Interaction Profile of Diphenyl Diselenide with Pharmacologically Significant Thiols
Abstract
:1. Introduction
2. Results and Discussion
| Compound | Structure |
|---|---|
| DPDS |  |
| CYS |  |
| GSH |  |
| DTT |  |
| DMPS |  |
| DMSA |  |
| Thiol | k2 [M−1s−1] | |
|---|---|---|
| DPDS | Ethanol | |
| CYS | 1059 ± 38 | 967 ± 13 |
| DMPS | 172 ± 12 | 160 ± 19 |
| DMSA | 116 ± 17 | 97 ± 13 |
| DTT | 970 ± 22 | 492 ± 12 |
| GSH | 163 ± 8 | 113 ± 11 |

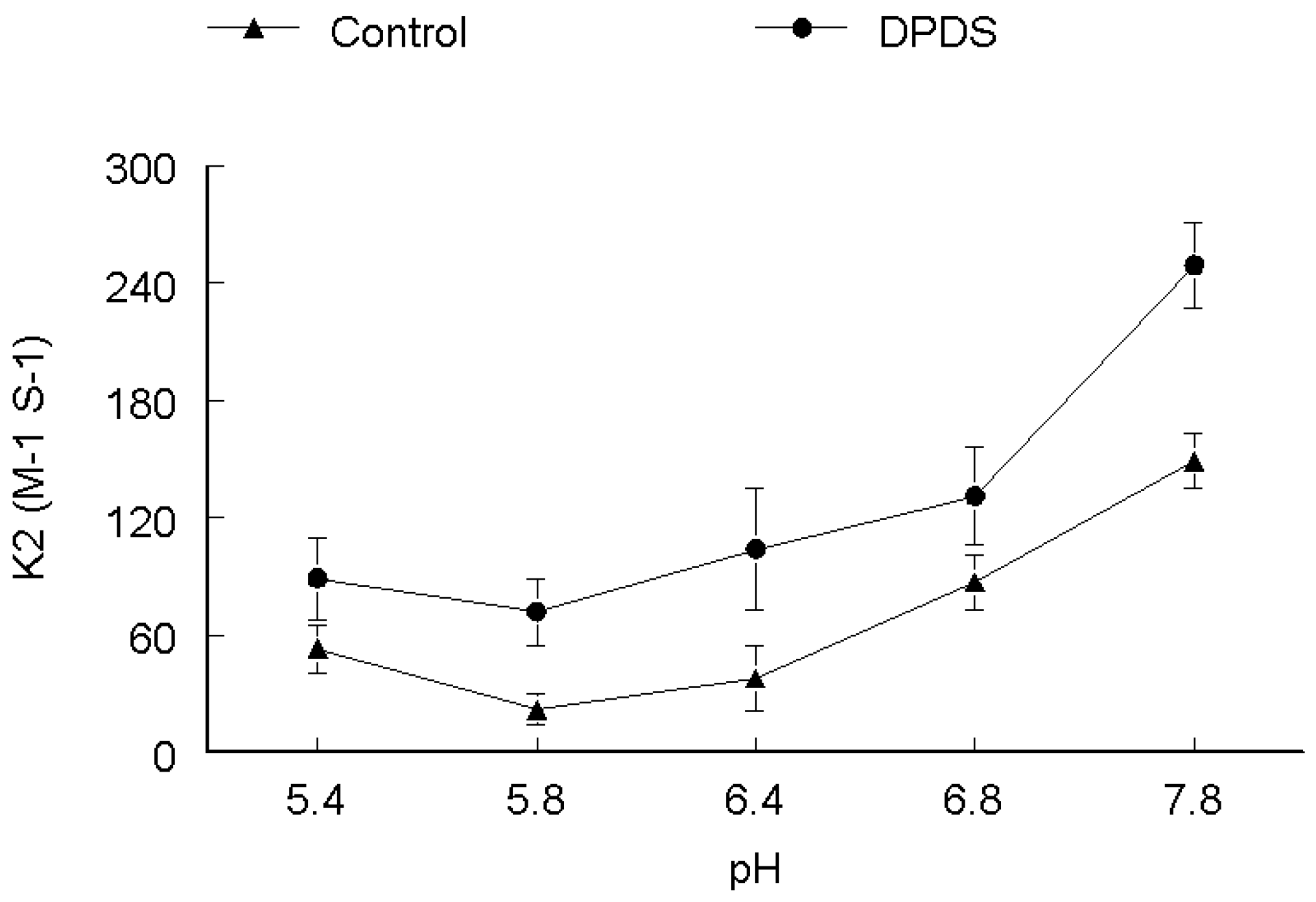
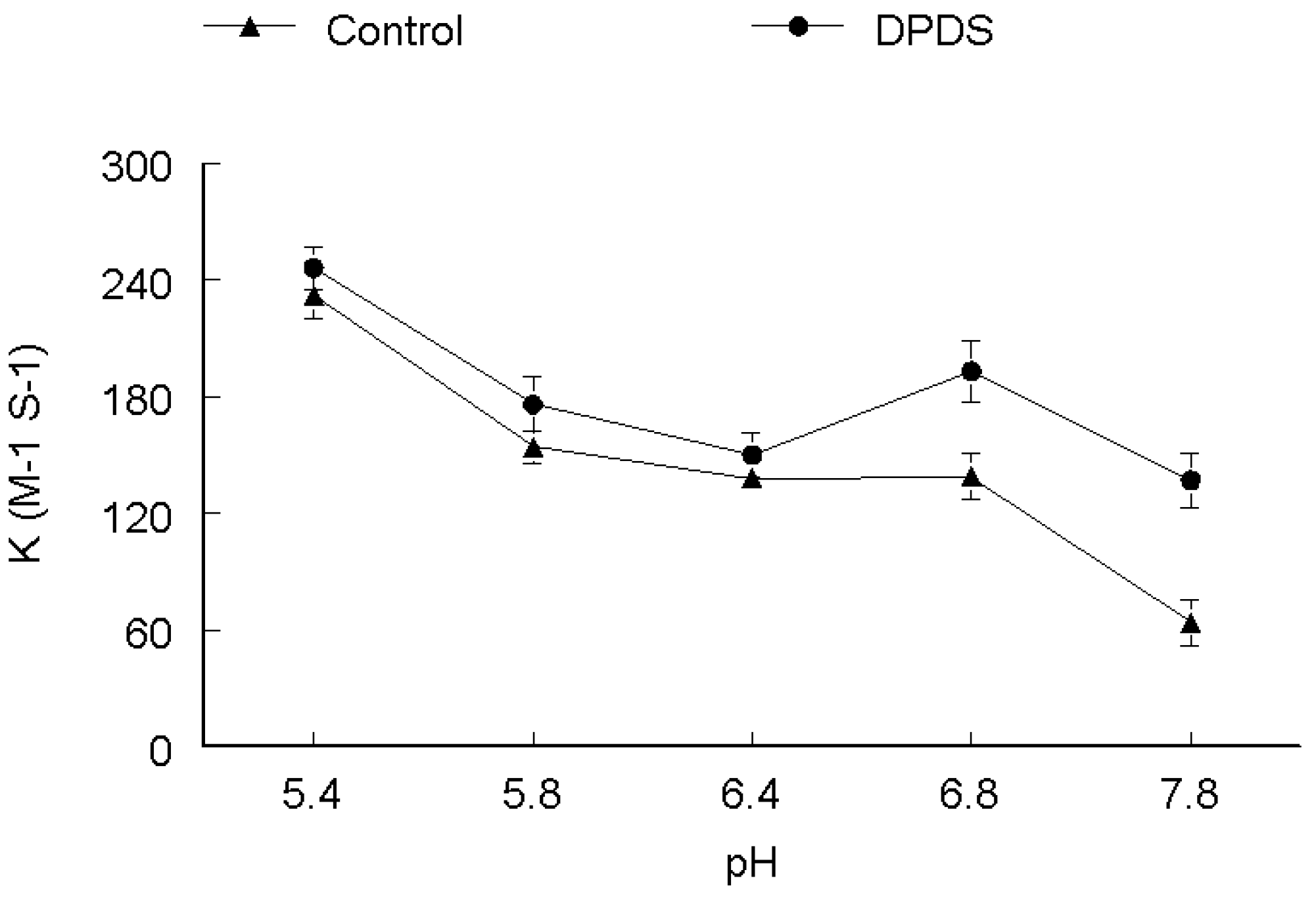
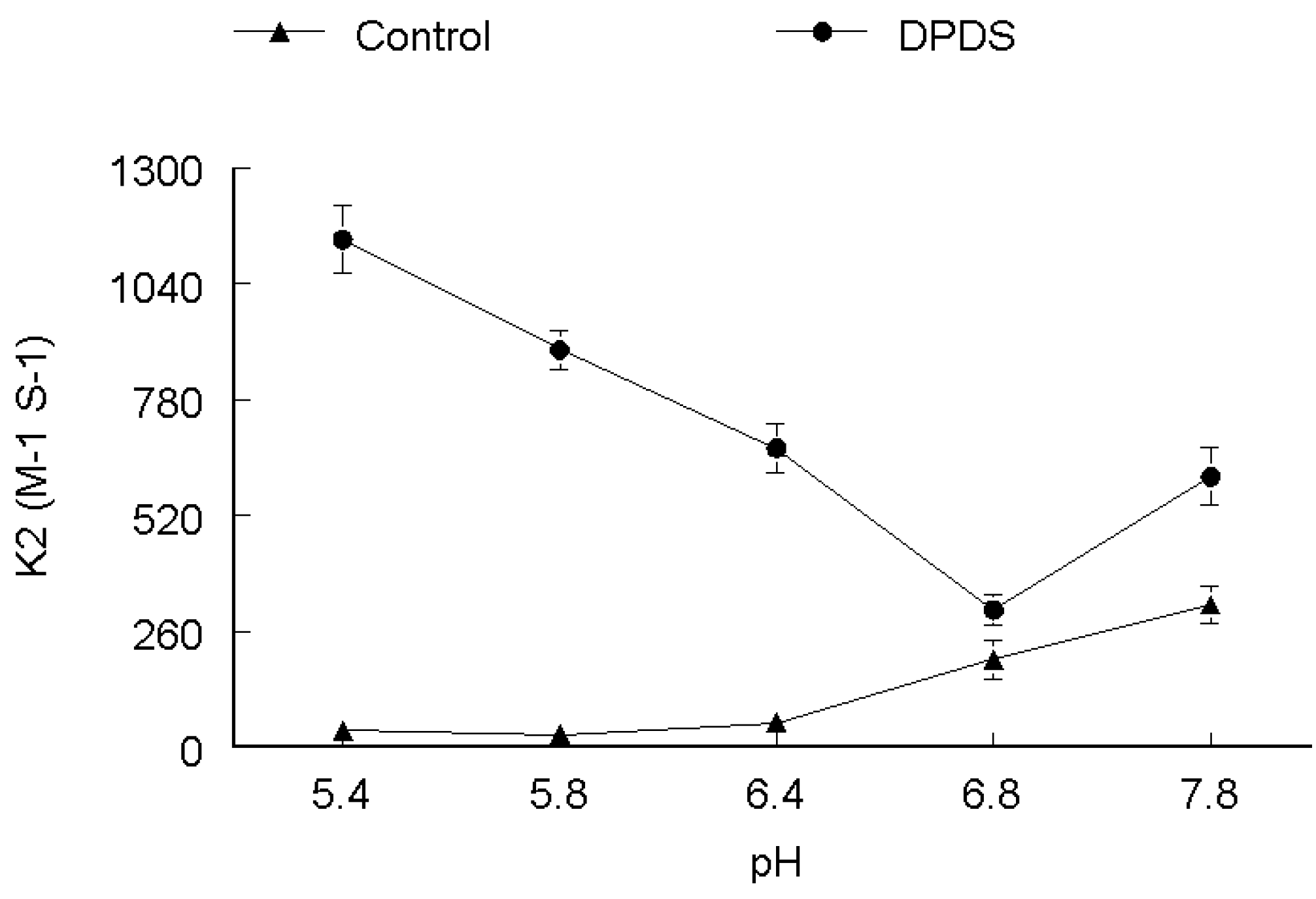

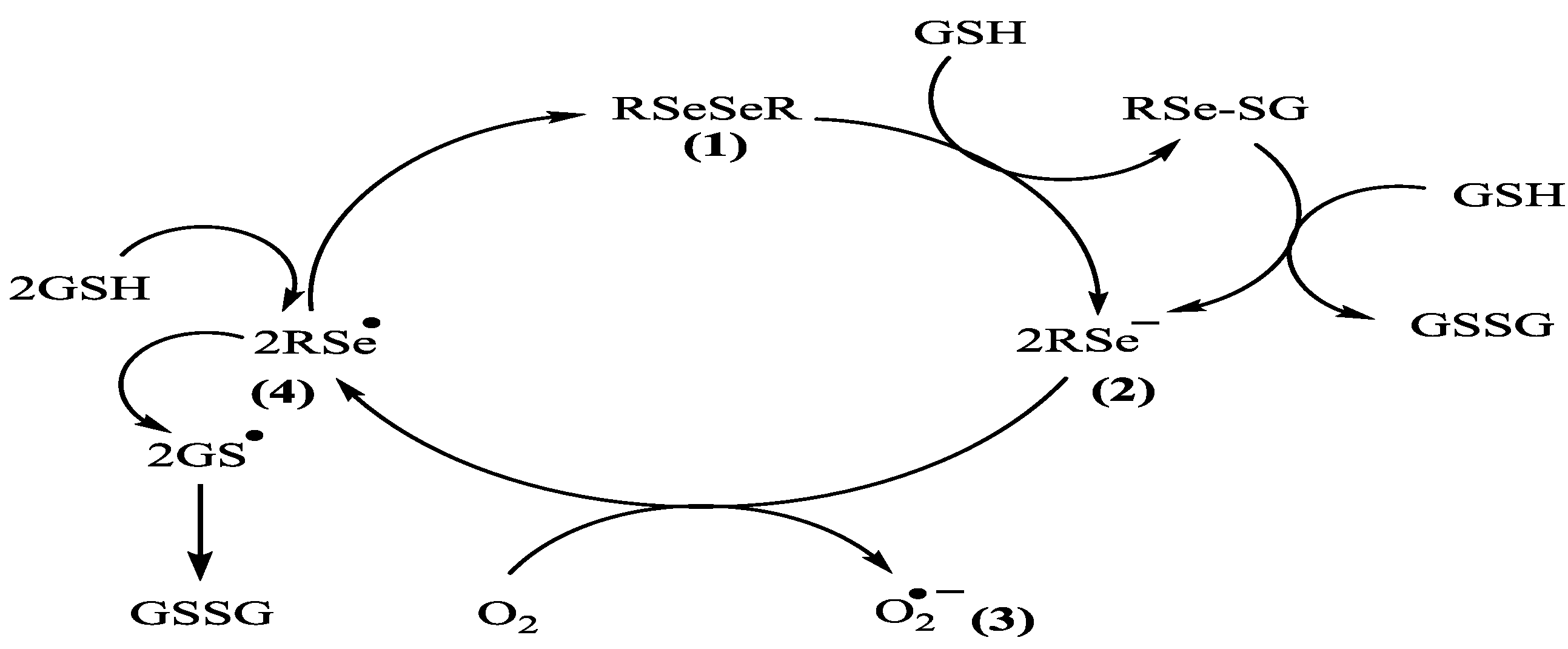
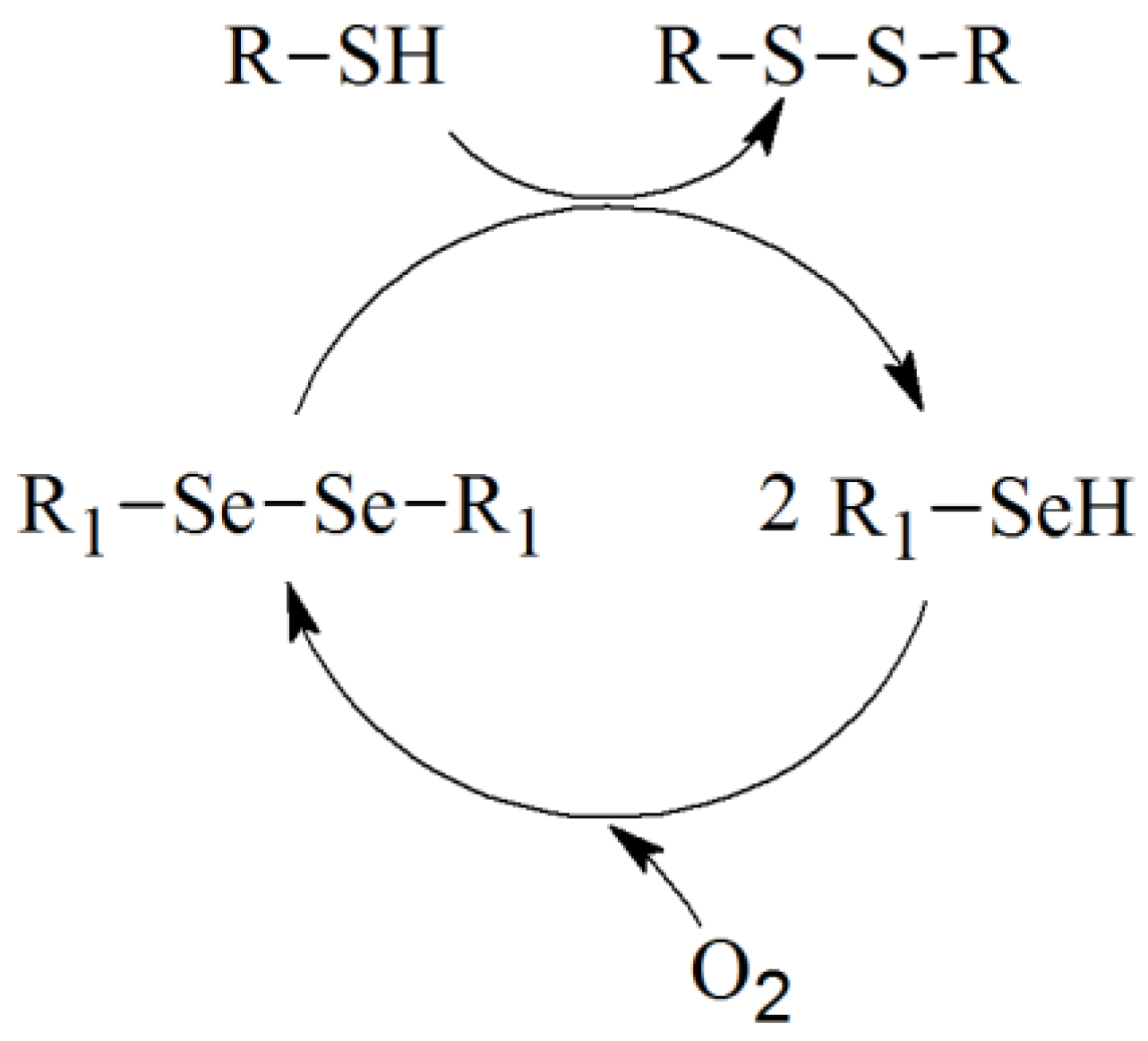
3. Experimental
3.1. Chemicals
3.2. DPDS Synthesis and Preparation
3.3. Preparation of Buffers
3.4. The Rate of Thiol Oxidation
4. Conclusions
Acknowledgments
Conflicts of Interest
- Sample Availability: Not Available.
References
- Kettle, A.J.; Winterbourn, C.C. A key regulator of neutrophil oxidant production. Redox. Rep. 1997, 3, 3–15. [Google Scholar]
- Nogueira, C.W.; Rocha, J.B. Toxicology and pharmacology of selenium: Emphasis on synthetic organoselenium compounds. Arch. Toxicol. 2011, 85, 1313–1359. [Google Scholar]
- de Freitas, A.S.; Prestes, A.S.; Wagner, C.; Sudati, J.H.; Alves, D.; Porciúncula, L.O.; Kade, I.J.; Rocha, J.B.T. Reduction of diphenyl diselenide and analogs by mammalian thioredoxin reductase is independent of their gluthathione peroxidase-like activity: A possible novel pathway for their antioxidant activity. Molecules 2010, 15, 7699–7714. [Google Scholar]
- Puntel, R.L.; Roos, D.H.; Folmer, V.; Nogueira, C.W.; Galina, A.; Aschner, M.; Rocha, J.B.T. Mitochondrial dysfunction induced by different organochalchogens is mediated by thiol oxidation and is not dependent of the classical mitochondrial permeability transition pore opening. Toxicol. Sci. 2010, 117, 133–143. [Google Scholar] [CrossRef]
- Rupil, L.L.; de Bem, A.F.; Roth, G. Diphenyl diselenide-modulation of macrophage activation: Down-regulation of classical and alternative activation markers. Innate Immun. 2012, 18, 627–637. [Google Scholar] [CrossRef]
- Posser, T.; de Paula, M.T.; Franco, J.L.; Leal, R.B.; Rocha, J.B.T. Diphenyl diselenide induces apoptotic cell death and modulates ERK1/2 phosphorylation in human neuroblastoma SH-SY5Y cells. Arch. Toxicol. 2010, 117, 645–651. [Google Scholar]
- de Bem, A.F.; Farina, M.; Portella, R.L.; Nogueira, C.W.; Dinis, T.C.P.; Laranjinha, J.A.N.; Almeida, L.M.; Rocha, J.B.T. Diphenyl diselenide, a simple glutathione peroxidase mimetic, inhibits human LDL oxidation in vitro. Atherosclerosis 2008, 201, 92–100. [Google Scholar] [CrossRef]
- Corte, C.L.D.; Soares, F.A.A.; Aschner, M.; Rocha, J.B.T. Diphenyl diselenide prevents methylmercury-induced mitochondrial dysfunction in rat liver slices. Tetrahedron 2012, , in press.. [Google Scholar]
- Goeger, D.E.; Ganther, H.E. Oxidation of dimethyl selenide to dimethyl selenoxide by microsomes from rat liver and flavin-containing monooxygenase from pig liver. Arch. Biochem. Biophys. 1994, 310, 448–451. [Google Scholar] [CrossRef]
- Björnstedt, M.; Odlander, B.; Kuprin, S. Selenite incubated with NADPH and mammalian thioredoxin reductase yelds selenide, which inhibits lipoxygenase and changes the electron spin resonance spectrum of the active site iron. Biochemistry 1996, 35, 8511–8516. [Google Scholar] [CrossRef]
- Park, H.S.; Park, E.; Kim, M.S.; Ahn, K.; Kim, I.Y.; Choi, E.J. Selenite inhibits the c-Jun N-terminal kinase/stress-activated protein kinase (JNK/SAPK) through a thiol redox mechanism. J. Biol. Chem. 2000, 275, 2527–2531. [Google Scholar]
- Gupta, N.; Porter, T.D. Inhibition of human squalene monooxygenase by selenium compounds. J. Biochem. Mol. Toxicol. 2001, 16, 18–23. [Google Scholar] [CrossRef]
- Rocha, J.B.T.; Saraiva, R.A.; Garcia, S.C.; Gravina, F.S.; Nogueira, C.W. Aminolevulinate dehydratase (δ-ALA-D) as marker protein of intoxication with metals and other pro-oxidant situations. Toxicol. Res. 2012, 1, 85–102. [Google Scholar]
- Maciel, E.N.; Bolzan, R.C.; Braga, A.L.; Rocha, J.B.T. Diphenyl diselenide and diphenyl ditelluride differentially affect d-aminolevulinate dehydratase from liver, kidney, and brain of mice. J. Biochem. Mol. Toxicol. 2000, 14, 310–319. [Google Scholar] [CrossRef]
- Daiber, A.; Zhou, M.; Bachscmid, M.; Ullrich, V. Ebselen as a peroxynitrite scavenger in vitro and ex vivo. Biochem. Pharmacol. 2000, 59, 53–160. [Google Scholar]
- Santi, C.; Santoro, S.; Battistelli, B.; Testaferri, L.; Tiecco, M. Preparation of the First Bench-Stable Phenyl Selenolate: An Interesting “On Water” Nucleophilic Reagent. Eur. J. Org. Chem. 2008, 32, 5387–5390. [Google Scholar]
- Battistelli, B.; Lorenzo, T.; Tiecco, M.; Santi, C. On-Water” Michael-Type Addition Reactions Promoted by PhSeZnCl. Eur. J. Org. Chem. 2011, 10, 1848–1851. [Google Scholar]
- Santoro, S.; Battistelli, B.; Testaferri, L.; Tiecco, M.; Santi, C. Vinylic Substitutions Promoted by PhSeZnCl: Synthetic and Theoretical Investigations. Eur. J. Org. Chem. 2009, 29, 4921–4925. [Google Scholar]
- Santi, C.; Battistelli, B.; Testaferri, L.; Tiecco, M. On water preparation of phenylselenoesters. Green Chem. 2012, 14, 1277–1280. [Google Scholar] [CrossRef]
- Santi, C.; Santoro, S.; Testaferri, L.; Tiecco, M. Simple zinc-mediated preparation of selenols. ChemInform 2008, 39, 1471–1474. [Google Scholar]
- Tidei, C.; Piroddi, M.; Galli, F.; Santi, C. Oxidation of thiols promoted by PhSeZnCl. Tetrahedron Lett. 2012, 53, 232–234. [Google Scholar] [CrossRef]
- Jocelyn, P.C. The Biochemistry of the SH Group; Academic Press: London, UK, 1972. [Google Scholar]
- Lindley, H. Study of the Kinetics of the Reaction between. Thiol Compounds and Chloroacetamide. J. Biochem. 1960, 74, 577–584. [Google Scholar]
- Burk, R.F. Selenium in Biology and Human Health; Springer-Verlag: New York, NY, USA, 1994. [Google Scholar]
- Nogueira, C.W.; Zeni, G.; Rocha, J.B.T. Organoselenium and organotellurium compounds: Toxicology and pharmacology. Chem. Rev. 2004, 104, 6255–6285. [Google Scholar] [CrossRef]
- Paulmier, C. Selenium Reagents and Intermediates in Organic Synthesis; Pergamon Press: New York, NY, USA, 1986; p. 463. [Google Scholar]
- Ellman, G.D. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Tissue sulphydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hassan, W.; Rocha, J.B.T. Interaction Profile of Diphenyl Diselenide with Pharmacologically Significant Thiols. Molecules 2012, 17, 12287-12296. https://doi.org/10.3390/molecules171012287
Hassan W, Rocha JBT. Interaction Profile of Diphenyl Diselenide with Pharmacologically Significant Thiols. Molecules. 2012; 17(10):12287-12296. https://doi.org/10.3390/molecules171012287
Chicago/Turabian StyleHassan, Waseem, and Joao Batista Teixeira Rocha. 2012. "Interaction Profile of Diphenyl Diselenide with Pharmacologically Significant Thiols" Molecules 17, no. 10: 12287-12296. https://doi.org/10.3390/molecules171012287
APA StyleHassan, W., & Rocha, J. B. T. (2012). Interaction Profile of Diphenyl Diselenide with Pharmacologically Significant Thiols. Molecules, 17(10), 12287-12296. https://doi.org/10.3390/molecules171012287





