Synthesis, Reactions and Antimicrobial Activities of 8-Ethoxycoumarin Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
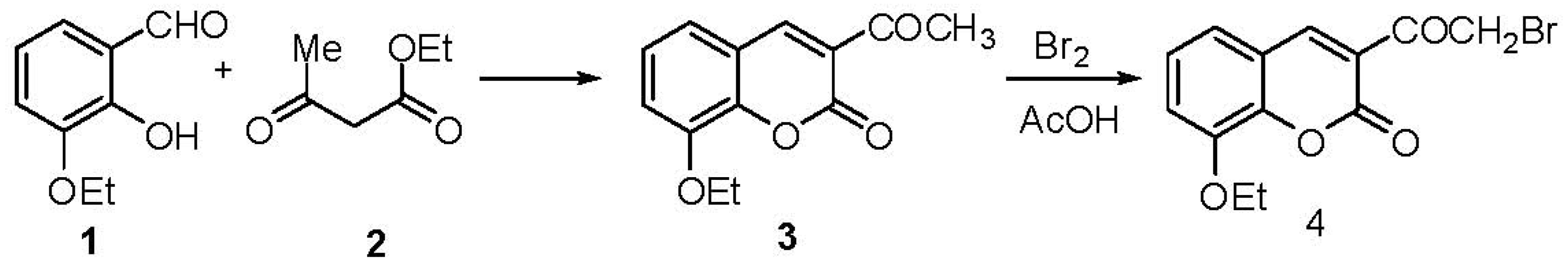
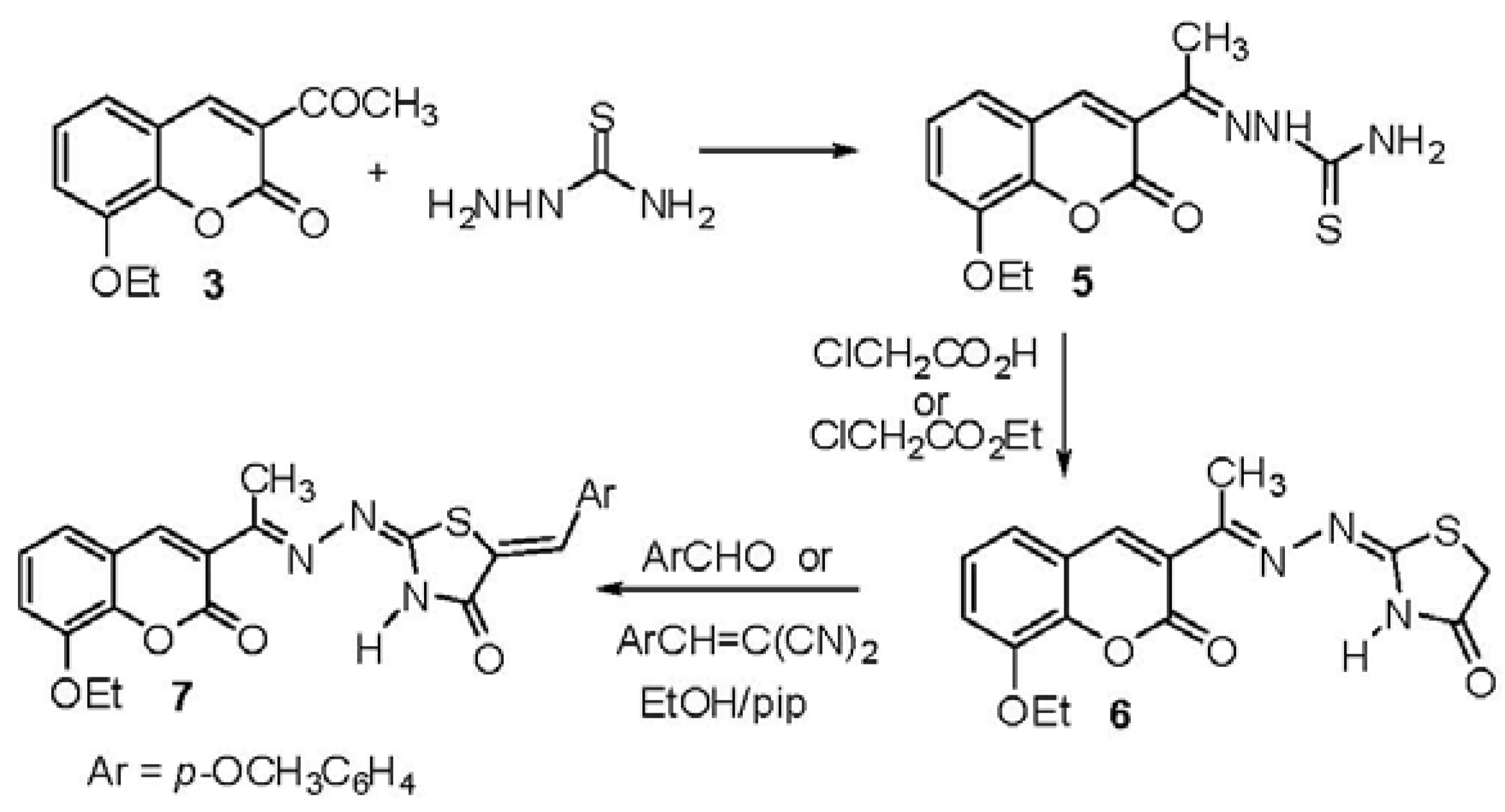
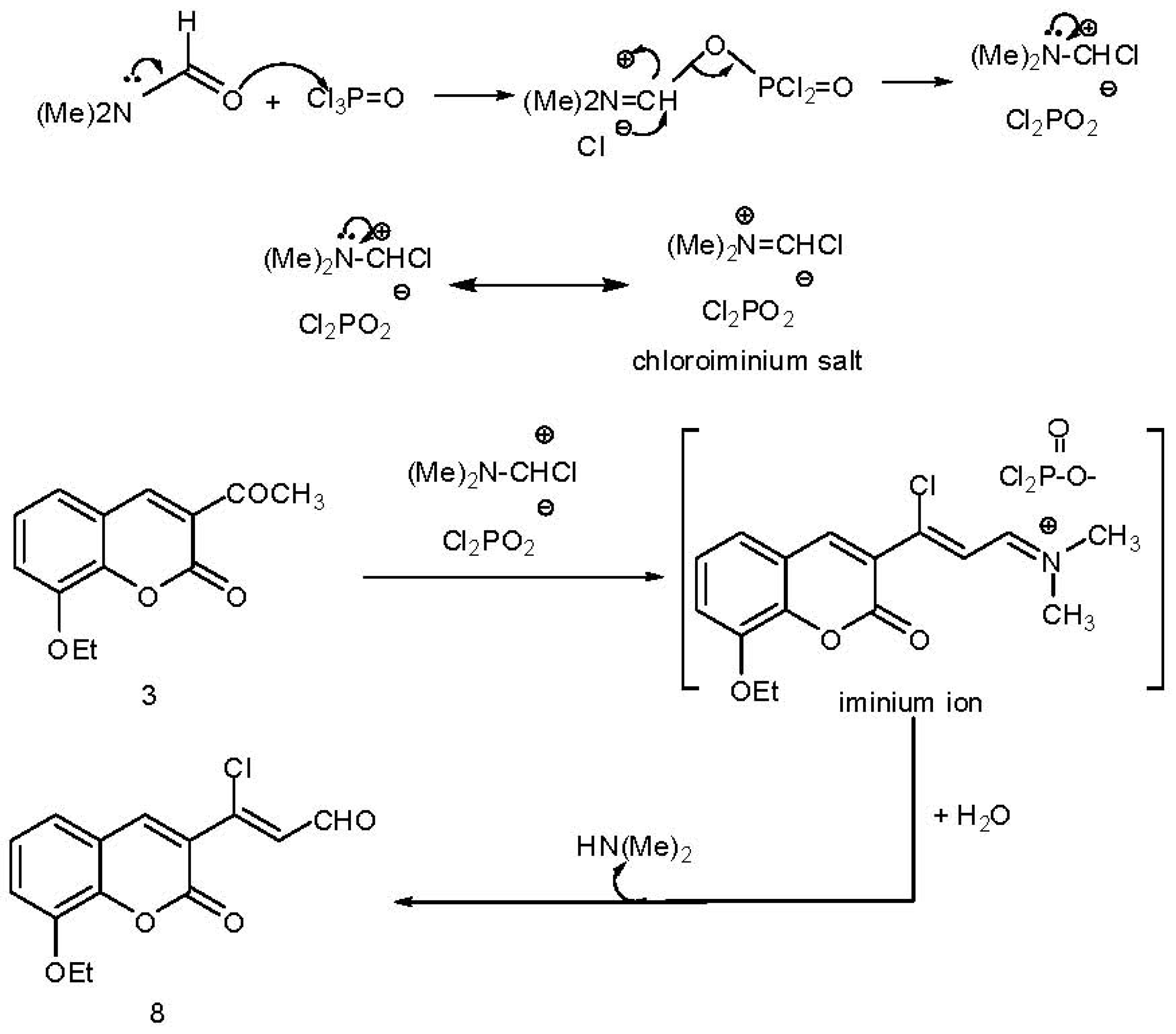

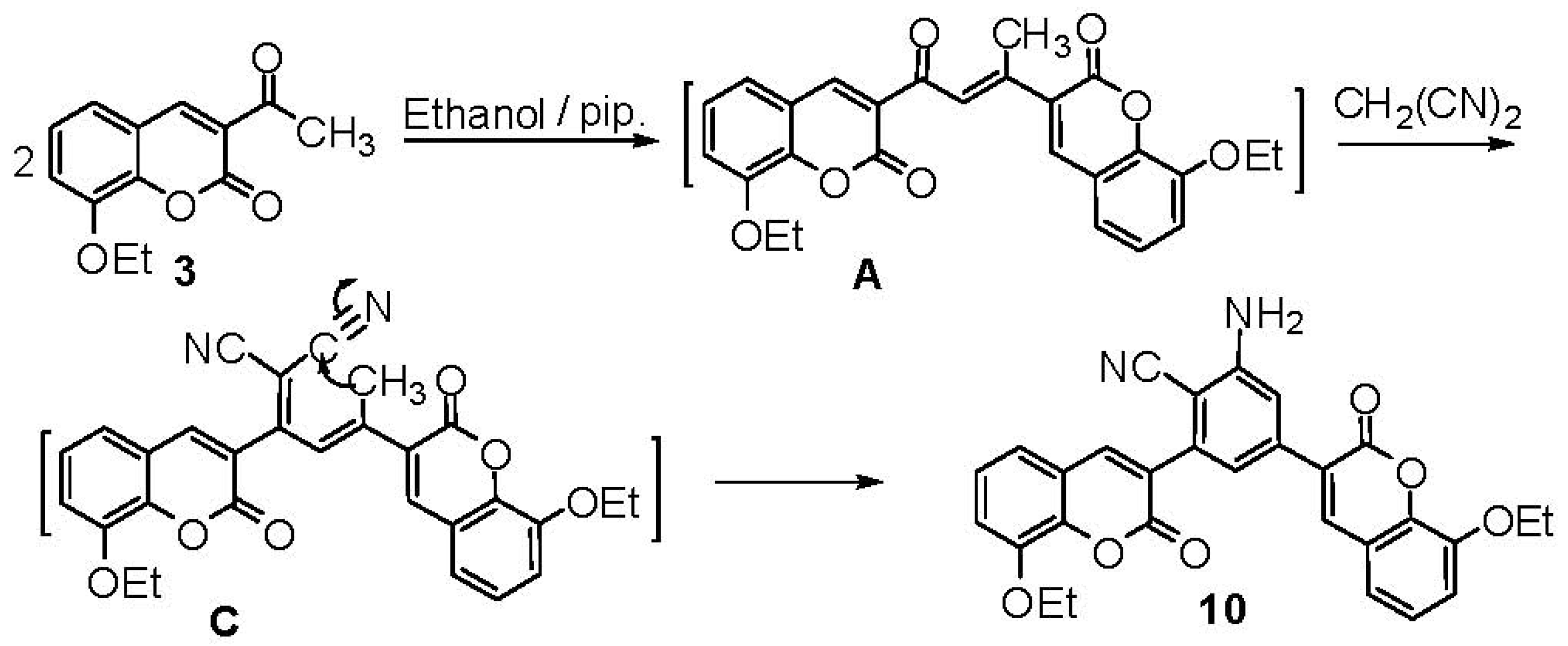
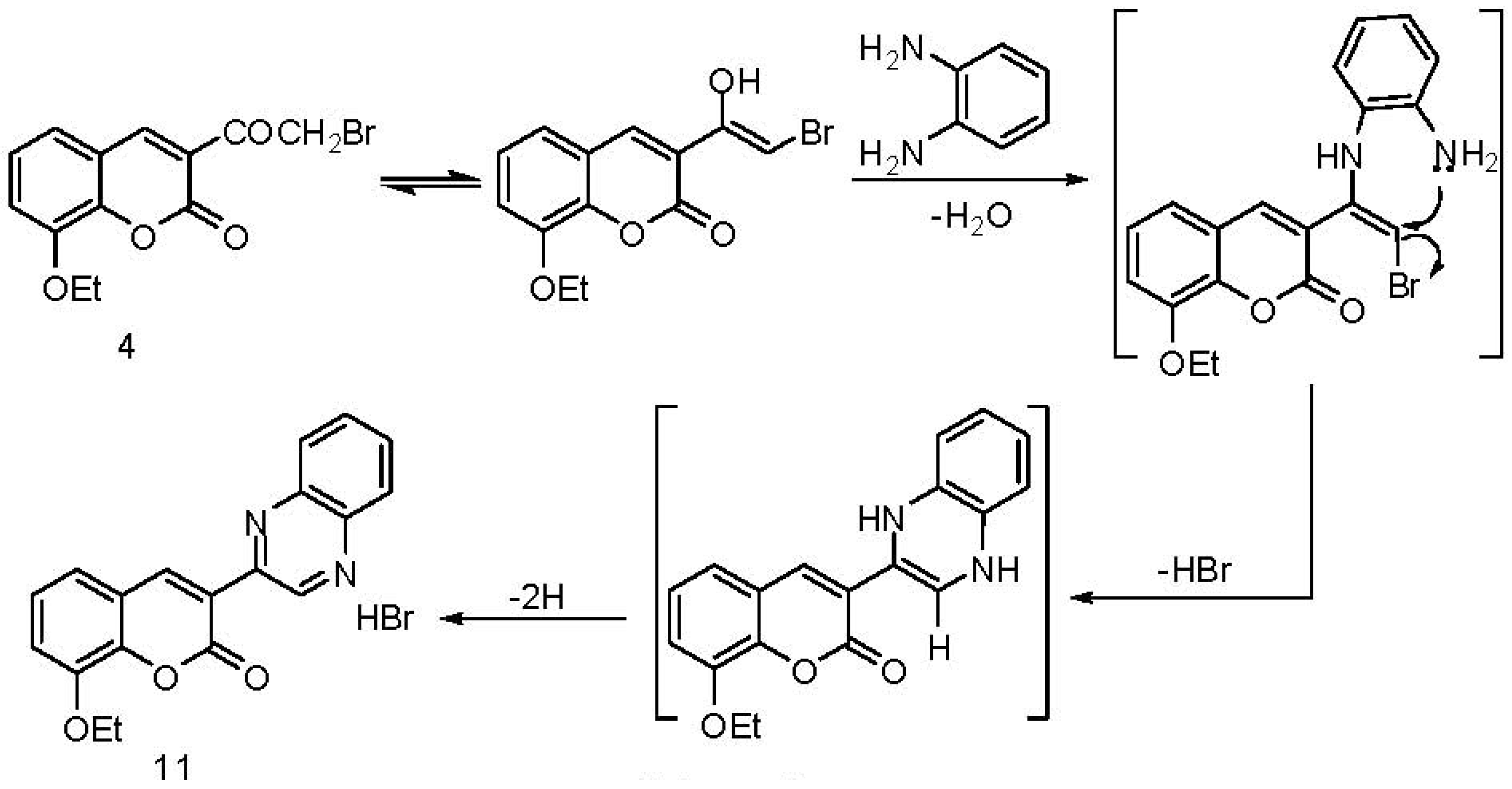

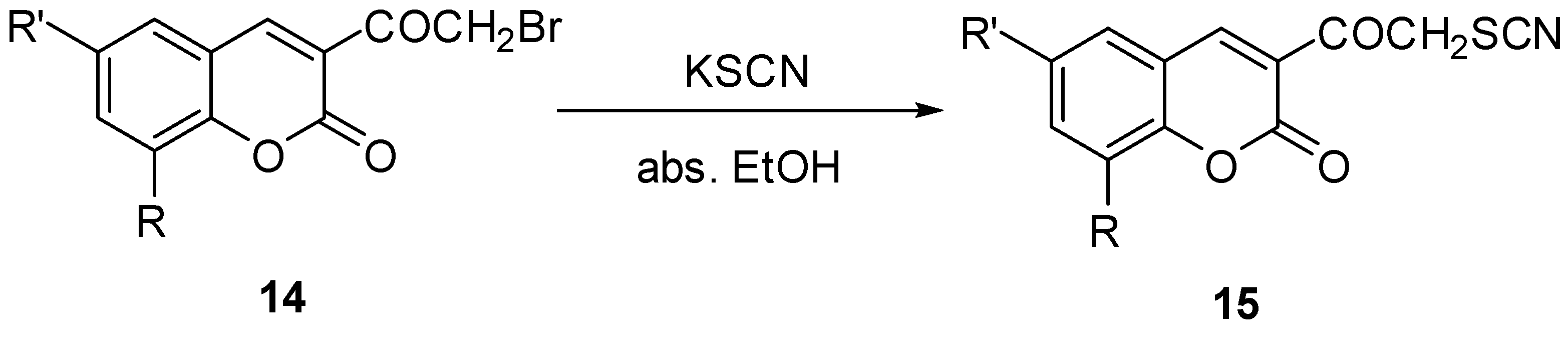

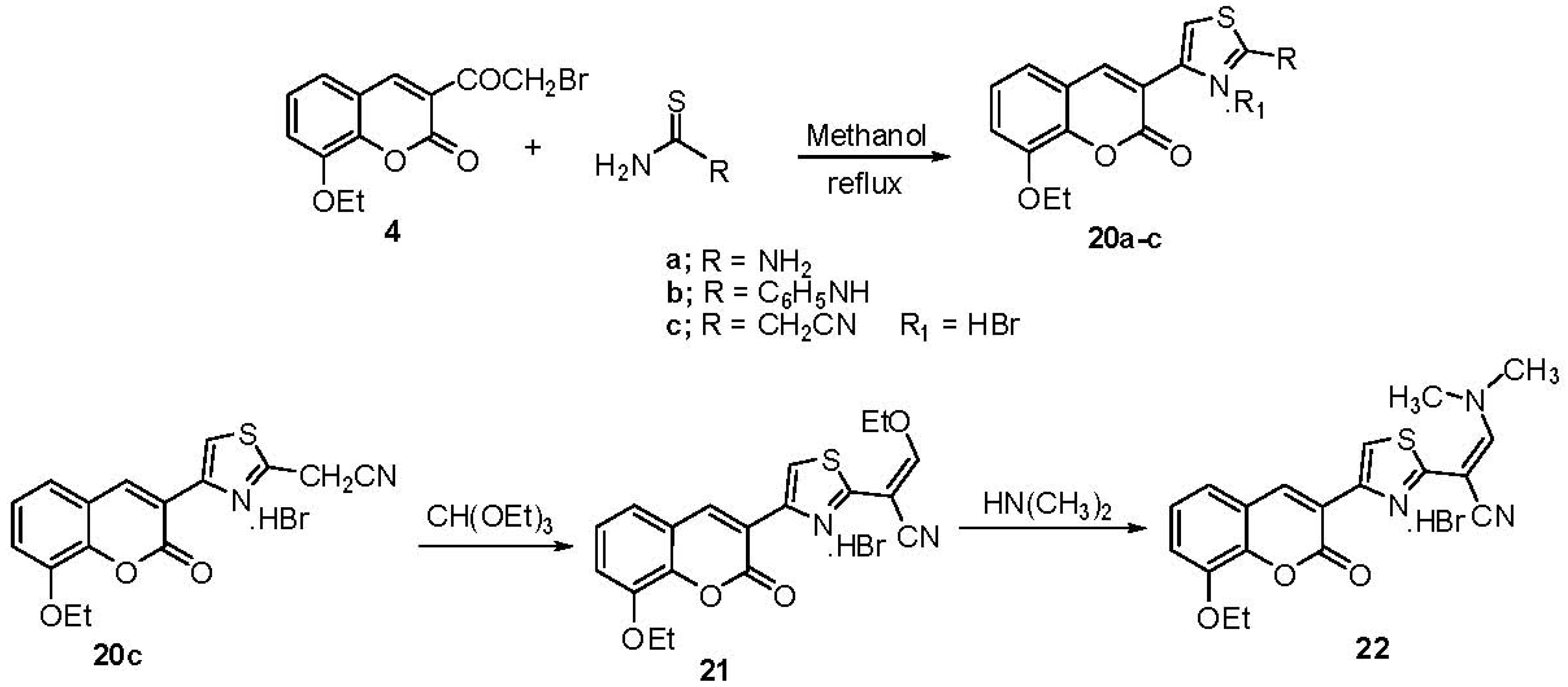

3. Experimental
3.1. General
3.2. Reaction of 20c with Aromatic Aldehydes
3.3. Antibacterial Activity
| Compd. No.a | Inhibition Zone Diameter in mm | |||||||
|---|---|---|---|---|---|---|---|---|
| Bordetella bronchiseptica ATCC 4617 | Escheri-chia coli ATCC 14169 | Bacillus pumilus ATCC 14884 | Bacillus Subtilis ATCC 6633 | Staph.aureus ATCC 29737 | Staph. Epidermidis ATCC 12228 | Candida albicans ATCC 10231 | Saccharomyces cerevisae ATCC 9080 | |
| 3 | 19 | 20 | 11 | 12 | 19 | 12 | 18 | 10 |
| 4 | 22 | 27 | 23 | 20 | 20 | 16 | 13 | 15 |
| 5 | 24 | 18 | 28 | 28 | 27 | 25 | 15 | 21 |
| 6 | 28 | 14 | 18 | 26 | 15 | 18 | 24 | 18 |
| 7 | 22 | 13 | 19 | 23 | 14 | 13 | 17 | 22 |
| 8 | NA | NA | NA | NA | NA | NA | NA | NA |
| 9 | NA | 13 | NA | NA | 10 | NA | NA | NA |
| 11 | NA | NA | NA | NA | NA | NA | NA | NA |
| 12 | NA | 14 | NA | NA | 14 | NA | NA | NA |
| 16 | 27 | 19 | 18 | 24 | 12 | 13 | 17 | 13 |
| 20a | NA | 10 | NA | NA | 10 | NA | NA | NA |
| 20b | NA | 11 | NA | NA | 12 | NA | NA | NA |
| 20c | 13 | 10 | 18 | 11 | 11 | 20 | 14 | 19 |
| 21 | 22 | 10 | 18 | 20 | 11 | 19 | 22 | 11 |
| 22 | 20 | 11 | 20 | 19 | 13 | 9 | 13 | 15 |
| 24 | 13 | 10 | NA | 21 | 10 | NA | NA | 12 |
| 25 | 8 | 14 | NA | 14 | 15 | NA | NA | 14 |
| 27a | 19 | 10 | 14 | 18 | 11 | 20 | 16 | 19 |
| 27b | 20 | 10 | 14 | 21 | 11 | 13 | 19 | 11 |
| 28 | 27 | 10 | 22 | 22 | 11 | 19 | 23 | 20 |
| Ampicillin* | 24 | 25 | 20 | 25 | 26 | 25 | - | - |
| Mycostatin* | - | - | - | - | - | - | 22 | 24 |
4. Conclusions
Conflict of Interest
- Sample Availability: Samples of the all compounds are available from the authors.
References and Notes
- Kennedy, R.O.; Thornes, R.D. Coumarins: Biology, Applications and Mode of Action; Wiley & Sons: Chichester, UK, 1997. [Google Scholar]
- Murray, R.D.H.; Mendez, J.; Brown, S.A. The Natureal Coumarins; Occurrence, Chemistry and Biochemistry; Wiley & Sons: New York, NY, USA, 1982. [Google Scholar]
- Egan, D.; Kennedy, R.O.; Moran, E.; Cox, D.; Prosser, E.; Thornes, R.D. The pharmacology, metabolism, analysis, and applications of coumarin and coumarin-related compounds. Drug Metab. Rev. 1990, 22, 503–529. [Google Scholar] [CrossRef]
- Gabor, M. The Pharmacology of Benzopyran Derivatives and Related Compounds; Akademiai Kiado: Budapest, Hungary, 1988; pp. 91–126. [Google Scholar]
- Guilbault, G.G. Practical Fluorescence. Theory, Methods and Techniques; Marcel Dekker: New York, NY, USA, 1973; p. 354. [Google Scholar]
- Rathbone, D.L.; Wang, D.; Su, Y.; Billington, D.C. Molecular recognition by fluorescent imprinted polymers. Tetrahedron Lett. 2000, 41, 123–126. [Google Scholar] [CrossRef]
- Abyshev, A.Z.; Gindin, V.A.; Semenov, E.V.; Agaev, E.M.; Abdulla-zade, A.A.; Guseinov, A.B. Structure and biological properties of 2H-1-benzopyran-2-one (coumarin) derivatives. Pharm. Chem. J. 2006, 40, 607–610. [Google Scholar] [CrossRef]
- Al-Bayati, R.I.; Hussain Al-Amiery, A.A.; Al-Majedy, Y.K. Design, synthesis and bioassay of novel coumarins. Afr. J. Pure Appl. Chem. 2010, 4, 74–86. [Google Scholar]
- Lee, S.; Sivakumar, K.; Shin, W.S.; Xie, F.; Wang, Q. Synthesis and anti-angiogenesis activity of coumarin derivatives. Bioorg. Med. Chem. Lett. 2006, 16, 4596–4599. [Google Scholar] [CrossRef]
- Monteiro, V.F.F.; Mathias, L.; Vieira, I.J.C.; Schripsema, J.; Braz-Filho, R. Prenylated coumarins, chalcone and new cinnamic acid and dihydrocinnamic acid derivatives from brosimum gaudichaudii. J. Braz. Chem. Soc. 2002, 13, 281–287. [Google Scholar] [CrossRef]
- Satyanarayana, V.S.V.; Sreevani, P.; Sivakumar, A.; Vijayakumar, V. Synthesis and antimicrobial activity of new Schiff bases containing coumarin moiety and their spectral characterization. ARKIVOC 2008, xvii, 221–233. [Google Scholar]
- Nofal, Z.M.; El-Zahar, M.I.; Abd El-Karim, S.S. Novel coumarin derivatives with expected biological activity. Molecules 2000, 5, 99–113. [Google Scholar] [CrossRef]
- Radanyi, C.; Bras, G.L.; Messaoudi, S.; Bouclier, C.; Peyrat, J.F.; Brion, J.D.; Marsaud, V.; Renoir, J.M.; Alami, M. Synthesis and biological activity of simplified denoviose-coumarins related to novobiocin as potent inhibitors of heat-shock protein 90 (hsp90). Bioorg. Med. Chem. Lett. 2008, 18, 2495–2498. [Google Scholar] [CrossRef]
- Ahmad, R.; Asad, M.; Siddiqui, Z.N.; Kumar, A. Screening of synthetic new heterocyclic derivatives of 3-formyl-4-hydroxycoumarin for anti-inflammatory activity in albino rats. J. Pharm. Res. Health Care 2009, 1, 46–62. [Google Scholar]
- Kostova, I. Synthetic and natural coumarins as cytotoxic agents. Curr. Med. Chem. Anticancer Agents 2005, 5, 29–46. [Google Scholar] [CrossRef]
- El-Agrody, A.M. Condensation Reactions of α-Cyanocinnamonitriles with Naphthols: Synthesis of Naphthopyranopyrimidines anda Naphthopyranone. J. Chem. Res.Synopses 1994, 280–281. [Google Scholar]
- El-Agrody, A.M.; Emam, H.A.; El-Hakim, M.H.; Abd El-Latif, M.S.; Fakery, A.H. Activated nitriles in heterocyclic synthesis: Synthesis of Pyrano[2,3-c]pyrimidine and Pyrano[3,2-e][1,2,4]triazolo[1,5-c]pyrimidine derivatives. J. Chem. Res. Synopses 1997, 320–321. [Google Scholar]
- Bedair, A.H.; El-Hady, N.A.; Abd El-Latif, M.S.; Fakery, A.H.; El-Agrody, A.M. 4-Hydroxycoumarin in heterocyclic synthesis-Part III. Synthesis of some new pyrano[2,3-c]pyrimidine, 2-substituted[1,2,4]triazolo[1,5-c]pyrimidine and pyrimido[1,6-b][1,2,4]triazine derivatives. Farmaco 2000, 55, 708–714. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; El-Hakim, M.H.; Abd El-Latif, M.S.; Fakery, A.H.; El-Sayed, E.S.M.; El-Ghareab, K.A. Synthesis of pyrano[2,3-c]pyrimidine and pyrano[3,2-e]-[1,2,4]triazolo[2,3-c]pyrimidine derivatives with promising antibacterial activity. Acta Pharm. 2000, 50, 111–120. [Google Scholar]
- Sayed, A.Z.; El-Hady, N.A.; El-Agrody, A.M. Condensation of α-cyanocinnamonitriles with 6-bromo-2-naphthol: synthesis of pyrano[2,3-c]pyrimidine and pyrano[3,2-e][1,2,4]triazolo[2,3-c]pyrimidine derivatives. J. Chem. Res. Synopses 2000, 164–166. [Google Scholar]
- El-Agrody, A.M.; Abd El-Latif, M.S.; El-Hady, N.A.; Fakery, A.H.; Bedair, A.H. Heteroaromatization with 4-Hydroxycoumarin Part II: Synthesis of Some New Pyrano[2,3-c]pyrimidines, [1,2,4]triazolo[1,5-c]pyrimidines and Pyrimido[1,6-b][1,2,4]triazine Derivatives. Molecules 2001, 6, 519–527. [Google Scholar] [CrossRef]
- Bedair, A.H.; Emam, H.A.; El-Hady, N.A.; Ahmed, K.A.R.; El-Agrody, A.M. Synthesis and antimicrobial activities of novel naphtho[2,1-b]pyran, pyrano[2,3-c]pyrimidine and pyrano[3,2-e][1,2,4]triazolo[2,3-c]-pyrimidine derivatives. II Farmaco 2001, 56, 965–973. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; Eid, F.A.; Emam, H.A.; Mohamed, H.M.; Bedair, A.H. Synthesis of 9-Methoxy and 9-Acetoxy-3-amino-1-(4-methoxyphenyl)-1H-benzo[f]chromene-2-carbonitriles via2-(iminopiperidin-1-yl-methyl)-3-(4-methoxyphenyl)acrylonitrile as Intermediate. Z. Naturforsch. B 2002, 57b, 579–585. [Google Scholar]
- Khafagy, M.M.; Abd El-Wahab, A.H.F.; Eid, F.A.; El-Agrody, A.M. Synthesis of halogen derivatives of benzo[h]chromene and benzo[a]anthracene with promising antimicrobial activities. Farmaco 2002, 57, 715–722. [Google Scholar] [CrossRef]
- Eid, F.A.; Bedair, A.H.; Emam, H.A.; Mohamed, H.M.; El-Agrody, A.M. Reaction of activated nitriles with methanolic piperidine and synthesis of 1H-Benzo[f]chromene, Diazabenzo[j]anthracene and Diazabenzo[a]- [1,2,4]triazolo[j]anthracene derivatives. Al-Azhar Bull. Sci. 2003, 14, 311–327. [Google Scholar]
- Abd-El-Aziz, A.S.; El-Agrody, A.M.; Bedair, A.H.; Christopher Corkery, T.; Ata, A. Synthesis of hyroxyquinoline derivatives, aminoheydroxychromene, aminocoumarin and their anitimicrobial activities. Heterocycles 2004, 63, 1793–1812. [Google Scholar] [CrossRef]
- Abd-El-Aziz, A.S.; Mohamed, H.M.; Mohammed, S.; Zahid, S.; Ata, A.; Bedair, A.H.; El-Agrody, A.M.; Harvey, P.D. Synthesis of novel coumarin and benzocoumarin derivatives and their biological and photophysical studies. J. Heterocycl. Chem. 2007, 44, 1287–1301. [Google Scholar] [CrossRef]
- Sabry, N.M.; Mohamed, H.M.; Motlaq, S.S.; El-Agrody, A.M. Synthesis of 4H-chromene, coumarin, 12H-chromeno[2,3-c]pyrimidine derivatives and some of their antimicrobial and cytotoxicity activities. Eur. J. Med. Chem. 2011, 46, 765–772. [Google Scholar] [CrossRef]
- Zhuravel, I.O.; Kovalenko, S.M.; Vlasov, S.V.; Chernykh, V.P. Solution phase synthesis of a combinatorial library of 3-[4-(coumarins-3-yl)-1,3-thiazol-2-yl carbamoyl]propanoic acid amides. Molecules 2005, 10, 444–456. [Google Scholar] [CrossRef]
- Czerney, P.; Jena, V.C.Z.; Hartmann, H. Heterocyclic substituted coumarins from β-chloropropeniminium salts. J. Prakt. Chem. 1982, 324, 255–266. [Google Scholar] [CrossRef]
- Koelsch, C.F.; Embree, H.D. Condensation of 3-acetylcoumarin with acetone and amines. J. Org. Chem. 1958, 23, 1606–1608. [Google Scholar] [CrossRef]
- O’Callaghan, C.N.; McMurry, T.B.H. Dimerisation of Kneovenagel condensation products obtained from simple unconjugated and α,β-unsaturated ketones. J. Chem. Res. Synopses 1999, 458–459. [Google Scholar]
- Zhou, J.F.; Gong, G.X.; An, L.T.; Liu, Y.; Zhu, F.X.; Zhu, Y.L.; Ji, S.J. An efficient synthesis of quinoxalines under catalyst free and microwave-irradiation conditions. Synlett 2008, 20, 3163–3166. [Google Scholar]
- Kumar, P.V.; Rao, V.R. Synthesis and biological activity of some 3-imidazo [1,2-a] pyridin-2-yl-chromen-2-one and 3-indolizin-2-yl-chromen-2-one. Indian J. Chem. Sect. B 2005, 44, 2120–2125. [Google Scholar]
- Ramanna, S.; Rao, V.R.; Kumari, T.S.; Rao, T.V.P. Synthesis of N-(4-2H-1-benzopyran-2-one)-2-thiazolyl)phthalimides. Phosphorous Sulfur Silicon 1995, 107, 197–204. [Google Scholar] [CrossRef]
- Islam, A.M.; Ali, F.M.; El-Sharief, A.M.Sh.; Bedair, A.H.; El-Masry, F.M. Some reactions of coumarins with hydrazine and ethylenediamine. Egypt J. Chem. 1983, 26, 233–239. [Google Scholar]
- Chunduru, R.; Sreenivasa, V.; Vedula, R.R. One pot synthesis of 3-[2-(arylamino)-thiazole-4-yl]coumarins in a three-component synthesis and a catalyst and solvent-free synthesis grinding. J. Chem. Res. 2010, 34, 50–53. [Google Scholar] [CrossRef]
- Santhosh, P.; Chunduru, V.S.R.; Rao, V.R. One pot synthesis of tri-substituted pyrazoles via multi-component approach. Chem. Heterocycl. Comp. 2011, 47, 448–451. [Google Scholar] [CrossRef]
- Czerney, P.; Jena, V.C.Z.; Hartmann, H. 3-α-Bromoacetylcoumarines as synthones for heterocyclic substituted coumarins. J. Prakt. Chem. 1983, 325, 551–560. [Google Scholar] [CrossRef]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by agar dilution. Clin. Microbiol. Infect. 2000, 6, 509–515. [CrossRef]
- National Committee for Clinical Laboratory Standards (NCCLS), Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, 5th ed; Approved Standard M7-A5. NCCLS: Wayne, PA, USA, 2000.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mohamed, H.M.; El-Wahab, A.H.F.A.; Ahmed, K.A.; El-Agrody, A.M.; Bedair, A.H.; Eid, F.A.; Khafagy, M.M. Synthesis, Reactions and Antimicrobial Activities of 8-Ethoxycoumarin Derivatives. Molecules 2012, 17, 971-988. https://doi.org/10.3390/molecules17010971
Mohamed HM, El-Wahab AHFA, Ahmed KA, El-Agrody AM, Bedair AH, Eid FA, Khafagy MM. Synthesis, Reactions and Antimicrobial Activities of 8-Ethoxycoumarin Derivatives. Molecules. 2012; 17(1):971-988. https://doi.org/10.3390/molecules17010971
Chicago/Turabian StyleMohamed, Hany M., Ashraf H. F. Abd El-Wahab, Kamal A. Ahmed, Ahmed M. El-Agrody, Ahmed H. Bedair, Fathy A. Eid, and Mostafa M. Khafagy. 2012. "Synthesis, Reactions and Antimicrobial Activities of 8-Ethoxycoumarin Derivatives" Molecules 17, no. 1: 971-988. https://doi.org/10.3390/molecules17010971
APA StyleMohamed, H. M., El-Wahab, A. H. F. A., Ahmed, K. A., El-Agrody, A. M., Bedair, A. H., Eid, F. A., & Khafagy, M. M. (2012). Synthesis, Reactions and Antimicrobial Activities of 8-Ethoxycoumarin Derivatives. Molecules, 17(1), 971-988. https://doi.org/10.3390/molecules17010971




