Reactivity and Synthetic Applications of 4,5-Dicyanopyridazine: An Overview †
Abstract
:1. Introduction
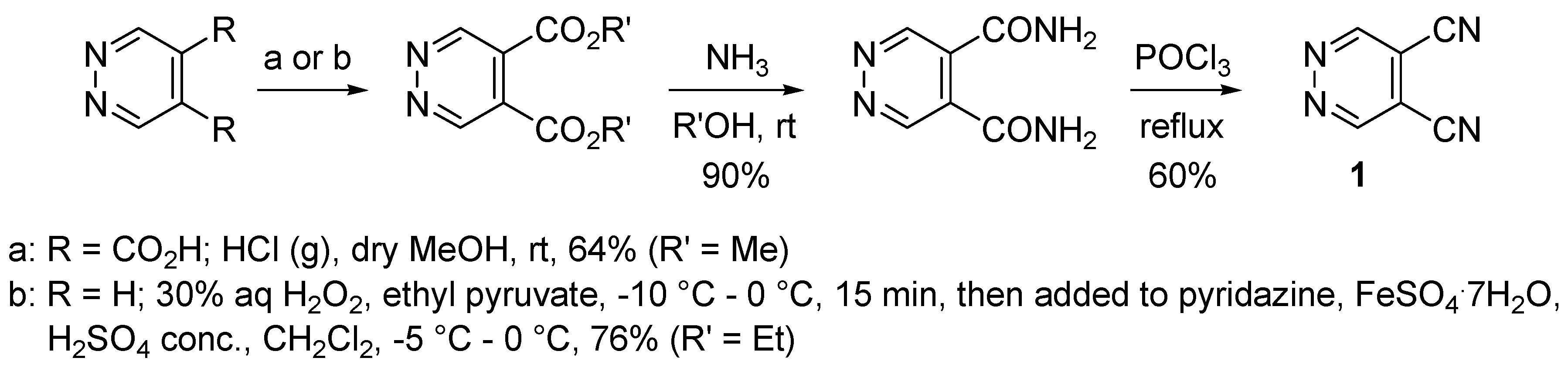
2. Syntheses of 4,5-Dicyanopyridazine (DCP)
3. Hetero Diels-Alder (HDA) Reactions
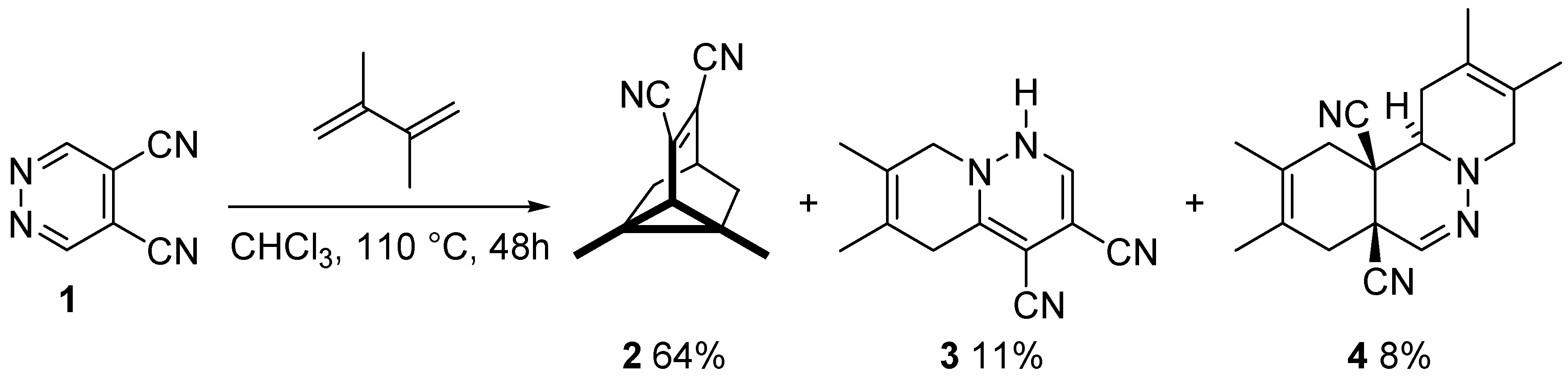
3.1. Reactions of DCP with Dienes: A Direct Access to Carbo- and Hetero-Cage Systems
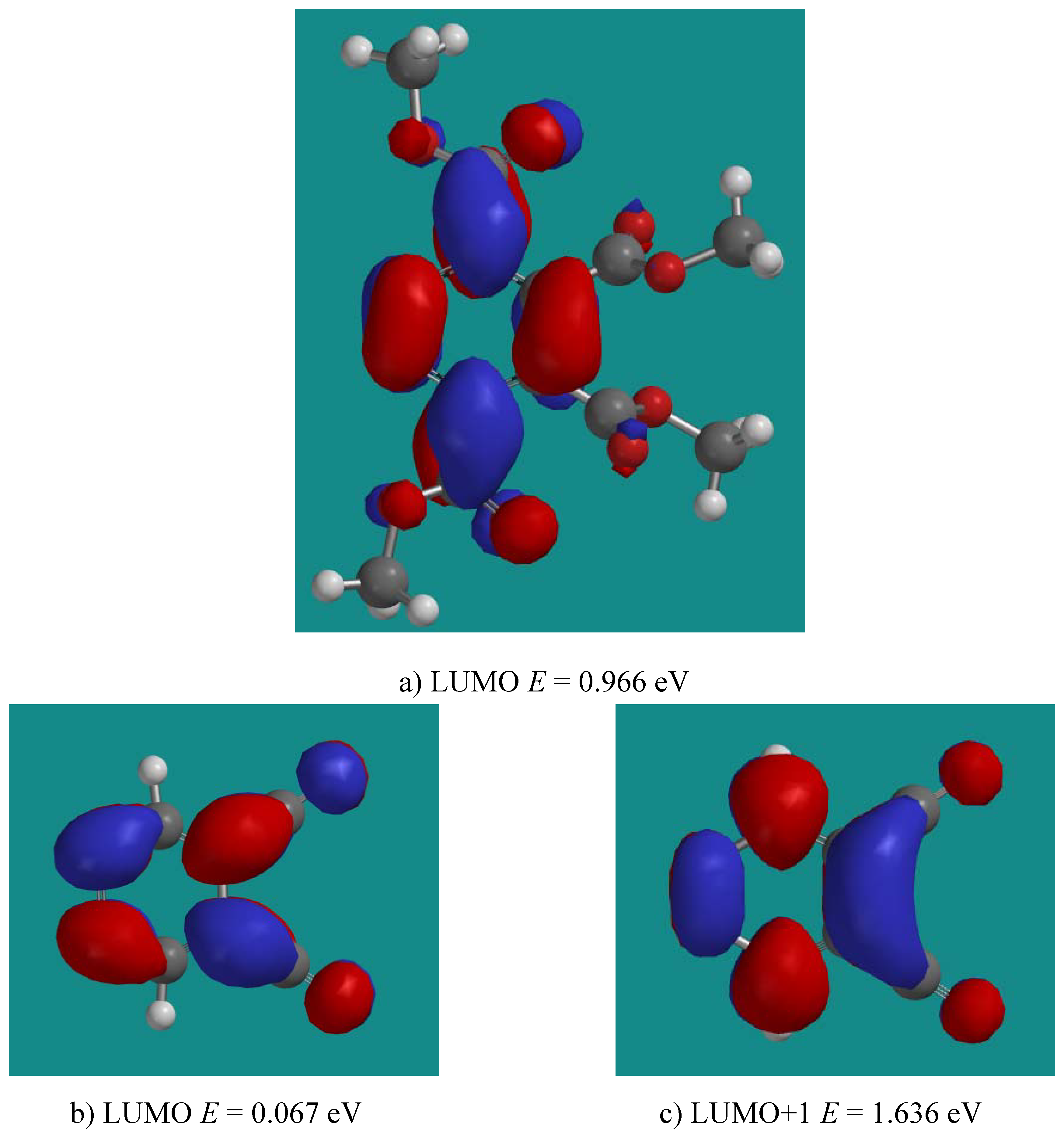
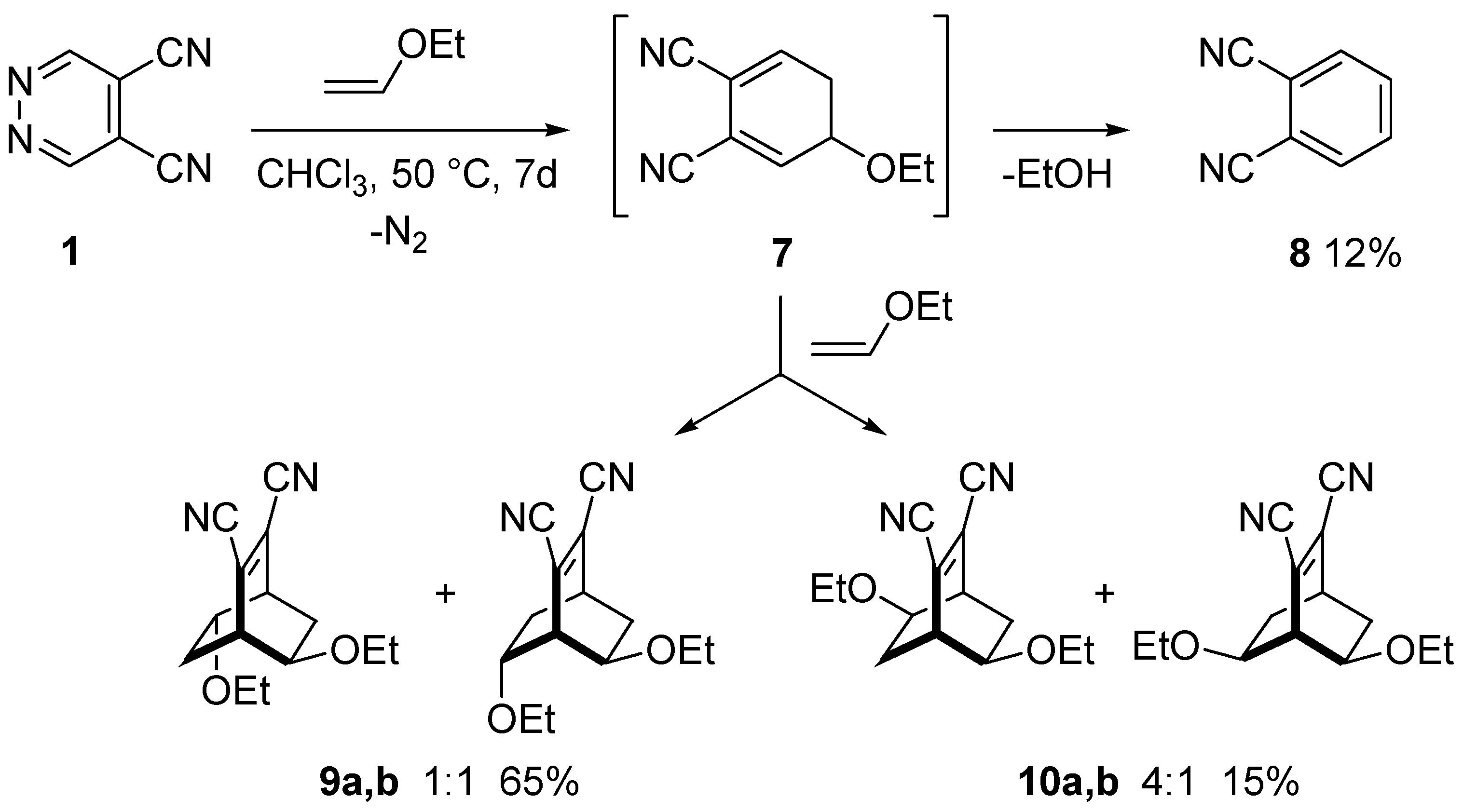
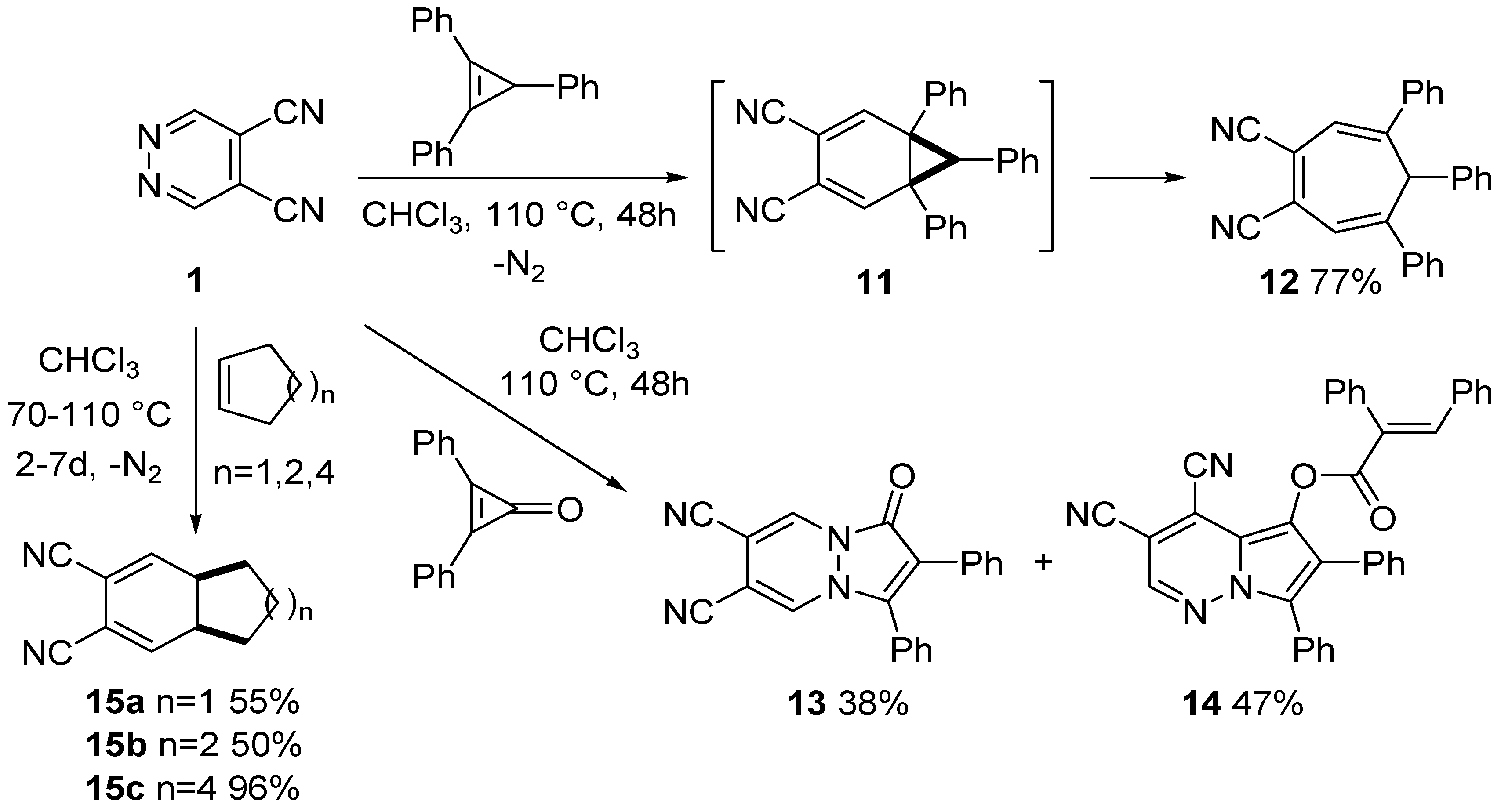
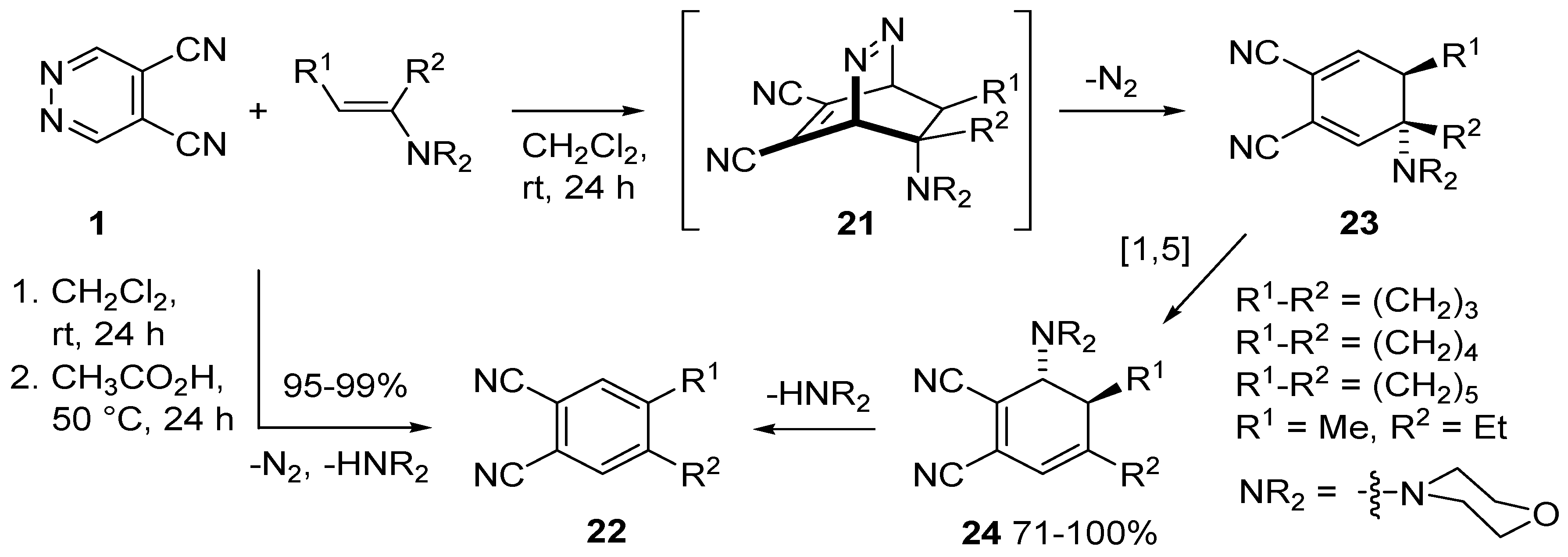
3.2. Reactions of DCP with Alkenes
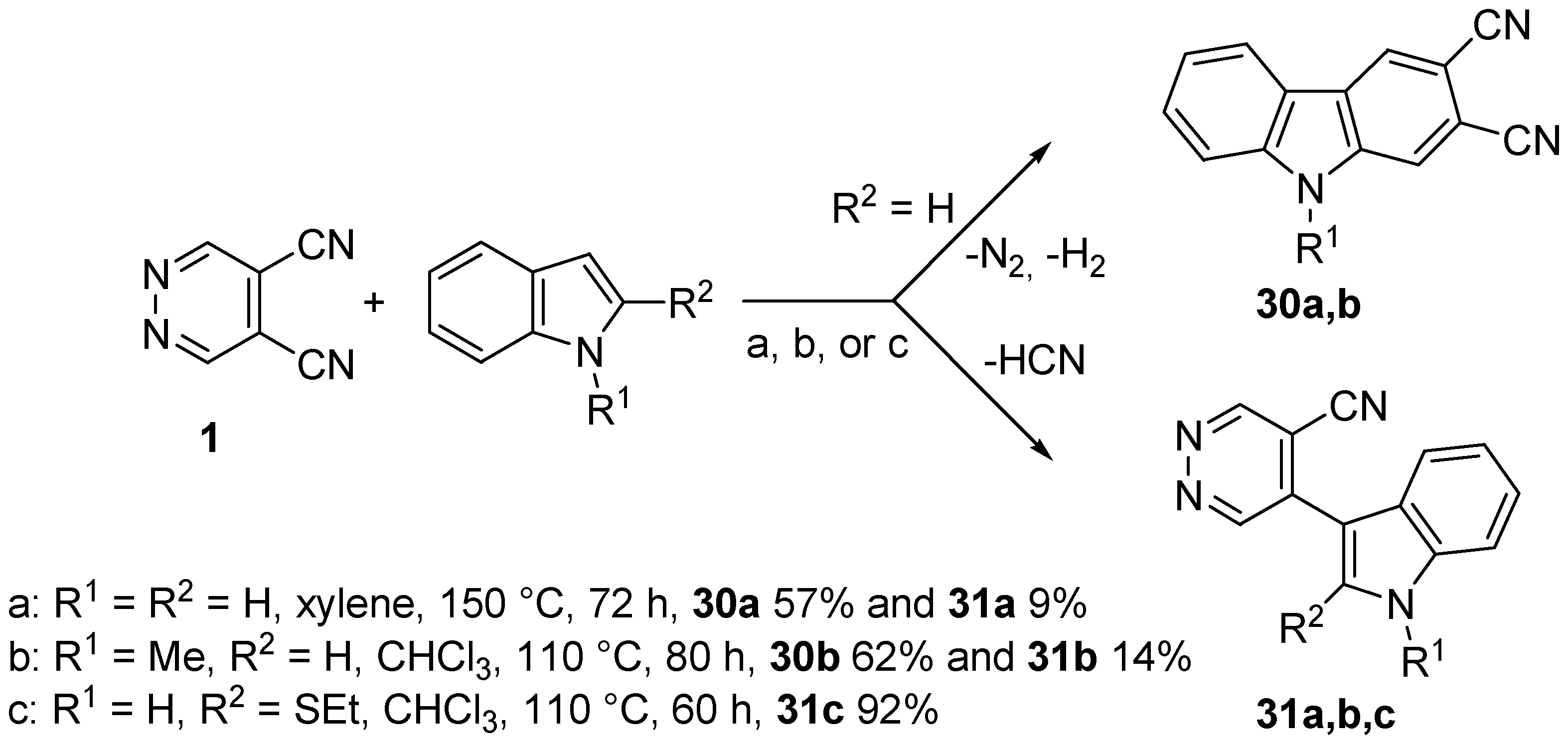
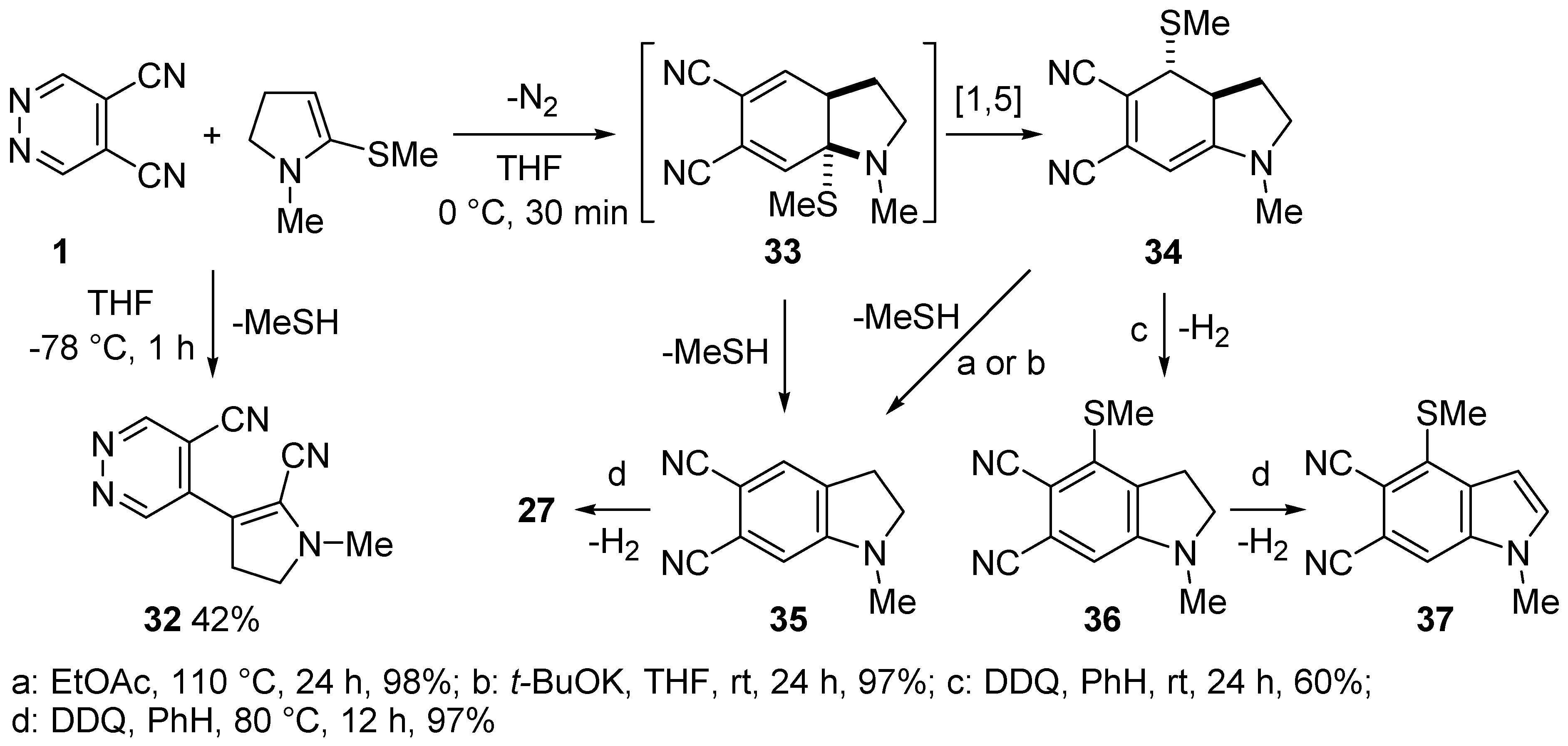
3.3. Reactions of DCP with Alkynes and Enamines
3.4. Reactions of DCP with Heterocyclic Dienophiles
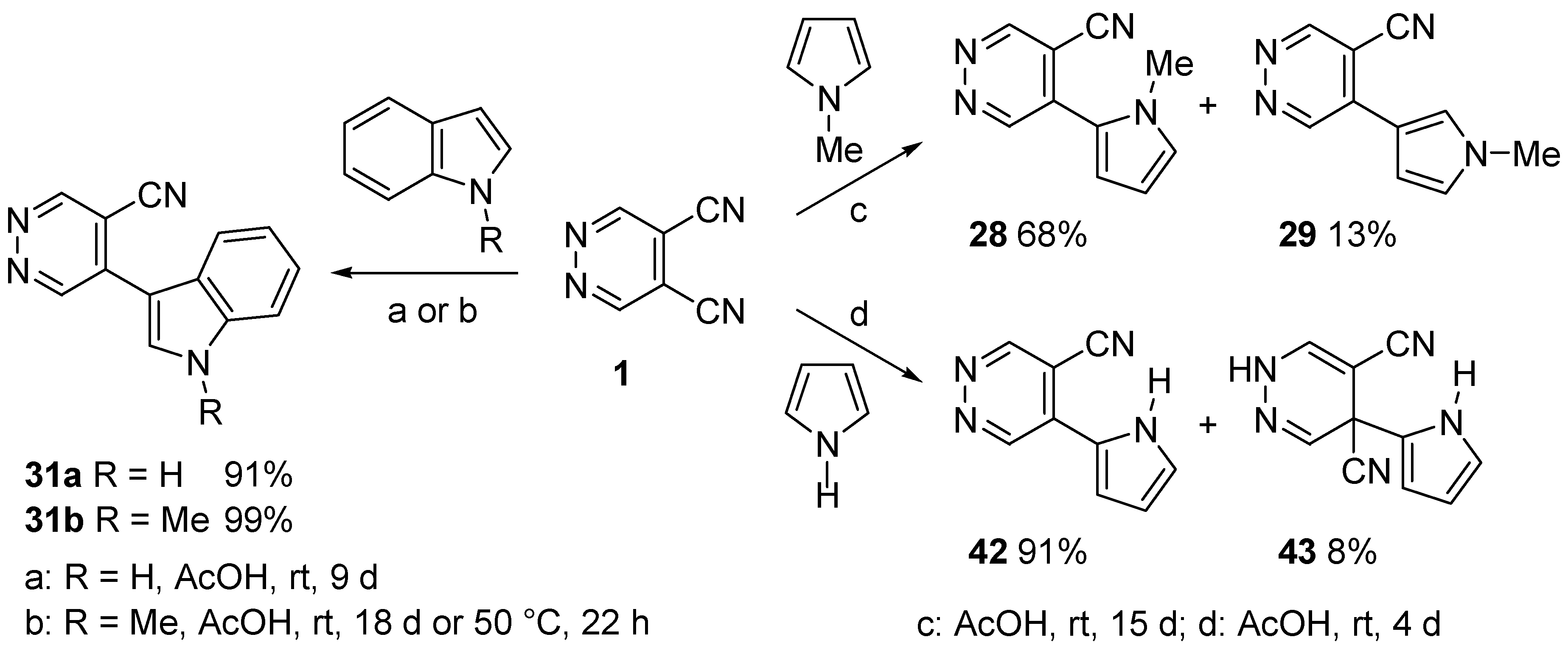
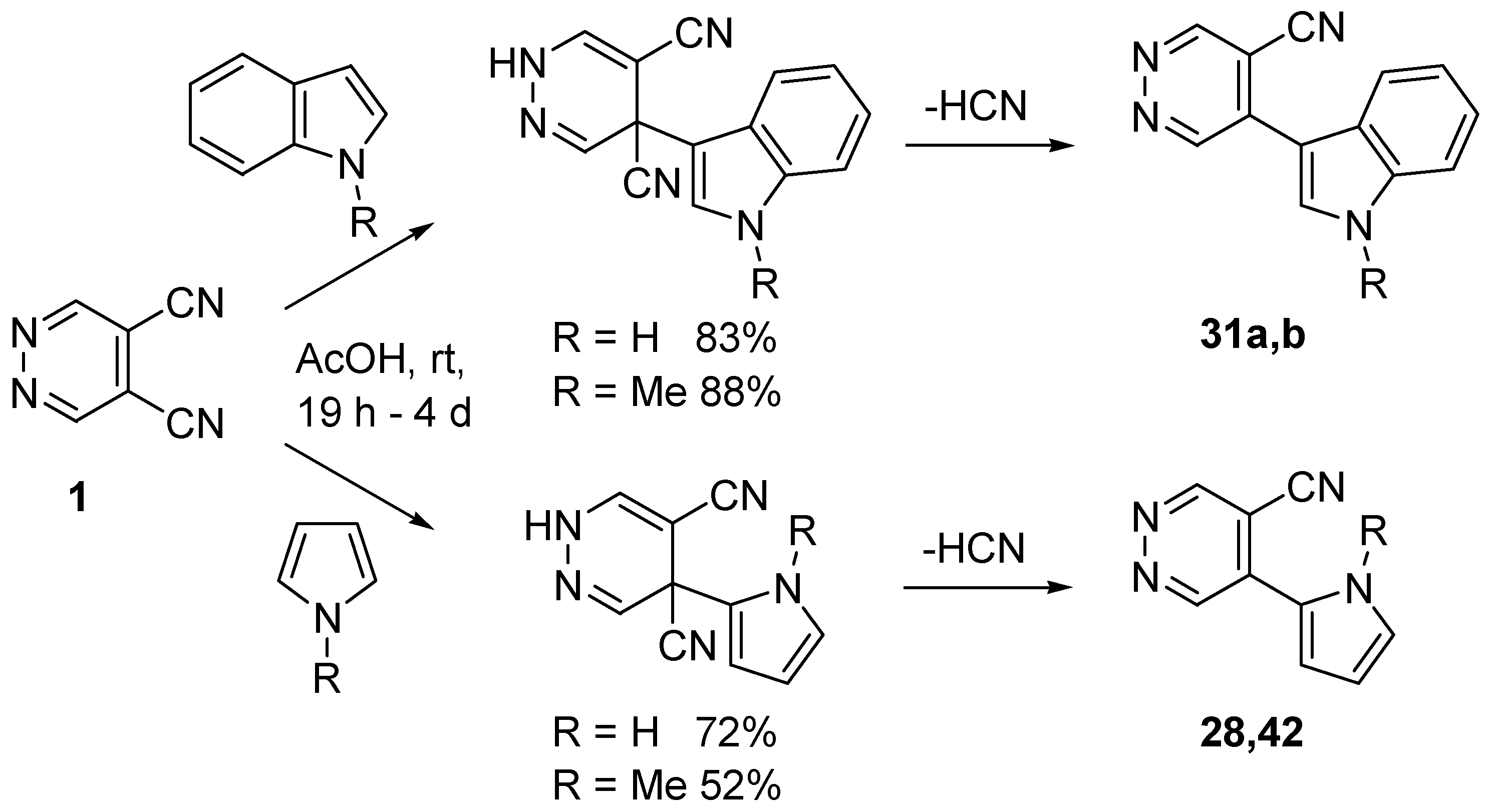
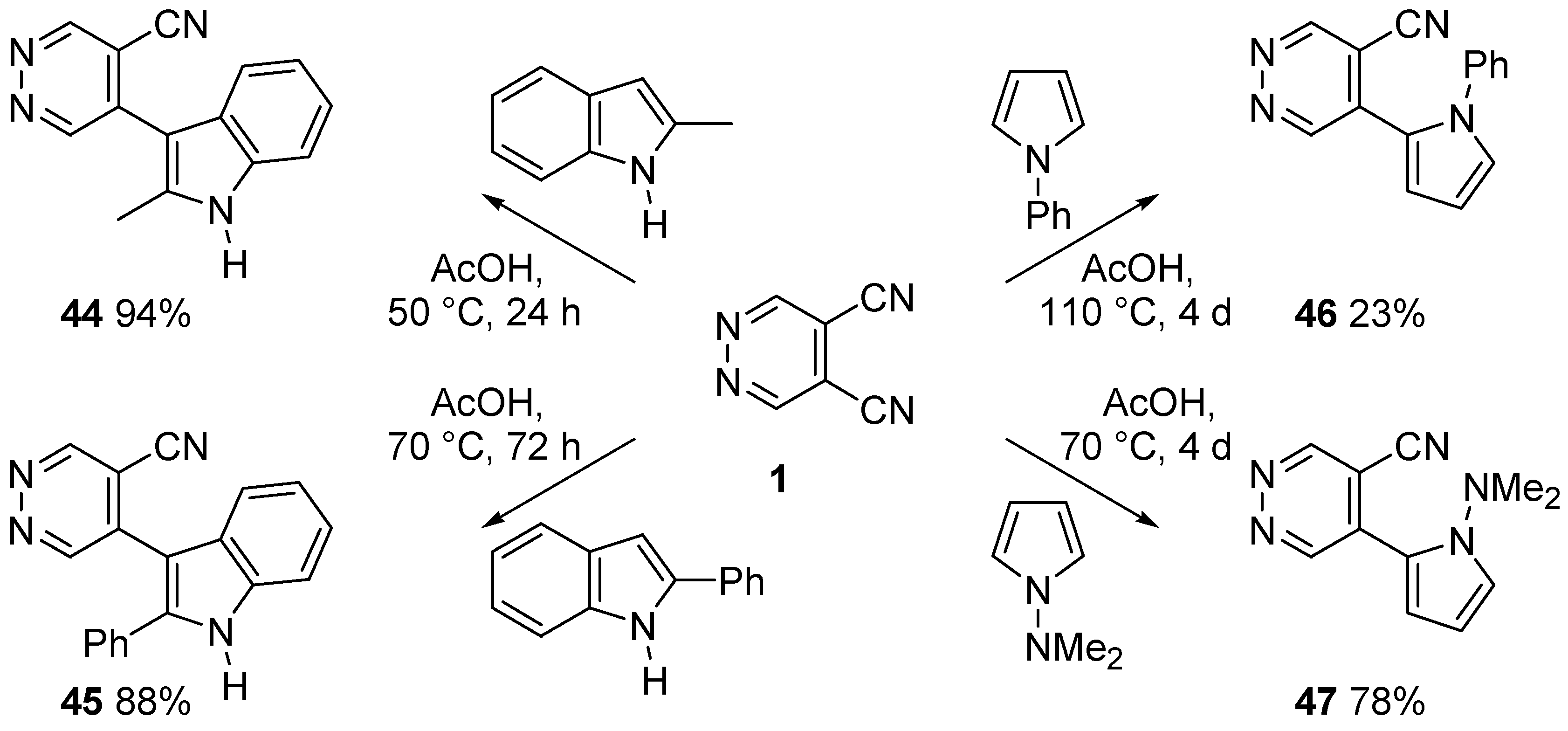
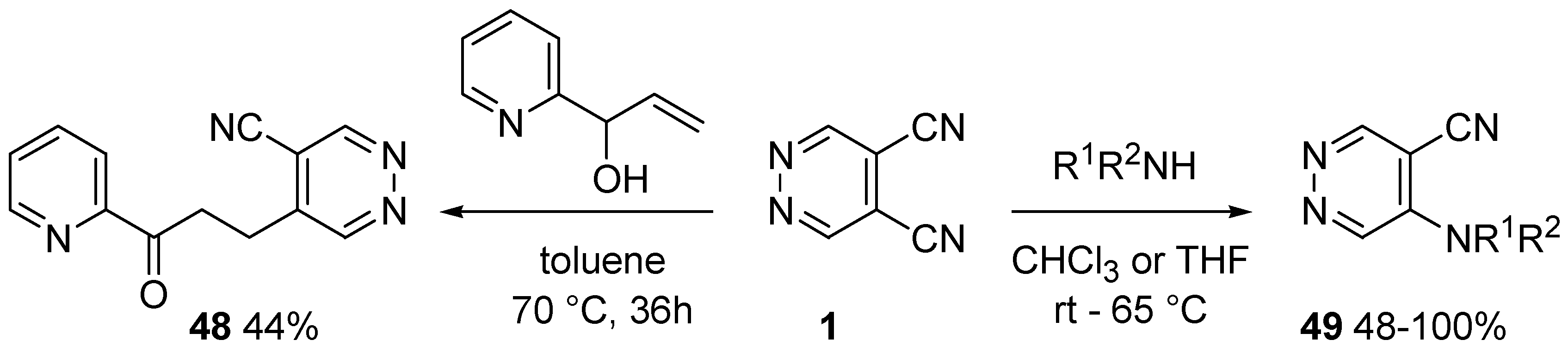
4. Nucleophilic Aromatic Substitutions on DCP
5. Synthetic applications
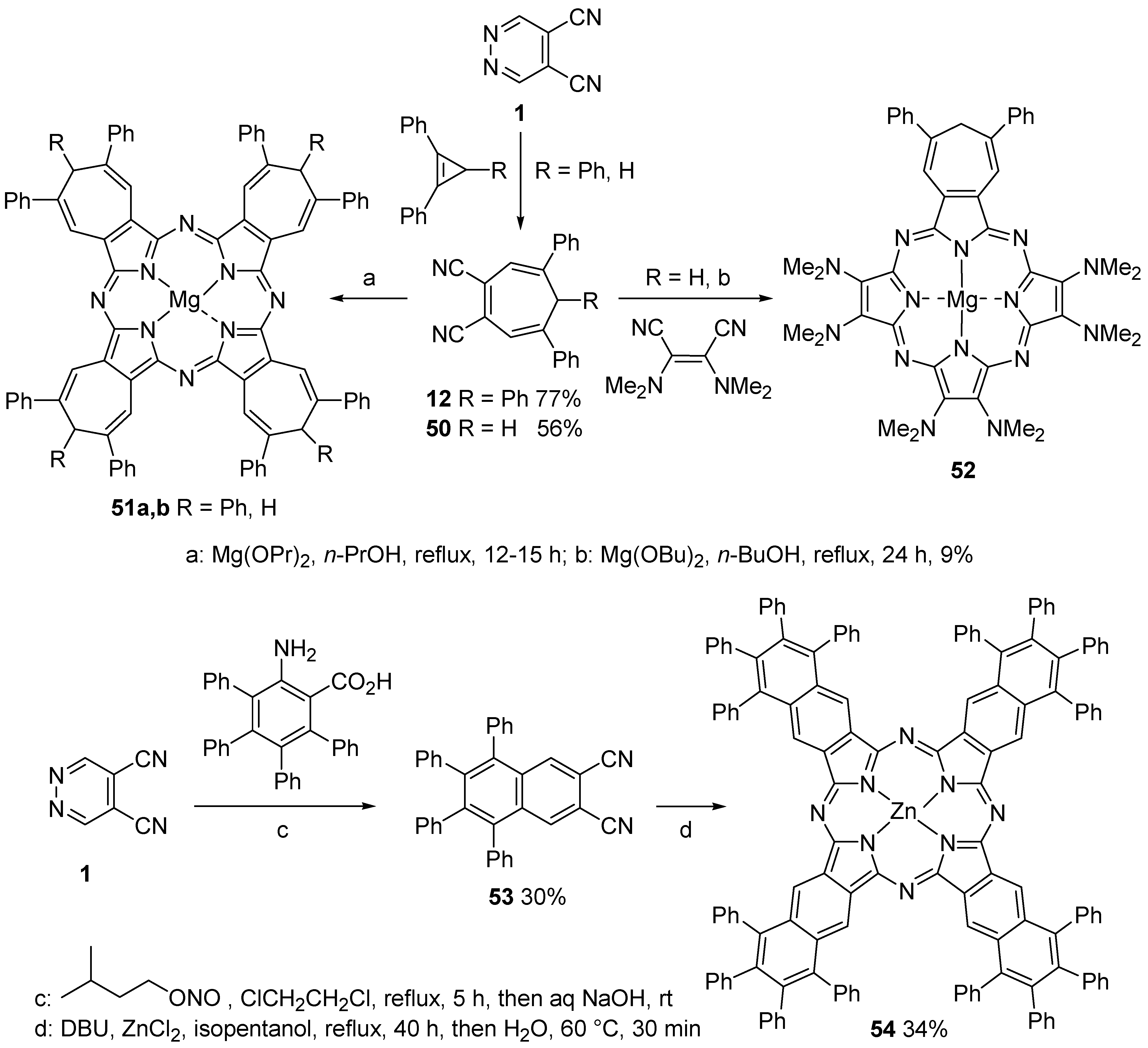
5.1. Synthesis of Phthalocyanine Analogues
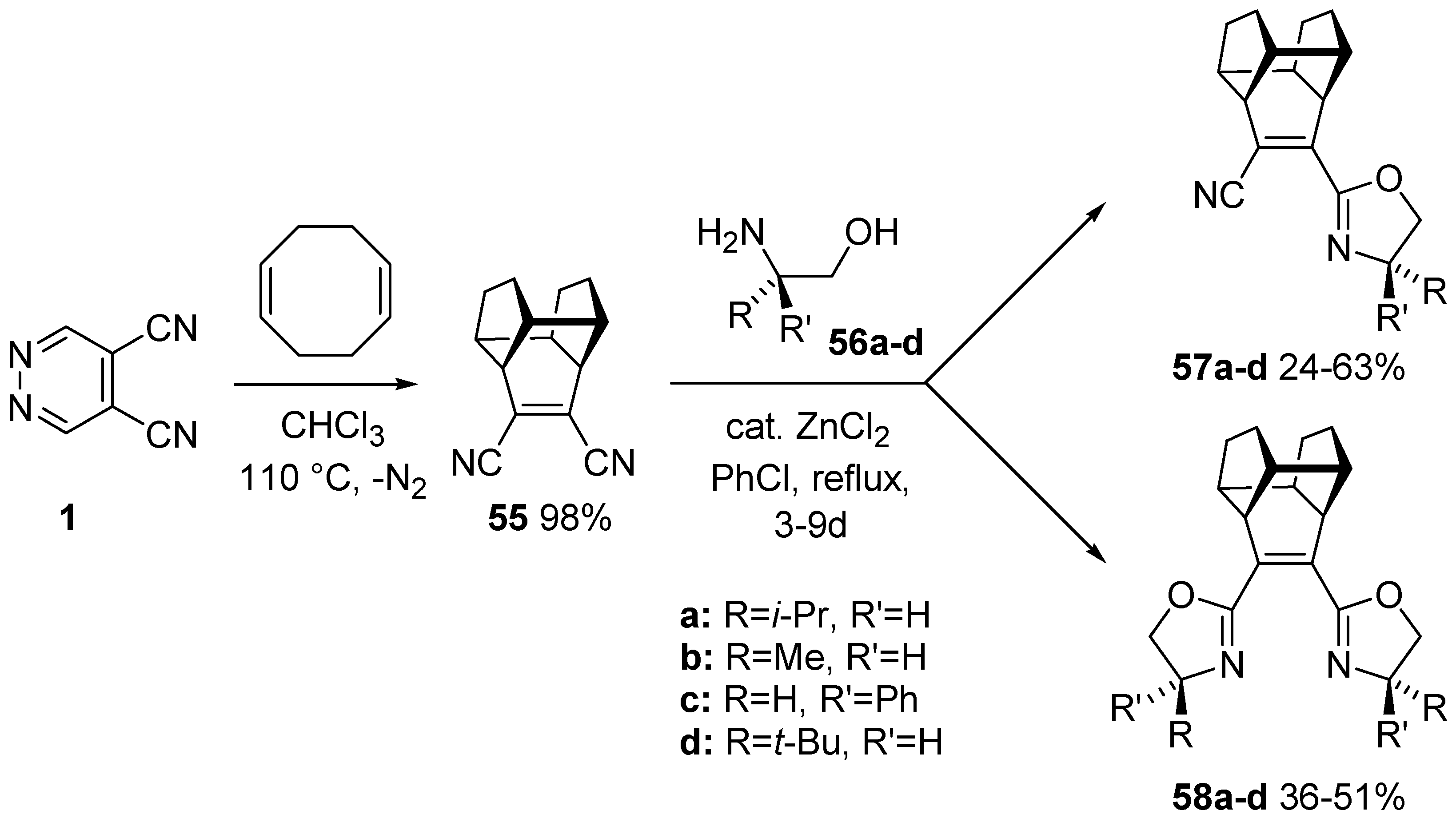
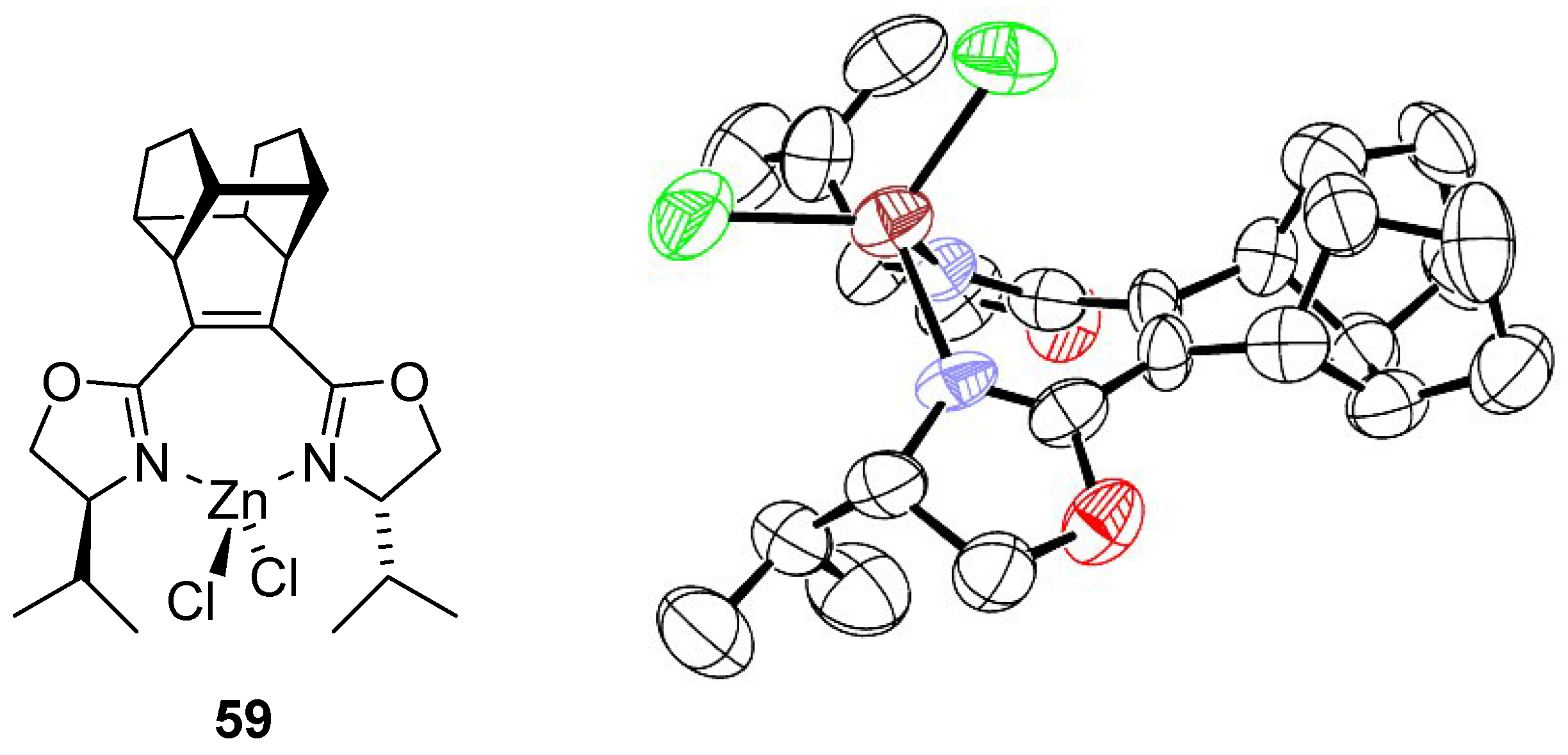
5.2. Synthesis of Enantiopure C2 Symmetric bis(2-Oxazolinyl) Cage Ligands
6. Conclusions
Acknowledgements
References
- Tišler, M.; Stanovnik, B. Pyridazines and their Benzo Derivatives. In Comprehensive Heterocyclic Chemistry; Boulton, A.J., McKillop, A., Eds.; Pergamon: Oxford, UK, 1984; Volume 3, pp. 1–56. [Google Scholar]
- Coates, W.J. Pyridazines and their Benzo Derivatives. In Comprehensive Heterocyclic Chemistry II; Boulton, A.J., Ed.; Pergamon: Oxford, UK, 1996; Volume 6, pp. 1–91. [Google Scholar]
- Maes, B.U.W.; Lemière, G.L.F. Pyridazines and their Benzo Derivatives. In Comprehensive Heterocyclic Chemistry III; Aitken, A., Ed.; Elsevier: Oxford, UK, 2008; Volume 8, pp. 1–116. [Google Scholar]
- Di Stefano, L.; Castle, R.N. The synthesis of pyridazino[4,5-d]pyridazines, pyrazino [2,3-d] pyridazines and a pyrimido [4,5-d] pyridazine. J. Heterocycl. Chem. 1968, 5, 53–59. [Google Scholar] [CrossRef]
- Heinisch, G.; Lötsch, G. Pyridazines XXIV. Application of Radicalic ethoxycarbonylation to the synthesis of pyridazine mono- and polycarboxylic acid esters. Tetrahedron 1985, 41, 1199–1205. [Google Scholar] [CrossRef]
- Minisci, F. Novel applications of free-radical reactions in preparative organic chemistry. Synthesis 1973, 1–24. [Google Scholar] [CrossRef]
- Minisci, F.; Fontana, F.; Vismara, E. Substitutions by nucleophilic free radicals: A new general reaction of heteroaromatic bases. J. Heterocycl. Chem. 1990, 27, 79–96. [Google Scholar] [CrossRef]
- Neunhoeffer, H.; Werner, G. Reaktion von pyridazincarbonestern mit 1-diäthylaminopropin. Tetrahedron Lett. 1972, 13, 1517–1518. [Google Scholar] [CrossRef]
- Neunhoeffer, H.; Werner, G. Reaktion von pyridazinen mit 1-diäthylaminopropin. Liebigs Ann. Chem. 1973, 437–442. [Google Scholar] [CrossRef]
- Neunhoeffer, H.; Werner, G. Reaktion von pyridazinen mit 1-methoxy-N,N-dimethylvinylamin. Liebigs Ann. Chem. 1973, 1955–1962. [Google Scholar]
- Jojima, T.; Takeshiba, H.; Konotsune, T. Pyridazines I. Novel intramolecular cycloaddition of 3-chloro-6-(2-allylphenoxy)pyridazines. Chem. Pharm. Bull. 1973, 20, 2191–2203. [Google Scholar] [CrossRef]
- Boger, D.L. Diels-Alder reactions of azadienes. Tetrahedron 1983, 39, 2869–2939. [Google Scholar] [CrossRef]
- Boger, D.L. Diels-Alder reactions of heterocyclic azadienes: Scope and applications. Chem. Rev. 1986, 86, 781–793. [Google Scholar] [CrossRef]
- Boger, D.L.; Weinreb, S.N. Heteroaromatic Azadienes. In Hetero Diels-Alder methodology in organic synthesis; Wasserman, H.H., Ed.; Academic: San Diego, CA, USA, 1987; pp. 300–357. [Google Scholar]
- Boger, D.L.; Patel, M. Recent applications of the inverse electron demand Diels-Alder reaction. Progr. Heterocycl. Chem. 1989, 1, 30–64. [Google Scholar]
- Benson, S.C.; Gross, J.L.; Snyder, J.K. Indole as a Dienophile in Inverse Electron Demand Diels-Alder Reactions: Reactions with 1,2,4-Triazines and 1,2-Diazines. J. Org. Chem. 1990, 55, 3257–3269. [Google Scholar] [CrossRef]
- Lantos, I.; Sheldrake, P.W.; Wells, A.S. Novel cage compounds from inter- and intra-molecular Diels-Alder reactions of heteroaromatic azadienes and methyl coumalate with cyclo-octa-1,5-diene. J. Chem. Soc. Perkin Trans. 1 1990, 1887–1890. [Google Scholar] [CrossRef]
- Neunhoeffer, H. 1,2,4-Triazines and their Benzo Derivatives. In Comprehensive Heterocyclic Chemistry; Boulton, A.J., McKillop, A., Eds.; Pergamon: Oxford, UK, 1984; Volume 3, pp. 422–429. [Google Scholar]
- Neunhoeffer, H. Tetrazines and Pentazines. In Comprehensive Heterocyclic Chemistry; Boulton, A.J., McKillop, A., Eds.; Pergamon: Oxford, UK, 1984; Volume 3, pp. 550–555. [Google Scholar]
- Neunhoeffer, H. 1,2,4-Triazines and their Benzo Derivatives. In Comprehensive Heterocyclic Chemistry II; Boulton, A.J., Ed.; Pergamon: Oxford, UK, 1996; Volume 6, pp. 533–538. [Google Scholar]
- Sauer, J. 1,2,4,5-Tetrazine. In Comprehensive Heterocyclic Chemistry II; Boulton, A.J., Ed.; Pergamon: Oxford, UK, 1996; Volume 6, pp. 912–944. [Google Scholar]
- Van der Plas, H.C. Cycloalkenopyridines by ring transformations of diazines and triazines. ARKIVOC 2008, (iii), 127–139. [Google Scholar]
- Saracoglu, N. Recent advances and applications in 1,2,4,5-tetrazine chemistry. Tetrahedron 2007, 63, 4199–4236. [Google Scholar] [CrossRef]
- Nesi, R.; Giomi, D.; Turchi, S. [4+2]- and [2+4]-Cycloaddition processes of 4,5-dicyanopyridazine with 2,3-dimethylbuta-1,3-diene. Tetrahedron 1994, 50, 9189–9194. [Google Scholar] [CrossRef]
- Tietze, L.F. Domino reactions in organic synthesis. Chem. Rev. 1996, 96, 115–136. [Google Scholar] [CrossRef] [PubMed]
- Winkler, J.D. Tandem Diels-Alder cycloadditions in organic synthesis. Chem. Rev. 1996, 96, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Adembri, G.; De Sio, F.; Nesi, R.; Scotton, M. Synthesis and properties of 1,4-dimethylpyridazino[4,5-d] pyridazines. J. Heterocycl. Chem. 1975, 12, 95–99. [Google Scholar] [CrossRef]
- Giomi, D.; Nesi, R.; Turchi, S.; Coppini, R. Tandem inverse electron-demand Diels-Alder reactions of 4,5-dicyanopyridazine with nonconjugated dienes: An excellent direct entry into carbo- and hetero-cage systems. J. Org. Chem. 1996, 61, 6028–6030. [Google Scholar] [CrossRef]
- Giomi, D.; Nesi, R.; Turchi, S.; Mura, E. Polyfunctionalized cage compounds by pericyclic domino processes of 4,5-dicyanopyridazine with dienes: Applications and limits. J. Org. Chem. 2000, 65, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Nesi, R.; Giomi, D.; Turchi, S.; Falai, A. 4,5-Dicyanopyridazine: A 1,2-diazine superheterodiene for [4+2] cycloaddition reactions. J. Chem. Soc. Chem. Commun. 1995, 2201–2202. [Google Scholar] [CrossRef]
- Turchi, S.; Giomi, D.; Capaccioli, C.; Nesi, R. Hetero Diels-Alder reactions of 4,5-dicyanopyridazine with alkenes. Tetrahedron 1997, 53, 11711–11720. [Google Scholar] [CrossRef]
- Turchi, S.; Nesi, R.; Giomi, D. Reactions of 4,5-dicyanopyridazine with alkynes and enamines: A new straightforward complementary route to 4-mono- and 4,5-disubstituted phthalonitriles. Tetrahedron 1998, 54, 1809–1816. [Google Scholar] [CrossRef]
- Nesi, R.; Turchi, S.; Giomi, D.; Corsi, C. Phthalonitriles by hetero Diels-Alder reactions of 4,5-dicyanopyridazine with enamines: Isolation and characterization of unprecedented intermediates. Tetrahedron 1998, 54, 10851–10856. [Google Scholar] [CrossRef]
- Sundberg, R.J. Pyrroles and their Benzo Derivatives: (iii) Synthesis and Applications. In Comprehensive Heterocyclic Chemistry; Bird, C.W., Cheeseman, G.W.H., Eds.; Pergamon: Oxford, UK, 1984; Volume 4, pp. 313–376. [Google Scholar]
- Sundberg, R.J. Pyrroles and their Benzo Derivatives: Synthesis. In Comprehensive Heterocyclic Chemistry II; Bird, C.W., Ed.; Pergamon: Oxford, UK, 1996; Volume 2, pp. 119–206. [Google Scholar]
- Gribble, G.W. Recent developments in indole ring synthesis–methodology and applications. J. Chem. Soc. Perkin Trans. 1 2000, 1045–1075. [Google Scholar] [CrossRef]
- Jones, R.A. Pyrroles and their Benzo Derivatives: Reactivity. In Comprehensive Heterocyclic Chemistry; Bird, C.W., Cheeseman, G.W.H., Eds.; Pergamon: Oxford, UK, 1984; Volume 4, pp. 261–268. [Google Scholar]
- Black, D.St.C. Pyrroles and their Benzo Derivatives: Reactivity. In Comprehensive Heterocyclic Chemistry II; Bird, C.W., Ed.; Pergamon: Oxford, UK, 1996; Volume 2, pp. 81–88. [Google Scholar]
- Giomi, D.; Cecchi, M. Study on direct benzoannelation of pyrrole and indole systems by domino reactions with 4,5-dicyanopyridazine. Tetrahedron 2002, 58, 8067–8071. [Google Scholar] [CrossRef]
- Giomi, D.; Cecchi, M. Domino reactions of 4,5-dicyanopyridazine with dihydroheterocycles: Synthetic and mechanistic features. J. Org. Chem. 2003, 68, 3340–3343. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.J. Synthesis of fused 1,2,4-triazines: 6- and 7-azapteridine and 6-azapurine ring systems. J. Org. Chem. 1985, 50, 2293–2298. [Google Scholar] [CrossRef]
- Ohba, S.; Konno, S.; Yamanaka, H. Studies on as-triazine derivatives. XVI. Reaction of 1,2,4-triazinecarbonitriles with carbanions. Chem. Pharm. Bull. 1991, 39, 486–488. [Google Scholar] [CrossRef]
- Ozaki, K.; Yamada, Y.; Oine, T. Studies on 4(1H)-quinazolinones. III. Some derivatizations of 2-ethoxycarbonylalkyl-1-substituted-4(1H)-quinazolinones. Chem. Pharm. Bull. 1983, 31, 2234–2243. [Google Scholar] [CrossRef]
- Hirano, H.; Lee, R.; Tada, M. The substitution reaction of pyrazine-2,3-dicarbonitrile derivatives with ammonia, amines, water and alcohols. J. Heterocycl. Chem. 1982, 19, 1409–1413. [Google Scholar] [CrossRef]
- Penney, J.M. Synthesis of aminopyridines via an unprecedented nucleophilic aromatic substitution of cyanopyridines. Tetrahedron Lett. 2004, 45, 2667–2669. [Google Scholar] [CrossRef]
- Landquist, J.K. Nucleophilic replacements in 4-cyanopyridinium salts. J. Chem. Soc. Perkin Trans. 1 1976, 454–456. [Google Scholar] [CrossRef]
- Haider, N.; Heinisch, G.; Moshuber, J. Pyridazines – 61. Unexpected reaction behavior of pyridazinecarbonitrile derivatives towards phenylmagnesium chloride. Tetrahedron 1991, 47, 8573–8578. [Google Scholar] [CrossRef]
- Hayashi, E.; Iinuma, M.; Utsunomiya, I.; Iijima, C.; Oishi, E.; Higashino, T. Grignard reactions, of 1-phthalazine-, 2-quinoxaline-, and 4-cinnoline-carbonitrile. Chem. Pharm. Bull. 1977, 25, 579–589. [Google Scholar] [CrossRef]
- Ames, D.E.; Bull, D. Preparation of cinnoline-3,4-dicarbonitrile and –dicarboxylic acid. Tetrahedron 1981, 37, 2489–2491. [Google Scholar] [CrossRef]
- Cecchi, M.; Micoli, A.; Giomi, D. Nucleophilic aromatic substitutions on 4,5-dicyanopyridazine. Pyrrole and indole systems as carbon nucleophiles. Tetrahedron 2006, 62, 12281–12287. [Google Scholar] [CrossRef]
- Giomi, D.; Piacenti, M.; Brandi, A. New reactivity of hydroxyallylpyridyl derivatives as C-3 carbon nucleophiles. Tetrahedron Lett. 2004, 45, 2113–2115. [Google Scholar] [CrossRef]
- Leznoff, C.C.; Lever, A.B.P. Phthalocyanines: Properties and Applications; VCH Publishers: New York, NY, USA, 1989; Volumes 1–4. [Google Scholar]
- Shirai, H.; Kobayashi, N. Phthalocyanines – Chemistry and Functions; IPC: Tokyo, Japan, 1997. [Google Scholar]
- Tanaka, M. Phthalocyanines—High performance pigments and their applications. In High performance pigments, 2nd ed.; Faulkner, E.B., Schwartz, R.J., Eds.; Wiley-VCH: Weinheim, Germany, 2009; pp. 275–291. [Google Scholar]
- Kobayashi, N.; Nonomura, T.; Nakai, K. A seven-membered carbon-ring-fused phthalocyanine analogue in which the π system changes during dehydrogenation/hydrogenation cycles. Angew. Chem. Int. Ed. 2001, 40, 1300–1303. [Google Scholar] [CrossRef]
- Ruggiero, A.; Fuchter, M.J.; Kokas, O.J.; Negru, M.; White, A.J.P.; Haycock, P.R.; Hoffman, B.M.; Barrett, A.G.M. A ‘push-pull’ tropylium-fused aminoporphyrazine. Tetrahedron 2009, 65, 9690–9693. [Google Scholar] [CrossRef]
- Lee, C.H.; Sooksimuang, T.; Mandal, B.K. Synthetic methodology towards novel polyphenylated naphthalocyanines. J. Porphyr. Phthalocyanines 2006, 10, 135–139. [Google Scholar] [CrossRef]
- McManus, H.A.; Guiry, P.J. Recent developments in the application of oxazoline-containing ligands in asymmetric catalysis. Chem. Rev. 2004, 104, 4151–4202. [Google Scholar] [CrossRef] [PubMed]
- Desimoni, G.; Faita, G.; Jørgensen, K.A. C2-Symmetric chiral bis(oxazoline) ligands in asymmetric catalysis. Chem. Rev. 2006, 106, 3561–3651. [Google Scholar] [CrossRef] [PubMed]
- Gant, T.G.; Meyers, A.I. The chemistry of 2-oxazolines (1985-present). Tetrahedron 1994, 50, 2297–2360. [Google Scholar] [CrossRef]
- Bolm, C.; Weickhardt, K.; Zehnder, M.; Ranff, T. Synthesis of optically active bis(2-oxazolines): Crystal structure of a 1,2-bis(2-oxazolinyl)benzene zinc chloride complex. Chem. Ber. 1991, 124, 1173–1180. [Google Scholar] [CrossRef]
- Cecchi, M.; Faggi, C.; Giomi, D. New enantiopure C2 symmetric bis(2-oxazolinyl)cage (Cage-Box) ligands from 4,5-dicyanopyridazine. Tetrahedron Asymmetry 2005, 16, 3998–4003. [Google Scholar] [CrossRef]
- Ho, T.L. Symmetry: A Basis for Synthesis Design; Wiley: New York, NY, USA, 1995; p. 474. [Google Scholar]





| Entry | Reagent | Time | Product | Yield % b [Ref.] |
|---|---|---|---|---|
| 1 | 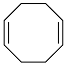 | 24 h | 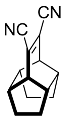 | 98 [28] |
| 2 | 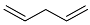 | 48 h | 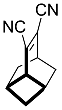 | 71 [28] |
| 3 | 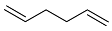 | 48 h | 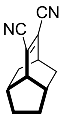 | 98 [28] |
| 4 |  | 48 h | 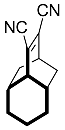 | 97 [28] |
| 5 | 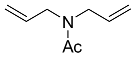 | 4 d | 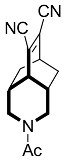 | 86 [28] |
| 6 | 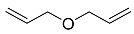 | 24 h | 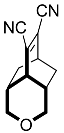 | 98 [28] |
| 7 |  | 72 h | 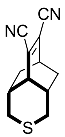 | 97 [28] |
| 8 | 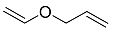 | 24 h | 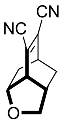 | 99 [29] |
| 9 | 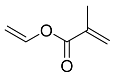 | 4 d | 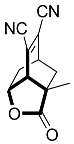 | 56 [29] c |
| 10 | 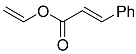 | 9 d | 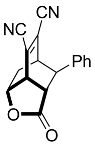 | 34 [29] |
| 11 | 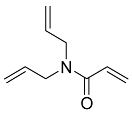 | 6 d | 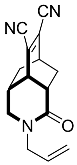 | 37 [29] |
| 12 | 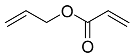 | 48 h | 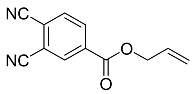 | 40 [29] |
| 13d | 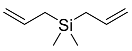 | 15 d | 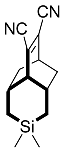 | 21 [29]d |
| 14 | 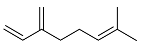 | 4 d | 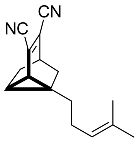 | 24 [29]e |
| 15 | 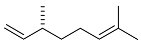 | 5 d | 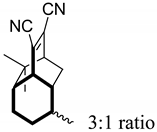 | 28 [29]e |
| 16 | 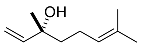 | 8 d | 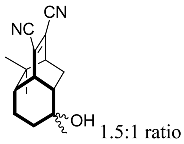 | 31 [29]e |
| 17 | 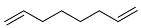 | 4 d | 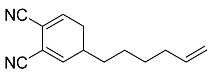 | 52 [29]f |
| 18 |  | 5 d | 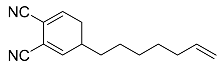 | 50 [29]f |
| 19 | 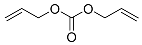 | 5 d |  | 52 [29]f |
| Entry | Reagent | Conditions | Product | Yield %b [Ref.] |
|---|---|---|---|---|
| 1 | 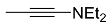 | CH2Cl2, 25 °C, 19 h | 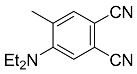 | 85 [32] |
| 2 | 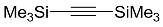 | CHCl3, 110 °C, 48 h | 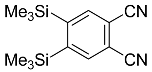 | 98 [32] |
| 3 | 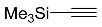 | CHCl3, 110 °C, 8 d | 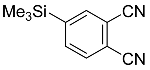 | 98 [32] |
| 4a | 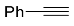 | xylene, 150 °C, 24 h | 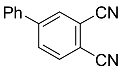 | 42 [32] |
| 4b | 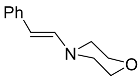 | CHCl3, 70 °C, 24 h | 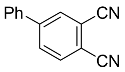 | 72 [32] |
| 5a |  | xylene, 150 °C, 5 d | 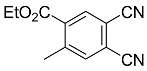 | 55 [32] |
| 5b | 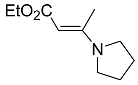 | CHCl3, 70 °C, 24 h | 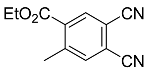 | 29 [32] |
| 6 | 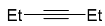 | xylene, 150 °C, 10 d | 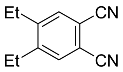 | 55 [32] |
| 7a | 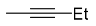 | xylene, 150 °C, 18 d | 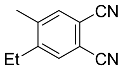 | 51 [32] |
| 7b 7c | 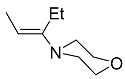 | CHCl3, 70 °C, 7 d CH2Cl2, rt, 24 h + AcOH, 50 °C, 24 h | 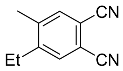 | 73 [32] 95 [33] |
| 8a | 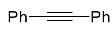 | xylene, 150 °C, 21 d | 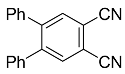 | 72 [32] |
| 8b | 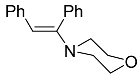 | CHCl3, 70 °C, 48 h | 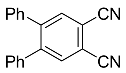 | 11 [32] |
| 9a 9b | 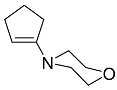 | CHCl3, 70 °C, 24 h CH2Cl2, rt, 24 h + AcOH, 50 °C, 24 h | 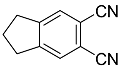 | 96 [32] 98 [33] |
| 10a 10b |  | CHCl3, 70 °C, 6 d CH2Cl2, rt, 24 h + AcOH, 50 °C, 24 h | 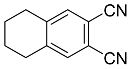 | 82 [32] 99 [33] |
| 11a 11b |  | CHCl3, 70 °C, 7 d CH2Cl2, rt, 24 h + AcOH, 50 °C, 24 h | 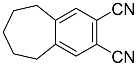 | 71 [32] 96 [33] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alfini, R.; Cecchi, M.; Giomi, D. Reactivity and Synthetic Applications of 4,5-Dicyanopyridazine: An Overview. Molecules 2010, 15, 1722-1745. https://doi.org/10.3390/molecules15031722
Alfini R, Cecchi M, Giomi D. Reactivity and Synthetic Applications of 4,5-Dicyanopyridazine: An Overview. Molecules. 2010; 15(3):1722-1745. https://doi.org/10.3390/molecules15031722
Chicago/Turabian StyleAlfini, Renzo, Marco Cecchi, and Donatella Giomi. 2010. "Reactivity and Synthetic Applications of 4,5-Dicyanopyridazine: An Overview" Molecules 15, no. 3: 1722-1745. https://doi.org/10.3390/molecules15031722




