Antimicrobial Activity of Some New Thioureides Derived from 2-(4-Chlorophenoxymethyl)benzoic Acid
Abstract
:Introduction
Results and Discussion
Synthesis
I. The synthesis of 2-(4-chlorophenoxymethyl)-benzoic acid (1)
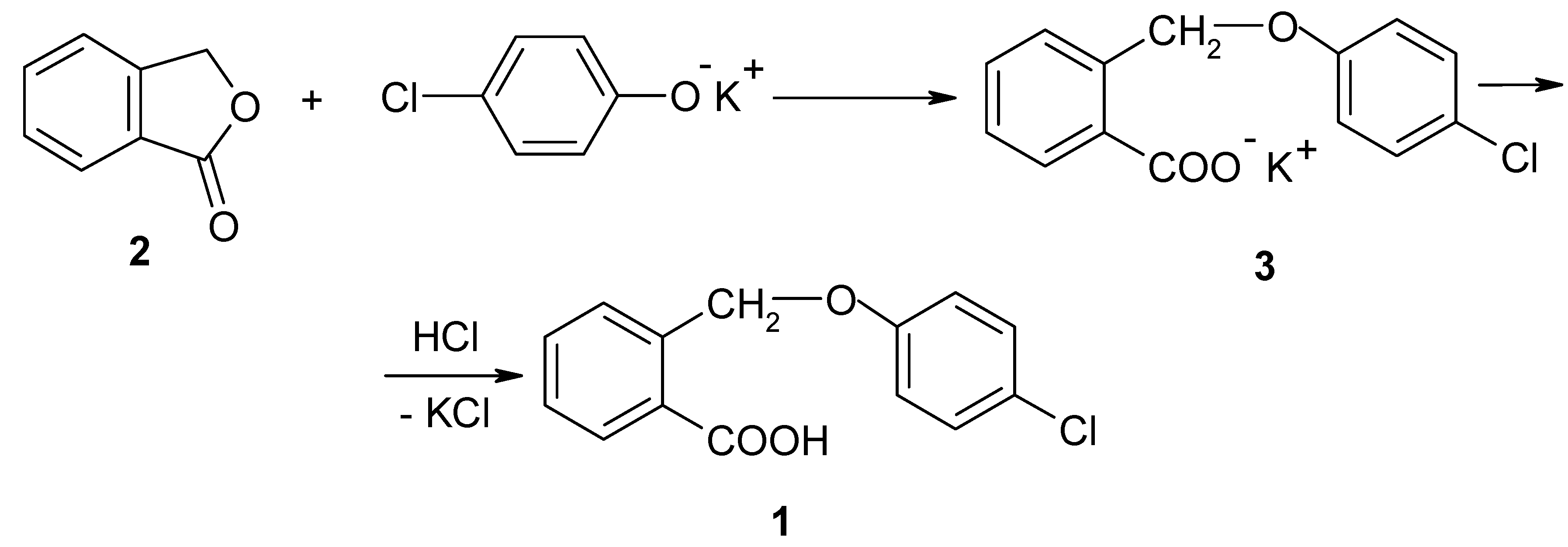
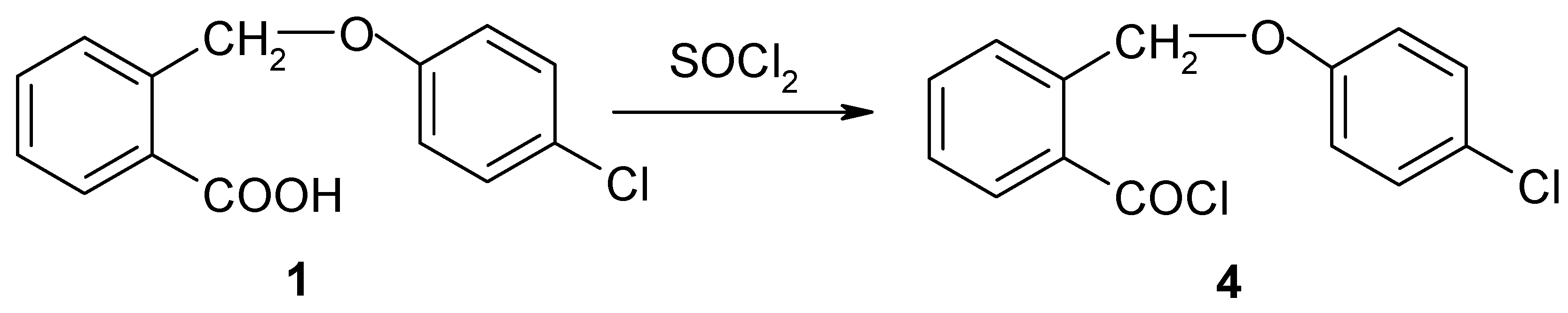
II. The synthesis of 2-(4-chloro-phenoxymethyl)-benzoyl chloride (4)
III. The synthesis of the new thioureides 5a-h

| No. | R | Molecular formula | Molecular weight | Melting point (0C) | Yield (%) |
|---|---|---|---|---|---|
| 5a | -C6H5 | C21H17ClN2O2S | 396.893 | 145.2- 147.6 | 64.3 |
| 5b | -C6H4CH3 (4) | C22H19Cl N2O2S | 410.913 | 135.7- 138.1 | 62.9 |
| 5c | -C6H4OCH3 (4) | C22H19ClN2O3S | 426.913 | 132.3- 135.4 | 69.4 |
| 5d | -C6H4Cl (4) | C21H16Cl2N2O2S | 431.336 | 154.8- 157.4 | 73.2 |
| 5e | -C6H3Cl2 (2,4) | C21H15Cl3N2O2S | 465.786 | 152.5- 155.0 | 71.6 |
| 5f | -C6H3Cl2 (2,5) | C21H15Cl3N2O2S | 465.786 | 152.6- 155.2 | 78.5 |
| 5g | -C6H3Cl2 (2,6) | C21H15Cl3N2O2S | 465.786 | 201.9- 204.6 | 65.7 |
| 5h | -C6H4Br (4) | C21H16ClBrN2O2S | 475.799 | 163.5- 165.2 | 75.3 |
 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| No. | R | C% | H% | N% | S% | ||||
| c. | e. | c. | e. | c. | e. | c. | e. | ||
| 5a. | -C6H5 | 63.49 | 63.36 | 4.28 | 4.25 | 7.05 | 6.99 | 8.06 | 7.94 |
| 5b. | -C6H4CH3 (4) | 64.24 | 63.99 | 4.62 | 4.60 | 6.81 | 6.78 | 7.78 | 7.82 |
| 5c. | -C6H4OCH3 (4) | 61.83 | 61.64 | 4.45 | 4.49 | 6.56 | 6.45 | 7.49 | 7.54 |
| 5d. | -C6H4Cl (4) | 58,42 | 58.70 | 3.70 | 3.70 | 6.49 | 6.37 | 7.42 | 7.41 |
| 5e. | -C6H3Cl2 (2,4) | 54.10 | 54.20 | 3.22 | 3.24 | 6.01 | 5.97 | 6.87 | 6.70 |
| 5f. | -C6H3Cl2 (2,5) | 54.10 | 54.03 | 3.22 | 3.29 | 6.01 | 5.92 | 6.87 | 6.71 |
| 5g. | -C6H3Cl2 (2,6) | 54.10 | 54.09 | 3.22 | 3.18 | 6.01 | 5.99 | 6.87 | 6.82 |
| 5h. | -C6H4Br (4) | 52.96 | 52.64 | 3.36 | 3.29 | 5.88 | 5.76 | 6.72 | 6.65 |
Spectral data
 | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R | H4- H7 | H8 | H10 | H11 | H13 | H14 | H17 | H18 | H19 | H20 | H21 | -NH- | ||
| -OCH3 | ||||||||||||||
 | 7.28- 7.68 m | 5.32 s | 7.01 d (7.9) | 7.30 d (7.9) | 7.30 d (7.9) | 7.01 d (7.9) | 7.28- 7.68 m | 7.28- 7.68 m | 7.28- 7.68 m | 7.28- 7.68 m | 7.28- 7.68 m | 11.84 s 12.40 s | - | |
 | 7.56- 7.62 m | 5.30 s | 7.31 d (8.9) | 7.00 d (9.0) | 7.00 d (9.0) | 7.31 d (8.9) | 7.45 d (8.6) | 7.20 d (8.2) | - | 7.20 d (8.2) | 7.45 d (8.6) | 11.79 s l12.33 sl | ||
 | 7.45- 7.66 m | 5.31 s | 7.02 d (8.9) | 7.46 d (9.0) | 7.46 d (9.0) | 7.02 d (8.9) | 7.32 d (8.8) | 6.96 d (8.8) | - | 6.96 d (8.8) | 7.32 d (8.8) | 11.79 s 12.26 s | ||
| 3.77 s | ||||||||||||||
 | 7.42- 7.63 m | 5.32 s | 7.00 d (9.0) | 7.30 d (9.0) | 7.30 d (9.0) | 7.00 d (9.0) | 7.46 d (8.8) | 7.62 d (8.8) | - | 7.62 d (8.8) | 7.46 d (8.8) | 11.90 s 12.38 s | - | |
 | 7.42- 7.68 m | 5.30 s | 6.99 d (9.1) | 7.30 d (9.1) | 7.30 d (9.1) | 6.99 d (9.1) | - | 7.73 d (2.3) | - | 7.81 dd (8.8; 2.3) | 7.96 d (8.8) | 12.12 s 12.43 s | - | |
 | 7.40- 7.70m | 5.30 s | 7.01 d (8.9) | 7.28 d (8.9) | 7.28 d (8.9) | 7.01 d (8.9) | - | 7.40-7.70 m | 7.39 dd (8.9; 2.6) | - | 8.11 d (2.6) | 12.16 s 12.53 s | - | |
 | 7.42-7.62 m | 5.28 s | 7.00 d (8.9) | 7.32 d (8.9) | 7.32 d (8.9) | 7.00 d (8.9) | - | 7.54 d (8.0) | 7.37 dd (8.0; 7.4) | 7.54 d (8.0) | - | 11.05 s 11.39 s | - | |
 | 7.45- 7.70 m | 5.34 s | 7.00 d (8.9) | 7.31 d (8.9) | 7.31 d (8.9) | 7.00 d (8.9) | 7.57 s | 7.57 s | - | 7.57 s | 7.57 s | 11.89 s 12.37 s | - | |
 | |||||||
|---|---|---|---|---|---|---|---|
| R |  |  |  |  | |||
| C1 | 169.57 | 170.05 | 170.02 | 169.98 | |||
| C2 | 134.99 | 131.04 | 133.37 | 131.1 | |||
| C3 | 137.64 | 135.64 | 135.24 | 136.8 | |||
| C4 | 127.56 | 124.19 | 127.9 | 126.1 | |||
| C5 | 133.10 | 129.22 | 131.05 | 133.3 | |||
| C6 | 128.26 | 128.41 | 128.56 | 127.9 | |||
| C7 | 128.17 | 127.88 | 128.44 | 127.9 | |||
| C8 | 67.73 | 67.79 | 67.8 | 67,82 | |||
| C9 | 156.83 | 157.0 | 157.05 | 157.03 | |||
| C10 | 116.37 | 116.44 | 116.50 | 116,49 | |||
| C11 | 130.73 | 129.05 | 129.25 | 129.3 | |||
| C12 | 125,9 | 124.68 | 125.9 | 128.5 | |||
| C13 | 130.73 | 129.05 | 129.25 | 129.3 | |||
| C14 | 116.37 | 116.44 | 116.50 | 116,49 | |||
| C15 | 178.65 | 178.83 | 179.05 | 179.18 | |||
| C16 | 134.9 | 135.22 | 130.7 | 135.3 | |||
| C17 | 123.77 | 124.09 | 124.74 | 124.7 | |||
| C18 | 128.88 | 128.55 | 113.78 | 128.4 | |||
| C19 | 124.58 | 133.32 | 157.44 | 130.2 | |||
| C20 | 128.88 | 128.55 | 113.78 | 128.4 | |||
| C21 | 123.77 | 124.09 | 124.74 | 124.7 | |||
| - | - | ||||||
| -OCH3 | 55.30 | ||||||
| R |  |  |  |  | |||
| C1 | 170.11 | 170.32 | 169.50 | 169.98 | |||
| C2 | 133.29 | 131.19 | 133.14 | 133.23 | |||
| C3 | 135.33 | 135.25 | 134.93 | 137.25 | |||
| C4 | 128.56 | 128.05 | 128.18 | 127.90 | |||
| C5 | 131.37 | 131.10 | 130.86 | 131.46 | |||
| C6 | 128.76 | 128.69 | 128.29 | 128.58 | |||
| C7 | 128.69 | 128.65 | 128.69 | 128.44 | |||
| C8 | 68.13 | 67.94 | 67.79 | 67.80 | |||
| C9 | 157.13 | 157.01 | 156.83 | 157.02 | |||
| C10 | 116.66 | 116.41 | 116.45 | 116.48 | |||
| C11 | 129.26 | 129.20 | 128.9 | 129.24 | |||
| C12 | 127.95 | 127.71 | 127.78 | 126.41 | |||
| C13 | 129.26 | 129.20 | 128.9 | 129.24 | |||
| C14 | 116.66 | 116.41 | 116.45 | 116.48 | |||
| C15 | 180.12 | 179.95 | 180.51 | 179.10 | |||
| C16 | 134.60 | 136.5 | 133.53 | 135.3 | |||
| C17 | 128.91 | 126.88 | 127.88 | 124.73 | |||
| C18 | 129.17 | 130.78 | 129.46 | 131.13 | |||
| C19 | 131.14 | 126.99 | 128.18 | 118.49 | |||
| C20 | 127.27 | 133.22 | 129.46 | 131.13 | |||
| C21 | 124.93 | 124.74 | 124.58 | 124.73 | |||
| - | - | - | - | ||||
| -OCH3 | |||||||
 | |||||||
|---|---|---|---|---|---|---|---|
| R | -CH2-O- | -CO-NH- | |||||
| ν CH2sym | ν CH2 asym | νC-O-Csym | νC-O-Casym | δ CH2 | ν C-N | ||
| -C6H5 | 2852.2 | 2921.7 | 1008.2 | 1243.0 | 1491.4 | 1553.6 | 1243.0 |
| -C6H4CH3 (4) | 2852.2 | 2921.7 | 1012.7 | 1243.9 | 1491.1 | 1550.6 | 1243.9 |
| -C6H4OCH3 (4) | 2829.0 | 2910.1 | 1036.6 | 1241.1 | 1488.5 | 1553.1 | 1241.1 |
| -C6H4Cl (4) | 2848.3 | 2929.4 | 1035.1 | 1243.1 | 1491.3 | 1554.3 | 1243.1 |
| -C6H3Cl2 (2,4) | 2856.0 | 2921.7 | 1036.0 | 1242.7 | 1491.5 | 1540.0 | 1242.7 |
| -C6H3Cl2 (2,5) | 2852.2 | 2921.7 | 1036.3 | 1243.6 | 1491.0 | 1552.7 | 1243.6 |
| -C6H3Cl2 (2,6) | 2856.0 | 2917.8 | 1036.5 | 1240.3 | 1486.9 | 1553.3 | 1240.3 |
| -C6H4Br (4) | 2852.2 | 2921.7 | 1033.4 | 1242.9 | 1490.5 | 1553.9 | 1242.9 |
| R | -CO-NH- | aromatic rings | -NH-CS-NH- | C-halogen | |||
| ν C=O | ν N-H | ν =CH | ν C=C | ν C=S | ν C-Cl | ν C-Br | |
| -C6H5 | 1682.9 | 3442.7 | 3018.1 | 1598.7 | 1166.7 | 749.5 | - |
| -C6H4CH3 (4) | 1683.5 | 3458.1 | 3029.7 | 1596.8 | 1164.0 | 749.1 | - |
| -C6H4OCH3 (4) | 1683.4 | 3436.9 | 3002.7 | 1598.6 | 1167.4 | 746.7 | - |
| -C6H4Cl (4) | 1679.8 | 3356.4 | 3029.7 | 1603.2 | 1151.4 | 744.2 | - |
| -C6H3Cl2 (2,4) | 1684.0 | 3381.1 | 3099.2 | 1584.2 | 1152.9 | 744.8 | - |
| -C6H3Cl2 (2,5) | 1679.1 | 3442.7 | 3025.9 | 1585.0 | 1168.2 | 751.6 | - |
| -C6H3Cl2 (2,6) | 1693.1 | 3439.4 | 3030.2 | 1597.8 | 1168.3 | 753.2 | - |
| -C6H4Br (4) | 1678.6 | 3360.7 | 3030.6 | 1600.7 | 1151.1 | 755.9 | 607.7 |
Qualitative antimicrobial activity screening

Quantitative antimicrobial activity assay
| Organisms | Compounds | |||||||
|---|---|---|---|---|---|---|---|---|
| 5a | 5b | 5c | 5d | 5e | 5f | 5g | 5h | |
| E. coli | > 1024 | 32 | 32 | 32 | > 1024 | > 1024 | 256 | 128 |
| S. enteritidis | 256 | > 1024 | > 1024 | 32 | > 1024 | > 1024 | 128 | 128 |
| P. aeruginosa | > 1024 | > 1024 | > 1024 | > 1024 | > 1024 | > 1024 | 32 | 32 |
| L. monocytogenes | > 1024 | > 1024 | > 1024 | > 1024 | 512 | 512 | > 1024 | > 1024 |
| S. aureus | 32 | 32 | 32 | 32 | 32 | 256 | 32 | 32 |
| B. subtilis | > 1024 | > 1024 | 1024 | > 1024 | > 1024 | > 1024 | > 1024 | > 1024 |
| C. albicans | 256 | 64 | 512 | 512 | > 1024 | > 1024 | 32 | 32 |
Flow cytometry and annexin V assay
| HeLa cells | ||||
|---|---|---|---|---|
| 1 (% necrosis) | 2 (% secondary necrosis) | 3 (% viable cells) | 4 (% apoptosis) | |
| Cell control | 3.42 | 0.70 | 95.00 | 0.19 |
| 5g | 13.89 | 9.10 | 84.00 | 0.15 |
| 5h | 6.77 | 0.70 | 95.00 | 0.15 |
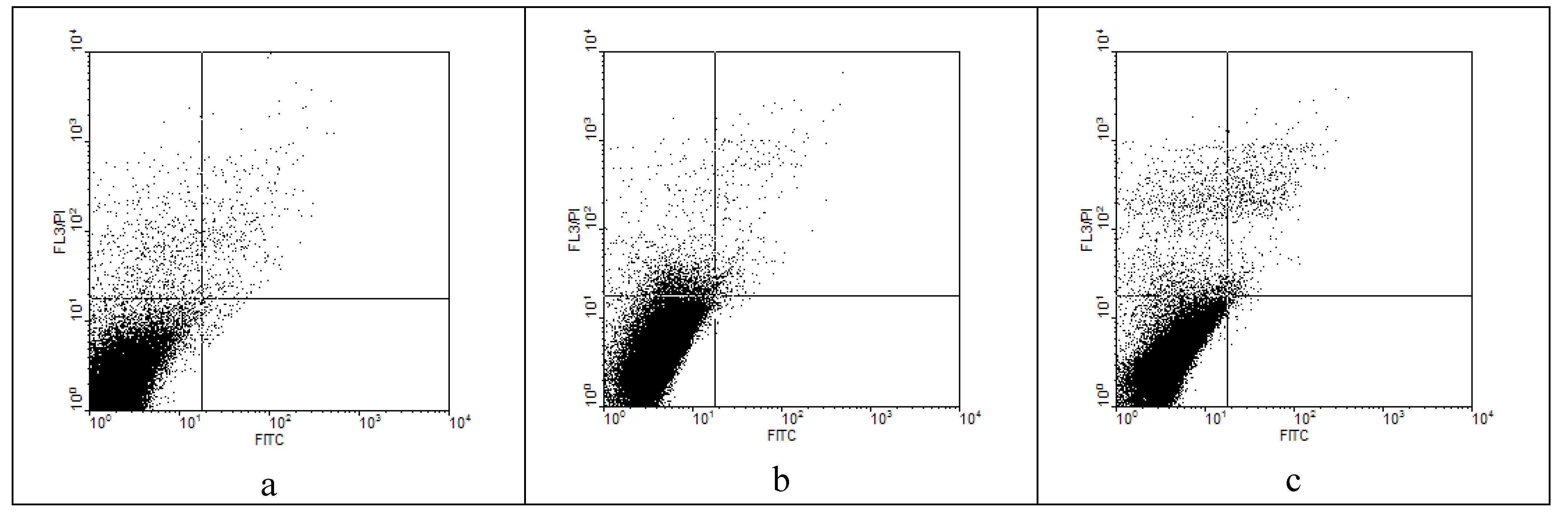
Conclusions
Experimental
General
Synthesis of 2-(4-chloro-phenoxymethyl)-benzoic acid (1)
Synthesis of 2-(4-chloro-phenoxymethyl)-benzoyl chloride (4)
General procedure for the synthesis of the new thioureides (5a)-(5h)
Microbial strains
Qualitative screening of the antimicrobial properties of the tested compounds
Quantitative assay of the antimicrobial activity
Flow cytometry and annexin V assay
References
- Limban, C.; Missir, Al.; Chiriţă, I. Noi tioureide ale acidului 2-fenoximetil-benzoic. Nota I. Farmacia 2000, 6, 73–78. [Google Scholar]
- Limban, C.; Missir, Al.; Chiriţă, I. Noi tioureide ale acidului 2-fenoximetil-benzoic. Nota II. Farmacia 2004, 5, 7–12. [Google Scholar]
- Limban, C.; Missir, Al.; Chiriţă, I.; Missir, C. The synthesis and characterization of some new thioureides of 2-(4-methylphenoxymethyl)-benzoic acid with potential pharmacological activity. In Romanian International Conference on Chemistry and Chemical Engineering-RICCCE XIV, Bucarest, Rumania, 22-24 September 2005; Vol.4. S-02-99.
- Limban, C.; Chiriţă, I.; Missir, Al. Synthesis and characterization of some new thioureides of 2-(4-methoxyphenoxymethyl)-benzoic acid with potential pharmacological activity. TimisoaraMed. J. 2005, 55, 42–44. [Google Scholar]
- Mutiu, A.; Alexiu, I.; Chivu, M.; Petica, M.; Anton, G.; Bleotu, C.; Diaconu, C.; Popescu, C.; Jucu, V.; Cernescu, C. Detection of human papillomavirus gene sequence in cell lines derived from laryngeal tumors. J. Cell. Mol. Med. 2001, 5, 49–59. [Google Scholar] [CrossRef]
- Diaconu, C.; Bleotu, C.; Chivu, M.; Alexiu, A.; Petrusca, D.; Anton, G.; Achim, R.; Ruta, S.M.; Cernescu, C. The development of larger cells that spontaneously escape senescence - a step during the immortalization of a human cancer cell line. J. Cell. Mol. Med. 2004, 8, 93–101. [Google Scholar] [CrossRef]
- Olar, R.; Badea, M.; Cristurean, E.; Lazar, V.; Cernat, R.; Balotescu, M.C. Thermal behavior, spectroscopic and biological characterization of Co (II), Zn (II) and Pt (II) complexes with N,N-dimethylbiguanide. J. Therm. Anal. Calorim. 2005, 80, 451–455. [Google Scholar] [CrossRef]
- Lazar, V.; Balotescu, M.C.; Moldovan, L.; Vasilescu, G.; Petrache, L.M.; Bulai, D.; Cernat, R. Comparative evaluation of qualitative and quantitative methods used in the study of antifungal and antibacterial activity of hydroalcoholic vegetal extracts. Roum. Biotechnol. Lett. 2005, 10, 2225–2232. [Google Scholar]
- Balotescu, M.C.; Oprea, E.; Petrache, L.M.; Bleotu, C.; Lazar, V. Antibacterial, antifungal and cytotoxic activity of salvia officinalis essential oil and tinctures. Roum. Biotechnol. Lett. 2005, 10, 2481–2481. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Seventeenth Informational Supplement, M100-S17, Vol. 27 No. 1, Replaces M100-S16, Vol. 26 No. 3, Informational Supplement; CLSI: Wayne, PA, USA, 2007. [Google Scholar]
- Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; Approved standard M7-A2; CLSI: Wayne, PA, USA, 1999. [Google Scholar]
- Balotescu, M.C.; Limban, C.; Missir, A.V.; Chirita, I.C.; Nitulescu, M.G. The Synthesis and Biological Activities Of Some New 2-(4-Methoxyphenoxymethyl)benzoic Acid Thioureides. Rev. Chim. (Bucuresti) 2007, 58, 1064–1068. [Google Scholar]
- Sample availability: Contact the authors.
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Limban, C.; Chifiriuc, M.-C.B.; Missir, A.-V.; Chiriţă, I.C.; Bleotu, C. Antimicrobial Activity of Some New Thioureides Derived from 2-(4-Chlorophenoxymethyl)benzoic Acid. Molecules 2008, 13, 567-580. https://doi.org/10.3390/molecules13030567
Limban C, Chifiriuc M-CB, Missir A-V, Chiriţă IC, Bleotu C. Antimicrobial Activity of Some New Thioureides Derived from 2-(4-Chlorophenoxymethyl)benzoic Acid. Molecules. 2008; 13(3):567-580. https://doi.org/10.3390/molecules13030567
Chicago/Turabian StyleLimban, Carmen, Mariana-Carmen Balotescu Chifiriuc, Alexandru-Vasile Missir, Ileana Cornelia Chiriţă, and Coralia Bleotu. 2008. "Antimicrobial Activity of Some New Thioureides Derived from 2-(4-Chlorophenoxymethyl)benzoic Acid" Molecules 13, no. 3: 567-580. https://doi.org/10.3390/molecules13030567






