Comparison of the Phenolic Compounds, Carotenoids and Tocochromanols Content in Wheat Grain under Organic and Mineral Fertilization Regimes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Kernel Characteristics
| System of fertilization | NPK | C | FYM | MBM | MBM+EM-1 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| mean | range | mean | range | mean | range | mean | range | mean | range | |
| 1000 kernels weight (g) | 34.52 ± 0.47a | 34.01–35.13 | 35.29 ± 0.39b | 34.63–35.64 | 33.85 ± 0.68ac | 32.77–34.58 | 33.83 ± 0.55ac | 33.24–34.48 | 33.79 ± 0.42c | 33.16–34.18 |
| Kernel length (mm) | 7.26 ± 0.39a | 5.59–8.19 | 7.15 ± 0.4b | 5.97–8.43 | 7.22 ± 0.39ab | 6.05–8.19 | 7.30 ± 0.37a | 5.97–8.14 | 7.26 ± 0.42a | 5.99–8.54 |
| Kernel width (mm) | 3.20 ± 0.24ab | 2.48–3.68 | 3.24 ± 0.26b | 2.51–3.86 | 3.15 ± 0.26a | 2.23–3.90 | 3.18 ± 0.26a | 2.36–3.83 | 3.15 ± 0.25a | 2.18–3.75 |
| Length/width ratio (-) | 2.27 ± 0.15a | 1.95–2.78 | 2.21 ± 0.42b | 4.76–7.09 | 2.30 ± 0.17ac | 1.88–2.98 | 2.30 ± 0.16a | 1.92–2.87 | 2.32 ± 0.18c | 1.91–2.94 |
| Kernel surface H (°) | 27.23 ± 0.95a | 23–28 | 27.90 ± 0.52b | 26–30 | 28.02 ± 2.12b | 25–31 | 27.93 ± 0.39b | 26–30 | 27.63 ± 0.96c | 22–30 |
| Kernel surface S (%) | 28.01 ± 3.42a | 12.5–35.55 | 30.52 ± 3.39b | 23.05–42.19 | 30.76 ± 3.64b | 23.83–51.56 | 29.64 ± 3.34c | 18.36–37.89 | 28.6 ± 3.38a | 18.36–41.41 |
| Kernel surface I (%) | 60.13 ± 3.06a | 52.73–74.74 | 58.24 ± 2.74bc | 49.22–63.8 | 57.7 ± 3.2b | 37.37–63.67 | 58.57 ± 2.75cd | 51.69–67.06 | 59.13 ± 3.08d | 50.91–69.01 |

2.2. Chemical Composition
| Compound * | System of fertilization | ||||
|---|---|---|---|---|---|
| NPK | C | FYM | MBM | MBM+EM-1 | |
| Phenolic compounds composition | |||||
| Free phenolics | 449.6 ± 14.5a | 509.0 ± 61.2ab | 506.8 ± 95.4ab | 515.6 ± 20.7ab | 530.2 ± 12.4b |
| Total phenolics – EE | 1249 ± 41.6a | 1277 ± 77.9ab | 1328 ± 33.0ab | 1282 ± 119.5ab | 1363 ± 32.7b |
| Total phenolics – ME | 1460 ± 143.5ab | 1739 ± 124.3c | 1487 ± 14.8ab | 1608 ± 122.9bc | 1414 ± 125.0a |
| Total phenolics | 2710 ± 101.9a | 3016 ± 202.3b | 2816 ± 47.8ac | 2891 ± 3.3bc | 2777 ± 157.7ac |
| Free/total phenolics | 16.60 | 16.88 | 18.00 | 17.83 | 19.09 |
| Phenolic acids | |||||
| ferulic | 538.8 ± 31.1a | 440.7 ± 35.0bc | 479.2 ± 51.6b | 420.5 ± 10.8c | 418.3 ± 4.2c |
| p-coumaric | 45.4 ± 2.5a | 32.6 ± 2.5b | 37.3 ± 0.3c | 38.4 ± 1.6c | 19.8 ± 0.2d |
| sinapic | 86.1 ± 19.0a | 77.8 ± 10.7ab | 88.6 ± 7.1a | 86.6 ± 15.8a | 68.3 ± 3.4b |
| vanillic | 10.8 ± 0.1a | 9.0 ± 0.1b | 8.9 ± 0.6b | 9.3 ± 0.8b | 7.5 ± 0.3c |
| p-OH benzoic | 5.7 ± 0.2a | 4.4 ± 0.1b | 4.4 ± 0.3b | 4.7 ± 0.0c | 3.5 ± 0.0d |
| sum of phenolic acids | 686.8 ± 14.7a | 564.6 ± 48.5c | 618.4 ± 59.9b | 559.5 ± 4.2c | 517.3 ± 1.4c |
| Compound * | System of fertilization | ||||
|---|---|---|---|---|---|
| NPK | C | FYM | MBM | MBM+EM−1 | |
| Carotenoids | |||||
| Soluble CAR | 1.54 ± 0.01a | 1.51 ± 0.18ab | 1.51 ± 0.15ab | 1.41 ± 0.01ab | 1.38 ± 0.01 b |
| Total CAR | 3.60 ± 0.03a | 3.59 ± 0.10a | 3.77 ± 0.12b | 3.87 ± 0.20b | 3.54 ± 0.09 a |
| Tocochromanols | |||||
| Soluble α-T | 13.49 ± 0.63a | 14.19 ± 0.38b | 14.02 ± 0.53ab | 15.09 ± 0.10c | 13.40 ± 0.39a |
| Total α-T | 14.61 ± 0.45a | 15.20 ± 0.05a | 14.93 ± 0.24a | 15.48 ± 1.10a | 13.56 ± 0.84b |
| Soluble β-T | 3.51 ± 0.09a | 3.58 ± 0.03a | 3.60 ± 0.08a | 3.53 ± 0.07a | 3.08 ± 0.11b |
| Total β-T | 5.73 ± 0.25a | 5.89 ± 0.16a | 6.10 ± 0.07a | 5.86 ± 0.14a | 5.15 ± 0.24b |
| Soluble α-T3 | 5.52 ± 0.10a | 5.50 ± 0.13ab | 5.63 ± 0.12b | 5.34 ± 0.26b | 5.16 ± 0.11c |
| Total α-T3 | 6.29 ± 0.48a | 5.98 ± 0.22a | 5.43 ± 0.12b | 6.08 ± 0.04a | 5.38 ± 0.30b |
| Soluble β-T3 | 17.95 ± 0.29ab | 18.30 ± 0.62bc | 17.76 ± 0.03ab | 18.56 ± 0.42c | 17.44 ± 0.51a |
| Total β-T3 | 18.86 ± 0.24ab | 19.14 ± 0.18a | 18.75 ± 0.38bc | 18.69 ± 0.33c | 17.55 ± 0.04d |
| Soluble T | 17.00 ± 0.53a | 17.77 ± 0.35b | 17.62 ± 0.60b | 18.62 ± 0.17c | 16.48 ± 0.50a |
| Total T | 20.34 ± 0.54a | 21.09 ± 0.18ab | 21.03 ± 0.12ab | 21.34 ± 1.35b | 18.71 ± 0.95c |
| Soluble T3 | 23.47 ± 0.77a | 23.80 ± 0.87a | 23.39 ± 0.08a | 23.90 ± 0.32a | 22.60 ± 0.87b |
| Total T3 | 25.15 ± 0.02a | 25.12 ± 0.34a | 24.18 ± 0.31b | 24.77 ± 0.47ab | 22.93 ± 0.19c |
| Soluble T+T3 | 40.47 ± 1.30a | 41.57 ± 1.22ab | 41.01 ± 0.68ab | 42.52 ± 0.48b | 39.08 ± 1.38c |
| Total T+T3 | 45.49 ± 0.53a | 46.21 ± 0.52a | 45.21 ± 0.43a | 46.11 ± 1.82a | 41.64 ± 1.15b |
| Soluble T3/T | 1.38 ± 0.08a | 1.34 ± 0.03ab | 1.33 ± 0.04ab | 1.28 ± 0.05b | 1.37 ± 0.03a |
| Total T3/T | 1.24 ± 0.03a | 1.19 ± 0.02ab | 1.15 ± 0.03b | 1.16 ± 0.06b | 1.23 ± 0.05a |
2.3. Discussion
3. Experimental
3.1. Plot Experiments
| Content of mineral compound | NPK | Compost | Farm Yard Manure | Meat and Bone Meal | Meat and Bone Meal + Effective Microorganisms (MBM+EM−1) |
|---|---|---|---|---|---|
| [kg·ha−1] | (C) | (FYM) | (MBM) | ||
| N | 90.0 | 71.0 | 51.0 | 99.8 | 99.8 |
| P | 31.0 | 29.0 | 12.1 | 59.7 | 59.7 |
| K | 83.0 | 62.0 | 49.0 | 6.2 | 6.2 |
| Mg | - | 16.0 | 8.0 | 3.0 | 3.0 |
| Ca | - | 52.0 | 34.0 | 28.5 | 28.5 |
| Na | - | 3.8 | 3.2 | 8.4 | 8.4 |
| Cu | - | 0.048 | 0.050 | 0.015 | 0.015 |
| Fe | - | 5.58 | 3.85 | 0.77 | 0.77 |
| Mn | - | 0.740 | 0.450 | 0.005 | 0.005 |
| Zn | - | 0.504 | 0.250 | 0.149 | 0.149 |
| Dose of fertilizer | 10 t·ha−1 | 10 t·ha−1 | 1.5 t·ha−1 | 1.5 t·ha−1 |
3.2. Grain Features Measurement
3.3. Extraction of Grain Phytochemicals
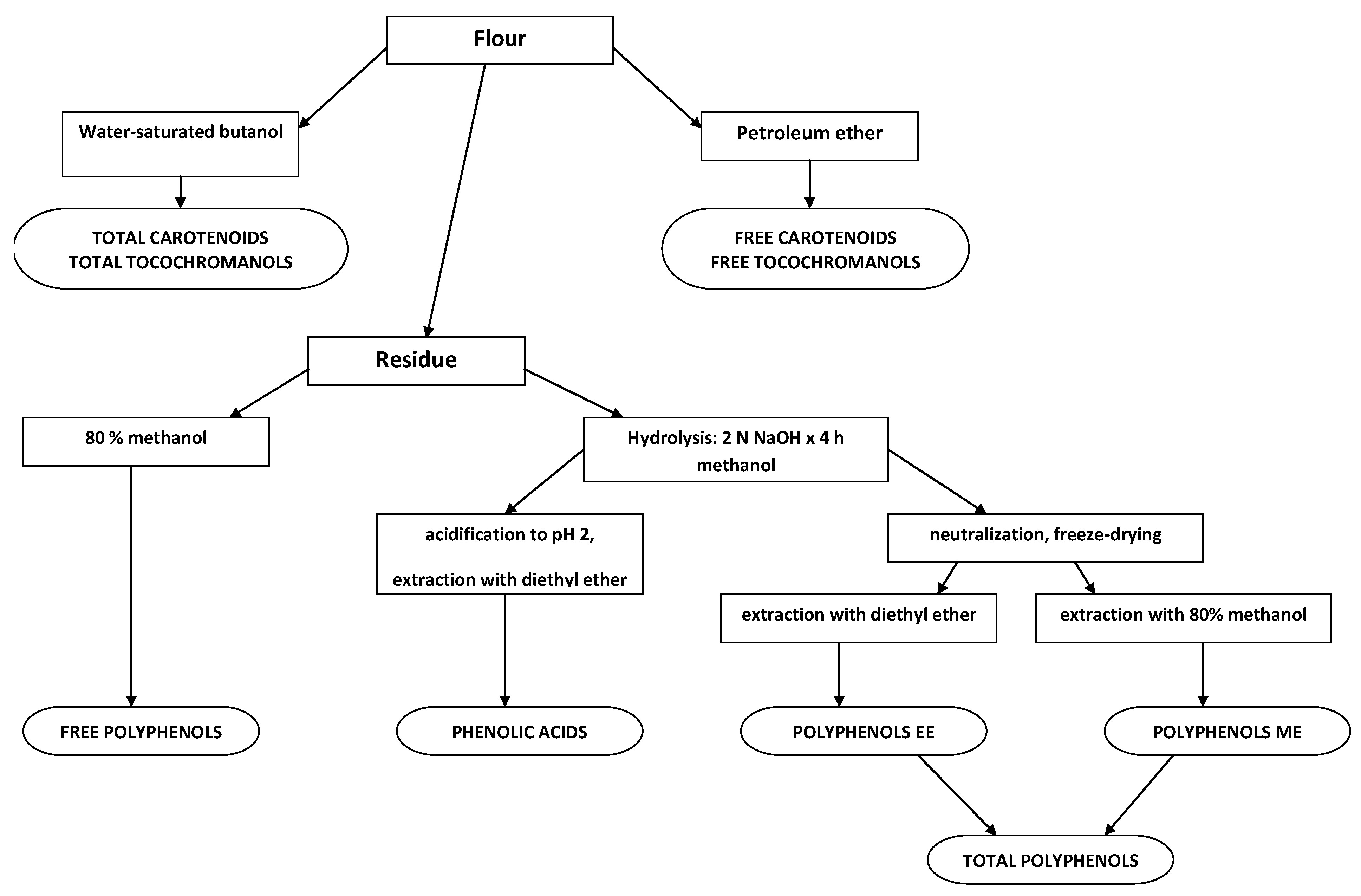
3.4. Determination of Total Carotenoids Content
3.5. Determination of Tocopherols and Tocotrienols Content
3.6. Determination of Total and Free Polyphenols Content
3.7. Determination of Phenolic Acids Content
3.8. Statistical Analysis
4. Conclusions
Acknowledgements
References
- Całyniuk, B.; Grochowska-Niedworok, E.; Białek, A.; Czech, N.; Kukielczak, A. Food guide pyramid—its past and present. Probl. Hig. Epidemiol. 2011, 92, 20–24. [Google Scholar]
- Liu, R.H. Whole grain phytochemicals and health. J. Cereal Sci. 2007, 46, 207–219. [Google Scholar] [CrossRef]
- Okarter, N.; Liu, C.-S.; Sorrells, M.E.; Liu, R.H. Phytochemical content and antioxidant activity of six diverse varieties of whole wheat. Food Chem. 2010, 119, 249–257. [Google Scholar] [CrossRef]
- Zieliński, H.; Kozłowska, H. Antioxidant activity and total phenolics in selected cereal grains and their different morphological fractions. J. Agric. Food Chem. 2000, 48, 2008–2016. [Google Scholar] [CrossRef]
- Moore, J.; Hao, Z.; Zhou, K.; Luther, M.; Costa, J.; Yu, L. Carotenoid, tocopherol, phenolic acid, and antioxidant properties of Maryland-grown soft wheat. J. Agric. Food Chem. 2005, 53, 6649–6657. [Google Scholar]
- Zhou, K.; Yin, J.-J.; Yu, L. Phenolic acid, tocopherol and carotenoid compositions, and antioxidant functions of hard red winter wheat bran. J. Agric. Food Chem. 2005, 53, 3916–3922. [Google Scholar] [CrossRef]
- Bonfil, D.J.; Czosnek, H.; Kafkafi, U. Changes in wheat seed storage protein fingerprint due to soil mineral content. Euphytica 1997, 95, 209–219. [Google Scholar] [CrossRef]
- Ishaq, M.; Ibrahim, M.; Lal, R. Tillage effect on nutrient uptake by wheat and cotton as influenced by fertilizer rate. Soil Till. Res. 2001, 62, 41–53. [Google Scholar] [CrossRef]
- Cabrera-Bosquet, L.; Albrizio, R.; Araus, J.L.; Nogués, S. Photosynthetic capacity of field-grown durum wheat under different N availabilities: A comparative study from leaf to canopy. Environ. Exp. Bot. 2009, 67, 145–152. [Google Scholar] [CrossRef]
- Winter, C.K.; Davis, S.F. Organic Foods. J. Food Sci. 2006, 71, 117–124. [Google Scholar] [CrossRef]
- Zuchowski, J.; Jonczyk, K.; Pecio, L.; Oleszek, W. Phenolic acid concentrations in organically and conventionally cultivated spring and winter wheat. J. Sci. Food Agric. 2011, 91, 1089–1095. [Google Scholar] [CrossRef]
- Choudhary, M.; Bailey, L.D.; Grant, C.A. Review of the use of swine manure in crop production: Effects on yield and composition and on soil and water quality. Waste Manag. Res. 1996, 14, 581–595. [Google Scholar]
- Prassad, P.V.V.; Satyanarayana, V.; Murthy, V.R.; Broote, K.J. Maximizing yields in rice-groundnut cropping sequence through integrated nutrient management. Field Crop Res. 2002, 75, 9–21. [Google Scholar] [CrossRef]
- Liu, X.; Herbert, S.J.; Jin, J.; Zhang, Q.; Wang, G. Responses of photosynthetic rates and yield/quality of main crops to irrigation and manure application in the black soil area of Northeast China. Plant Soil 2004, 261, 55–60. [Google Scholar] [CrossRef]
- Yang, C.; Yang, L.; Yang, Y.; Ouyang, Z. Rice root growth and nutrient uptake as influenced by organic manure in continuously and alternately flooded paddy soils. Agric. Water Manag. 2004, 70, 67–81. [Google Scholar] [CrossRef]
- Chen, L.; Kivelä, J.; Helenius, J.; Kangas, A. Meat bone meal as fertilizer for barley and oat. Agric. Food Sci. 2011, 20, 235–244. [Google Scholar] [CrossRef]
- Jeng, A.; Haraldsen, T.K.; Vagstad, N.; Grønlund, A. Meat and bone meal as nitrogen fertilizer to cereals in Norway. Agric. Food Sci. 2004, 13, 268–275. [Google Scholar] [CrossRef]
- Jeng, A.S.; Haraldsen, T.K.; Grønlund, A.; Pedersen, P.A. Meat and bone meal as nitrogen and phosphorus fertilizer to cereals and rye grass. Nutr. Cycl. Agroecosys. 2006, 76, 183–191. [Google Scholar]
- Mondini, C.; Cayuela, M.L.; Sinicco, T.; Sánchez-Monedero, M.A.; Bertolone, E.; Bardi, L. Soil application of meat and bone meal. Short-term effects on mineralization dynamics and soil biochemical and microbiological properties. Soil Biol. Biochem. 2004, 40, 462–474. [Google Scholar]
- Nogalska, A.; Czapla, J.; Skwierawska, M. The effect of increasing doses of meat-and-bone meal on the yield and macronutrient content of perennial ryegrass (Lolium perrene L.). Pol. J. Nat. Sci. 2011, 26, 5–13. [Google Scholar]
- Piskier, T. Reaction of spring wheat to the application of bio-stimulators and soil absorbents. J. Res. Appl. Agric. Eng. 2006, 51, 136–138. [Google Scholar]
- Javaid, A.; Shah, M.B.M. Growth and yield response of wheat to EM (effective microorganisms) and parthenium green manure. Afr. J. Biotechnol. 2010, 9, 3373–3381. [Google Scholar]
- Taie, H.A.A.; El-Mergawi, R.; Radwan, S. Isoflavonoids, flavonoids, phenolic acids profiles and antioxidant activity of soybean seeds as affected by organic and bioorganic fertilization. Am. Eurasian J. Agric. Environ. Sci. 2008, 4, 207–213. [Google Scholar]
- Omar, N.F.; Hassan, S.A.; Yusoff, U.K.; Abdullah, N.A.P.; Wahab, P.E.M.; Sinniah, U.R. Phenolics, flavonoids, antioxidant activity and cyanogenic glycosides of organic and mineral-base fertilized cassava tubers. Molecules 2012, 17, 2378–2387. [Google Scholar] [CrossRef]
- Hussain, A.; Larsson, H.; Olsson, M.E.; Kuktaite, R.; Grausgruber, H.; Johansson, E. Is organically produced wheat a source of tocopherols and tocotrienols for health food? Food Chem. 2012, 132, 1789–1795. [Google Scholar] [CrossRef]
- Stracke, B.A.; Eitel, J.; Watzl, B.; Mader, P.; Rufer, C.E. Influence of the production method of phytochemical concentrations in whole wheat (Triticum aestivum L.): A comparative study. J. Agric. Food Chem. 2009, 57, 10116–10121. [Google Scholar]
- Dholakia, B.B.; Ammiraju, J.S.S.; Singh, H.; Lagu, M.D.; Röder, M.S.; Rao, V.S.; Dhaliwal, H.S.; Ranjekar, P.K.; Gupta, V.S.; Weber, W.E. Molecular marker analysis of kernel size and shape in bread wheat. Plant Breeding 2003, 122, 392–395. [Google Scholar]
- Ramya, P.; Chaubal, A.; Kulkarni, K.; Gupta, L.; Kadoo, N.; Dhaliwal, H.S.; Chhuneja, P.; Lagu, M.; Gupta, V. QTL mapping of 1000-kernel weight, kernel length, and kernel width in bread wheat (Triticum aestivum L.). J. Appl. Genet. 2010, 51, 421–429. [Google Scholar]
- Konopka, I.; Tańska, M.; Pszczółkowska, A.; Fordoński, G.; Kozirok, W.; Olszewski, J. The effect of water stress an wheat kernel size, color and protein composition. Pol. J. Nat. Sci. 2007, 22, 157–171. [Google Scholar] [CrossRef]
- Altenbach, S.B.; DuPont, F.; Kothari, K.; Chan, R.; Johnson, E.; Lieu, D. Temperature, water and fertilizer influence the timing of key events during grain development in a US spring wheat. J. Cereal Sci. 2003, 37, 9–20. [Google Scholar] [CrossRef]
- Abdel-Aal, E-S.M.; Rabalski, I. Bioactive compounds and their antioxidant capacity in selected primitive and modern wheat species. Open Agric. J. 2008, 2, 7–14. [Google Scholar] [CrossRef]
- Dinelli, G.; Segura-Carretero, A.; Di Silvestro, R.; Marotti, I.; Fu, S.; Benedettelli, S.; Ghiselli, L.; Fernadez-Gutierrez, A. Determination of phenolic compounds in modern and old varieties of durum wheat using liquid chromatography coupled with time-of-flight mass spectrometry. J. Chromatogr. A 2009, 1216, 7229–7240. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Phenolic compounds in cereal grains and their health benefits. Cereal Food World 2007, 52, 105–111. [Google Scholar]
- Abdel-Aal, E.S.M.; Young, J.C.; Rabalski, I. Anthocyanin composition in black, blue, pink, purple, and red cereal grains. J. Agric. Food Chem. 2006, 54, 4696–4704. [Google Scholar] [CrossRef]
- Chen, Y.; Ross, A.B.; Aman, P.; Kamal-Eldin, A. Alkylresorcinols as markers of whole grain wheat and rye in cereal products. J. Agric. Food Chem. 2004, 52, 8242–8246. [Google Scholar] [CrossRef]
- Kulawinek, M.; Jaromin, A.; Kozubek, A.; Żarnowski, R. Alkylresorcinols in selected polish rye and wheat cereals and whole-grain cereal products. J. Agric. Food Chem. 2008, 15, 7236–7242. [Google Scholar]
- Kulawinek, M.; Kozubek, A. Quantitative determination of alkylresorcinols in cereal grains: Independence of the length of the aliphatic side chain. J. Food Lipids 2008, 15, 251–262. [Google Scholar] [CrossRef]
- Platani, C.; Beleggia, R.; Digesù, A.M.; Fares, C.; Moscaritolo, S.; D’Egidio, M.G.; Cattivelli, L. Characterization of lignans content in cereals. In From Seed to Pasta: the Durum Wheat Chain, Proccedings of the International Durum Wheat Symposium, Bologna, Italy, June-July 2008; International Durum Wheat Symposium: Bologna, Italy, 2008. Available online: http://www.fromseedtopasta2008.it/PDF%20POSTER%202/P.%207.2_Platani%20et%20al.pdf (accessed on 9 October 2012). Poster 7.2:1. [Google Scholar]
- Rispail, N.; Morris, P.; Webb, K.J. Phenolic compounds: Extraction and analysis. In Lotus japonicus Handbook, 1st ed.; Márquez, A.J., Ed.; Springer: Dordrecht, The Netherlands, 2005; pp. 349–354. [Google Scholar]
- Litvinenko, V.I.; Makarov, V.A. The alkaline hydrolysis of flavonoid glycosides. Chem. Nat. Compd. 1969, 5, 305–306. [Google Scholar] [CrossRef]
- Everette, J.D.; Bryant, Q.M.; Green, A.M.; Abbey, Y.A.; Wangila, G.W.; Walker, R.B. Thorough study of reactivity of various compound classes toward the Folin-Ciocalteu reagent. J. Agric. Food Chem. 2010, 58, 8139–8144. [Google Scholar] [CrossRef]
- Glick, B.R. The enhancement of plant growth by free-living bacteria. Can. J. Microbiol. 1995, 41, 109–117. [Google Scholar] [CrossRef]
- Kern, S.M.; Bennett, R.N.; Mellon, F.A.; Kroon, P.A.; Garcia-Conesa, M.T. Absorption of hydroxycinnamates in humans after high-bran cereal consumption. J. Agric. Food Chem. 2003, 51, 6050–6055. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar]
- Suproniene, S.; Mankeviciene, A.; Kadziene, G.; Feiziene, D.; Feiza, V.; Semaskiene, R.; Dabkevicius, Z. The effect of different tillage and fertilization practices on the mycoflora of wheat grains. Agric. Food Sci. 2011, 20, 315–326. [Google Scholar]
- Benoit, I.; Danchin, E.G.J.; Bleichrodt, R.J.; de Vries, R.P. Biotechnological applications and potential of fungal feruloyl esterases based on prevalence, classification and biochemical diversity. Biotechnol. Lett. 2008, 30, 387–396. [Google Scholar] [CrossRef]
- Mayer, A.M. Polyphenol oxidases in plants and fungi: Going places? A review. Phytochemistry 2008, 67, 2318–2331. [Google Scholar] [CrossRef]
- Fazary, A.E.; Ju, Y.-H. Feruloyl esterases as biotechnological tools: Current and future perspectives. Acta Biochim. Biophys. Sin. 2007, 39, 811–828. [Google Scholar] [CrossRef]
- Panfili, G.; Fratianni, A.; Irano, M. Normal phase high-performance liquid chromatography method for the determination of tocopherols and tocotrienols in cereals. J. Agric. Food Chem. 2003, 51, 3940–3944. [Google Scholar] [CrossRef]
- DellaPenna, D. A decade of progress in understanding vitamin E synthesis in plants. J. Plant Physiol. 2005, 162, 729–737. [Google Scholar] [CrossRef]
- Hussain, A. Quality of organically produced wheat from diverse origin. Acta Universitatis agriculturae Sueciae 2012, 18, 1–92. [Google Scholar]
- Dörmann, P. Functional diversity of tocochromanols in plants. Planta 2007, 225, 269–276. [Google Scholar] [CrossRef]
- Falk, J.; Munné-Bosch, S. Tocochromanol functions in plants: antioxidation and beyond. J. Exp. Bot. 2010, 61, 1549–1566. [Google Scholar] [CrossRef]
- Mène-Saffrané, L.; Jones, A.D.; DellaPenna, D. Plastochromanol-8 and tocopherols are essential lipid-soluble antioxidants during seed desiccation and quiescence in Arabidopsis. Proc. Natl. Acad. Sci. USA 2010, 107, 17815–17820. [Google Scholar] [CrossRef]
- Britz, S.J.; Kremer, D.F. Warm temperatures or drought during seed maturation increase free alpha-tocopherol in seeds of soybean (Glycine max L, Merr.). J. Agric. Food Chem. 2002, 50, 6058–6063. [Google Scholar]
- Oh, M.M.; Carey, E.E.; Rajashekar, C.B. Environmental stresses induce healthpromoting phytochemicals in lettuce. Plant Physiol. Bioch. 2009, 47, 578–583. [Google Scholar] [CrossRef]
- Konopka, I.; Kozirok, W.; Tańska, M. Wheat endosperm hardness. Part I. Relationships to colour of kernel cross-section. Eur. Food Res. Technol. 2005, 220, 11–19. [Google Scholar]
- Konopka, I.; Czaplicki, S.; Rotkiewicz, D. Differences in content and composition of free lipids and carotenoids in flour of spring and winter wheat cultivated in Poland. Food Chem. 2006, 95, 290–300. [Google Scholar] [CrossRef]
- Konopka, I.; Kozirok, W.; Rotkiewicz, D. Lipids and carotenoids of wheat grain and flour and attempt of correlating them with digital image analysis of kernel surface and cross-section. Food Res. Int. 2004, 37, 429–438. [Google Scholar] [CrossRef]
- Szymański, N.; Patterson, R.A. Effective microorganisms (EM) wastewater systems. In Proceedings of Best Management Proccedings of One-site ‘03 Conference, Armidale, Australia; 2003; pp. 347–354. [Google Scholar]
- Valarini, P.J.; Alvarez, M.C.D.; Gasco, J.M.; Guerrero, F.; Tokeshi, H. Assessment soil properties by organic matter and EM microorganisms incorporation. Rev. Bras. Ciênc. Solo 2003, 27, 519–525. [Google Scholar]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef]
- Kaneko, S.; Nagamine, T.; Yamada, T. Esterification of endosperm lutein with fatty acids during the storage of wheat seeds. Biosci. Biotechnol. Biochem. 1995, 59, 1–4. [Google Scholar] [CrossRef]
- Irmak, S.; Jonnala, R.S.; MacRitchie, F. Effect of genetic variaton on phenolic acid and policosanol contents of Pegaso wheat lines. J. Cereal Sci. 2008, 48, 20–26. [Google Scholar] [CrossRef]
- Craft, N.E. Relative solubility, stability, and absorptivity of lutein and β-carotene in organic solvents. J. Agric. Food Chem. 1992, 40, 431–434. [Google Scholar]
- Gimeno, E.; Castellote, A.I.; Lamuela-Raventos, R.M.; Torre, M.C.; Lopez-Sabater, M.C. Rapid determination of vitamin E in vegetable oils by reversed-phase high-performance liquid chromatography. J. Chromatogr. A 2000, 881, 251–254. [Google Scholar] [CrossRef]
- Ribereau-Gayon, P. Conspectus of the phenolic constituents. In Plant Phenolics; Heywood, V.H., Ed.; Hafner Publishing Co: New York, NY, USA, 1972; pp. 1–23. [Google Scholar]
- Ogrodowska, D.; Czaplicki, S.; Zadernowski, R.; Mattila, P.; Hellström, J.; Naczk, M. Phenolic acids in seeds and products obtained from Amaranthus cruentus. J. Food Nutr. Res. 2012, 51, 96–101. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Konopka, I.; Tańska, M.; Faron, A.; Stępień, A.; Wojtkowiak, K. Comparison of the Phenolic Compounds, Carotenoids and Tocochromanols Content in Wheat Grain under Organic and Mineral Fertilization Regimes. Molecules 2012, 17, 12341-12356. https://doi.org/10.3390/molecules171012341
Konopka I, Tańska M, Faron A, Stępień A, Wojtkowiak K. Comparison of the Phenolic Compounds, Carotenoids and Tocochromanols Content in Wheat Grain under Organic and Mineral Fertilization Regimes. Molecules. 2012; 17(10):12341-12356. https://doi.org/10.3390/molecules171012341
Chicago/Turabian StyleKonopka, Iwona, Małgorzata Tańska, Alicja Faron, Arkadiusz Stępień, and Katarzyna Wojtkowiak. 2012. "Comparison of the Phenolic Compounds, Carotenoids and Tocochromanols Content in Wheat Grain under Organic and Mineral Fertilization Regimes" Molecules 17, no. 10: 12341-12356. https://doi.org/10.3390/molecules171012341






