Phenolic and Theobromine Contents of Commercial Dark, Milk and White Chocolates on the Malaysian Market
Abstract
:Introduction
Results and Discussion
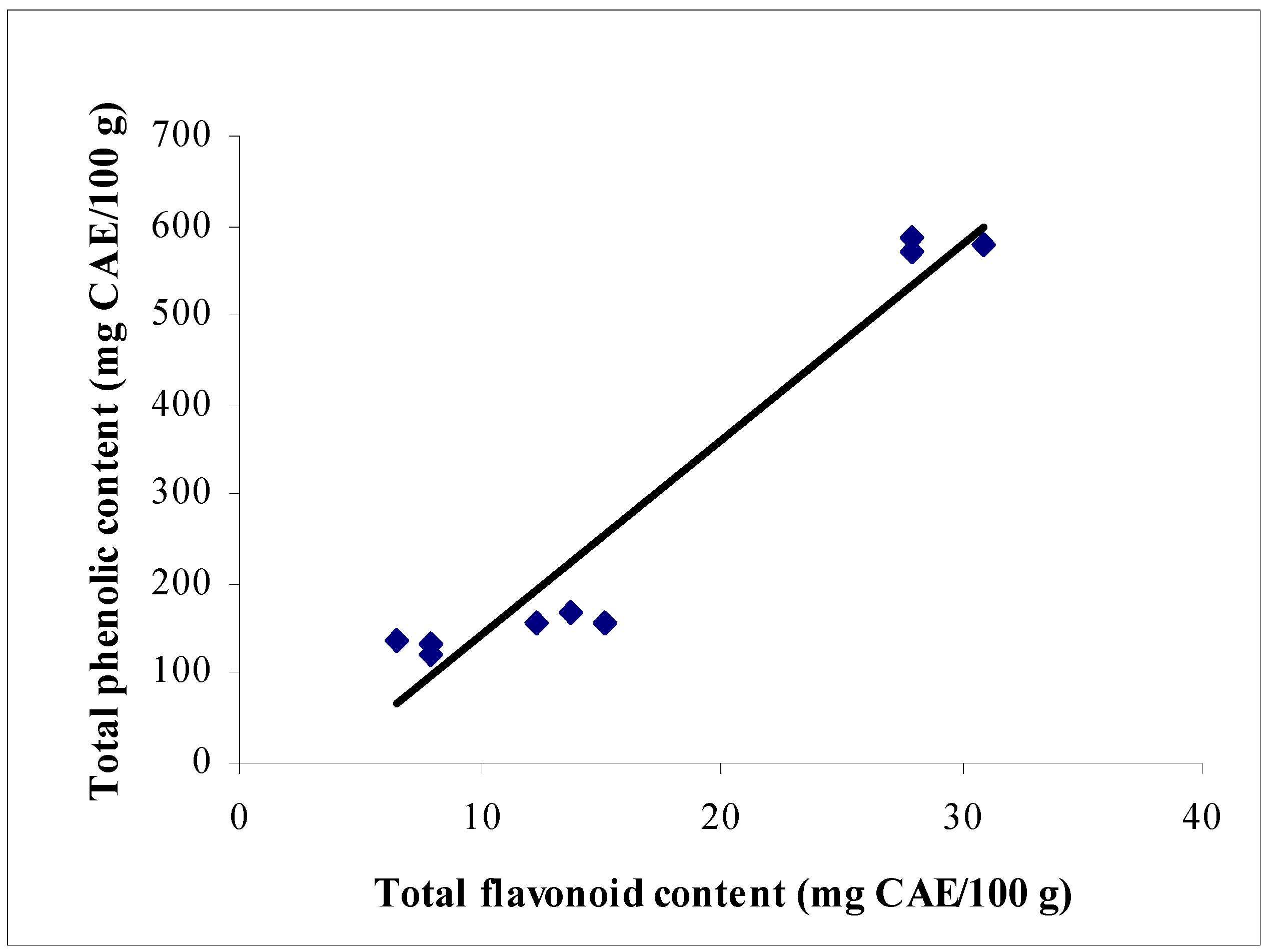
Total phenolic and flavonoid contents
| Type of chocolate | Total phenolic content (mg CAE/100 g chocolate) | Total flavonoid content (mg CAE /100 g chocolate) |
|---|---|---|
| Dark | 578.64 ± 5.04 | 28.30 ± 1.92 |
| Milk | 160.46 ± 6.58 | 13.48 ± 1.54 |
| White | 126.39 ± 7.86 | 7.70 ± 0.55 |
Catechin and epicatechin contents
Methylxanthine contents
| Type of chocolate | mg/100 g chocolate |
|---|---|
| Dark | 883.11 ± 3.54 |
| Milk | 125.154 ± 0.98 |
| White | BDL |
Conclusions
Experimental
Sample collection and preparation
Chemicals
Sample extraction
Determination of total phenolic content
Determination of flavonoid content
Analysis of individual polyphenols and methylxanthines by reversed-phase high performance liquid chromatography (HPLC)
Acknowledgements
References
- Hertog, M.G.L.; Kromhout, D.; Aravanis, C.; Blackburn, H.; Buzina, R.; Fidanza, F.; Giampaoli, S.; Jansen, A.; Menotti, A.; Nedeljkovic, S.; Pekkarinen, M.; Simic, B.S.; Toshima, H.; Feskens, E.J.M.; Hollman, P.C.H.; Katan, M.B. Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Arch. Int. Med. 1995, 155, 381–386. [Google Scholar] [CrossRef]
- Knekt, P.; Jarvinen, R.; Reunanen, A.; Maatela, J. Flavonoid intake and coronary mortality in Finland: A cohort study. Brit. Med. J. 1996, 312, 478–481. [Google Scholar]
- Amin, I.; Faizul, H.A.; Azli, R. Effect of cocoa powder extract on plasma glucose levels in hyperglycemic rats. Nutr. Food Sci. 2004, 34, 116–121. [Google Scholar] [CrossRef]
- Ruzaidi Azli, M.M.; Amin, I.; Nawalyah, A.G.; Hamid, M.; Faizul, H.A. The effect of Malaysian cocoa extract on glucose levels and lipid profiles in diabetic rats. J. Ethnopharmacol. 2005, 98, 55–60. [Google Scholar] [CrossRef]
- Ruzaidi Azli, M.M.; Abbe Maleyki, M.J.; Amin, I.; Nawalyah, A.G.; Muhajir, H. Protective effect of polyphenol-rich extract prepared from Malaysian cocoa (Theobroma cacao) on glucose levels and lipid profiles in streptozotocin-induced diabetic rats. J. Sci. Food Agric. 2008, 88, 1442–1447. [Google Scholar] [CrossRef]
- Abbe Maleyki, M.J.; Amin, I.; Chong, P.P.; Muhajir, H.; Syed Hasbullah, S.K. Effects of cocoa extract on glucometabolism, oxidative stress, and antioxidant enzymes in obese-diabetic (Ob-db) rats. J. Agric. Food Chem. 2008, 56, 7877–7884. [Google Scholar] [CrossRef]
- Taubert, D.; Roesen, R.; Schömig, E. Effect of cocoa and tea intake on blood pressure – A Meta-analysis. Arch. Intern. Med. 2007, 167, 626–634. [Google Scholar] [CrossRef]
- Giuseppe, D.R.; Castelnuovo, D.A.; Centritto, F.; Zito, F.; Curtis, D.A.; Costanzo, S.; Vohnout, B.; Sieri, S.; Krogh, V.; Donati, M.B.; Gaetano, D.G.; Iacoviello, L. Regular consumption of dark chocolate is associated with low serum concentrations of C-reactive protein in a healthy Italian population. J. Nutr. 2008, 138, 1939–1945. [Google Scholar]
- Hammerstone, J.F.; Lazarus, S.A.; Mitchell, A.E.; Rucker, R.; Schmitz, H.H. Identification of procyanidins in cocoa (Theobroma cacao) and chocolate using high-perfomance liquid chromatography/ mass spectrometry. J. Agric. Food Chem. 1999, 47, 490–496. [Google Scholar]
- Kim, H.; Keeney, P.G. (-)-Epicatechin content in fermented and unfermented cocoa beans. J. Food Sci. 1984, 49, 1090–1092. [Google Scholar] [CrossRef]
- Kelm, M.A.; Johnson, J.C.; Robbins, R.J.; Hammerstone, J.F.; Schmitz, H.H. High-performance liquid chromatography separation and purification of cacao (Theobroma cacao L.) procyanidins according to degree of polymerization using a diol stationary phase. J. Agric. Food Chem. 2006, 54, 1571–1576. [Google Scholar]
- Tomas-Barberan, F.A.; Cienfuegos-Jovellanos, E.; Marin, A.; Muguerza, B.; Gil Izquierdo, A.; Cerdaa, B.; Zafrilla, P.; Morillas, J.; Mulero, J.; Ibarra, A.; Pasamar, M.; Ramoan, D.; Espin, J.C. A new process to develop a cocoa powder with higher flavonoid monomer content and enhanced bioavailability in healthy humans. J. Agric. Food Chem. 2007, 55, 3926–3935. [Google Scholar] [CrossRef]
- Rios, L.Y.; Gonthier, M.P.; Remesy, C.; Mila, I.; Lapierre, C.; Lazarus, S.A.; Williamson, G.; Scalbert, A. Chocolate intake increases urinary excretion of polyphenol-derived phenolic acids in healthy human subjects. Am. J. Clin. Nutr. 2003, 77, 912–918. [Google Scholar]
- Adamson, G.E.; Lazarus, S.A.; Mitchell, A.E.; Prior, R.L.; Cao, G.; Jacobs, P.H.; Kremers, B.G.; Hammerstone, J.F.; Rucker, R.B.; Ritter, K.A.; Schmitz, H.H. HPLC method for the quantification of procyanidins in cocoa and chocolate samples and correlation to total antioxidant capacity. J. Agric. Food Chem. 1999, 47, 4184–4188. [Google Scholar] [CrossRef]
- Sanbongi, C.; Osakabe, N.; Natsume, M.; Takizawa, T.; Gomi, S.; Osawa, T. Antioxidative polyphenols isolated from Theobroma cacao. J. Agric. Food Chem. 1998, 46, 454–457. [Google Scholar] [CrossRef]
- Natsume, M.; Osakabe, N.; Yamagishi, M.; Takizawa, T.; Nakamura, T.; Miytake, H.; Hatano, T.; Yoshida, T. Analyses of polyphenols in cocoa liquor, cocoa, and chocolate by normal-phase and reversed-phase HPLC. Biosci. Biotechnol. Biochem. 2000, 64, 2581–2587. [Google Scholar] [CrossRef]
- Stavric, B. Methylxanthines: Toxicity to humans, theobromine, paraxanthine and the combined effects of methylxanthines. Food Chem. Toxicol. 1988, 26, 725–733. [Google Scholar] [CrossRef]
- Shively, C.A.; Tarka, S.M. Methylxanthine composition and consumption patterns of cocoa and chocolate products. Prog. Clin. Biol. Res. 1984, 158, 149–78. [Google Scholar]
- Smit, H.J.; Gaffan, E.A.; Rogers, P.J. Methylxanthines are the psychopharmacologically active constituents of chocolate. Psychopharmacology 2004, 176, 412–419. [Google Scholar] [CrossRef]
- Zoumas, B.L.; Kreiser, W.E.; Martin, R.A. Theobromine and caffeine content of chocolate products. J. Food Sci. 1980, 45, 314–316. [Google Scholar] [CrossRef]
- Blauch, J.L.; Tarka, S.M. HPLC determination of caffeine and theobromine in coffee, tea, and instant hot cocoa mixes. J. Food Sci. 1983, 48, 745–747. [Google Scholar] [CrossRef]
- Craig, W.J.; Nguyen, T.T. Caffeine and theobromine levels in cocoa and carob products. J. Food Sci. 1984, 49, 302–303. [Google Scholar] [CrossRef]
- DeVries, J.W.; Johnson, K.D.; Heroff, J.C. HPLC determination of caffeine and theobromine content of various natural and red dutch cocoas. J. Food Sci. 1981, 46, 1968–1969. [Google Scholar] [CrossRef]
- Ramli, N.; Rahman, S.A.; Hassan, O.; Yatim, A.M.; Said, M.; Siang, L.L.; Fong, N.W. Caffeine and theobromine levels in chocolate couverture and coating products. Mal. J. Nutr. 2000, 6, 55–63. [Google Scholar]
- Ramli, N.; Yatim, A.M.; Said, M.; Heng, C.H. HPLC determination of methylxanthine and polyphenols levels in cocoa and chocolate products. Malay. J. Anal. Sci. 2001, 7, 377–386. [Google Scholar]
- Gu, L.; House, S.E.; Wu, X.; Ou, B.; Prior, R.L. Procyanidin and catechin contents and antioxidant capacity of cocoa and chocolate products. J. Agric. Food Chem. 2006, 54, 4057–4061. [Google Scholar] [CrossRef]
- Cooper, K.A.; Campos-Gimenez, E.; Alvarez, D.J.; Nagy, K.; Donovan, J.L.; Williamson, G. Rapid reverse-phase ultra-performance liquid chromatography analysis of the major cocoa polyphenols and inter-relationship of their concentration in chocolates. J. Agric. Food Chem. 2007, 55, 2841–2847. [Google Scholar] [CrossRef]
- Grassi, D.; Lippi, C.; Necozione, S.; Desideri, G.; Ferri, C. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am. J. Clin. Nutr. 2004, 81, 611–614. [Google Scholar]
- Cooper, K.A.; Campos-Gimenez, C.; Alvarez, D.J.; Rytz, A.; Nagy, K.; Williamson, G. Predictive relationship between polyphenol and nonfat cocoa solids content of chocolate. J. Agric. Food Chem. 2008, 56, 260–265. [Google Scholar]
- Wollgast, J.; Anklam, E. Review on polyphenols in Theobroma cacao: Changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]
- Bracco, U.; Grailhe, N.; Rostango, W.; Egli, R. Analytical evaluation of cocoa curing in the Ivory Coast. J. Sci. Food Agric. 1969, 20, 713–717. [Google Scholar] [CrossRef]
- Hansen, C.E.; del Olmo, M.; Burri, C. Enzyme activities in cocoa beans during fermentation. J. Sci. Food Agric. 1998, 77, 273–281. [Google Scholar] [CrossRef]
- Miller, K.B.; Hurst, W.J.; Payne, M.J.; Stuart, D.A.; Apgar, J.; Sweigart, D.S.; Ou, B. Impact of alkalization on the antioxidant and flavanol content of commercial cocoa powders. J. Agric. Food Chem. 2008, 56, 8527–8533. [Google Scholar] [CrossRef]
- Lee, K.W.; Kim, Y.J.; Lee, H.J.; Lee, C.Y. Cocoa has more phenolic phytochemicals and a higher antioxidant capacity than teas and red wine. J. Agric. Food Chem. 2003, 51, 7292–7295. [Google Scholar] [CrossRef]
- Ministry of Agriculture, Fisheries, and Food (MAFF). Survey of caffeine and other methylxanthines in energy drinks and other caffeine-containing products (updated). Food Surveillance Information Sheet, 144. 1998. [Google Scholar]
- Caudle, A.G.; Gu, Y.; Bell, L.N. Improved analysis of theobromine and caffeine in chocolate food products formulated with cocoa powder. Food Res. Int. 2001, 34, 599–603. [Google Scholar] [CrossRef]
- Michener, W.; Rozin, P. Pharmacological versus sensory factors in the satiation of chocolate craving. Physiol. Behav. 1994, 56, 419–422. [Google Scholar] [CrossRef]
- Mumford, G.K.; Benowitz, N.L.; Evans, S.M.; Kaminski, B.J.; Preston, K.L.; Sannerud, C.A.; Silverman, K.; Grifliths, R.R. Absorption rate of methylxanthines following capsules, cola and chocolate. Eur. J. Clin. Pharmacol. 1996, 51, 319–325. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Sample Availability: Samples are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Meng, C.C.; Jalil, A.M.M.; Ismail, A. Phenolic and Theobromine Contents of Commercial Dark, Milk and White Chocolates on the Malaysian Market. Molecules 2009, 14, 200-209. https://doi.org/10.3390/molecules14010200
Meng CC, Jalil AMM, Ismail A. Phenolic and Theobromine Contents of Commercial Dark, Milk and White Chocolates on the Malaysian Market. Molecules. 2009; 14(1):200-209. https://doi.org/10.3390/molecules14010200
Chicago/Turabian StyleMeng, Cheng Chia, Abbe Maleyki Mhd Jalil, and Amin Ismail. 2009. "Phenolic and Theobromine Contents of Commercial Dark, Milk and White Chocolates on the Malaysian Market" Molecules 14, no. 1: 200-209. https://doi.org/10.3390/molecules14010200




