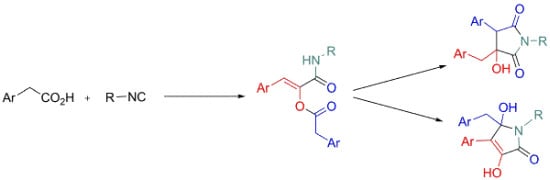
3. Experimental
3.1. General
1H-NMR and 13C-NMRs were recorded on a Varian MERCURYplus 300 instrument at 300/75 MHz. Chemical shifts are reported in p.p.m. using TMS (0.00 ppm) as internal standard for 1H spectra and the residual solvent peak for 13C ones. Coupling constants (J) are reported in Hz. HRMS were recorded with a Micromass AutoSpec instrument at the Parque Cientifico Tecnologico of the University of Burgos (Spain). Microwave heating was performed in a CEM Discover apparatus equipped with infrared detector for temperature measurement. All reagents have been bought from Fluka or Aldrich.
3.2. General Procedure for the Synthesis of Compounds 3
The acid (1 mmol) and the isocyanide (1 mmol) were dissolved in dry dichloromethane (DCM, 1 mL) in a closed MW vessel and heated at 100 °C for 30 min using 150 W power. The resulting mixture was then diluted with DCM (9 mL) and washed with saturated sodium bicarbonate (2 × 5 mL). The organic layer was dried with sodium sulphate, filtered, concentrated in vacuo and purified by flash chromatography with PE/EtOAc eluent.
(Z)-3-(Butylamino)-3-oxo-1-phenylprop-1-en-2-yl 2-phenylacetate (3a). 1H-NMR (CDCl3) δ 0.87 (t, J = 7.1 Hz, 3H), 1.15–1.33 (m, 4H), 3.13 (q, J = 6.9 Hz, 2H), 3.83 (s, 2H), 5.61 (br s, 1H), 7.22–7.33 (m, 5H), 7.34–7.46 (m, 6H); 13C-NMR (CDCl3) δ 168.01, 162.47, 139.46, 132.87, 132.51, 129.68, 129.57, 129.34, 129.28, 128.82, 128.08, 124.20, 41.89, 39.59, 31.46, 20.07, 13.89; HRMS: calcd. for C21H23NO3 337.1678, found 337.1670.
(Z)-3-(Butylamino)-1-(4-chlorophenyl)-3-oxoprop-1-en-2-yl 2-(4-chlorophenyl)acetate (3b). 1H-NMR (CDCl3) δ 0.90 (t, J = 7.1 Hz, 3H), 1.15–1.35 (m, 4H), 3.20 (q, J = 6.9 Hz, 2H), 3.80 (s, 2H), 5.53 (br s, 1H), 7.10–7.40 (m, 9H); 13C-NMR (CDCl3) δ 167.35, 161.93, 139.79, 135.04, 134.10, 130.89, 130.74, 130.65, 130.57, 129.32, 128.92, 122.61, 40.90, 39.50, 31.34, 19.95, 13.72; HRMS: calcd. for C21H21Cl2NO3 405.0898, found 405.0892.
(Z)-3-(Butylamino)-1-(3-methoxyphenyl)-3-oxoprop-1-en-2-yl 2-(3-methoxyphenyl)acetate (3c). 1H-NMR (CDCl3) δ 0.87 (t, J = 7.0 Hz, 3H), 1.12–1.30 (m, 4H), 3.12 (q, J = 6.6 Hz, 2H), 3.79 (s, 3H), 3.80 (br s, 5H), 5.53 (t, J = 5.3 Hz, 1H), 6.80–7.05 (m, 6H), 7.15–7.35 (m, 3H); 13C-NMR (CDCl3) δ 167.83, 162.26, 160.34, 159.75, 139.51, 134.25, 133.79, 130.41, 129.85, 124.30, 122.07, 121.65, 115.23, 115.03, 114.94, 113.61, 55.45, 55.39, 41.96, 39.60, 31.45, 20.10, 13.92; HRMS: calcd. for C23H27NO5 397.1889, found 397.1891.
(Z)-3-(Benzylamino)-3-oxo-1-phenylprop-1-en-2-yl 2-phenylacetate (3d). 1H-NMR (CDCl3) δ 3.81 (s, 2H), 4.35 (d, J = 5.9 Hz, 2H), 5.81 (br s, 1H), 7.10–7.40 (m, 16H); 13C-NMR (CDCl3) δ 168.06, 162.47, 139.26, 137.84, 132.64, 132.43, 129.77, 129.44, 129.29, 128.89, 128.88, 128.06, 127.92, 127.75, 124.73, 43.82, 41.92 (two C-H aromatic signals overlapped); HRMS: calcd. for C24H21NO3 371.1521, found 371.1540.
(Z)-3-(Benzylamino)-1-(4-chlorophenyl)-3-oxoprop-1-en-2-yl 2-(4-chlorophenyl)acetate (3e). 1H-NMR (CDCl3) δ 3.75 (s, 2H), 4.38 (d, J = 5.7 Hz, 2H), 6.13 (t, J = 5.7 Hz, 1H), 7.10–7.35 (m, 14H); 13C-NMR (CDCl3) δ 167.45, 161.96, 139.53, 137.40, 135.19, 133.96, 130.64, 130.57, 130.53, 129.26, 128.95, 128.81, 127.77, 123.18, 43.73, 40.85 (two C-H aromatic signals overlapped); HRMS: calcd. for C24H19Cl2NO3 439.0742, found 439.0740.
(Z)-3-(Benzylamino)-1-(3-methoxyphenyl)-3-oxoprop-1-en-2-yl 2-(3-methoxyphenyl)acetate (3f). 1H-NMR (CDCl3) δ 3.69 (s, 3H), 3.76 (br s, 5H), 4.32 (d, J = 5.9 Hz, 2H), 5.97 (t, J = 5.8 Hz, 1H), 6.70–7.30 (m, 14H); 13C-NMR (CDCl3) δ 167.99, 162.40, 160.17, 159.73, 139.39, 137.83, 134.01, 133.64, 130.24, 129.84, 128.79, 127.70, 127.60, 124.71, 122.10, 121.44, 115.28, 115.04, 114.93, 113.58, 55.36, 55.33, 43.70, 41.82; HRMS: calcd. for C26H25NO5 431.1733, found 431.1715.
(Z)-Benzyl 3-(3-phenyl-2-(2-phenylacetoxy)acrylamido)propanoate (3g). 1H-NMR (CDCl3) δ 2.57 (t, J = 6.0 Hz, 2H), 3.54 (q, J = 6.1 Hz, 2H), 3.83 (s, 2H), 5.13 (s, 2H), 6.50 (br s, 1H), 7.12–7.49 (m, 16H); 13C NMR (CDCl3) δ 172.72, 168.28, 162.55, 139.49, 135.72, 132.67, 132.33, 129.80, 129.77, 129.35, 129.20, 128.86, 128.80, 128.62, 128.44, 127.96, 124.24, 66.84, 41.61, 35.25, 33.81; HRMS: calcd. for C27H25NO5 443.1733, found 443.1740.
(Z)-Benzyl 3-(3-(4-chlorophenyl)-2-(2-(4-chlorophenyl)acetoxy)acrylamido)propanoate (3h). 1H-NMR (CDCl3) δ 2.61 (t, J = 6.0 Hz, 2H), 3.58 (q, J = 6.0 Hz, 2H), 3.79 (s, 2H), 5.13 (s, 2H), 6.68 (t, J = 6.0 Hz, 1H), 7.11 (s, 1H), 7.19 (s, 5H), 7.23–7.41 (m, 8H); 13C-NMR (CDCl3) δ 172.96, 167.87, 162.20, 140.00, 135.62, 135.29, 134.05, 131.10, 130.95, 130.86, 130.69, 129.33, 129.04, 128.88, 128.66, 128.40, 122.91, 66.90, 40.75, 35.22, 33.70; HRMS: calcd. for C27H23Cl2NO5 511.0953, found 511.1001.
(Z)-Benzyl 3-(3-(3-methoxyphenyl)-2-(2-(3-methoxyphenyl)acetoxy)acrylamido)propanoate (3i). 1H-NMR (CDCl3) δ 2.55 (t, J = 6.1 Hz, 2H), 3.52 (q, J = 6.3 Hz, 2H), 3.76 (s, 3H), 3.77 (s, 3H), 3.81 (s, 2H), 5.12 (s, 2H), 6.46 (t, J = 5.9 Hz, 1H), 6.81–6.99 (m, 6H), 7.16 (t, J = 8.0 Hz, 1H), 7.21 (s, 1H), 7.24–7.40 (m, 6H); 13C-NMR (CDCl3) δ 172.59, 168.20, 162.44, 160.17, 159.72, 139.61, 135.72, 134.01, 133.60, 130.19, 129.81, 128.83, 128.60, 128.44, 124.30, 122.10, 121.90, 115.37, 115.25, 115.02, 113.44, 66.82, 55.42, 55.39, 41.53, 35.27, 33.77; HRMS: calcd. for C29H29NO7 503.1944, found 503.1958.
3.3. General Procedure for the Synthesis of Compounds 9
Compound 3 (0.5 mmol) and triethylamine (0.5 mmol) were dissolved in dry benzene (1 mL) in a closed MW vessel and heated at 90 °C for 30 min with 100 W power. The resulting suspension was diluted with Et2O (2 mL) and filtered. The solid was then washed with PE/Et2O 1:1 (5 mL) and dried under vacuum or alternatively was purified by flash chromatography with PE/EtOAc eluent.
3-Benzyl-1-butyl-3-hydroxy-4-phenylpyrrolidine-2,5-dione (9a). 1H-NMR (CDCl3) δ 0.91 (t, J = 7.2 Hz, 3H), 1.18–1.33 (m, 2H), 1.48–1.60 (m, 2H), 2.60–2.90 (br s, 1H), 3.09 (d, J = 13.5 Hz, 1H), 3.26 (d, J = 13.5 Hz, 1H), 3.53 (t, J = 7.3 Hz, 2H), 4.05 (s, 1H), 6.90–6.98 (m, 2H), 7.25–7.38 (m, 8H); 13C-NMR (CDCl3) δ 178.61, 175.65, 134.29, 132.18, 130.70, 129.97, 129.08, 129.02, 128.48, 127.93, 77.14, 54.62, 44.18, 39.09, 29.82, 20.18, 13.74; HRMS: calcd. for C21H23NO3 337.1678, found 337.1670.
1-Butyl-3-(4-chlorobenzyl)-4-(4-chlorophenyl)-3-hydroxypyrrolidine-2,5-dione (9b). 1H-NMR (CDCl3) δ 0.92 (t, J = 7.2 Hz, 3H), 1.18–1.32 (m, 2H), 1.47–1.60 (m, 2H), 2.84 (s, 1H), 3.04 (d, J = 13.7 Hz, 1H), 3.19 (d, J = 13.7 Hz, 1H), 3.54 (t, J = 7.3 Hz, 2H), 3.94 (s, 1H), 6.88 (d, J = 8.4 Hz, 2H), 7.19 (d, J = 8.4 Hz, 2H), 7.25–7.35 (m, 4H); 13C-NMR (CDCl3) δ 178.50, 175.31, 134.57, 134.10, 132.49, 131.95, 131.36, 130.32, 129.27, 129.12, 53.78, 43.12, 39.21, 29.77, 20.14, 13.77 (one signal covered by solvent residual peak); HRMS: calcd. for C21H21Cl2NO3 405.0898, found 405.0887.
1-Butyl-3-hydroxy-3-(3-methoxybenzyl)-4-(3-methoxyphenyl)pyrrolidine-2,5-dione (9c). 1H-NMR (CDCl3) δ 0.92 (t, J = 7.3 Hz, 3H), 1.20–1.35 (m, 2H), 1.48–1.63 (m, 2H), 2.43 (s, 1H), 3.07 (d, J = 13.5 Hz, 1H), 3.23 (d, J = 13.5 Hz, 1H), 3.55 (t, J = 7.4 Hz, 2H), 3.73 (s, 3H), 3.78 (s, 3H), 4.05 (s, 1H), 6.40–6.60 (m, 2H), 6.75–6.95 (m, 4H), 7.20–7.30 (m, 2H); 13C-NMR (CDCl3) δ 178.47, 175.57, 160.05, 159.96, 135.70, 133.34, 130.12, 130.08, 122.89, 121.94, 116.46, 115.88, 113.95, 113.15, 77.02, 55.38, 55.34, 54.36, 43.94, 39.09, 29.81, 20.17, 13.79; HRMS: calcd. for C23H27NO5 397.1889, found 397.1871.
1,3-Dibenzyl-3-hydroxy-4-phenylpyrrolidine-2,5-dione (9d). 1H-NMR (CDCl3) δ 3.08 (d, J = 13.5 Hz, 1H), 3.26 (d, J = 13.5 Hz, 1H), 4.06 (s, 1H), 4.67 (d, J = 14.1 Hz, 1H), 4.73 (d, J = 14.1 Hz, 1H), 5.30 (s, 1H), 6.80–6.94 (m, 2H), 7.12–7.42 (m, 13H); 13C-NMR (CDCl3) δ 178.05, 175.32, 135.43, 134.01, 131.86, 130.63, 129.88, 129.12, 128.95, 128.84, 128.62, 128.30, 127.94, 54.61, 44.02, 42.86 (two C–H aromatic signals overlapped); HRMS: calcd. for C24H21NO3 371.1521, found 371.1508.
1-Benzyl-3-(4-chlorobenzyl)-4-(4-chlorophenyl)-3-hydroxypyrrolidine-2,5-dione (9e). 1H-NMR (CDCl3) δ 1H-NMR (300 MHz, CDCl3) δ 2.73 (s, 1H), 2.99 (d, J = 13.7 Hz, 1H), 3.17 (d, J = 13.7 Hz, 1H), 3.94 (s, 1H), 4.66 (d, J = 13.8 Hz, 1H), 4.72 (d, J = 13.8 Hz, 1H), 6.82 (d, J = 8.4 Hz, 2H), 7.06 (d, J = 8.4 Hz, 2H), 7.15–7.35 (m, 9H); 13C-NMR (CDCl3) δ 178.05, 174.88, 135.14, 134.62, 134.00, 132.22, 131.83, 131.28, 130.24, 129.25, 129.15, 128.99, 128.79, 128.47, 76.91, 53.98, 43.18, 42.93; HRMS: calcd. for C24H19Cl2NO3 439.0742, found 439.0737.
1-Benzyl-3-hydroxy-3-(3-methoxybenzyl)-4-(3-methoxyphenyl)pyrrolidine-2,5-dione (9f). 1H-NMR (CDCl3) δ 2.22 (s, 1H), 3.07 (d, J = 13.5 Hz, 1H), 3.21 (d, J = 13.5 Hz, 1H), 3.64 (s, 3H), 3.73 (s, 3H), 4.07 (s, 1H), 4.68 (d, J = 14.1 Hz, 1H), 4.74 (d, J = 14.1 Hz, 1H), 6.33–6.37 (m, 1H), 6.46 (d, J = 7.6 Hz, 1H), 6.76–6.86 (m, 4H), 7.15–7.24 (m, 2H), 7.27–7.38 (m, 5H); 13C-NMR (CDCl3) δ 177.98, 175.17, 160.06, 160.02, 135.57, 135.46, 133.18, 130.20, 130.12, 128.96, 128.79, 128.26, 122.85, 121.99, 116.34, 115.28, 114.39, 113.33, 55.39, 55.30, 54.56, 43.92, 42.85 (aliphatic quaternary C signal covered by residual solvent peak); HRMS: calcd. for C26H25NO5 431.1733, found 431.1731.
Benzyl 3-(3-benzyl-3-hydroxy-2,5-dioxo-4-phenylpyrrolidin-1-yl)propanoate (9g). 1H-NMR (CDCl3) δ 2.24 (s, 1H), 2.68 (t, J = 7.2 Hz, 2H), 3.11 (d, J = 13.5 Hz, 1H), 3.21 (d, J = 13.5 Hz, 1H), 3.79–3.95 (m, 2H), 4.05 (s, 1H), 5.11 (s, 2H), 6.87–6.95 (m, 2H), 7.22–7.40 (m, 13H); 13C-NMR (CDCl3) δ 178.16, 175.37, 170.59, 135.55, 134.11, 131.78, 130.64, 129.90, 129.08, 129.05, 128.78, 128.66, 128.61, 128.54, 127.95, 77.08, 67.05, 54.59, 43.84, 34.93, 32.04; HRMS: calcd. for C27H25NO5 443.1733, found 443.1718.
Benzyl 3-(3-(4-chlorobenzyl)-4-(4-chlorophenyl)-3-hydroxy-2,5-dioxopyrrolidin-1-yl)propanoate (9h). 1H-NMR (CDCl3) δ 2.67 (s, 1H), 2.70 (t, J = 6.6 Hz, 2H), 3.03 (d, J = 13.8 Hz, 1H), 3.13 (d, J = 13.8 Hz, 1H), 3.80–3.95 (m, 3H), 5.09 (s, 2H), 6.84 (dm, J = 8.4 Hz, 2H), 7.17 (dm, J = 8.4 Hz, 2H), 7.25 (dm, J = 8.4 Hz, 2H), 7.31 (dm, J = 8.4 Hz, 2H), 7.33–7.38 (m, 5H); 13C-NMR (CDCl3) δ 177.91, 174.99, 170.67, 135.44, 134.62, 134.07, 132.51, 132.00, 131.38, 130.07, 129.33, 129.26, 129.14, 128.83, 128.71, 67.20, 53.98, 42.85, 35.15, 31.95 (aliphatic quaternary C signal covered by residual solvent peak); HRMS: calcd. for C27H23Cl2NO5 511.0953, found 511.0938.
Benzyl 3-(3-hydroxy-3-(3-methoxybenzyl)-4-(3-methoxyphenyl)-2,5-dioxopyrrolidin-1-yl)propano-ate (9i). 1H-NMR (CDCl3) δ 2.18 (s, 1H), 2.69 (td, J = 7.1, 2.3 Hz, 2H), 3.09 (d, J = 13.5 Hz, 1H), 3.17 (d, J = 13.5 Hz, 1H), 3.73 (s, 3H), 3.78 (s, 3H), 3.88 (t, J = 6.9 Hz, 2H), 4.05 (s, 1H), 5.11 (s, 2H), 6.42–6.45 (m, 1H), 6.52 (d, J = 7.5 Hz, 1H), 6.77–6.86 (m, 4H), 7.15–7.30 (m, 2H), 7.32–7.38 (m, 5H); 13C-NMR (CDCl3) δ 178.00, 175.17, 170.62, 160.09, 160.07, 135.64, 135.61, 133.10, 130.26, 130.14, 128.82, 128.70, 128.65, 122.91, 121.83, 116.57, 115.81, 114.16, 113.20, 77.00, 67.10, 55.44, 55.41, 54.66, 43.91, 35.02, 32.07; HRMS: calcd. for C29H29NO7 503.1944, found 503.1933.
3.4. General Procedure for the Synthesis of Compounds 10
Compound 3 (0.5 mmol) was suspended in dry t-BuOH (2 mL) and sodium t-butoxide (0.55 mmol) was added; the solution turned yellow immediately and a TLC control showed complete disappearance of starting material. The reaction was quenched with 0.5 M NH4H2PO4 (5 mL) and extracted with Et2O (2 × 5 mL). The combined organics were anhydrified with sodium sulphate, filtered and concentrated in vacuo. The crude was taken up in DCM (3 mL), a white/yellow solid crashed down and it was collected by filtration. Alternatively the crude was purified by flash chromatography with PE/EtOAc eluent.
5-Benzyl-1-butyl-3,5-dihydroxy-4-phenyl-1H-pyrrol-2(5H)-one (10a). 1H-NMR (DMSO-d6) δ 0.93 (t, J = 7.2 Hz, 3H), 1.27–1.43 (m, 2H), 1.57–1.75 (m, 2H), 3.19 (d, J = 13.8 Hz, 1H), 3.28 (d, J = 13.7 Hz, 1H), 3.32–3.47 (m, 2H), 6.50 (s, 1H), 6.69 (dd, J = 6.6, 2.9 Hz, 2H), 7.01–7.12 (m, 3H), 7.28 (t, J = 7.2 Hz, 1H), 7.44 (t, J = 7.8 Hz, 2H), 8.01 (dd, J = 8.4, 1.0 Hz, 2H), 10.02 (s, 1H); 13C-NMR (DMSO-d6) δ 164.70, 144.39, 135.54, 132.89, 129.61, 128.63, 127.87, 127.69, 127.05, 126.84, 118.97, 90.78, 41.30, 39.09, 31.09, 20.36, 14.10; HRMS: calcd. for C21H23NO3 337.1678, found 337.1690.
1-Butyl-5-(4-chlorobenzyl)-4-(4-chlorophenyl)-3,5-dihydroxy-1H-pyrrol-2(5H)-one (10b). 1H-NMR (DMSO-d6) δ 0.92 (t, J = 7.3 Hz, 3H), 1.27–1.42 (m, 2H), 1.55–1.74 (m, 2H), 3.14 (d, J = 13.8 Hz, 1H), 3.30 (d, J = 13.8 Hz, 1H), 3.39 (t, J = 7.2 Hz, 2H), 6.61 (s, 1H), 6.68 (d, J = 8.4 Hz, 2H), 7.15 (d, J = 8.3 Hz, 2H), 7.51 (d, J = 8.7 Hz, 2H), 7.99 (d, J = 8.7 Hz, 2H), 10.35 (s, 1H); 13C-NMR (DMSO-d6) δ 164.08, 145.04, 134.27, 131.40, 131.21, 131.09, 130.91, 128.92, 128.43, 127.55, 117.29, 90.18, 30.82, 20.06, 13.80 (two signals covered by solvent residual peak); HRMS: calcd. for C21H21Cl2NO3 405.0898, found 405.0879.
1-Butyl-3,5-dihydroxy-5-(3-methoxybenzyl)-4-(3-methoxyphenyl)-1H-pyrrol-2(5H)-one (10c). 1H-NMR (DMSO-d6) δ 0.92 (t, J = 7.3 Hz, 3H), 1.25–1.40 (m, 2H), 1.55–1.75 (m, 2H), 3.16 (d, J = 13.7 Hz, 1H), 3.24 (d, J = 13.7 Hz, 1H), 3.36 (t, J = 7.5 Hz, 2H), 3.53 (s, 3H), 3.76 (s, 3H), 6.21 (m, 1H), 6.32 (d, J = 7.7 Hz, 1H), 6.50 (s, 1H), 6.64 (dd, J = 7.9, 2.2 Hz, 1H), 6.87 (dd, J = 7.5, 2.4 Hz, 1H), 7.00 (t, J = 7.8 Hz, 1H), 7.36 (t, J = 8.0 Hz, 1H), 7.58 (m, 1H), 10.10 (s, 1H); 13C-NMR (DMSO-d6) δ 164.38, 158.99, 158.37, 144.64, 136.84, 133.99, 129.28, 128.48, 121.75, 119.97, 118.44, 114.62, 113.17, 112.22, 111.81, 90.47, 54.92, 54.55, 41.06, 30.82, 20.07, 13.84 (one signal covered by residual solvent peak); HRMS: calcd. for C23H27NO5 397.1889, found 397.1881.
1,5-Dibenzyl-3,5-dihydroxy-4-phenyl-1H-pyrrol-2(5H)-one (10d). 1H-NMR (DMSO-d6) δ 3.23 (d, J = 12.3 Hz, 1H), 3.33 (d, J = 12.3 Hz, 1H), 4.54 (d, J = 15.7 Hz, 1H), 4.74 (d, J = 15.7 Hz, 1H), 6.59 (dd, J = 7.7, 1.6 Hz, 2H), 6.70 (s, 1H), 6.98–7.12 (m, 3H), 7.19–7.36 (m, 4H), 7.39–7.52 (m, 4H), 7.99–8.07 (m, 2H), 10.13 (s, 1H); 13C-NMR (DMSO-d6) δ 164.85, 144.30, 138.70, 135.19, 132.74, 129.45, 128.31, 128.08, 127.98, 127.50, 126.71, 126.64, 126.44, 119.18, 90.62, 42.29, 41.56 (two C-H aromatic signals overlapped); HRMS: calcd. for C24H21NO3 371.1521, found 371.1515.
1-Benzyl-5-(4-chlorobenzyl)-4-(4-chlorophenyl)-3,5-dihydroxy-1H-pyrrol-2(5H)-one (10e). 1H-NMR (DMSO-d6) δ 3.19 (d, J = 13.6 Hz, 1H), 3.35 (d, J = 13.6 Hz, 1H), 4.54 (d, J = 15.6 Hz, 1H), 4.74 (d, J = 15.6 Hz, 1H), 6.59 (d, J = 8.4 Hz, 2H), 6.81 (s, 1H), 7.11 (d, J = 8.3 Hz, 2H), 7.20–7.35 (m, 3H), 7.43 (d, J = 7.2 Hz, 2H), 7.52 (d, J = 8.7 Hz, 2H), 8.01 (d, J = 8.7 Hz, 2H), 10.47 (s, 1H); 13C-NMR (DMSO-d6) δ 164.52, 144.88, 138.50, 134.08, 131.34, 131.23, 131.17, 131.07, 129.06, 128.44, 128.07, 128.02, 127.50, 126.71, 117.95, 90.29, 42.23, 40.80; HRMS: calcd. for C24H19Cl2NO3 439.0742, found 439.0749.
1-Benzyl-3,5-dihydroxy-5-(3-methoxybenzyl)-4-(3-methoxyphenyl)-1H-pyrrol-2(5H)-one (10f). 1H-NMR (DMSO-d6) δ 3.20 (d, J = 13.8 Hz, 1H), 3.30 (d, J = 13.8 Hz, 1H), 3.51 (s, 3H), 3.76 (s, 3H), 4.50 (d, J = 15.7 Hz, 1H), 4.70 (d, J = 15.7 Hz, 1H), 6.17 (br s, 1H), 6.22 (d, J = 7.6 Hz, 1H), 6.63 (dd, J = 7.9, 2.2 Hz, 1H), 6.69 (s, 1H), 6.88 (dd, J = 7.8, 2.2 Hz, 1H), 6.95 (t, J = 7.3 Hz, 1H), 7.18–7.46 (m, 6H), 7.57–7.63 (m, 2H), 10.19 (s, 1H); 13C-NMR (DMSO-d6) δ 164.80, 159.00, 158.38, 144.51, 138.60, 136.66, 133.95, 129.30, 128.44, 128.11, 127.97, 126.65, 121.85, 120.10, 119.09, 114.60, 113.25, 112.39, 111.99, 90.58, 54.94, 54.58, 42.28, 41.54; HRMS: calcd. for C26H25NO5 431.1733, found 431.1748.
Benzyl 3-(2-benzyl-2,4-dihydroxy-5-oxo-3-phenyl-2,5-dihydro-1H-pyrrol-1-yl)propanoate (10g). 1H-NMR (CDCl3) δ 1.40–1.60 (br s, 1H), 2.72 (dt, J = 16.8, 5.3 Hz, 1H), 3.14 (ddd, J = 16.5, 8.8, 5.9 Hz, 1H), 3.25 (d, J =13.8 Hz, 1H), 3.39 (d, J = 13.8 Hz, 1H), 3.65–3.85 (m, 2H), 4.14 (br s, 1H), 5.06 (d, J = 12.3 Hz, 1H), 5.12 (d, J = 12.3 Hz, 1H), 6.70–6.75 (m, 2H), 7.05–7.15 (m, 3H), 7.25–7.35 (m, 6H), 7.45 (t, J = 7.6 Hz, 2H), 8.02 (d, J = 7.4 Hz, 2H); 13C-NMR (CDCl3) δ 173.29, 165.56, 142.12, 135.43, 134.46, 131.51, 129.67, 128.86, 128.81, 128.73, 128.29, 128.21, 127.96, 127.28, 120.90, 92.03, 67.28, 42.25, 35.51, 32.59 (one C-H aromatic signal overlapped); HRMS: calcd. for C27H25NO5 443.1733, found 443.1742.
Benzyl 3-(2-(4-chlorobenzyl)-3-(4-chlorophenyl)-2,4-dihydroxy-5-oxo-2,5-dihydro-1H-pyrrol-1-yl)-propanoate (10h). 1H-NMR (DMSO-d6) δ 2.65–2.80 (m, 2H), 3.14 (d, J = 13.8 Hz, 1H), 3.30 (d, J = 13.8 Hz, 1H), 3.60–3.80 (m, 2H), 5.12 (s, 2H), 6.67 (d, J = 8.4 Hz, 2H), 6.70 (s, 1H), 7.14 (d, J = 8.4 Hz, 2H), 7.29–7.44 (m, 5H), 7.49 (d, J = 8.8 Hz, 2H), 7.98 (d, J = 8.7 Hz, 2H), 10.5 (s, 1H); 13C-NMR (DMSO-d6) δ 170.94, 164.31, 144.72, 136.02, 134.06, 131.29, 131.15, 131.13, 131.03, 128.92, 128.40, 128.11, 128.02, 127.93, 127.57, 117.98, 90.16, 65.68, 34.46, 33.08; HRMS: calcd. for C27H23Cl2NO5 511.0953, found 511.0955.
Benzyl 3-(2,4-dihydroxy-2-(3-methoxybenzyl)-3-(3-methoxyphenyl)-5-oxo-2,5-dihydro-1H-pyrrol-1-yl)-propanoate (10i). 1H-NMR (CDCl3) δ 2.70 (dt, J = 16.6, 5.2 Hz, 1H), 3.05–3.15 (m, 1H), 3.22 (d, J = 14.0 Hz, 1H), 3.40 (d, J = 14.0 Hz, 1H), 3.57 (s, 3H), 3.65–3.85 (m, 2H), 3.84 (s, 3H), 4.11 (br s, 1H), 5.06 (d, J = 12.0 Hz, 1H), 5.12 (d, J = 12.0 Hz, 1H), 6.24 (s, 1H), 6.37 (d, J = 7.6 Hz, 1H), 6.65 (dd, J = 8.2, 2.1 Hz, 1H), 6.83–6.92 (m, 1H), 7.00 (t, J = 7.9 Hz, 1H), 7.24–7.41 (m, 6H), 7.56–7.67 (m, 2H); 13C NMR (CDCl3) δ 173.27, 165.52, 159.80, 159.24, 142.45, 135.90, 135.45, 132.81, 129.76, 129.16, 128.84, 128.71, 128.70, 122.00, 120.90, 120.74, 114.92, 113.78, 113.55, 113.35, 91.97, 67.26, 55.47, 55.17, 42.18, 35.48, 32.62; HRMS: calcd. for C29H29NO7 503.1944, found 503.1962.