Comparision of the Cytotoxic Effects of Birch Bark Extract, Betulin and Betulinic Acid Towards Human Gastric Carcinoma and Pancreatic Carcinoma Drug-sensitive and Drug-Resistant Cell Lines
Abstract
:1. Introduction
2. Results and Discussion
2.1. Quantitative and qualitative analysis of betulin, betulinic acid, and birch bark extract
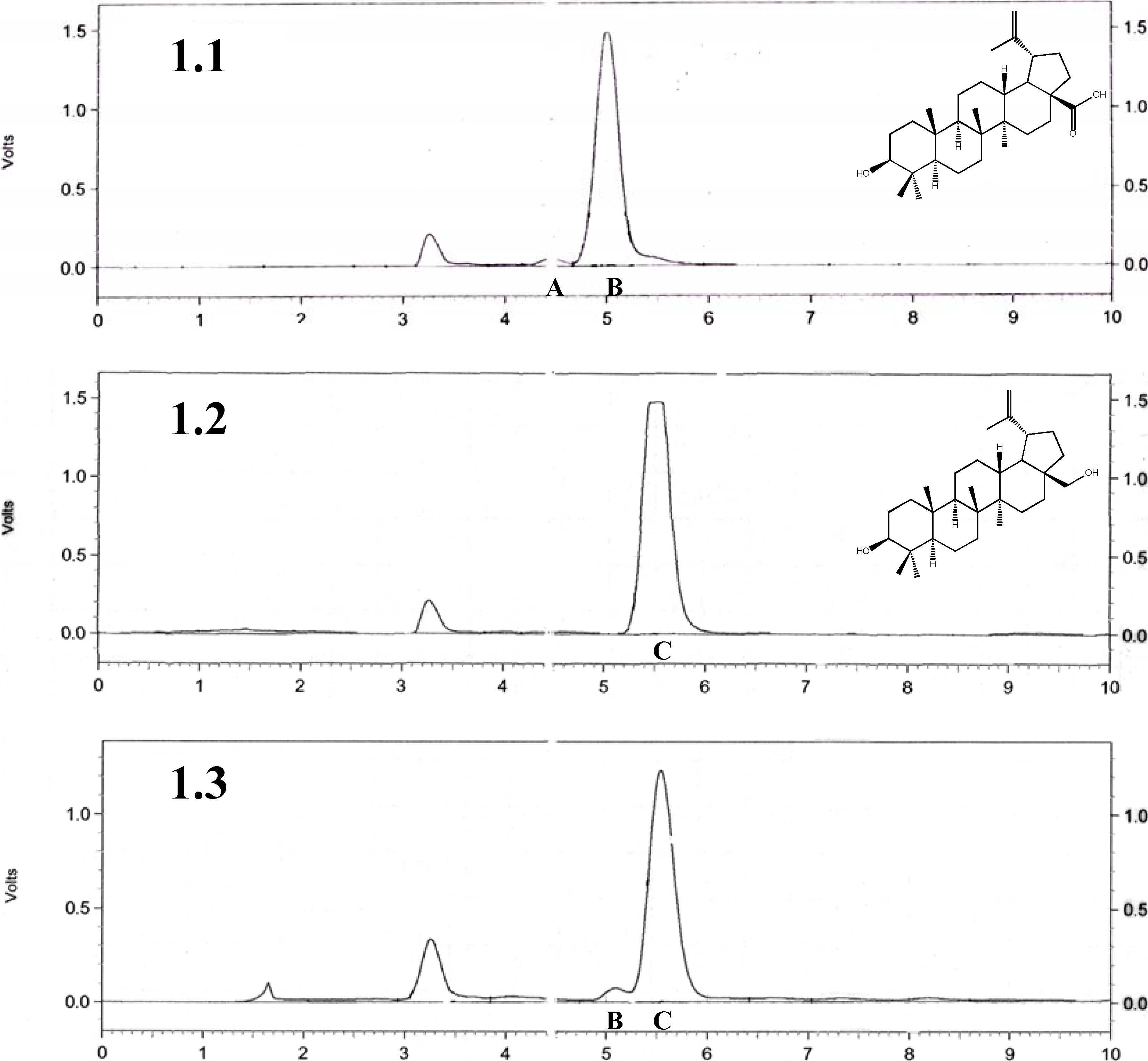

2.2. Comparison of the anticancer activity of the betulin, betulinic acid and birch bark extract

| Studied cell line | Betulin IC50 [μM] | Extract IC50 [μM] | Betulinic acid IC50 [μM] | Comparison between substances P value |
|---|---|---|---|---|
| 181P | 21.09 | 25.26 | 7.42 | 0.0182 |
| 181RNOV | 20.62 | 9.07 | 3.13 | 0.0182 |
| 181RDB | 26.50 | 23.03 | 7.96 | 0.0186 |
| Comparison between parental and resistant lines P value | 0.0273 | 0.0273 | 0.0665 | |
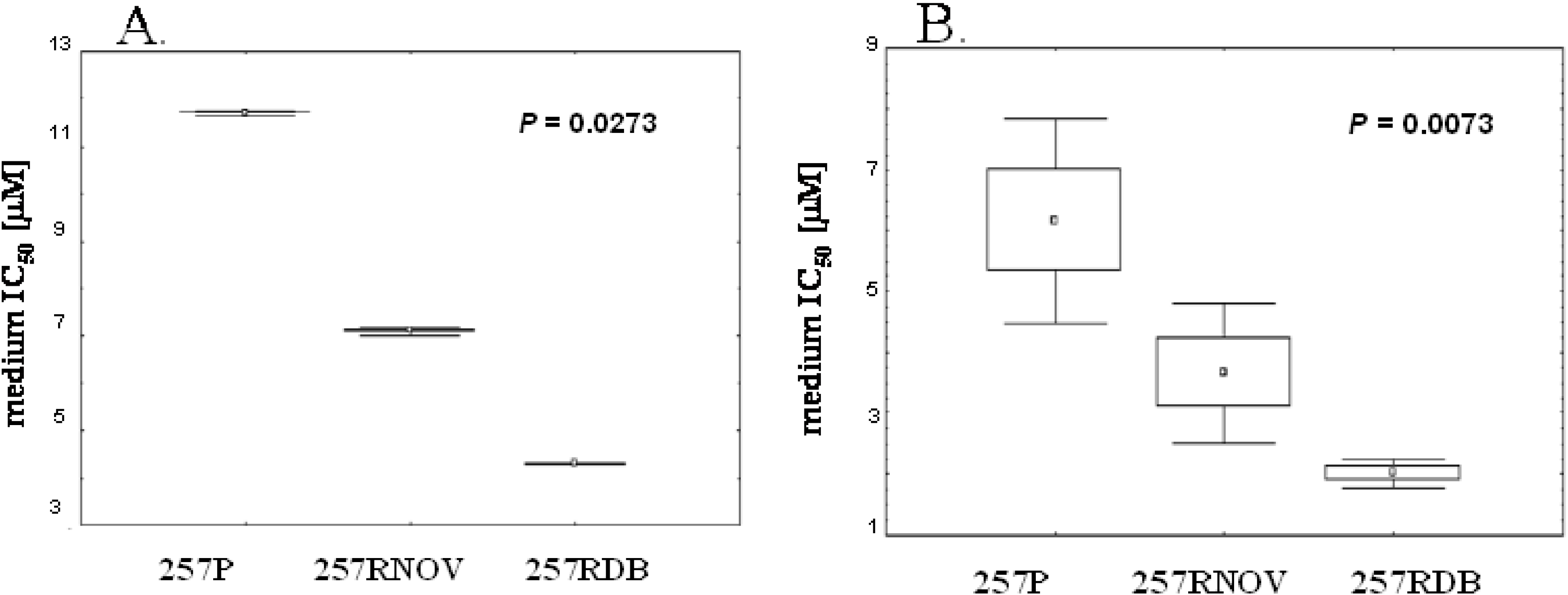
| 257P | 18.74 | 11.66 | 6.16 | 0.0062 |
| 257RNOV | 12.25 | 7.08 | 3.66 | 0.0073 |
| 257RDB | 10.97 | 4.29 | 2.01 | 0.0069 |
| Comparison between parental and resistant lines P value | 0.0273 | 0.0273 | 0.0273 |
3. Experimental
3.1. Materials and methods
3.2. Plant material
3.3. Preparation of the birch bark extract
3.4. Purification of the betulin from the extract
3.5. HPLC analysis
3.6. NMR analysis
3.7. Mass spectroscopy analysis
3.8. Cell lines
| Cell line | Origin | Selection agent | Supposed resistance mechanisms |
|---|---|---|---|
| EPP85-181P | Pancreatic carcinoma | ||
| EPP85-181RNOV | Mitoxantrone | Topo II | |
| EPP85-181RDB | Daunorubicin | MDR1/P-gp | |
| EPG85-257P | Gastric carcinoma | ||
| EPG85-257RNOV | Mitoxantrone | BCRP, GPC3, Topo II, TAP | |
| EPG85-257RDB | Daunorubicin | MDR1/P-gp |
3.9. Cell culture
3.10. Cell proliferation assay
3.11. Statistical analysis
4. Conclusions
Acknowledgements
References and Notes
- Siddik, Z.H. Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 22, 7265–7279. [Google Scholar] [CrossRef]
- Lage, H. ABC-transporters: implications on drug resistance from microorganisms to human cancers. Int. J. Antimicrob. Agents 2003, 22, 188–199. [Google Scholar] [CrossRef]
- Materna, V.; Stege, A.; Surowiak, P.; Priebsch, A.; Lage, H. RNA interference-triggered reversal of ABCC2-dependent cisplatin resistance in human cancer cells. Biochem. Biophys. Res. Commun. 2006, 348, 153–157. [Google Scholar] [CrossRef]
- Kowalski, P.; Surowiak, P.; Lage, H. Reversal of different drug-resistant phenotypes by an autocatalytic multitarget multiribozyme directed against the transcripts of the ABC transporters MDR1/P-gp, MRP2, and BCRP. Mol. Ther. 2005, 11, 508–522. [Google Scholar] [CrossRef]
- Mantwill, K.; Kohler-Vargas, N.; Bernshausen, A.; Bieler, A.; Lage, H.; Kaszubiak, A.; Surowiak, P.; Dravits, T.; Treiber, U.; Hartung, R.; Gansbacher, B.; Holm, P.S. Inhibition of the multidrug-resistant phenotype by targeting YB-1 with a conditionally oncolytic adenovirus: implications for combinatorial treatment regimen with chemotherapeutic agents. Cancer. Res. 2006, 66, 7195–7202. [Google Scholar] [CrossRef]
- Pisha, E.; Chai, H.; Lee, I.S.; Chagwedera, T.E.; Farnsworth, N.R.; Cordell, G.A.; Beecher, C.W.; Fong, H.H.; Kinghorn, A.D.; Brown, D.M. Discovery of betulinic acid as a selective inhibitor of human melanoma that functions by induction of apoptosis. Nat. Med. 1995, 1, 1046–1051. [Google Scholar] [CrossRef]
- Schmidt, M.L.; Kuzmanoff, K.L.; Ling-Indeck, L.; Pezzuto, J.M. Betulinic acid induces apoptosis in human neuroblastoma cell lines. Eur. J. Cancer. 1997, 33, 2007–2010. [Google Scholar] [CrossRef]
- Wick, W.; Grimmel, C.; Wagenknecht, B.; Dichgans, J.; Weller, M. Betulinic acid-induced apoptosis in glioma cells: A sequential requirement for new protein synthesis, formation of reactive oxygen species, and caspase processing. J. Pharmacol. Exp. Ther. 1999, 289, 1306–1312. [Google Scholar]
- Fulda, S.; Friesen, C.; Los, M.; Scaffidi, C.; Mier, W.; Benedict, M.; Nunez, G.; Krammer, P.H.; Peter, M.E.; Debatin, K.M. Betulinic acid triggers CD95 (APO-1/Fas)- and p53-independent apoptosis via activation of caspases in neuroectodermal tumors. Cancer. Res. 1997, 57, 4956–4964. [Google Scholar]
- Ji, Z.N.; Ye, W.C.; Liu, G.G.; Hsiao, W.L. 23-Hydroxybetulinic acid-mediated apoptosis is accompanied by decreases in bcl-2 expression and telomerase activity in HL-60 Cells. Life. Sci. 2002, 72, 1–9. [Google Scholar] [CrossRef]
- Thurnher, D.; Turhani, D.; Pelzmann, M.; Wannemacher, B.; Knerer, B.; Formanek, M.; Wacheck, V.; Selzer, E. Betulinic acid: a new cytotoxic compound against malignant head and neck cancer cells. Head Neck 2003, 25, 732–740. [Google Scholar] [CrossRef]
- Kessler, J.H.; Mullauer, F.B.; de Roo, G.M.; Medema, J.P. Broad in vitro efficacy of plant-derived betulinic acid against cell lines derived from the most prevalent human cancer types. Cancer Lett. 2007, 251, 132–145. [Google Scholar] [CrossRef]
- Zuco, V.; Supino, R.; Righetti, S.C.; Cleris, L.; Marchesi, E.; Gambacorti-Passerini, C.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not on normal cells. Cancer Lett. 2002, 175, 17–25. [Google Scholar] [CrossRef]
- Tan, Y.; Yu, R.; Pezzuto, J.M. Betulinic acid-induced programmed cell death in human melanoma cells involves mitogen-activated protein kinase activation. Clin. Cancer Res. 2003, 9, 2866–2875. [Google Scholar]
- Fernandes, J.; Castilho, R.O.; da Costa, M.R.; Wagner-Souza, K.; Coelho Kaplan, M.A.; Gattass, C.R. Pentacyclic triterpenes from Chrysobalanaceae species: cytotoxicity on multidrug resistant and sensitive leukemia cell lines. Cancer Lett. 2003, 190, 165–169. [Google Scholar] [CrossRef]
- Jung, G.R.; Kim, K.J.; Choi, C.H.; Lee, T.B.; Han, S.I.; Han, H.K.; Lim, S.C. Effect of betulinic acid on anticancer drug-resistant colon cancer cells. Basic. Clin. Pharmacol. Toxicol. 2007, 101, 277–285. [Google Scholar] [CrossRef]
- Fulda, S.; Debatin, K.M. Sensitization for anticancer drug-induced apoptosis by betulinic Acid. Neoplasia 2005, 7, 162–170. [Google Scholar] [CrossRef]
- Sawada, N.; Kataoka, K.; Kondo, K.; Arimochi, H.; Fujino, H.; Takahashi, Y.; Miyoshi, T.; Kuwahara, T.; Monden, Y.; Ohnishi, Y. Betulinic acid augments the inhibitory effects of vincristine on growth and lung metastasis of B16F10 melanoma cells in mice. Br. J. Cancer 2004, 90, 1672–1678. [Google Scholar] [CrossRef]
- Surowiak, P.; Drag, M.; Materna, V.; Lage, H.; Dietel, M. Betulinic acid reveal stronger cytotoxic activity toward normal melanocyte cell line (NHEM-neo) than drug-resistant and drug-sensitive melanoma cell lines (MeWo). Mol. Med. Rep. 2009, (in press). [Google Scholar]
- Chintharlapalli, S.; Papineni, S.; Ramaiah, S.K.; Safe, S. Betulinic acid inhibits prostate cancer growth through inhibition of specificity protein transcription factors. Cancer Res. 2007, 67, 2816–2823. [Google Scholar] [CrossRef]
- Hata, K.; Hori, K.; Takahashi, S. Differentiation- and apoptosis-inducing activities by pentacyclic triterpenes on a mouse melanoma cell line. J. Nat. Prod. 2002, 65, 645–648. [Google Scholar] [CrossRef]
- Kellner, U.; Hutchinson, L.; Seidel, A.; Lage, H.; Danks, M.K.; Dietel, M.; Kaufmann, S.H. Decreased drug accumulation in a mitoxantrone-resistant gastric carcinoma cell line in the absence of P-glycoprotein. Int. J. Cancer 1997, 71, 817–824. [Google Scholar] [CrossRef]
- Lage, H.; Dietel, M. Multiple mechanisms confer different drug-resistant phenotypes in pancreatic carcinoma cells. J. Cancer Res. Clin. Oncol. 2002, 128, 349–357. [Google Scholar] [CrossRef]
- Osheroff, N.; Corbett, A.H.; Robinson, M.J. Mechanism of action of topoisomerase II-targeted antineoplastic drugs. Adv. Pharmacol. 1994, 29B, 105–126. [Google Scholar] [CrossRef]
- Wichert, A.; Stege, A.; Midorikawa, Y.; Holm, P.S.; Lage, H. Glypican-3 is involved in cellular protection against mitoxantrone in gastric carcinoma cells. Oncogene 2004, 23, 945–955. [Google Scholar] [CrossRef]
- Gyorffy, B.; Surowiak, P.; Kiesslich, O.; Denkert, C.; Schafer, R.; Dietel, M.; Lage, H. Gene expression profiling of 30 cancer cell lines predicts resistance towards 11 anticancer drugs at clinically achieved concentrations. Int. J. Cancer 2006, 118, 1699–1712. [Google Scholar] [CrossRef]
- Zhao, G.; Yan, W.; Cao, D. Simultaneous determination of betulin and betulinic acid in white birch bark using RP-HPLC. J. Pharm. Biomed. Anal. 2007, 43, 959–962. [Google Scholar] [CrossRef]
- Kim, D.S.H.L.; Chen, Z.D.; Nguyen, V.T.; Pezzuto, J.M.; Qiu, S.X.; Lu, Z.Z. A concise semi-synthetic approach to betulinic acid from betulin. Synth. Comm. 1997, 27, 1607–1612. [Google Scholar] [CrossRef]
- Karpova, M.B.; Sanmun, D.; Henter, J.I.; Smirnov, A.F.; Fadeel, B. Betulinic acid, a natural cytotoxic agent, fails to trigger apoptosis in human Burkitt's lymphoma-derived B-cell lines. Int. J. Cancer 2006, 118, 246–252. [Google Scholar] [CrossRef]
- Denault, J.B.; Drag, M.; Salvesen, G.S.; Alves, J.; Heidt, A.B.; Deveraux, Q.; Harris, J.L. Small molecules not direct activators of caspases. Nat. Chem. Biol. 2007, 3, 519. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Drag, M.; Surowiak, P.; Drag-Zalesinska, M.; Dietel, M.; Lage, H.; Oleksyszyn, J. Comparision of the Cytotoxic Effects of Birch Bark Extract, Betulin and Betulinic Acid Towards Human Gastric Carcinoma and Pancreatic Carcinoma Drug-sensitive and Drug-Resistant Cell Lines. Molecules 2009, 14, 1639-1651. https://doi.org/10.3390/molecules14041639
Drag M, Surowiak P, Drag-Zalesinska M, Dietel M, Lage H, Oleksyszyn J. Comparision of the Cytotoxic Effects of Birch Bark Extract, Betulin and Betulinic Acid Towards Human Gastric Carcinoma and Pancreatic Carcinoma Drug-sensitive and Drug-Resistant Cell Lines. Molecules. 2009; 14(4):1639-1651. https://doi.org/10.3390/molecules14041639
Chicago/Turabian StyleDrag, Marcin, Pawel Surowiak, Malgorzata Drag-Zalesinska, Manfred Dietel, Hermann Lage, and Józef Oleksyszyn. 2009. "Comparision of the Cytotoxic Effects of Birch Bark Extract, Betulin and Betulinic Acid Towards Human Gastric Carcinoma and Pancreatic Carcinoma Drug-sensitive and Drug-Resistant Cell Lines" Molecules 14, no. 4: 1639-1651. https://doi.org/10.3390/molecules14041639
APA StyleDrag, M., Surowiak, P., Drag-Zalesinska, M., Dietel, M., Lage, H., & Oleksyszyn, J. (2009). Comparision of the Cytotoxic Effects of Birch Bark Extract, Betulin and Betulinic Acid Towards Human Gastric Carcinoma and Pancreatic Carcinoma Drug-sensitive and Drug-Resistant Cell Lines. Molecules, 14(4), 1639-1651. https://doi.org/10.3390/molecules14041639




