Staphylococcus aureus (MRSA). Eye 2005, 19, 284–291. [Google Scholar] [CrossRef]
- Brul, S.; Coote, P. Preservative agents in foods: Mode of action and microbial resistance mechanisms. Int. J. Food Microbiol. 1999, 50, 1–17. [Google Scholar] [CrossRef]
- Zink, D.L. The impact of consumer demands and trends on food processing. Emerging Infect. Dis. 1997, 3, 467–469. [Google Scholar] [CrossRef]
- Wasserman, S.I.; Marquardt, D.L. Anaphlaxis in Allergy: Principles and Practice, 3rd ed; CV Mosby: St. Louis, MO, USA, 1988; p. 1365. [Google Scholar]
- Kodicek, E. The effect of unsaturated fatty acids on Gram-positive bacteria. Symp. Soc. Exp. Biol. 1988, 3, 217–232. [Google Scholar]
- Pollock, M.R. The effects of long-chain fatty acids on the growth of Haemophilus pertussis and other organisms. Symp. Soc. Exp. Biol. 1949, 3, 193–216. [Google Scholar]
- Kitahara, T.; Koyama, N.; Matsuda, J.; Aoyama, Y.; Hirakata, Y.; Kamihira, S.; Kohno, S.; Nakashima, M.; Sasaki, H. Antimicrobial activity of saturated fatty acids and fatty amines against methicillin-resistant Staphylococcus aureus. Biol. Pharm. Bull. 2004, 27, 1321–1326. [Google Scholar] [CrossRef]
- Cheesbrough, M. Medical Laboratory Manual for Tropical Countries: Microbiology; Tropical Health Technology: Oxford, UK, 1984; pp. 196–205. [Google Scholar]
- Gatsing, D.; Mbah, J.A.; Garba, I.H.; Tane, P.; Djemgou, P.; Nji-Nkah, B.F. An Antisalmonellal agent from the leaves of Glossocalyx brevipes Benth (Monimiaceae). Pak. J. Biol. Sci. 2006, 9, 84–87. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the title compound are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, Y.-S.; Kim, H.; Jung, E.; Kim, J.-H.; Hwang, W.; Kang, E.-J.; Lee, S.; Ha, B.-J.; Lee, J.; Park, D. A Novel Antibacterial Compound from Siegesbeckia glabrescens. Molecules 2012, 17, 12469-12477. https://doi.org/10.3390/molecules171112469
Kim Y-S, Kim H, Jung E, Kim J-H, Hwang W, Kang E-J, Lee S, Ha B-J, Lee J, Park D. A Novel Antibacterial Compound from Siegesbeckia glabrescens. Molecules. 2012; 17(11):12469-12477. https://doi.org/10.3390/molecules171112469
Chicago/Turabian StyleKim, Young-Soo, Hyungil Kim, Eunsun Jung, Jang-Hyun Kim, Wangtaek Hwang, Eun-Ju Kang, Sanghyun Lee, Byung-Jo Ha, Jongsung Lee, and Deokhoon Park. 2012. "A Novel Antibacterial Compound from Siegesbeckia glabrescens" Molecules 17, no. 11: 12469-12477. https://doi.org/10.3390/molecules171112469
APA StyleKim, Y.-S., Kim, H., Jung, E., Kim, J.-H., Hwang, W., Kang, E.-J., Lee, S., Ha, B.-J., Lee, J., & Park, D. (2012). A Novel Antibacterial Compound from Siegesbeckia glabrescens. Molecules, 17(11), 12469-12477. https://doi.org/10.3390/molecules171112469






