Synthesis and Biological Activity of Some Novel Derivatives of 4-Amino-3-(D-galactopentitol-1-yl)-5-mercapto-1,2,4-triazole
Abstract
:Introduction
Results and Discussion
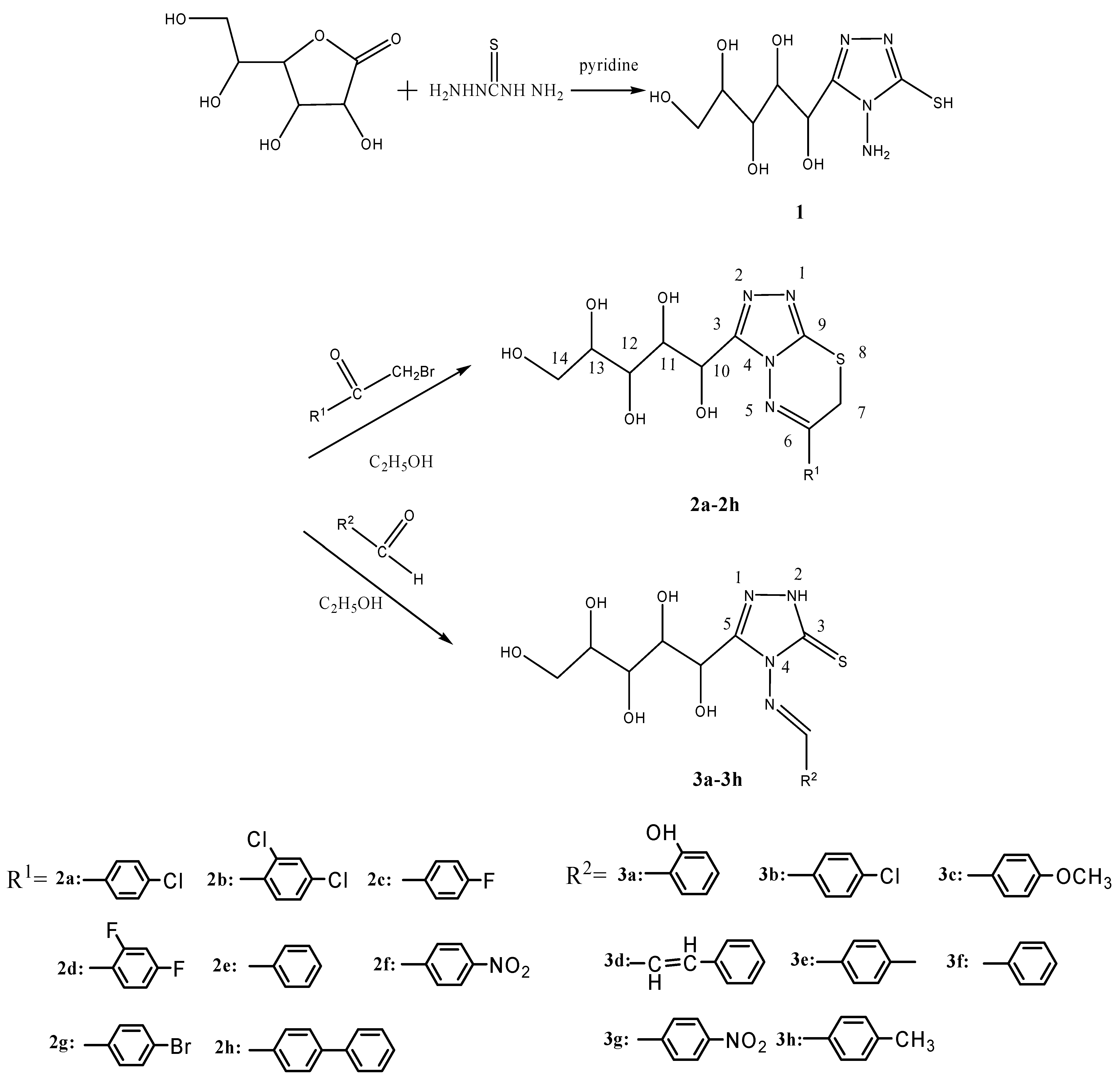
Synthesis
IR spectra of the title compounds
1H-NMR spectra of the title compounds
13C-NMR spectra of the title compounds
Biological evaluation

| Compd. | Radish | Wheat | ||
|---|---|---|---|---|
| 10 ppm | 100 ppm | 10 ppm | 100 ppm | |
| 1 | 100/23.5/3.8 | 85/40.1/-1.3 | 85/-3.4/20.9 | 75/-28.1/8.7 |
| 2a | 30/-54.7/-74.9 | -100% | 85/-74.5/-72.4 | -100% |
| 2b | 35/-27/-52.2 | -100% | 85/-41.9/-52.6 | -100% |
| 2c | 20/-56.1/-77.4 | -100% | 95/-79.5/-78.1 | -100% |
| 2d | 25/-44.5/-61.1 | -100% | 100/-69/-65.9 | -100% |
| 2e | 65/4.9/-21.6 | 75/6.2/-28.8 | 100/8.2/12.9 | 80/3.1/7.3 |
| 2f | -100% | -100% | 85/-95.5/-95.6 | -100% |
| 2g | 35/-37.2/-65.7 | -100% | 90/-52.6/-56.4 | -100% |
| 2h | 15/-31.8/-58.1 | -100% | 95/-15.2/-43.5 | 95/-96.6/-96.1 |
| 3a | 30/-17.5/-34.7 | 30/-14.4/-36.7 | 85/19/-2.0 | 85/6.9/-1.2 |
| 3b | 5/-27.8/-77.8 | -100% | 5/-18.1/-83.6 | -100% |
| 3c | 5/-12.5/-61.1 | 5/-100/-7.2 | 100/-85.8/-83.4 | 100/-91.8/-100 |
| 3d | 5/-69.6/-91.7 | -100% | 95/-91.4/-92 | 75/-92.7/-100 |
| 3e | 15/0.3/-46.7 | 15/-50.2/-78.1 | 60/-9.5/-12.3 | 55/-91.4/-91.6 |
| 3f | 65/-11.6/-34.4 | -100% | 55/-79.4/-85.2 | -100% |
| 3g | 40/-32.9/-31.5 | -100% | 85/-36.1/-28.7 | -100% |
| 3h | 30/-96.4/-98.1 | -100% | 65/-83.8/-78.5 | -100% |
| Reference | 90/0/0 | 90/0/0 | 100/0/0 | 100/0/0 |
Experimental
General
Preparation of 4-amino-3-(D-galactopentitol-1-yl)-5-mercapto-1,2,4-triazole (1).
General method for the preparation of 6-aryl-3-(D-galactopentitol-1-yl)-7H-1,2,4-triazolo [3,4-b] [1,3,4] thiadiazines 2a~2h
General method for the preparation of 4-(arylmethylidene)amino-5-(D-galactopentitol-1-yl)-3- mercapto-4H- 1,2,4-triazoles 3a~3h
| Compd. | Molecular formula | Colour | Yield/% | M.p./°C | Anal./% Found (calc.) | ||
|---|---|---|---|---|---|---|---|
| C | H | N | |||||
| 2a | C15H17ClN4O5S | white | 75.6 | 195~197 | 45.21(44.95) | 4.12(4.27) | 14.31(13.98) |
| 2b | C15H16Cl2N4O5S | yellow | 68.7 | 101~103 | 41.07(41.39) | 3.64(3.70) | 12.69(12.87) |
| 2c | C15H17FN4O5S | white | 79.2 | 208~210 | 46.78(46.87) | 4.71(4.46) | 14.79(14.58) |
| 2d | C15H16F2N4 O5S | white | 66.1 | 175~177 | 44.95(44.77) | 4.25(4.01) | 13.74(13.92) |
| 2e | C15H18N4O5S | white | 49.4 | 200~202 | 49.04(49.17) | 4.86(4.95) | 15.17(15.29) |
| 2f | C15H17N5O7S | brown | 48.7 | 178~180 | 44.08(43.79) | 4.46(4.17) | 17.35(17.02) |
| 2g | C15H17BrN4O5S | white | 57.3 | 183~185 | 40.22(40.46) | 3.72(3.85) | 12.28(12.58) |
| 2h | C21H22N4O5S | brown | 61.2 | 103~105 | 56.86(57.00) | 5.24(5.01) | 12.59(12.66) |
| 3a | C14H18N4O6S | white | 68.2 | 206~208 | 45.46(45.40) | 4.81(4.90) | 15.25(15.13) |
| 3b | C14H17ClN4O5S | white | 72.5 | 218~220 | 43.12(43.25) | 4.50(4.41) | 14.29(14.41) |
| 3c | C15H20N4O6S | white | 71.8 | 221~223 | 46.98(46.87) | 5.36(5.24) | 14.38(14.57) |
| 3d | C16H20N4O5S | white | 72.6 | 192~193 | 50.36(50.52) | 5.47(5.30) | 14.90(14.73) |
| 3e | C22H30N8O10S2 | yellow | 75.1 | 218~220 | 41.72(41.90) | 4.56(4.79) | 17.82(17.77) |
| 3f | C14H18N4O5S | white | 65.4 | 203~205 | 47.71(47.45) | 5.24(5.12) | 15.57(15.81) |
| 3g | C14H17N5O7S | brown | 63.5 | 228~229 | 42.21(42.10) | 4.36(4.29) | 17.78(17.54) |
| 3h | C15H20N4O5S | white | 75.8 | 220~222 | 48.82(48.90) | 5.31(5.47) | 15.26(15.21) |
| Compd. | IR (ν/cm-1) |
|---|---|
| 2a 2b 2c 2d 2e 2f 2g 2h 3a 3b 3c 3d 3e 3f 3g 3h | 3417(OH), 2978(CH2), 1634(C=N), 1511(N=C-S), 679(C-S-C). 3417(OH), 2968(CH2), 1632(C=N), 1469(N=C-S), 614(C-S-C). 3417(OH), 2921(CH2), 1630(C=N), 1453(N=C-S), 624(C-S-C). 3416(OH), 2958(CH2), 1615(C=N), 1469(N=C-S), 617(C-S-C). 3421(OH), 2921(CH2), 1630(C=N), 1470(N=C-S), 668(C-S-C). 3438(OH), 2976(CH2), 1634(C=N), 1470(N=C-S), 664(C-S-C). 3413(OH), 2934(CH2), 1631(C=N), 1465(N=C-S), 680(C-S-C). 3440(OH), 2946(CH2), 1632(C=N), 1469(N=C-S), 697(C-S-C). 3417(OH), 2936(CH2), 1621(C=N), 1277(C=S). 3415(OH), 2925(CH2), 1626(C=N), 1292(C=S). 3416(OH), 2970(CH2), 1631(C=N), 1252(C=S). 3416(OH), 2975(CH2), 1627(C=N), 1247(C=S). 3414(OH), 2921(CH2), 1622(C=N), 1273(C=S). 3261(OH), 2933(CH2), 1587(C=N), 1272(C=S). 3535(OH), 2873(CH2), 1582(C=N), 1234(C=S). 3363(OH), 2920(CH2), 1604(C=N), 1281(C=S). |
| Compd. | 1H-NMR (δ, ppm) | 13C-NMR (δ, ppm) |
|---|---|---|
| 2a | 3.40-6.03 (m, 13H, C-H, O-H), 7.68-8.11 (m, 4H, Ar-H). | 22.86, 62.90, 65.91, 68.83, 69.71, 71.12, 129.24, 129.68, 131.86, 137.26, 141.97, 155.06, 155.65. |
| 2b | 3.37-5.12 (m, 13H, C-H, O-H), 7.61-7.88 (m, 3H, Ar-H). | 25.83, 62.88, 65.28, 69.00, 69.81, 71.45, 128.01, 129.76, 132.47, 132.61, 133.39, 136.21, 140.46, 154.54, 154.64. |
| 2c | 3.34-5.29 (m, 13H, C-H,O-H), 7.40-8.13 (m, 4H, Ar-H). | 22.88, 62.92, 65.26, 69.02, 69.82, 71.44, 116.24, 130.11, 140.25, 153.49, 154.55, 162.54, 165.86. |
| 2d | 3.33-5.25 (m, 13H, C-H,O-H), 7.30-7.94 (m, 3H, Ar-H). | 25.06, 62.90, 65.26, 69.02, 69.83, 71.46, 105.23, 112.74, 119.57, 132.14, 140.32, 151.50, 154.61, 162.58, 165.92. |
| 2e | 3.39-5.31 (m, 13H, C-H,O-H), 7.56-8.06 (m, 5H, Ar-H). | 22.94, 62.91, 65.60, 68.93, 69.78, 71.31, 127.72, 129.06, 132.11, 133.27, 141.26, 154.80, 155.64. |
| 2f | 3.35-5.76 (m, 13H, C-H,O-H), 8.22-8.42 (m, 4H, Ar-H). | 23.01, 62.54, 66.06, 73.70, 75.71, 81.18, 108.36, 123.96, 128.78, 139.43, 149.13, 152.61, 153.92. |
| 2g | 3.35-5.29 (m, 13H, C-H,O-H), 7.78-7.99 (m, 4H, Ar-H). | 22.88, 63.08, 65.44, 69.19, 69.97, 71.57, 125.78, 129.72, 132.20, 132.94, 140.44, 153.70, 154.76. |
| 2h | 3.34-5.30 (m, 13H, C-H,O-H), 7.44-8.14 (m, 9H, Ar-H). | 23.00, 63.10, 65.43, 69.19, 70.02, 71.66, 127.04, 127.29, 128.42, 128.48, 129.27, 132.59, 138.99, 140.56, 143.38, 154.24, 154.74. |
| 3a | 3.35-5.54 (m, 11H, C-H, C-OH), 6.91-7.87 (m, 4H, Ar-H), 10.10 (s, 1H, CH=N), 10.44 (s, 1H, Ar-OH), 13.79 (s, 1H, HN-C). | 62.94, 65.73, 69.08, 69.82, 70.57, 116.57, 118.38, 119.58, 127.84, 134.03, 152.06, 158.37, 160.46, 161.38. |
| 3b | 3.32-5.29 (m, 11H, C-H, C-OH), 7.60-7.96 (m, 4H, Ar-H), 10.03 (s, 1H, CH=N), 13.84 (s, 1H, HN-C). | 62.90, 65.63, 69.10, 69.81, 70.41, 129.27, 130.31, 131.25, 137.09, 152.30, 161.18, 161.36. |
| 3c | 3.32-5.26 (m, 11H, C-H, C-OH), 3.85-3.88 (m, 3H, CH3), 7.08-7.87 (m, 4H, Ar-H), 9.73(s, 1H, CH=N), 13.76 (s, 1H, HN-C). | 55.53, 62.89, 65.58, 69.06, 69.83, 70.39, 114.59, 124.68, 130.65, 152.10, 161.29, 162.75, 163.26. |
| 3d | 3.35-5.26 (m, 11H, C-H, C-OH), 7.11-7.47 (m, 5H, Ar-H), 7.72-7.76 (m, 2H, CH=CH), 9.56-9.59 (d, 1H, CH=N), 13.79 (s, 1H, HN-C). | 62.913, 65.42, 69.128, 69.76, 70.43, 123.65, 127.98, 128.98, 130.15, 135.00, 146.10, 151.99, 161.24, 165.65. |
| 3e | 3.32-5.91 (m, 22H, C-H, C-OH), 7.09-7.87 (m, 4H, Ar-H), 10.01-10.13 (m, 2H, CH=N), 13.51 (s, 2H, HN-C). | 62.90, 65.68, 69.11, 69.83, 70.43, 129.17, 135.70, 152.43, 160.86, 161.40. |
| 3f | 3.31-5.27 (m, 11H, C-H, C-OH), 7.53-7.92 (m, 5H, Ar-H), 9.96 (s, 1H, CH=N), 13.81 (s, 1H, HN-C). | 62.89, 65.60, 69.13, 69.84, 70.46, 128.62, 129.06, 132.26, 132.45, 152.20, 161.36, 162.94. |
| 3g | 3.45-5.32(m, 11H, C-H, C-OH), 7.85-8.41(m, 4H, Ar-H), 10.41(s, 1H, CH=N), 13.92(s, 1H, HN-C). | 62.92, 65.69, 69.08, 69.80, 70.39, 124.20, 129.71, 138.43, 149.37, 152.60, 158.50, 161.46. |
| 3h | 2.40-2.51(m, 3H, CH 3), 3.32-5.27(m, 11H, C-H, C-OH), 7.35-7.81(m, 4H, Ar-H), 9.86(s, 1H, CH=N), 13.80(s, 1H, HN-C). | 21.25, 62.94, 65.61, 69.17, 69.89, 70.50, 128.69, 129.61, 129.69, 142.81, 152.18, 161.39, 163.15. |
Plant growth-regulating activity tests: materials and preparation of the test solutions
Method for plate testing the plant growth-regulating activities of compounds 1, 2a~2h, 3a~3h
Acknowledgements
References
- Chadha, V. K.; Ranwa, N. S.; Dadheech, P. K. Synthesis and screening of biological activity of triazolothiadiazines and triazolothiadiazoles. J. Phytol. Res. 1998, 11, 201–202. [Google Scholar]
- Sakata, M.; Shirakawa, Y.; Kamata, N.; Hiroshino, Y.S.; Jie, O. Y. Synthesis and antibacterial activity of some new s-triazolo[3,4-b]-1,3,4- thiadiazine derivatives. J. Heterocycl. Chem. 2000, 37, 269–271. [Google Scholar] [CrossRef]
- Nadkarni, B. A.; Kamat, V. R.; Khadse, B. G. Synthesis and anthelmintic activity of 3,6-disubstituted -7H-s-triazolo[3,4-b][1,3,4] thiadiazines. Arzneim. Forsch. 2001, 51, 569–573. [Google Scholar]
- Holla, B. S.; Akberali, P. M.; Shivananda, M. K. Studies on nitrophenylfuran derivatives: part XII. synthesis, characterization, antibacterial and antiviral activities of some nitrophenyl-furfurylidene- 1,2,4-triazolo[3,4-b]-1,3,4-thiadiazines. Farmaco 2001, 56, 919–927. [Google Scholar] [CrossRef]
- Hui, X. P.; Zhang, Y.; Xu, P. F.; Wang, Q.; Zhang, Q.; Zhang, Z. Y. Syntheses and antibacterial activities of novel 3-substituted-6-(4-chlorophenyl)-7-(1H-1,2,4-triazole-1-yl)-1',2',4'-triazolo [3,4-b]-1",3",4"-thiadiazines. Chin. J. Org. Chem. 2005, 25, 700–704. [Google Scholar]
- Holla, B. S.; Rao, B. S.; Sarojini, B. K.; Akberali, P. M.; Kumari, N. S. Synthesis and studies on some new fluorine containing triazolothiadiazines as possible antibacterial, antifungal and anticancer agents. Eur. J. Med. Chem. 2006, 41, 657–663. [Google Scholar] [CrossRef]
- Holla, B. S.; Rao, B.S.; Shridhara, K.; Akberali, P. M. Studies on arylfuran derivatives Part XI. Synthesis, characterisation and biological studies on some Mannich bases carrying 2,4-dichloro- phenylfurfural moiety. Farmaco 2000, 55, 338–344. [Google Scholar] [CrossRef]
- Marina, K.; Anastssia, M.; Panagiotis, M.; Nicole, P.; Spyroula, P. G.; Christophe, P.; Myriam, W.; Erik, D. C. Synthesis and antiviral activity evaluation of some new 6-substituted 3-(1-adamantyl)- 1,2,4- triazolo[3,4-b][1,3,4] thiadiazoles. Farmaco 2002, 57, 253–257. [Google Scholar] [CrossRef]
- Jin, J. Y.; Zhang, L. X.; Chen, X. X.; Zhang, A. J.; Zhang, H. L. Syntheses and biological activities of 6-aryl-3-(3-hydroxypropyl)-7H-1,2,4-triazolo[3,4-b][1,3,4]thiadiazines. Molecules 2007, 12, 297–303. [Google Scholar] [CrossRef]
- Sun, X. H.; Liu, Y. F. Studies on synthesis of 1,3-diaminothiourea. Huaxue Tongbao 1999, 62, 43–48. [Google Scholar]
- Awad, L. F.; El Asir, E. S. Synthesis and conformational analysis of seco C- nucleosides and their diseco double-headed analogues of the 1,2,4-triazole, 1,2,4-triazolo[3,4-b]1,3,4-thiadiazole. Carbohyd. Res. 1998, 312, 9–22. [Google Scholar] [CrossRef]
- Shi, H. J.; Wang, Z. Y.; Shi, H. X. Study on the intramolecular Mannich reaction of 3-aryl-4-amino-5-mercapto-1, 2, 4-triazoles. Synth. Commun. 1998, 28, 3937–3941. [Google Scholar]
- Zhou, S. N.; Zhang, L. X.; Jin, J. Y.; Zhang, A. J.; Lei, X. X.; Lin, J. S.; He, J. W.; Zhang, H. L. Synthesis and biological activities of some novel triazolothiadiazines and Schiff bases derived from 1, 2, 4-triazole. Phosphorus, Sulfur Silicon Relat. Elem. 2007, 182, 419–432. [Google Scholar] [CrossRef]
- Wu, T. X.; Li, Zh. J.; Zhao, J. C. A facile method for the synthesis of 4-amino- 5-hydrocarbon-2,4-dihydro-3H-1,2,4-triazole-3-thione Schiff bases. Chem. J. Chin. Univ. 1998, 19, 1617–1619. [Google Scholar]
- Liu, F. M.; Wang, B. L. Synthesis of trifluoromethyl-substituted 1,2,4-triazole Mannich bases. Chin. J. Org. Chem. 2000, 20, 738–742. [Google Scholar]
- Zhang, X.; Tan, Zhang. L.; Xiao, M. Synthesis and fungicidal activity of Schiff base of triazole and thiophene. Chin. J. Pest. Sci. 2005, 7, 353–356. [Google Scholar]
- Zhang, A. J.; Zhang, L. X.; Ding, J. Ch. Synthesis and NMR Study of 4-(Arylmethylidene)-amino- 5-aryl-3-mercapto-4H-1,2,4-triazoles. Chin. J. Org. Chem. 2003, 25, 442–444. [Google Scholar]
- Zhang, L. X.; Zhang, A. J.; Lei, X. X.; Zou, K. H.; Ng, S. W. 4-Amino-3-(1,2,3,4,5-penta- hydroxypentyl)-1,2,4-1H-triazole-5(4H)-thione. Acta Cryst. 2004, E60, o613–o615. [Google Scholar]
- Zou, K. H.; Zhang, L. X.; Zhou, S. N.; Zhang, A. J.; Jin, J. Y.; Yin, P. Syntheses and crystal structures of 4-amino-3-(3-hydroxypropyl)-1H-1,2,4-triazole-5(4H)-thione and 6-(4-biphenylyl)- 3-(3-hydroxypropyl)-7H-1,2,4-triazolo [3,4-b] [1,3,4]thiadiazine. Chin. J. Struct. Chem. 2006, 25, 1517–1523. [Google Scholar]
- Zhang, A. J.; Zhang, L. X.; Xiong, Y.; Xu, D. J.; Li, X. J. Studies on synthesis and chromatography of 6-aryl-3-(D-glucopentitol-1-yl)-7H-1,2,4-triazolo[3,4-b][1,3,4]thiadiazines. Chin. J. Org. Chem. 2003, 23, 456–460. [Google Scholar]
- Sample Availability: Available form the authors.
© 2007 by MDPI (www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Jin, J.-y.; Zhang, L.-x.; Zhang, A.-j.; Lei, X.-X.; Zhu, J.-H. Synthesis and Biological Activity of Some Novel Derivatives of 4-Amino-3-(D-galactopentitol-1-yl)-5-mercapto-1,2,4-triazole. Molecules 2007, 12, 1596-1605. https://doi.org/10.3390/12081596
Jin J-y, Zhang L-x, Zhang A-j, Lei X-X, Zhu J-H. Synthesis and Biological Activity of Some Novel Derivatives of 4-Amino-3-(D-galactopentitol-1-yl)-5-mercapto-1,2,4-triazole. Molecules. 2007; 12(8):1596-1605. https://doi.org/10.3390/12081596
Chicago/Turabian StyleJin, Jian-yu, Li-xue Zhang, An-jiang Zhang, Xin-Xiang Lei, and Jiang-Hai Zhu. 2007. "Synthesis and Biological Activity of Some Novel Derivatives of 4-Amino-3-(D-galactopentitol-1-yl)-5-mercapto-1,2,4-triazole" Molecules 12, no. 8: 1596-1605. https://doi.org/10.3390/12081596




