One Pot Synthesis of α-Aminophosphonates Containing Bromo and 3,4,5-Trimethoxybenzyl Groups under Solvent-free Conditions
Abstract
:Introduction
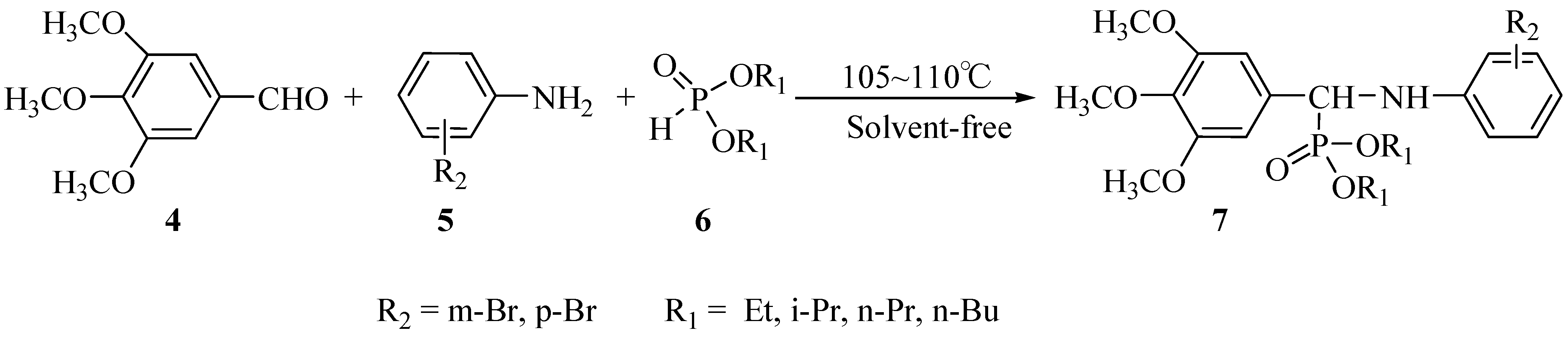
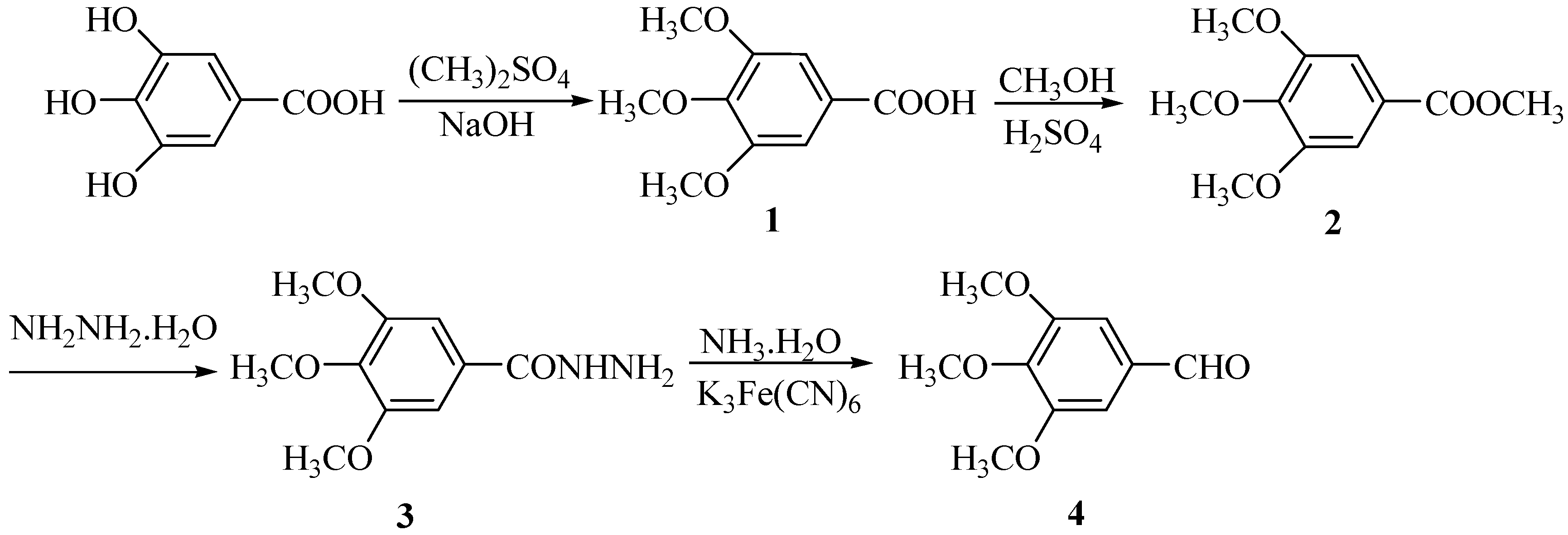
Results and Discussion
| Entry | Dialkyl phosphate: amine: aldehyde (molar ratio) | Reaction temperature (°C) | Reaction time (h) | Yield (%) |
|---|---|---|---|---|
| 1 | 1:1:1 | 85~90 | 2 | 37.5 |
| 2 | 1:1:1 | 95~100 | 2 | 61.2 |
| 3 | 1:1:1 | 105~110 | 2 | 65.6 |
| 4 | 1:1:1 | 115~120 | 2 | 56.3 |
| 5 | 1.2:1:1 | 105~110 | 2 | 61.8 |
| 6 | 2:1:1 | 105~110 | 2 | 60.3 |
| Entry | Dialkyl phosphate: amine: aldehyde (molar ratio) | Catalyst | Amount of catalyst (mol %) | Reaction time (min.) | Yield (%) |
|---|---|---|---|---|---|
| 1 | 1:1:1 | - | - | 120 | 65.6 |
| 2 | 1:1:1 | - | - | 30 | 41.0 |
| 3 | 1:1:1 | BF3·Et2O | 5 | 30 | 66.6 |
| 4 | 1:1:1 | BF3·Et2O | 10 | 30 | 76.0 |
| 5 | 1:1:1 | BF3·Et2O | 15 | 30 | 71.7 |
| Compounds | Substitutents | With Catalyst | Without Catalyst | |||
|---|---|---|---|---|---|---|
| R1 | R2 | Reaction time (min.) | Yield (%) | Reaction time (h) | Yield (%) | |
| 7a | Et | p-Br | 30 | 76.0 | 2 | 65.6 |
| 7b | Et | m-Br | 30 | 75.9 | 2 | 61.5 |
| 7c | i-Pr | p-Br | 30 | 82.0 | 2 | 67.8 |
| 7d | i-Pr | m-Br | 30 | 81.0 | 2 | 71.0 |
| 7e | n-Bu | p-Br | 30 | 76.1 | 2 | 62.5 |
| 7f | n-Bu | m-Br | 30 | 70.6 | 2 | 61.4 |
| 7g | n-Pr | p-Br | 30 | 83.5 | 2 | 74.2 |
| 7h | n-Pr | m-Br | 30 | 75.8 | 2 | 61.3 |
Crystal Structure Analysis of 7g.

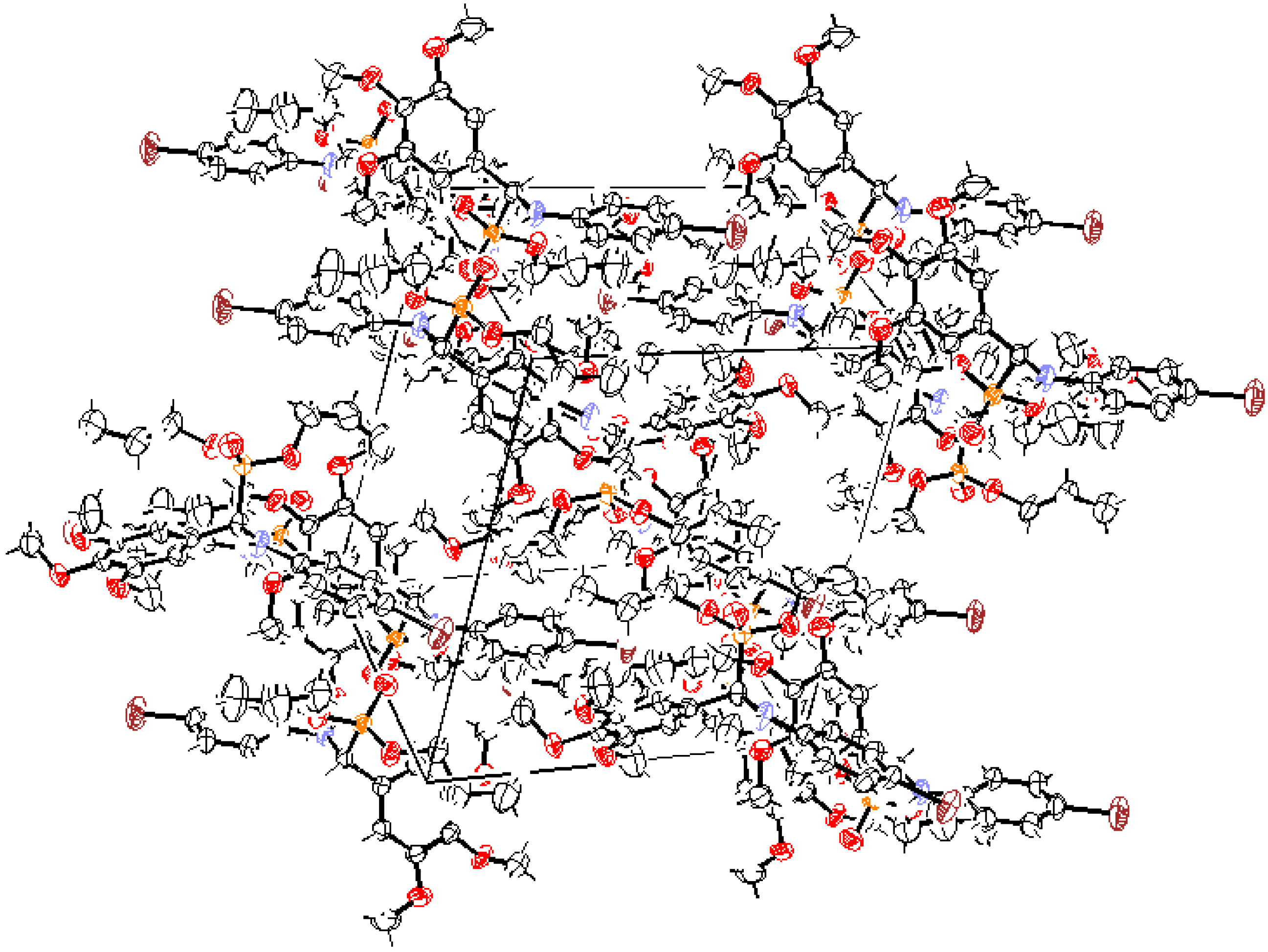
| D-H…A | d(D-H)/nm | d(H-A)/nm | d(D-A)/nm |  DHA(°) DHA(°) |
| N(1)-H(1)…O(4) | 0.860 | 2.118 | 2.960 | 165.94 |
Conclusions
Experimental
General
General procedure for the preparation of products 7a-h
Yields and physicochemical properties:
X-Ray diffraction experiment
Acknowledgments
References
- Kafarski, P.; Lejczak, B. Aminophosphonic acids of potential medical importance. Curr. Med. Chem. Anti-Cancer Agents 2001, 1, 301–312. [Google Scholar] [CrossRef]
- De Lombaert, S.; Blanchard, L.; Tan, J.; Sakane, Y.; Berry, C.; Ghai, R.D. Non-peptidic inhibitors of neutral endopeptidase 24.11 - 1. Discovery and optimization of potency. Bioorg. Med. Chem. Lett. 1995, 5, 145–150. [Google Scholar] [CrossRef]
- Kafarski, P.; Lejczak, B. Biological activity of aminophosphonic acids. Phosphorus Sulfur 1991, 63, 193–215. [Google Scholar] [CrossRef]
- Atherton, F. R.; Hassall, C. H.; Lambert, R. W. Synthesis and structure-activity relationships of antibacterial phosphono-peptides incorporating (1-aminoethyl) phosphonic acid and (amino- methyl)phosphonic acid. J. Med. Chem. 1986, 29, 29–40. [Google Scholar] [CrossRef]
- Zhang, M.-H.; Li, F.-G. Production Status Quo and Opportunity of Bromine-contained Pesticide (I). Fine Special. Chem. 2005, 13, 1–3. [Google Scholar]
- Sepulveda-Boza, S.; Walizei, G. H.; Rezende, M. C.; Vásquez, Y.; Mascayano, C.; Mejías, L. The preparation of new isoflavones. Synth. Commun. 2001, 31, 1933–1940. [Google Scholar] [CrossRef]
- Zhao, J.; Song, J.-Y.; Wang, Ch.-J.; Liu, L.; Sun, X.-Q. Synthesis of 3,4,5-Trimethoxy- benzoylsolanesylamide. Chin. J. Appl. Chem. 2003, 20, 95–97. [Google Scholar]
- Li, D.-J.; Ge, Zh.-H. Synthesis and Characterization of 3,4,5-Trimethoxylbenzylformyl- hydrazones. Chin. J. Syn. Chem. 2004, 12, 487–490. [Google Scholar]
- Ji, Y.-F.; Zong, Zh.-M.; Wei, X.-Y. Synthesis of 3, 4, 5- Trimethoxybenzaldehyde. Chin. J. Appl. Chem. 2001, 18, 581–583. [Google Scholar]
- Fields, E. K. The Synthesis of Esters of Substituted Amino Phosphonic Acids. J. Am. Chem. Soc. 1952, 74, 1528–1531. [Google Scholar] [CrossRef]Cherkasov, R. A.; Galkin, V. I. The Kabachnik-Fields reaction: synthetic potential and the problem of the mechanism. Russ. Chem. Rev. 1998, 67, 857–882. [Google Scholar]
- Mauthner, F.; Clarke, H.T. Org. Synth. Coll.; Gilman, H., Ed.; Wiley: New Work, 1941; Vol. 1, p. 537. [Google Scholar]
- Labouta, I. M.; Hassan, A.M.; Aboulwafa, O. M.; Kader, O. Synthesis of Some Substituted Benzimidazoles with Potential Antimicrobial Activity. Monatsh. Chem. 1989, 120, 571–574. [Google Scholar] [CrossRef]
- Xu, K.-X. The Manual of Fine Chemical Raw Materials and Organic Intermediates [M]; Chemical Industrial Press of China: Beijing, P.R. China, 1998. (in Chinese) [Google Scholar]
- Huang, R.-Q.; Wang, H.-L.; Zhou, J. The Preparation of Organic Intermediates; Chemical Industrial Press of China: Beijing, P.R. China, 2001. (in Chinese) [Google Scholar]
- Sheldrick, G. M. SHELXTL V5.1, Software Reference Manual. Bruker AXS, Inc.: Madison, Wisconsin, USA, 1997. [Google Scholar]
- Wilson, A. J. International Table for X-ray Crystallography; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992; Vol. C, Tables 6.1.1.4 (pp. 500-502) and 4.2.6.8 (pp. 219-222), respectively. [Google Scholar]
- Li, S. Z.; Wang, D. M.; Jiao, S. M. Pesticide Experiment Methods-Fungicide Sector; Agriculture Press of China: Beijing, P.R. China, 1991; pp. 93–94. [Google Scholar]
- Sample Availability: Samples of the compounds are available from authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Li, C.; Song, B.; Yan, K.; Xu, G.; Hu, D.; Yang, S.; Jin, L.; Xue, W.; Lu, P. One Pot Synthesis of α-Aminophosphonates Containing Bromo and 3,4,5-Trimethoxybenzyl Groups under Solvent-free Conditions. Molecules 2007, 12, 163-172. https://doi.org/10.3390/12020163
Li C, Song B, Yan K, Xu G, Hu D, Yang S, Jin L, Xue W, Lu P. One Pot Synthesis of α-Aminophosphonates Containing Bromo and 3,4,5-Trimethoxybenzyl Groups under Solvent-free Conditions. Molecules. 2007; 12(2):163-172. https://doi.org/10.3390/12020163
Chicago/Turabian StyleLi, Caihong, Baoan Song, Kai Yan, Gangfang Xu, Deyu Hu, Song Yang, Linhong Jin, Wei Xue, and Ping Lu. 2007. "One Pot Synthesis of α-Aminophosphonates Containing Bromo and 3,4,5-Trimethoxybenzyl Groups under Solvent-free Conditions" Molecules 12, no. 2: 163-172. https://doi.org/10.3390/12020163






