Docking Studies and Anti-inflammatory Activity of β-Hydroxy-β-arylpropanoic Acids
Abstract
:Introduction
Results and Discussion
Synthesis of studied compounds
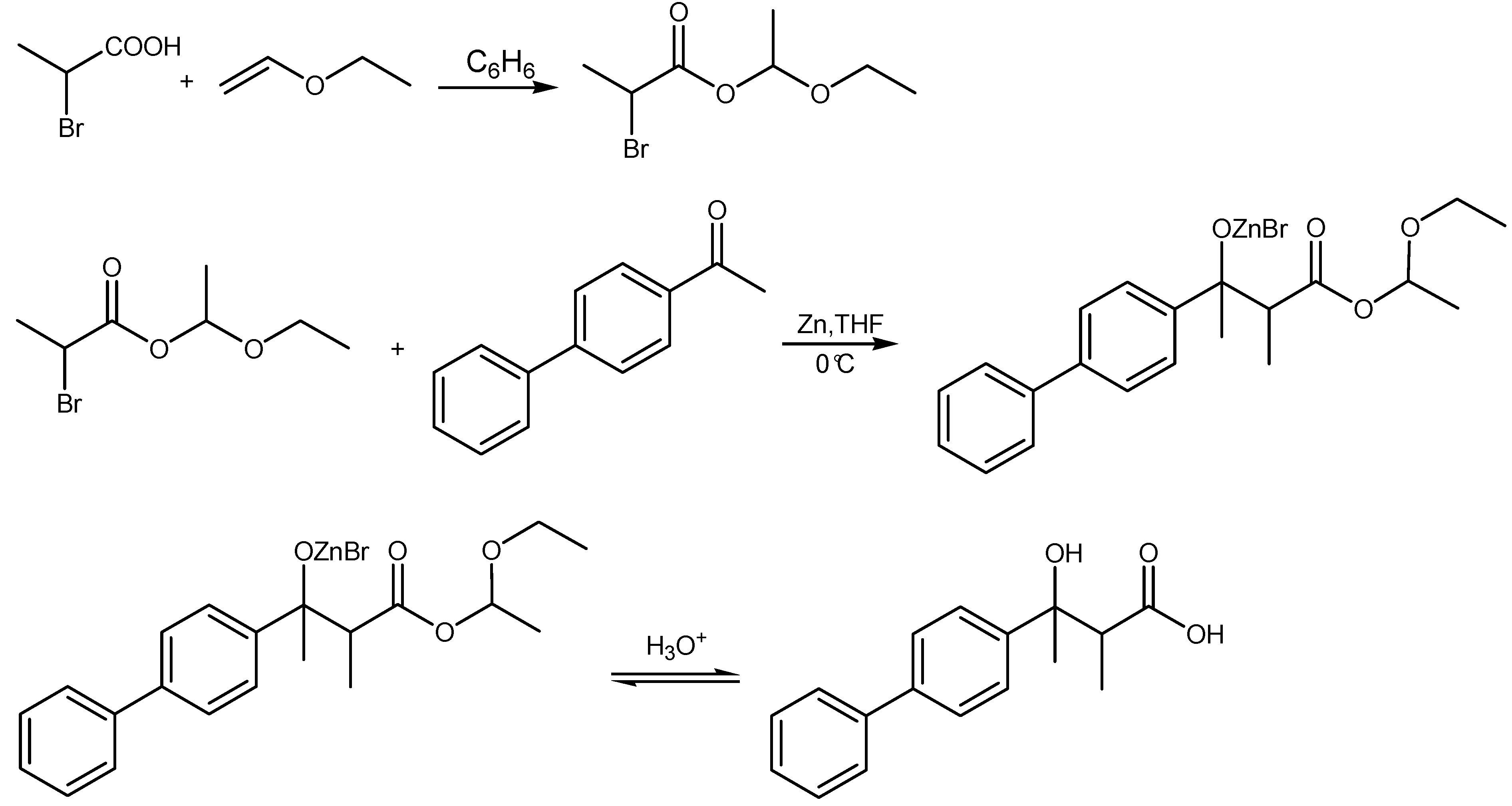
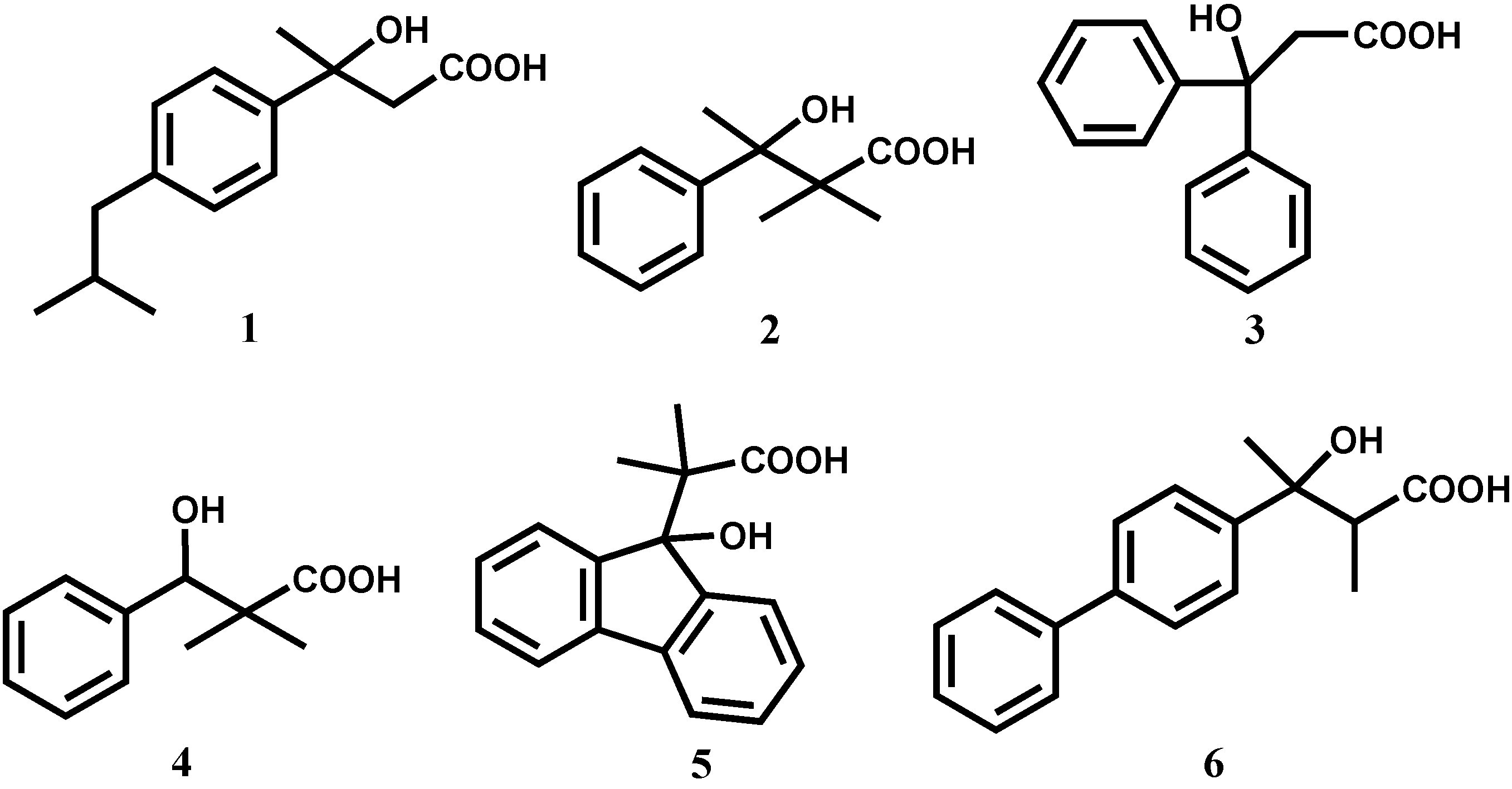
3-hydroxy-2,2-dimethyl-3-phenylbutanoic acid (2),
3-hydroxy-3,3-diphenylpropanoic acid (3),
3-hydroxy-2,2-dimethyl-3-phenylpropanoic acid (4),
2-(9-(9-hydroxyfluorenyl))-2-methylpropanoic acid (5) and
3-hydroxy-2-methyl-3-(4-diphenylyl)butanoic acid (6).
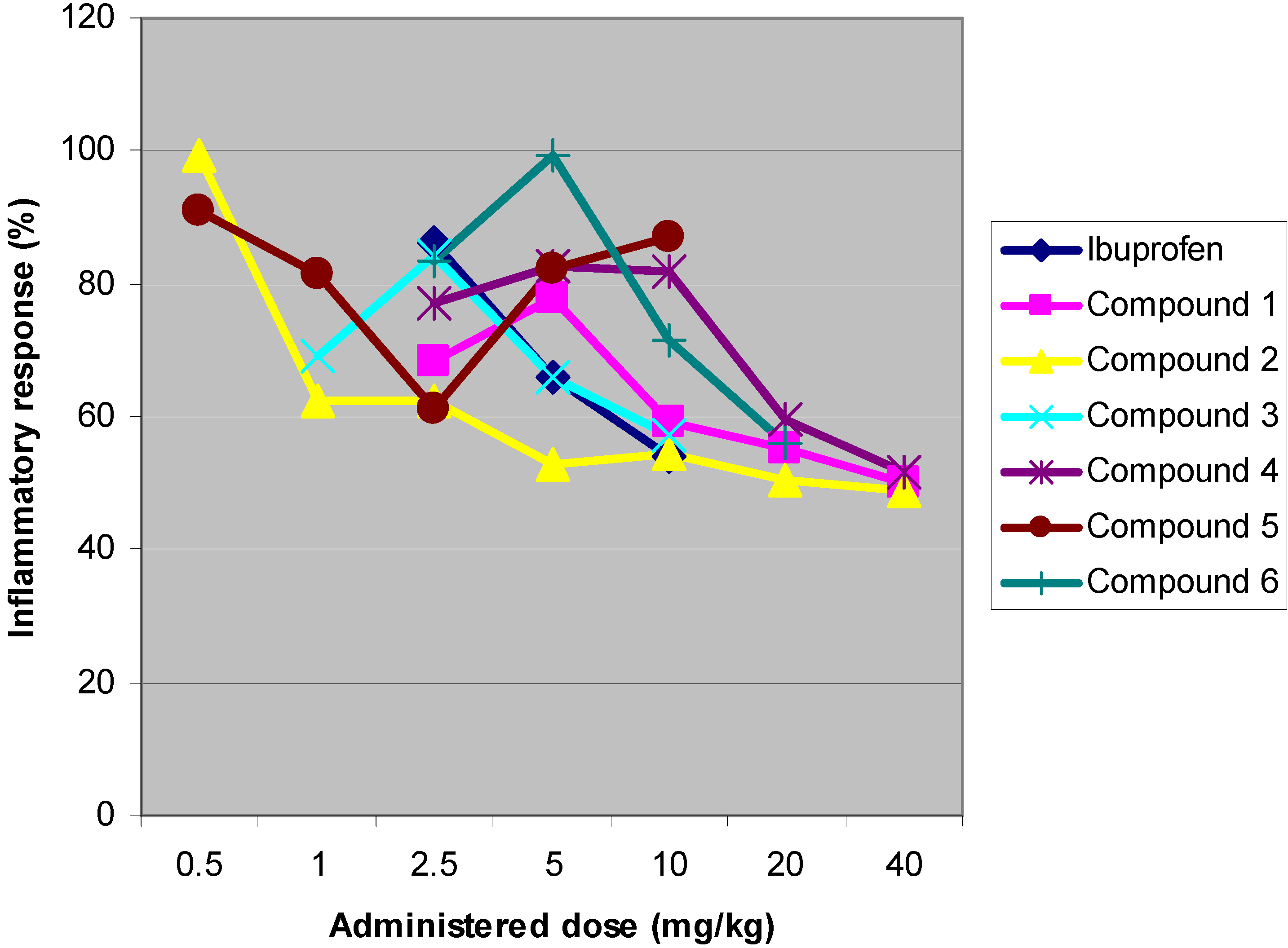
Anti-inflammatory activity: carrageenan-induced paw-oedema
Gastric tolerability
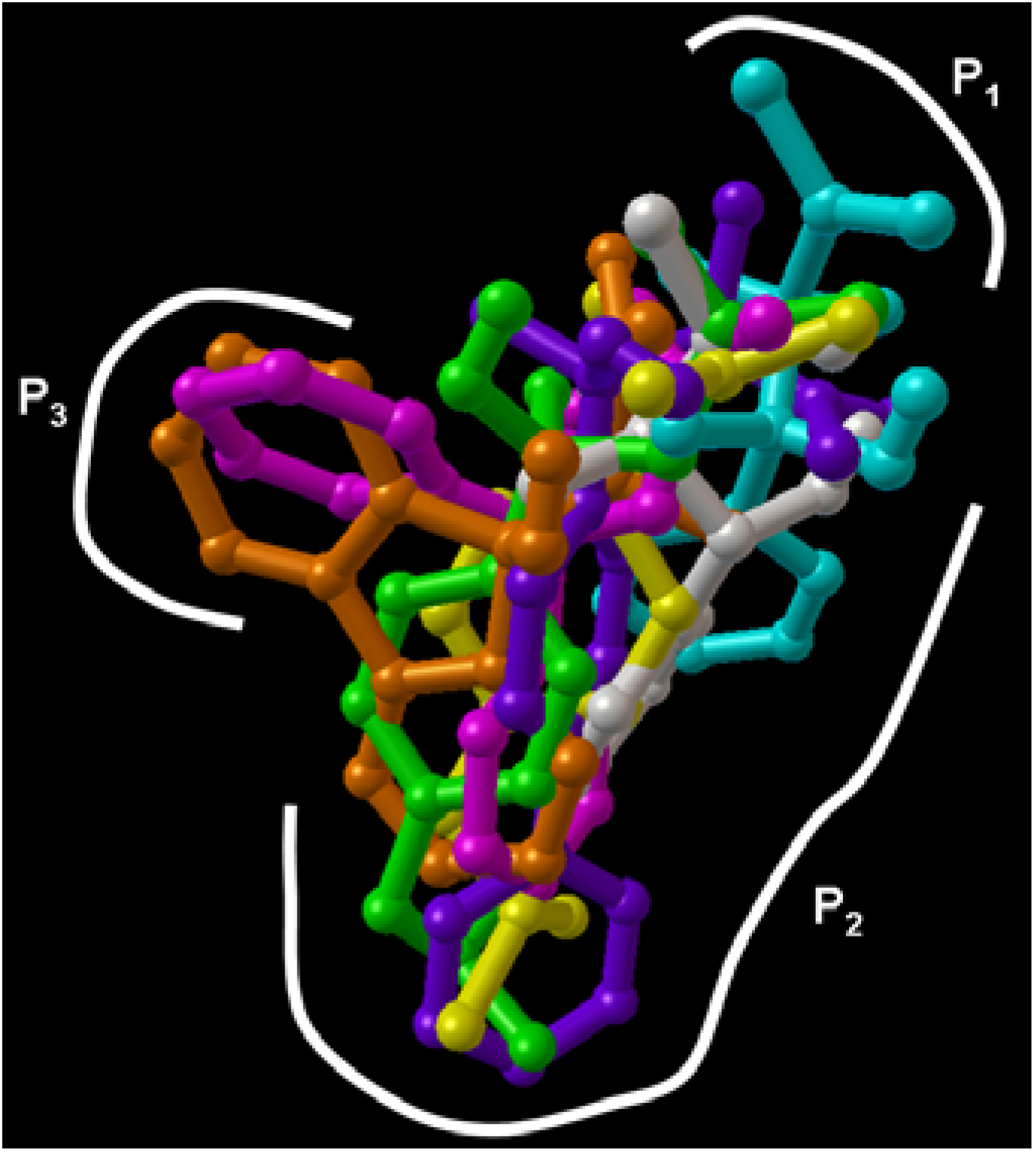
Molecular docking experiments
| Compound | Stereoisomer | Binding energy (kcal/mol) | ED50 (μM/Kg)* | |
|---|---|---|---|---|
| COX-1 | COX-2 | |||
| Ibuprofen | R | -6.47 | -6.21 | 51.7 |
| S | -6.74 | -6.05 | ||
| 1 | R | -7.70 | -7.52 | 138.7 |
| S | -7.58 | -7.20 | ||
| 2 | R | -6.84 | -5.89 | 81.3 |
| S | -7.15 | -6.34 | ||
| 3 | -7.39 | -7.17 | 50.0 | |
| 4 | R | -6.58 | -6.05 | 217.1 |
| S | -6.78 | -5.92 | ||
| 5 | -8.18 | -7.94 | 14.2 | |
| 6 | R,R | -8.59 | -8.00 | 70.1** |
| S,S | -8.61 | -8.35 | ||
| R,S | -9.02 | -8.38 | ||
| S,R | -8.69 | -8.57 | ||


| Compound No. of Rule-of-five violations TPSA (Ǻ) at pH 7.4 |
|---|
| Ibuprofen 0 40.13 |
| Compounds: |
| 1,2,3,4,5 and 6 0 60.36 |
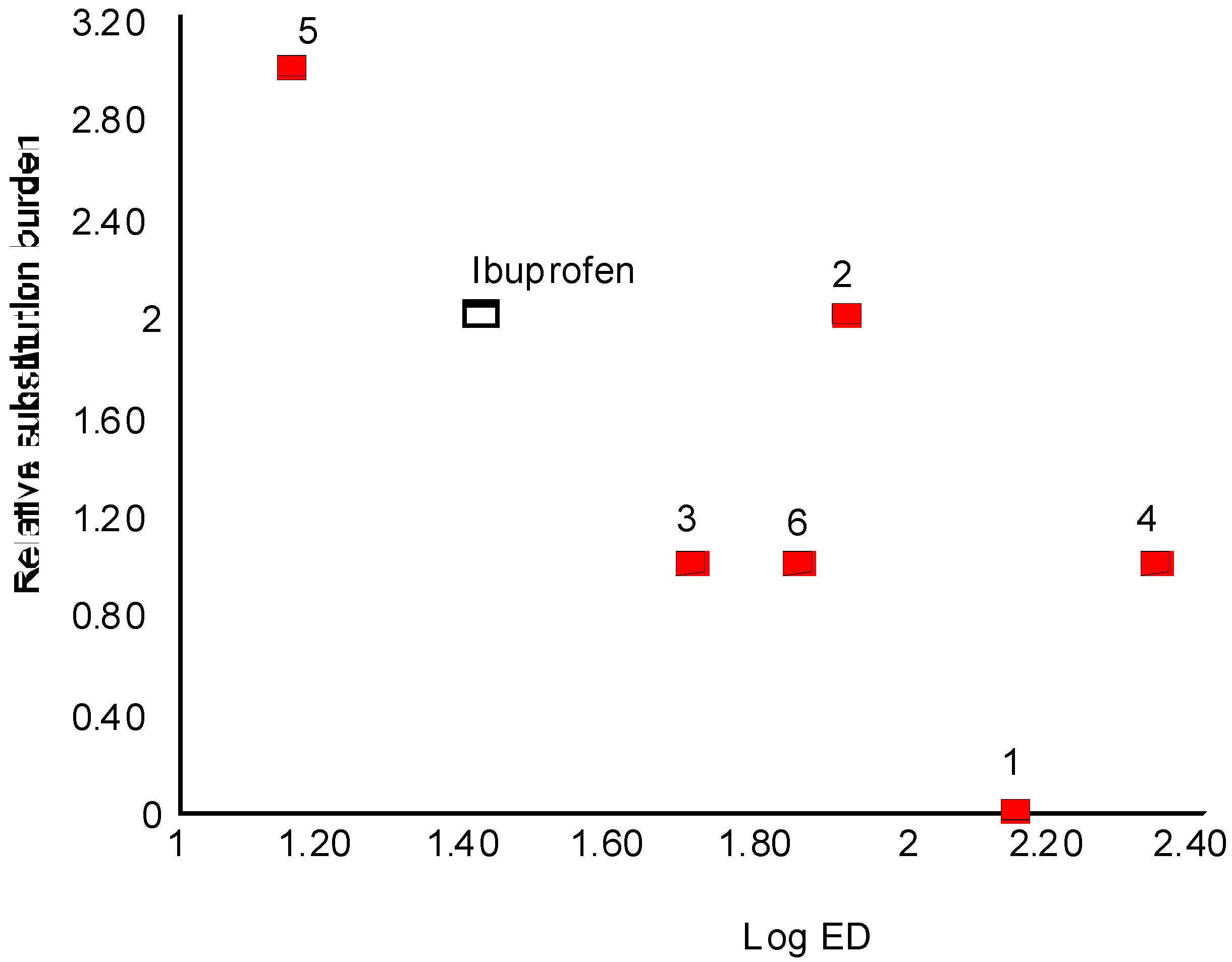
Structure-activity relationship
| Compound | log ED50 | Relative extent of substitution |
|---|---|---|
| Ibuprofen | 1.412 | 2 |
| 1 | 2.142 | 0 |
| 2 | 1.910 | 2 |
| 3 | 1.699 | 1 |
| 4 | 2.337 | 1 |
| 5 | 1.152 | 3 |
| 6 | 1.846 | 1 |
Conclusions
Experimental
General
Synthesis of 3-Hydroxy-2-methyl-3-(4-biphenylyl)butanoic acid (6)
Animal studies
Anti-inflammatory activity: carrageenan-induced rat paw oedema test

Gastric tolerability test
Statistical analysis
Acknowledgements
References and Notes
- Botting, R. M. Cyclooxygenase: Past, present and future. A tribute to John R. Vane (1027-2004). J. Therm. Bio. 2006, 31, 208–219. [Google Scholar] [CrossRef]
- Kiefer, W.; Dannhardt, G. Novel Insights and Therapeutical Applications in the Field of Inhibitors of COX-2. Curr. Med. Chem. 2004, 11, 3147–3161. [Google Scholar] [CrossRef]
- Goodsell, D. S. The Molecular Perspective: Cyclooxygenase-2. Oncologist 2000, 5, 169–171. [Google Scholar] [CrossRef]
- Dilber, S.P.; Žižak, Ž.S.; Stojković, T.P.; Juranić, Z.D.; Drakulić, B.J.; Juranić, I.O. Antiproliferative Activity of β-Hydroxy-β-arylalkanoic Acids. Int. J. Mol. Sci. 2007, 8, 214–228. [Google Scholar] [CrossRef]
- Morris, G. M.; Goodsell, D. S.; Halliday, R. S.; Huey, R.; Hart, W. E.; Belew, R. K.; Olson, A. J. Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J. Comput. Chem. 1998, 19, 1639–1662. [Google Scholar] [CrossRef]
- Selinsky, B. S.; Gupta, K.; Sharkey, K.; Loll, P. J. Structural Analysis of NSAID Binding by Prostaglandin H2 Synthase: Time-Dependent and Time-Independent Inhibitors Elicit Identical Enzyme Conformations. Biochemistry-US 2001, 40, 5172–5180. [Google Scholar] [CrossRef]
- CS ChemOffice Version 7.0 Ultra; Cambridge Soft Corporation, Software Publishers Association: Washington D.C., USA.
- Rosati, O.; Curini, M.; Marcotullio, M. C.; Macchiarulo, A.; Perfumi, M.; Mattioli, L.; Rismondo, F.; Cravotto, G. Synthesis, docking studies and anti-inflammatory activity of 4,5,6,7-tetrahydro-2H-indazole derivatives. Bioorg. Med. Chem. 2007, 15, 3463–3473. [Google Scholar] [CrossRef]
- Smith, D.A.; Van der Waterbeemd, H. Pharmacokinetics and metabolism in early drug discovery. Curr. Opin. Chem. Biol. 1999, 3, 373–378. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.; Feeney, P. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- PSA plugin, Marvin 4.1.13; ChemAxon (http://www.chemaxon.com), 2007
- Ertl, P.; Rohde, B.; Selzer, P. Fast Calculation of Molecular Polar Surface Area as a Sum of Fragment-Based Contributions and Its Application to the Prediction of Drug Transport Properties. J. Med. Chem. 2000, 43, 3714–371. [Google Scholar] [CrossRef]
- Palm, K.; Stenberg, P.; Luthman, K.; Artursson, P. Polar molecular surface properties predict the intestinal absorption of drugs in humans. Pharm. Res. 1997, 14, 568–571. [Google Scholar] [CrossRef]
- Norinder, U.; Osterberg, T.; Artursson, P. Theoretical calculation and prediction of intestinal absorption of drugs in humans using MolSurf parameterization and PLS statistics. Eur. J. Pharm. Sci. 1999, 8, 49–56. [Google Scholar] [CrossRef]
- Oyanagui, Y.; Sato, S. Inhibition by nilvadipine of ischemic and carrageenan paw edema as well as of superoxide radical production from neutrophils and xanthine oxidase. Arzneimittel-Forsch. 1991, 41, 469–474. [Google Scholar]
- Litchfield, J.T.; Wilcoxon, F.A. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 1949, 96, 99. [Google Scholar]
- Adami, E.; Marazzi-Uberti, E.; Turba, C. Pharmacological research on gefarnate, a new synthetic isoprenoid with an anti-ulcer action. Arch. Int. Pharmacodyn. Ther. 1964, 147, 113–145. [Google Scholar]
- Sample Availability: Samples of the compounds 1-6 are available from the authors (contact S.P. Dilber; sandad@pharmacy.bg.ac.yu)
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Dilber, S.P.; Dobric, S.L.; Juranic, Z.D.; Markovic, B.D.; Vladimirov, S.M.; Juranic, I.O. Docking Studies and Anti-inflammatory Activity of β-Hydroxy-β-arylpropanoic Acids. Molecules 2008, 13, 603-615. https://doi.org/10.3390/molecules13030603
Dilber SP, Dobric SL, Juranic ZD, Markovic BD, Vladimirov SM, Juranic IO. Docking Studies and Anti-inflammatory Activity of β-Hydroxy-β-arylpropanoic Acids. Molecules. 2008; 13(3):603-615. https://doi.org/10.3390/molecules13030603
Chicago/Turabian StyleDilber, Sanda P., Silva Lj. Dobric, Zorica D. Juranic, Bojan D. Markovic, Sote M. Vladimirov, and Ivan O. Juranic. 2008. "Docking Studies and Anti-inflammatory Activity of β-Hydroxy-β-arylpropanoic Acids" Molecules 13, no. 3: 603-615. https://doi.org/10.3390/molecules13030603
APA StyleDilber, S. P., Dobric, S. L., Juranic, Z. D., Markovic, B. D., Vladimirov, S. M., & Juranic, I. O. (2008). Docking Studies and Anti-inflammatory Activity of β-Hydroxy-β-arylpropanoic Acids. Molecules, 13(3), 603-615. https://doi.org/10.3390/molecules13030603




