Synthesis
On the basis of these studies in the present work we report simple and mild solvent-free microwave mediated methodologies for the synthesis of pyrazoles derivatives from α,β-unsaturated carbonyl compounds.
Table 1.
Physical and chemical properties of products
a-
c obtained under microwave activation.
![Molecules 12 01482 i001]()
Table 1.
Physical and chemical properties of products a-c obtained under microwave activation. ![Molecules 12 01482 i001]()
| Compound | R | R’ | mp, °C a | bp, °C b (mmHg) | 1H-NMR data, δ/ppm |
|---|
| a | C6H5 | CH3 | 124-126* | - | 2.31 (3H, s, CH3C(3)), 6.35 (1H, s, C(4)H), 7.30-7.75 (5H, aromatic) |
| b | ![Molecules 12 01482 i002]() | CH3 | - | 165-170 (0.1) | 0.95 (6H, s, (CH3)2C), 1.42 (3H, s, CH3C=C), 2.31 (3H, s, CH3C(3)), 5.80 (1H, s, C(4)H) |
| c | C6H5 | C6H5 | 200-202** | - | 6.85 (1H, s, C(4)H), 7.32-7.74 (10H, aromatic) |
We investigated in detail three carbonyl substrates:
trans-4-phenyl-3-buten-2-one, β-ionone and
trans-chalcone. Compounds
a-
c (
Table 1) have been obtained under microwave activation following two methodologies (MW1 and MW2).
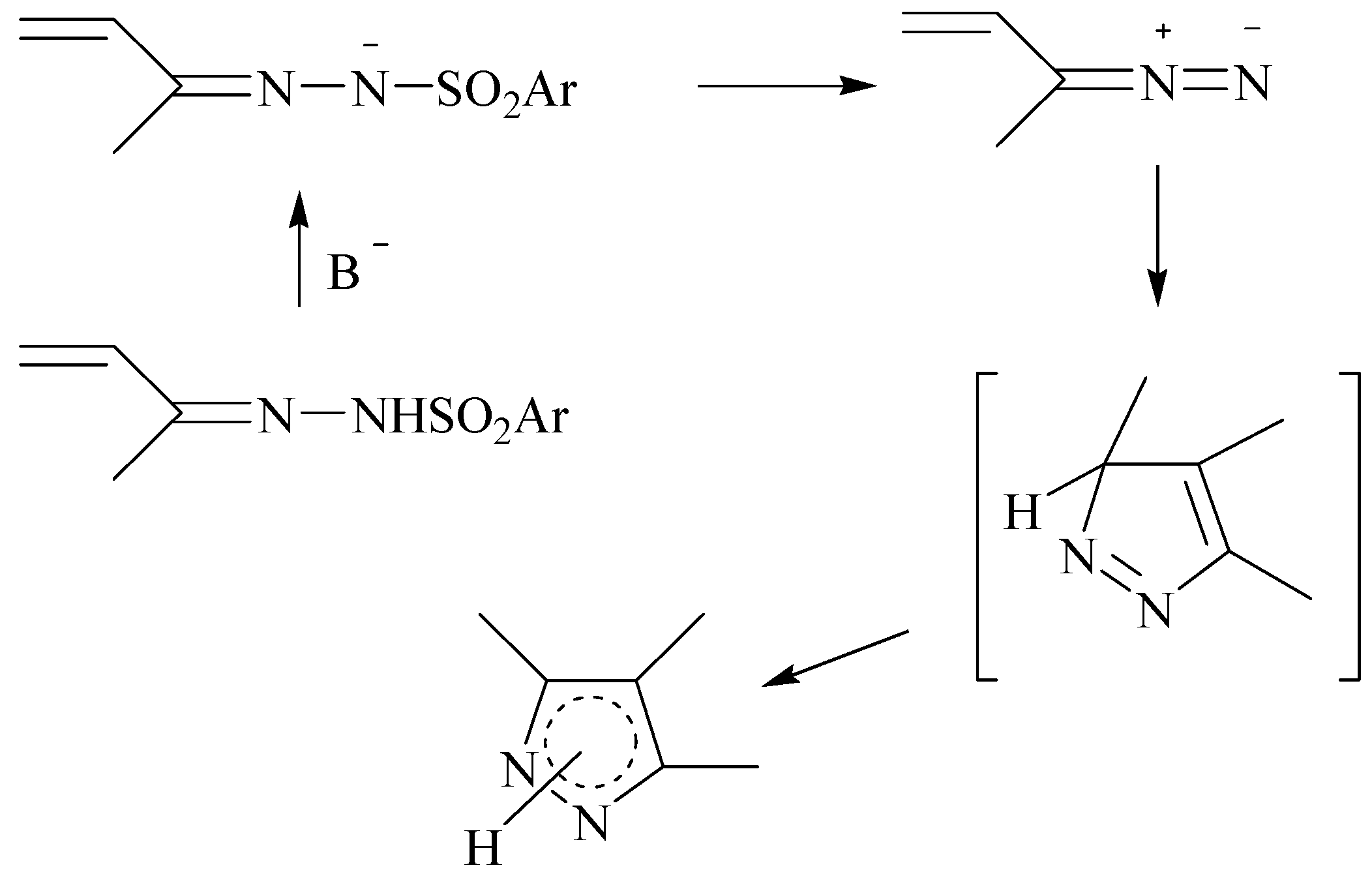
The first approach to synthesize the pyrazole derivatives (MW1) refers to the 1,3-cycloaddition of the corresponding tosylhydrazones of the carbonyl compounds selected, using a base in dry media (
Scheme 2). Tosylhydrazones were prepared following the classical procedure (ketone plus tosylhydrazine in methanol at reflux) [
12].
Scheme 2.
Reaction scheme for the synthesis of pyrazoles a-c from tosylhydrazones.
Scheme 2.
Reaction scheme for the synthesis of pyrazoles a-c from tosylhydrazones.
To fine-tune the method, and using the synthesis of compound
a from the tosylhydrazone of trans-4-phenyl-3-buten-2-one as a model reaction, several experiments were carried out under microwave activation using a base impregnated on mineral solid supports, a neat base in the reaction mixture, a base with an energy transfer agent such as
N,N-dimethylformamide or graphite. The most remarkable results are reported in
Table 2.
Table 2.
Operative conditions and relative yields for the synthesis of product
a from tosylhydrazone.
![Molecules 12 01482 i003]()
Table 2.
Operative conditions and relative yields for the synthesis of product a from tosylhydrazone. ![Molecules 12 01482 i003]()
| Entry | Method | Time, min. | Temp., °C | Yield, % |
|---|
| A | K2CO3 (8:1)* | 45 | 150 | n.r. |
| B | K2CO3/KOH (1/1:1)* | 45 | 145 | 84 |
| C | KF/Al2O3 (1/1:1)* | 10 | 145 | 65** |
| D | K2CO3 (4:1)* + ETI1 | 10 | 150 | 62** |
| E | K2CO3 (4:1)* + ETI2 | 1 | 130 | 95 |
| F | K2CO3 (2:1)* + ETI2 | 1 | 130 | 95 |
In general, the simple mixing of neat reagents gave better results than mineral solid supports in terms of reaction times and yields. Only the use of K
2CO
3 as a base, without any energy transfer improver, did not lead to any product. The addition of an energy transfer improver enhanced the reaction speed and yields and allowed us to reduce both the reaction temperature and amount of the base. The best performing conditions resulted from neat reagents in the presence of anhydrous K
2CO
3 (substrate/base 1:2) and a catalytic amount of
N,N-dimethylformamide (DMF) as an energy transfer improver [
13].
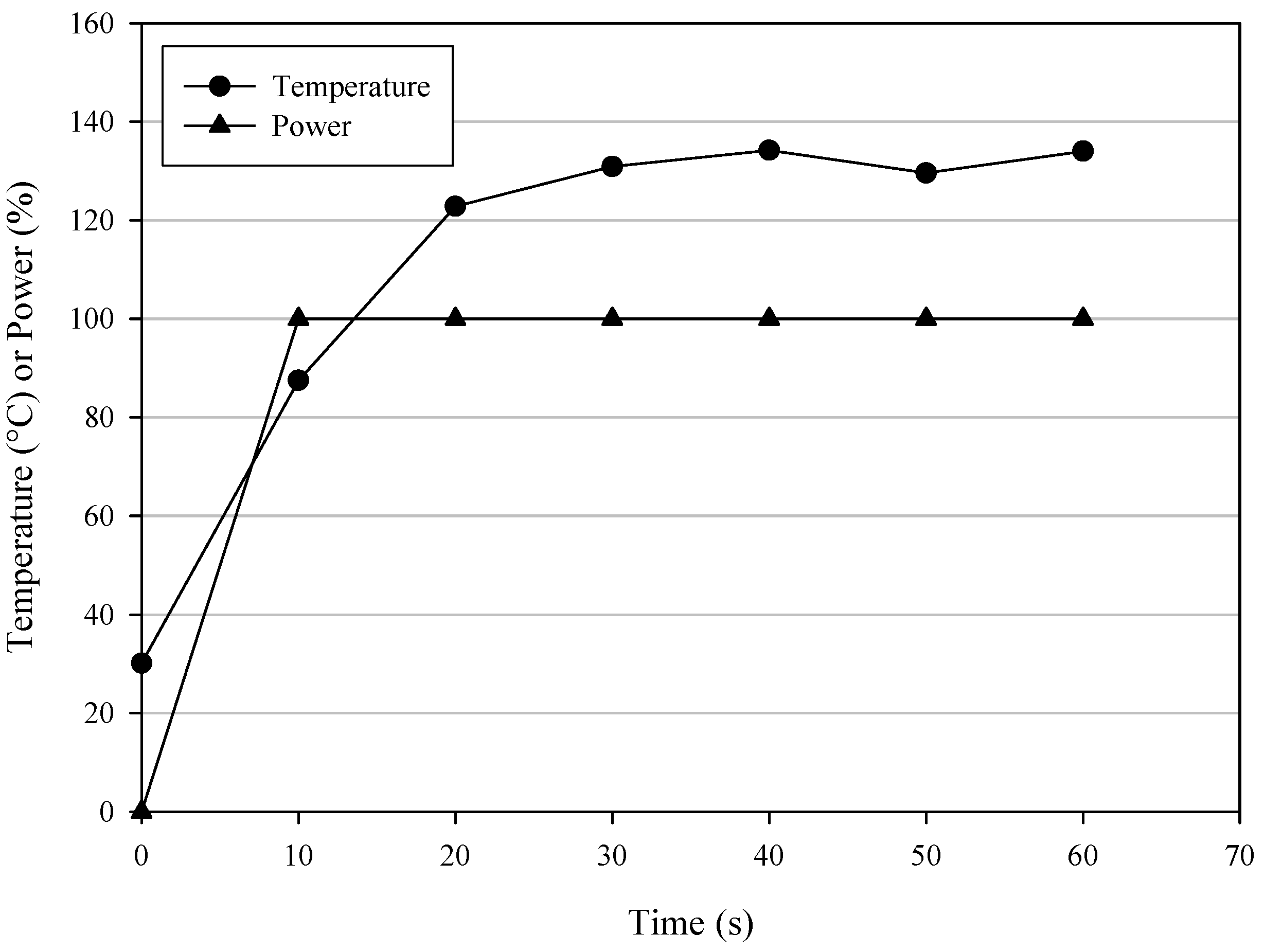
Figure 1 shows the temperature and power profiles corresponding to entry F in
Table 2 for the MW1 synthetic protocol of product
a. This method was then applied to the synthesis of all compounds. The results are reported in
Table 3.
Figure 1.
Graph of temperature and power %
vs time for the synthesis of pyrazole
a following MW1 synthetic approach under the entry F (
Table 2) operative conditions.
Figure 1.
Graph of temperature and power %
vs time for the synthesis of pyrazole
a following MW1 synthetic approach under the entry F (
Table 2) operative conditions.
In order to evaluate how microwave irradiation can affect cyclization, the best performing method (entry F) was also carried out under conventional heating (thermostated oil bath) under the same conditions in terms of reagents quantities and reaction vessel, in order to obtain compound a. Moreover, due to the slower temperature increase of conventional heating, reaction time was considered starting from when the temperature setpoint (130 °C) was reached. No product was detected after one minute of conventional treatment at 130 °C. When conventional heating was extended TLC monitoring of the oil bath reaction mixture revealed that the substrate started reacting only after 10 minutes, but then some degradation compounds were also detected.
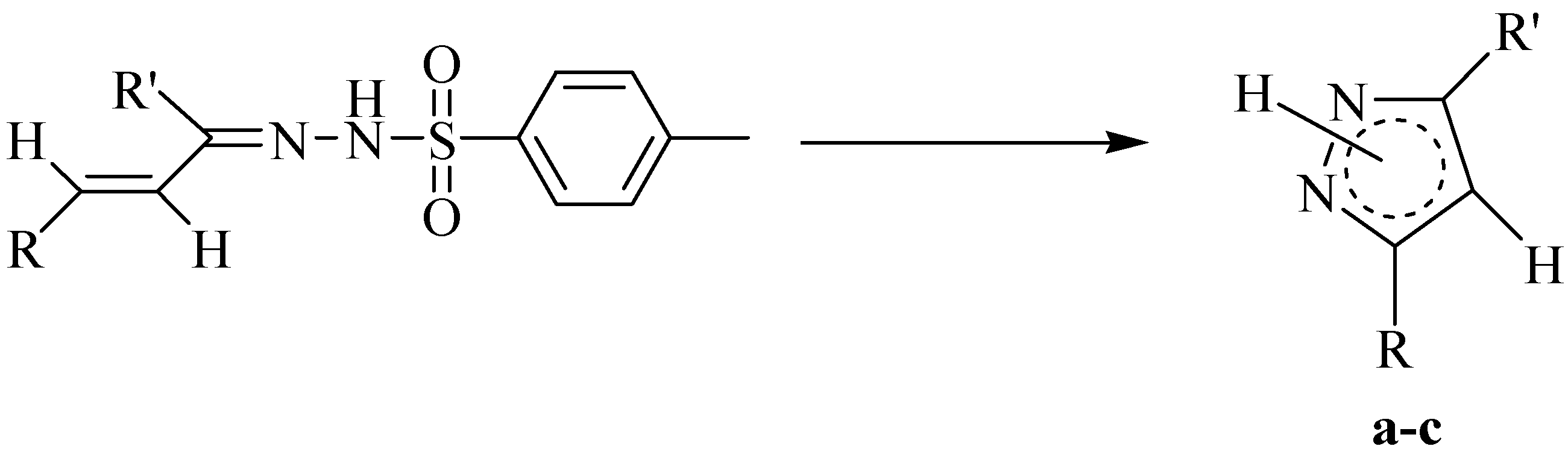
In the second phase of the work (MW2), as a further improvement and simplification, a “one-pot” approach to the synthesis of the desired heterocyclic products was investigated, removing the tosylhydrazone formation step. Hence, starting directly from the carbonyl precursors (
Scheme 3) and using the catalytic conditions which gave the best results in the previous approach (entry
F = K
2CO
3/substrate 2:1, 130 °C, DMF as energy transfer improver), the synthesis of the pyrazole derivatives
a-
c was studied at different reaction times, monitoring the product formation by TLC. The results obtained from this last synthetic pathway are reported in
Table 4. As an example,
Figure 2 shows the temperature and power profiles for MW2 synthetic protocol of product
a.
Table 3.
Solvent free microwave-mediated synthesis (MW1) of compounds
a-
c under the best catalytic conditions (entry
F –
Table 2) from tosylhydrazones.
Scheme 3.
Reaction scheme for the synthesis of pyrazoles a-c starting directly from carbonyl precursors.
Scheme 3.
Reaction scheme for the synthesis of pyrazoles a-c starting directly from carbonyl precursors.
Table 4.
MW-assisted “one pot synthesis” (MW2) of compounds a-c from carbonyl precursors.
Figure 2.
Graph of temperature and power %
vs time for the synthesis of pyrazole
a following MW2 synthetic approach under the entry F (
Table 2) operative conditions.
Figure 2.
Graph of temperature and power %
vs time for the synthesis of pyrazole
a following MW2 synthetic approach under the entry F (
Table 2) operative conditions.
With a simple mixing of the reagents (ketone, tosylhydrazide, K2CO3 and DMF) and with a maximum reaction time of five minutes all the compounds were obtained in good yields, very close to those obtained using the best catalytic conditions of MW1 (Yield % entry F: MW1/one pot: compound a = 95/95, compound b = 88/84, compound c = 65/65).
Again, in order to evaluate how microwave irradiation affected the cyclization, the “one pot” synthesis of compound a was also carried out under conventional heating (thermostated oil bath) under the same conditions in terms of reagents quantities and reaction vessel used. No product was detected after 3 minutes of treatment at 130 °C. The reaction in oil bath was also monitored for longer reaction times and after 10 minutes of treatment at 130 °C some degradation products were detected.
Modelling
The software Concerto 4.0 (Vector Fields, U.K.) has been used to numerically simulate the heating behaviour of the reactants mixture in the MW single mode applicator. The numerical simulation helped explaining the different yields and cleanness measured when using different reactants in the same experimental conditions.
The study of microwave heating of the reactants was conducted only for the MW-assisted one pot synthesis of products
a-
c (
Table 3). The use of the Concerto software allowed us to determine the SAR (Specific Absorption Rate, in W/kg), and thus to describe how heat is generated within the reaction vessel. The calculated SAR in the mid horizontal section of the reaction vessel is reported in the thermal plots shown in
Figure 3, as a function of compounds
a-
c.
Figure 3.
SAR in the mid section of the reaction vessel centrally positioned in the single mode applicator (300W forward power).
Figure 3.
SAR in the mid section of the reaction vessel centrally positioned in the single mode applicator (300W forward power).
The SAR distribution shows non-homogenous heating in the case of stationary load (power generation higher in the right side of the load), which, however, can be compensated by stirring. This distribution is due to the presence of the load inside the cavity, which is not equipped with plungers or movable short circuits to alter the electric field distribution. As a matter of fact, the load perturbs the empty cavity electric field distribution, having the maximum intensity region move away from the centre of the sample. This unwanted effect, on the other side, results useful in case of sample stirring (rotation), since it allows a much higher homogeneity of power generation with respect to a configuration in which the maximum of the electric field would be in the exact centre of the load. Modeling results show also that the solid reaction mixture for product a synthesis presents a 30% lower power generation in the examined section. However, it was experimentally observed that this particular sample (a) is the one that lead to the cleanest products and higher yield reaction. This behaviour could be ascribed to the easier and more accurate temperature control of the reaction in the case of lower power generation by the reactants (temperature gradients are reduced). On the other hand, in this case the overall process energy efficiency is lower, suggesting that an improved stirring and homogenisation could lead to higher yields for products b and c as well provided the synthesis conditions can be effectively controlled, minimising localised over- or under- heating.
Green metrics evaluation
A comparative quantitative assessment of the “greenness” of the new processes here proposed, in comparison to the classical one [
10], was conducted with a rational approach of the chemical sustainability of a reaction (“Green metrics”). For this the “Environmental Assessment Tool for Organic Syntheses” software (EATOS) was used [
14,
15]. This software, which takes into account the mass economy as well as the environmental and human health impact of the materials involved, has been applied to assess the eco-sustainability of the competing processes. EATOS elaborates all data relative to all materials used in the process (reagents, solvents, auxiliary materials, products and secondary products) and supplies an evaluation histogram. It is also possible to compare two or more chemical processes placing side by side their histograms and standardizing the quantities. As an example, in
Figure 4 the histograms related to the environmental impact of the synthesis of compound
a using both the conventional route in methanol [
10], and the newly proposed microwave approaches are reported. As the order of magnitude of the environmental impact of the classic procedure is much higher, the green metrics relative to MW1 and MW2 processes are also shown in detail in
Figure 5.
Figure 4.
Evaluation histograms of classic versus MW-mediated solvent-free approaches, MW1 and MW2 (one pot), for the synthesis of compound a. S-1= inverse selectivity (or mass index). E = environmental factor. EIin and EIout represent respectively S-1 and E weighted in function of the “weighting factors”: costs, risk, human toxicity, eco- toxicology. PEI represents Possible Environmental Impact.
Figure 4.
Evaluation histograms of classic versus MW-mediated solvent-free approaches, MW1 and MW2 (one pot), for the synthesis of compound a. S-1= inverse selectivity (or mass index). E = environmental factor. EIin and EIout represent respectively S-1 and E weighted in function of the “weighting factors”: costs, risk, human toxicity, eco- toxicology. PEI represents Possible Environmental Impact.
Figure 5.
Evaluation histograms of MW mediated solvent-free approaches, MW1 and MW2 (one pot), for the synthesis of compound a. S-1= inverse selectivity (or mass index). E = environmental factor. EIin and EIout represent respectively S-1 and E weighted in function of the “weighting factors” (costs, risk, human toxicity, ecotoxicology). PEI represents Possible Environmental Impact.
Figure 5.
Evaluation histograms of MW mediated solvent-free approaches, MW1 and MW2 (one pot), for the synthesis of compound a. S-1= inverse selectivity (or mass index). E = environmental factor. EIin and EIout represent respectively S-1 and E weighted in function of the “weighting factors” (costs, risk, human toxicity, ecotoxicology). PEI represents Possible Environmental Impact.
The graphical results shown in
Figure 4 suggest that the performance indicators of our innovative methods are considerably improved from a green chemistry point of view. A further confirmation can be given by the rapidity of the process which is not considered by EATOS but which is important in a green chemistry procedure context in terms of reduction of energy consumption.