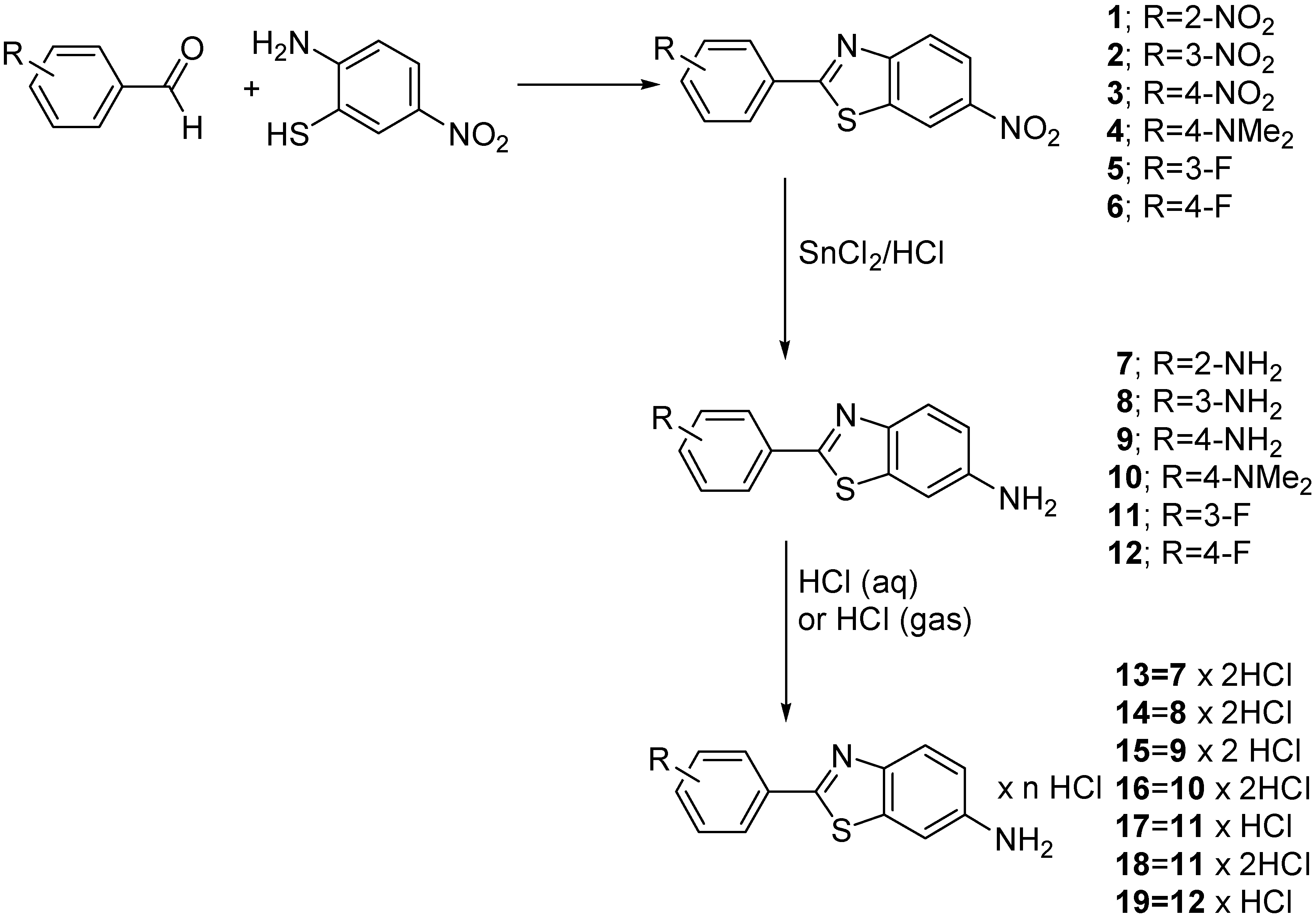
Experimental
General
Melting points were determined on a Kofler hot stage microscope and are uncorrected. IR spectra were recorded on a Nicolet Magna 760 spectrophotometer in KBr discs.
1H- and
13C-NMR spectra were recorded on either a Varian Gemini 300 or a Brucker Avance DPX 300 spectrometer. Chemical shifts (δ) are reported in parts per million (ppm) relative to tetramethylsilane (TMS) as an internal standard, and coupling constants (
J) are given in hertz (Hz). Elemental analyses were carried out in the Microanalitical laboratory at the Rugjer Boskovic Institute. All compounds were routinely checked by TLC with Merck silica gel 60F-254 glass plates. 2-Amino-5-nitrothiophenole was prepared according to the procedure described for preparation of 2-amino-5-cyanothiophenole [
9] from 6-nitro-benzothiazole.
Synthesis of 6-nitro-2-(substituted-phenyl)benzothiazoles 1-6
Procedure A: To a boiling solution of the appropriate substituted benzaldehyde (0.02 mol) in pyridine (20 mL), a solution of 2-amino-5-nitrothiophenole (0.02 mol) in pyridine (20 mL) was added dropwise, and the stirred reaction mixture was refluxed for 20 h. The mixture was then poured into 300 ml of 2 M hydrochloric acid and after cooling overnight the obtained crystalline product was filtered off and crystallized from appropriate solvent. Using this procedure the following compounds were prepared.
6-Nitro-2-(2-nitrophenyl)benzothiazole (1). Yield: 40.0% after crystallisation from xylene; mp 164-165°C; IR cm-1: 1569, 1517, 1348, 842, 756, 749, 721; 1H-NMR (DMSO-d6) δ: 9.33 (d, 1H, J = 2.0 Hz), 8.40 (dd, 1H, J = 2.0 Hz, J = 8.9 Hz), 8.26 (d, 1H, J = 8.9 Hz), 8.16 (d, 1H, J = 8.3 Hz), 8.06 (d, 1H, J = 8.1 Hz), 7.96-7.87 (m, 2H); Anal. Calcd. for C13H7N3O4S: C, 51.83; H, 2.34; N, 13.95; Found: C, 51.68; H, 2.40; N, 14.03.
6-Nitro-2-(3-nitrophenyl)benzothiazole (2). Yield: 70.9% (from xylene); mp 214-215 °C; IR cm-1: 1601, 1538, 1514, 1340, 829, 752, 739, 720; Anal. Calcd. for C13H7N3O4S: C, 51.83; H, 2.34; N, 13.95; Found: C, 51.99; H, 2.11; N, 13.90.
6-Nitro-2-(4-nitrophenyl)benzothiazole (3) Yield: 67.1% (from xylene); mp 256-257 °C; IR cm-1: 1606, 1517, 1346, 854, 754, 719; Anal. Calcd. for C13H7N3O4S: C, 51.83; H, 2.34; N, 13.95; Found: C, 51.92; H, 2.29; N, 13.88.
6-Nitro-2-(4-N,N-dimethylaminophenyl)benzothiazole (4). Yield: 66.1% (from xylene); mp 262-263 °C; IR cm-1: 1610, 1594, 1474, 1434, 1330, 810, 752; 1H-NMR (DMSO-d6) δ: 9.09 (s, 1H), 8.30 (d, 1H, J = 9.0 Hz), 8.05 (d, 1H, J = 9.0 Hz), 7.96 (d, 2H, J = 8.8 Hz), 6.85 (d, 2H, J = 8.8 Hz), 3.05 (s, 12H); Anal. Calcd for C15H13N3O2S: C, 60.18; H, 4.38; N, 14.04; Found: C, 60.41; H, 4.44; N, 14.30.
6-Nitro-2-(3-fluorophenyl)benzothiazole (5). Yield: 45.7% (from toluene); mp 199-200 °C; IR cm-1: 1588, 1515, 1448, 1343, 809, 785, 753; 1H-NMR (DMSO-d6) δ: 9.24 (d, 1H, J = 2.1 Hz), 8.35 (dd, 1H, J = 8.9 Hz, J = 2.3 Hz), 8.24 (d, 1H, J = 8.9 Hz), 7.99-7.93 (m, 2H), 7.65 (m, 1H), 7.49 (m, 1H); Anal. Calcd. for C13H7FN2O2S: C, 56.93; H, 2.57; N, 10.21; Found: C, 57.20; H, 2.63; N, 10.00.
6-Nitro-2-(4-fluorophenyl)benzothiazole (6). Yield: 58.2% (from xylene); mp 183-185 °C; IR cm-1: 1592, 1510, 1474, 1334, 839, 751; 1H-NMR (DMSO-d6) δ: 9.23 (d, 1H, J = 2.4 Hz), 8.36 (dd, 1H, J = 8.9 Hz, J = 2.4 Hz), 8.25-8.20 (m, 3H), 7.46 (dd, 2H, J = 8.8 Hz, J = 10.8 Hz); 13C-NMR (DMSO-d6) δ: 172.6, 164.6 (d, J = 251 Hz), 157.4, 144.6, 135.4, 130.4 (d, J = 9.3 Hz, 2C), 129.0, 123.3, 122.1, 119.7, 116.8 (d, J = 22.4 Hz, 2C); Anal. Calcd for C13H7FN2O2S: C, 56.93; H, 2.57; N, 10.21; Found: C, 56.74; H, 2.65; N, 10.02.
Synthesis of 6-amino-2-(substituted-phenyl) benzothiazoles 7-12
Procedure B: A solution of tin(II) chloride dihydrate (9 g, 0.04 mol), concd. HCl (18 mL) and methanol (18 mL) was added to the appropriate 6-nitro-2-(nitrophenyl)benzothiazole 1-3 (0.005 mol), or 0.01 mol of the compounds 4-6. The mixture was stirred and refluxed for 15 min. Methanol was removed by vacuum evaporation and the residue was dissolved in water. After basification to pH > 9 with 20% NaOH the product was extracted with ether and dried (MgSO4). The solvent was removed under reduced pressure and the white solid obtained was crystallised from the appropriate solvent. The following compounds were prepared by thus method:
6-Amino-2-(2-aminophenyl)benzothiazole (7). Yield: 74.5% (after crystallisation from toluene); mp 185-187 °C; IR cm-1: 3438, 3351, 3299, 1611, 1487, 817, 754; 1H-NMR (DMSO-d6) δ: 7.62 (d, 1H, J = 8.6 Hz), 7.47 (d, 1H, J = 7.6 Hz), 7.13 (s, 2H), 7.10 (d, 1H, J = 8.0 Hz), 7.04 (s, 1H), 6.79 (d, 1H, J = 8.3 Hz), 6.72 (m, 1H), 6.58 (m, 1H, J = 7.5 Hz), 5.42 (s, 2H); Anal. Calcd for C13H11N3S: C, 64.70; H, 4.59; N, 17.41; Found: C, 64.83; H, 4.63; N, 17.29.
6-Amino-2-(3-aminophenyl)benzothiazole (8). Yield: 68.2% (from xylene); mp 202-205 °C; IR cm-1: 3406, 3300, 3200, 1621, 1602, 1485, 815, 788; 1H-NMR (DMSO-d6) δ: 7.62 (d, 1H, J = 8.6 Hz), 7.19 (s, 1H), 7.13-7.02 (m, 3H), 6.74 (d, 1H, J = 8.6 Hz), 6.62 (d, 1H, J = 7.6 Hz), 5.44 (s, 2H), 5.35 (s, 2H); 13C-NMR (DMSO-d6) δ: 161.4, 149.2, 147.2, 145.1, 136.0, 134.0, 129.5, 122.9, 115.7, 114.9, 114.0, 111.3, 103.7; Anal. Calcd for C13H11N3S: C, 64.70; H, 4.59; N, 17.41; Found: C, 64.75; H, 4.42; N, 17.38.
6-Amino-2-(4-aminophenyl)benzothiazole (9). Yield: 68.8% (toluene); mp 263-265 °C; IR cm-1: 3456, 3365, 3304, 3193, 1621, 1606, 1488, 822; 1H-NMR (DMSO-d6) δ: 7.74 (d, 2H, J = 8.3 Hz), 7.67 (d, 1H, J = 8.6 Hz), 7.14 (s, 1H), 6.83 (d, 2H, J = 8.3 Hz), 5.84 (s, 2H), 5.43 (s, 2H); 13C-NMR (DMSO-d6) δ: 161.6, 151.0, 146.4, 145.4, 135.3, 127.8 (2C), 122.0, 120.9, 114.4, 113.5 (2C), 104.0; Anal. Calcd for C13H11N3S: C, 64.70; H, 4.59; N, 17.41; Found: C, 64.58; H, 4.50; N, 17.62.
6-Amino-2-(4-N,N-dimethylaminophenyl)benzothiazole (10). Yield: 68.7% (from xylene); mp 230-233°C; IR cm-1: 3459, 3306, 3188, 1610, 1489, 813; 1H-NMR (DMSO-d6) δ: 7.78 (d, 2H, J = 8.9 Hz), 7.60 (d, 1H, J = 8.6 Hz), 7.07 (d, 1H, J = 2.1 Hz), 6.81 (d, 2H, J = 8.9 Hz), 6.75 (dd, 1H, J = 2.1 Hz, J = 8.6 Hz), 5.36 (s, 2H), 3.02 (s, 6H); 13C-NMR (DMSO-d6) δ: 161.4, 151.4, 146.5, 145.4, 135.4, 127.5 (2C), 122.2, 120.9, 114.5, 111.8 (2C), 104.0, 39.7 (2C); Anal. Calcd for C15H15N3S: C, 66.88; H, 5.61; N, 15.60; Found: C, 66.95; H, 5.72; N, 15.52.
6-Amino-2-(3-fluorophenyl)benzothiazole (11). Yield: 74.6% (toluene-petroleum ether, 1:1, v/v); mp 127-128 °C; IR cm-1: 3408, 3312, 3218, 1602, 1442, 852, 827, 793; 1H-NMR (DMSO-d6) δ: 7.76-7.68 (m, 3H), 7.54 (m, 1H), 7.31 (m, 1H), 7.08 (s, 1H), 6.79 (d, 1H, J = 8.6 Hz), 5.57 (s, 2H); 13C-NMR (DMSO-d6) δ: 162.4 (d, J = 244 Hz), 158.7, 147.8, 144.9, 136.5, 135.6 (d, J = 8.2 Hz), 131.3 (d, J = 8.4 Hz), 123.4, 122.6 (d, J = 2.6 Hz), 116.8 (d, J = 21.0 Hz), 115.3, 112.5 (d, J = 23.0 Hz), 103.5. Anal. Calcd for C13H9FN2S: C, 63.92; H, 3.71; N, 11.47; Found: C, 63.73; H, 3.80; N, 11.52.
6-Amino-2-(4-fluorophenyl)benzothiazole (12). Yield: 79.3% (from toluene-petroleum ether, 1:1, v/v); mp 156-157 °C; IR cm-1: 3407, 3314, 3218, 1606, 1487, 839, 827, 801; 1H-NMR (DMSO-d6) δ: 7.97 (dd, 2H, J = 8.6 Hz, J = 5.3 Hz), 7.66 (d, 1H, J = 8.6 Hz), 7.33 (dd, 2H, J = 8.6 Hz, J = 10.3 Hz), 7.07 (s, 1H), 6.77 (d, 1H, J = 8.6 Hz), 5.50 (s, 2H); 13C-NMR (DMSO-d6) δ: 163.2 (d, J = 247 Hz), 159.3, 147.6 145.2, 136.5, 130.2, 128.7 (d, J = 7.7 Hz), 123.3, 116.3 (d, J = 21.8 Hz), 115.3, 103.8; Anal. Calcd for C13H9FN2S: C, 63.92; H, 3.71; N, 11.47; Found: C, 63.78; H, 3.85; N, 11.55.
Synthesis of amine hydrochloride salts 13-19.
Procedure C: The amines 7-12 (0.005 mol) were dissolved in a warm mixture of ethanol and xylene (1:1, v/v) followed by the addition of concd. HCl (1.0 mL). The contents were stirred in a stoppered flask for 4 h. The precipitated pure crystals were collected by filtration, washed with dry ether, and dried in vacuo. The following compounds were prepared by this method:
6-Amino-2-(2-aminophenyl)benzothiazole dihydrochloride (13). Yield: 96.6%; mp 224-227 °C; IR cm-1: 2816, 2662, 2524, 1486, 1463, 980, 767; 1H-NMR (DMSO-d6) δ: 8.11 (s, 1H), 8.07 (d, 1H, J = 8.6 Hz), 7.64 (d, 1H, J = 8.0 Hz), 7.49 (d, 1H, J = 8.6 Hz), 7.24 (dd, 1H, J = 8.0 Hz, J = 7.3 Hz), 6.29 (d, 1H, J = 8.3 Hz), 6.69 (dd, 1H, J = 7.7 Hz, J = 7.3), 4.78 (br s); 13C-NMR (DMSO-d6) δ: 169.6, 152.7, 144.2, 133.7, 132.3, 130.4, 129.2 (2C), 123.2, 122.3, 118.8, 117.1, 115.7; Anal. Calcd for C13H13Cl2N3S: C, 49.69; H, 4.18; N, 13.37; Cl, 22.56; Found: C, 49.55; H, 4.15; N, 13.37; Cl, 22.38.
6-Amino-2-(3-aminophenyl)benzothiazole dihydrochloride (14). Yield: 97.4%; mp 253-255 °C; IR cm-1: 2896, 2605, 1595, 1521, 1456, 818, 786; 1H-NMR (DMSO-d6) δ: 8.05 (d, 1H, J = 8.6 Hz), 7.97 (s, 1H), 7.88 (s, 1H), 7.80 (d, 1H, J = 7.7 Hz), 7.53 (dd, 1H, J = 8.0 Hz, J = 7.7 Hz), 7.40 (d, 1H, J = 8.8 Hz), 7.32 (d, 1H, J = 7.7 Hz); 13C-NMR (DMSO-d6) δ: 167.2, 152.4, 135.5, 134.3, 133.8, 131.0, 130.5, 126.1, 125.8, 124.0, 122.2, 121.1, 116.9; Anal. Calcd for C13H13Cl2N3S: C, 49.69; H, 4.18; N, 13.37; Cl, 22.56; Found: C, 49.70; H, 4.10; N, 13.35; Cl, 22.63.
6-Amino-2-(4-aminophenyl)benzothiazole dihydrochloride (15). Yield: 99.3%; mp 247-248 °C; IR cm-1: 2852, 2554, 1605, 1507, 1485, 974, 822; 1H-NMR (DMSO-d6) δ: 7.95-7.92 (m, 2H), 7.79 (d, 2H, J = 8.7 Hz), 7.36 (d, 1H, J = 8.3 Hz), 6.76 (d, 2H, J = 8.7 Hz), 4.41 (br s); 13C-NMR (DMSO-d6) δ: 168.5, 152.7, 143.5, 135.1 (2C), 129.4 (2C), 128.9, 126.1, 123.2, 122.0, 119.4, 116.9; Anal. Calcd for C13H13Cl2N3S: C, 49.69; H, 4.18; N, 13.37; Cl, 22.56; Found: C, 49.68; H, 4.06; N, 13.37; Cl, 22.64.
6-Amino-2-(4-N,N-dimethylaminophenyl)benzothiazole dihydrochloride (16). Yield: 99.5%; mp 240-242 °C; IR cm-1: 2796, 2568, 2453, 1607, 1519, 841; 1H-NMR (DMSO-d6) δ: 8.06 (s, 1H), 7.99 (d, 1H, J = 8.6 Hz), 7.89 (d, 2H, J = 8.9 Hz), 7.44 (d, 1H, J = 8.6 Hz), 6.85 (d, 2H, J = 8.9 Hz), 3.01 (s, 6H); 13C-NMR (DMSO-d6) δ: 169.8, 152.0, 150.2, 134.5, 128.9 (2C), 127.6, 122.6, 122.4, 121.6, 116.5, 114.7 (2C), 41.2 (2C); Anal. Calcd for C15H17Cl2N3S: C, 52.63; H, 5.01; N, 12.27; Cl, 20.71; Found: C, 52.60; H, 4.97; N, 12.23; Cl, 20.80.
6-Amino-2-(3-fluorophenyl)benzothiazole monohydrochloride (17). Yield: 96.1%; mp 206-207 °C; IR cm-1: 2806, 2589, 1585, 1514, 1265, 848, 787; 1H-NMR (DMSO-d6) δ: 8.06 (d, 1H, J = 8.6 Hz), 7.99 (s, 1H), 7.89-7.83 (m, 2H), 7.60 (dd, 1H, J = 7.3 Hz, J = 7.0 Hz), 7.44-7.38 (m, 2H), 3.95 (br s, 3H); 13C-NMR (DMSO-d6) δ: 166.1, 162.4 (d, J = 245 Hz), 151.5, 135.5, 134.7 (d, J = 8.2 Hz), 132.0, 131.6 (d, J = 8.3 Hz), 123.8, 123.4 (d, J = 2.6 Hz), 121.4, 118.2 (d, J = 21.0 Hz), 115.4, 113.4 (d, J = 22.4 Hz); Anal. Calcd for C13H10ClFN2S: C, 55.62; H, 3.60; N, 9.98; Cl, 12.63; Found: C, 55.43; H, 3.56; N, 9.79; Cl, 12.81.
6-Amino-2-(4-fluorophenyl)benzothiazole monohydrochloride (19). Yield: 91.3%; mp 245-247 °C; IR cm-1: 3058, 2854, 2586, 1602, 1488, 1237, 839, 816; 1H-NMR (DMSO-d6) δ: 8.13 (dd, 2H, J = 5.6 Hz, J = 8.3 Hz), 8.06 (d, 1H, J = 8.6 Hz), 7.97 (s, 1H), 7.45-7.37 (m, 3H), 3.63 (br s); 13C-NMR (DMSO-d6) δ: 166.7, 163.9 (d, J = 245 Hz), 151.8, 135.5, 131.0, 129.5 (d, J = 8.9 Hz, 2C), 129.1, 123.6, 121.1, 116.5 (d, J = 22.2 Hz, 2C), 115.3; Anal. Calcd for C13H10ClFN2S: C, 55.62; H, 3.60; N, 9.98; Cl, 12.63; Found: C, 55.45; H, 3.56; N, 9.87; Cl, 12.78.
Procedure D: The amines 7-12 (0.005 mol) were dissolved in a warm mixture of ethanol and xylene (1:1, v/v). A stream of dry hydrogen chloride was passed through the solution for 1 h while cooling to room temperature. The contents were then stirred in a stoppered flask for 2 h. The precipitated pure crystals were collected by filtration, washed with dry ether, and dried in vacuo. The following compound was prepared by this method:
6-Amino-2-(3-fluorophenyl)benzothiazole dihydrochloride (18). Yield: 97.2%; mp 209-210 °C; IR cm-1: 3045, 2798, 2590, 2541, 1588, 1486, 861, 795; 1H-NMR (DMSO-d6) δ: 8.17-8.14 (m, 2H), 7.94-7.88 (m, 2H), 7.64 (dd, 1H, J = 7.6 Hz, J = 6.3 Hz), 7.53 (d, 1H, J = 8.6 Hz), 7.46 (m, 1H), 4.45 (br s); 13C-NMR (DMSO-d6) δ: 167.2, 162.4 (d, J = 244 Hz), 152.4, 135.3, 134.6 (d, J = 8.2 Hz), 131.6 (d, J = 8.4 Hz), 129.7, 123.8, 123.6, 122.3, 118.4 (d, J = 20.9 Hz), 117.2, 113.5 (d, J = 21.4 Hz); Anal. Calcd for C13H11Cl2FN2S: C, 49.22; H, 3.50; N, 8.83; Cl, 22.35; Found: C, 49.45; H, 3.50; N, 8.98; Cl, 22.30.
Cell culture
Five different human cell lines were used for cytotoxicity screening: colon adenocarcinoma (CaCo-2), cervical adenocarcinoma (HeLa), laryngeal carcinoma (Hep-2), mammary adenocarcinoma (MCF7), lung fibroblast (WI-38). Cells were seeded in tissue culture flasks (Sarstedt, Numbrecht, Germany) in Dulbecco's Modified Eagle medium (DMEM) (Gibco, Paisley, Scotland) supplemented with 10% foetal bovine serum (FBS; Gibco), 2 mM glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin in a humidified atmosphere with 5% CO2 at 37 °C. The cultures were passaged at preconfluent densities with the use of 0.025% trypsin (Institute of Immunology, Zagreb, Croatia).
Cytotoxicity assays
For the experiments the cells were seeded onto 96-well microtiter plates at a density of 2 x 10
3 cells per well and allowed to adhere for 24 h before the benzothiazoles were added over a concentration range of 1-100 µM. Stock solutions of benzothiazoles were dissolved in DMSO (Sigma, Deisenhofen, Germany) at the concentration of 0.1 M and were diluted with medium without bovine calf serum. The DMSO concentration was adjusted to be the same in test and control plates (1%). All drug solutions were freshly prepared on the day of testing. After incubation with benzothiazoles for 72 h, cell growth was established according to the results of MTT assay [
10] - Sigma, Deisenhofen, Germany. Each experiment was made in quadruplicate and repeated three times. The absorbency was measured on a microplate reader at 570 nm. (Labsystems, Finland) The percentage of growth (PG) of the cell lines was calculated according to the following expression:
where mean Tz = the average of optical density measurements before exposure of cells to the test compound, mean Ti = the average of optical density measurements after the desired period of time, and mean C = the average of optical density measurements after the desired period of time with no exposure of cells to the test compound. The results are expressed as IC
50, which is the concentration necessary for 50% inhibition. The IC
50 values for each compound are calculated from dose response curves using linear regression analysis by fitting the test concentrations that give PG values above and below the reference value (i.e. 50%). Each result is a mean of IC
50 ± SD value from three separate experiments.