Synthesis and Cyclizations of N-(2,3-, 3,4- and 3,5-Dimethylphenyl)-β-alanines
Abstract
:Introduction
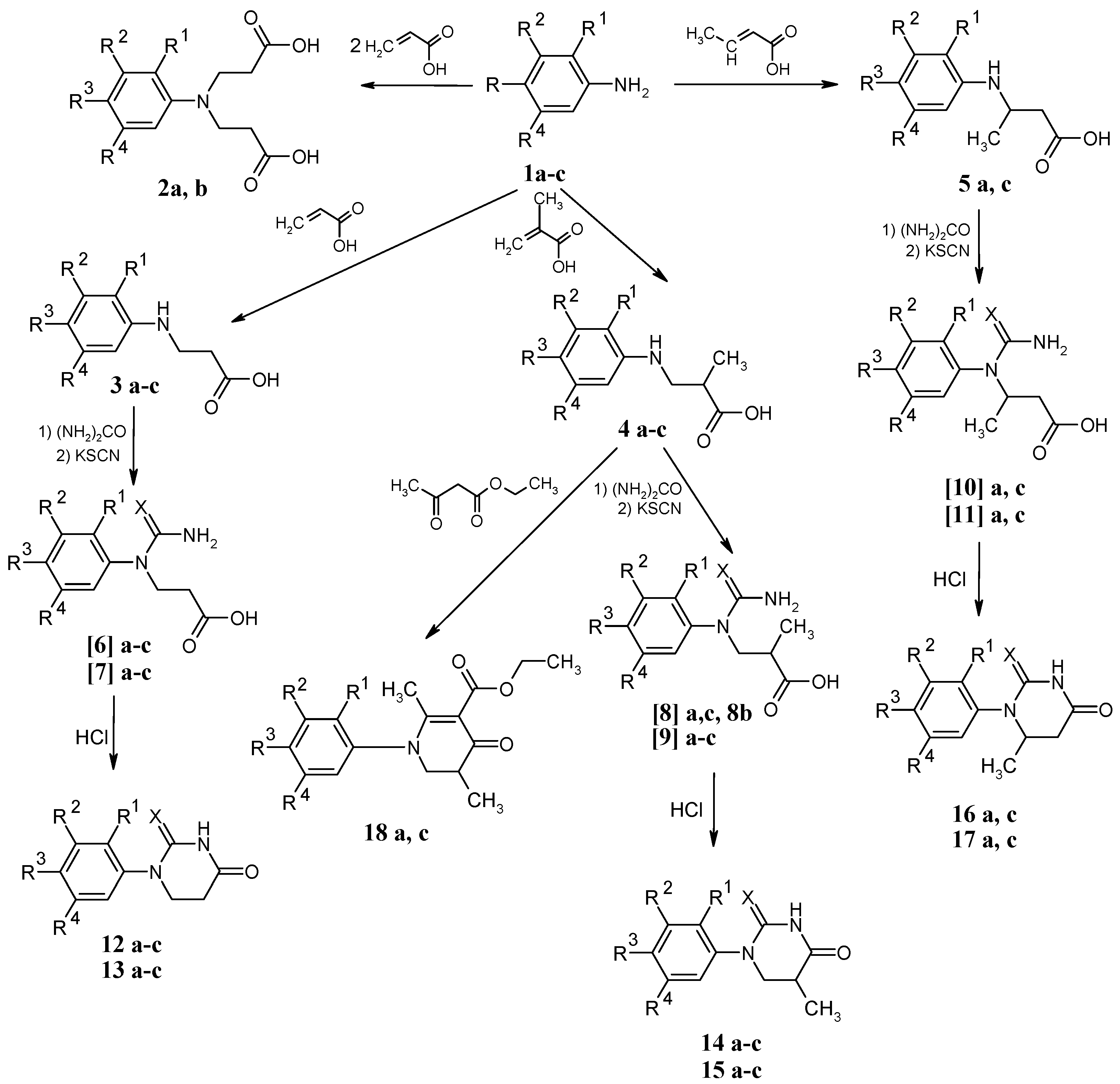
Results and Discussion
Conclusions
Experimental
General
| Compounds | 1H-NMR (solvent) | 13C-NMR (solvent) | IR (KBr tabl.) | |||
|---|---|---|---|---|---|---|
| 2a | (d6-DMSO) 2.35 (s, 3H, 2’-CH3Ar), 2.45 (s, 3H, 3’-CH3Ar), 2.30-2.65 (m, 4H, CH2CO), 3.65-3.95 (m, 4H, CH2NCH2), 7.30-7.90 (m, 3H, Ar), 11.14 (bs, 2H, COOH). | – | – | |||
| 2b | (d6-DMSO) 2.30 (s, 6H, 3’,4’-CH3Ar), 2.44-2.80 (m, 4H, CH2CO), 3.66-3.98 (m, 4H, CH2NCH2), 7.40-7.70 (m, 3H, Ar), 10.60 (bs, 2H, COOH). | – | – | |||
| 3a | (d6-acetone) 1.98 (s, 3H, 2’-CH3Ar), 2.18 (s, 3H, 3’-CH3Ar), 2.50-2.72 (m, 2H, CH2CO), 3.32-3.51 (m, 2H, NCH2), 6.40-7.10 (m, 3H, Ar). | – | – | |||
| 3b | (d6-DMSO) δ: 2.29 (s, 6H, 3’,4’-CH3Ar), 2.74-3.45 (m, 2H, CH2CO), 3.40-3.62 (m, 2H, NCH2), 7.45-7.60 (m, 3H, Ar), 9.66 (bs, 1H, COOH). | – | – | |||
| 3c | (d6-DMSO) 2.30 (s, 6H, 3’,5’-CH3Ar), 2.70-2.95 (m, 2H, CH2CO), 3.34-3.60 (m, 2H, NCH2), 7.02-7.25 (m, 3H, Ar), 10.25 (bs, 1H, COOH). | – | – | |||
| 4a | (d6-acetone) 1.24 (d, J = 6.9 Hz, 3H, CH CH3), 2.02 (s, 3H, 2’-CH3Ar), 2.21 (s, 3H, 3’-CH3Ar), ), 2.65-3.06 (m, 1H, CH), 3.13-3.60 (m, 2H, NCH2), 6.50-7.10 (m, 3H, Ar). | – | – | |||
| 4b | (d6-DMSO) 1.29 (d, J = 6.9 Hz, 3H, CH CH3), 2.28 (s, 6H, 3’,4’-CH3Ar), 2.75-3.10 (m, 1H, CH), 3.16-3.65 (m, 2H, NCH2), 7.25-7.50 (m, 3H, Ar), 10.66 (bs, 1H, COOH). | – | – | |||
| 4c | (CDCl3) 1.25 (d, J = 7.02 Hz, 3H, CHCH3), 2.23 (s, 6H, 3’,5’-CH3Ar), 2.74-2.90 (m, 1H, CH), 3.14-3.27 (m, 2H, CH2N), 6.25-6.45 (m, 3H, Ar), 7.5 (bs, 1H, COOH). | (CDCl3) 14.84 (CHCH3), 21.47 (3’,5’-CH3Ar), 39.23 (CH), 47.09 (CH2), 111.39 (Ar-C-2’,6’), 120.22 (Ar-C-4’), 139.01 (Ar-C-3’,5’), 147.44 (Ar-C-1’), 181.17 (CO). | – | |||
| 5a | (d6-DMSO) 1.36 (d, J = 6.5 Hz, 3H, CH CH3), 2.34 (s, 3H, 2’-CH3Ar), 2.37 (s, 3H, 3’-CH3Ar), 2.60-3.25 (m, 2H, CH2CO), 3.65-4.15 (m, H, CH), 7.45-7.65 (m, 3H, Ar), 10.67 (bs, 1H, COOH). | – | – | |||
| 5c | (d6-DMSO) 1.35 (d, J = 6.6 Hz, 3H, CH CH3), 2.36 (s, 6H, 3’,5’-CH3Ar), 2.50-3.15 (m, 2H, CH2CO), 3.65-4.10 (m, 1H, CH), 7.02-7.32 (m, 3H, Ar), 10.70 (bs, 1H, COOH). | – | – | |||
| 8b | (d6-DMSO) 1.01 (d, J = 7.0 Hz, 3H, CHCH3), 2.31 (s, 6H, 3’, 4’-CH3Ar), 2.37-2.56 (m, 1H, CHCO), 3.58-3.79 (m, 2H, CH2N), 6.99-7.23 (m, 3H, Ar), 12.20 (bs, 1H, NH). | (d6-DMSO) 14.58 (5-CH3), 18.84 (4’-CH3), 19.35 (3’-CH3), 38.33 (C-5), 51.23 (C-6), 125.16 (Ar-C-6’), 128.80 (Ar-C-2’), 130.33 (Ar-C-5’), 134.83 (Ar-C-4’), 137.37, (Ar-C-3’), 139.90 (Ar-C-1’), 157.41 (C-2), 175.93 (C-4). | 3328.16 (NH), 1692.11 (C=O), 1631.71 (C=O). | |||
| 12a | (d6-DMSO) 2.01 (s, 3H, 2’-CH3Ar), 2.21 (s, 3H, 3’-CH3Ar), 2.60-2.89 (m, 2H, CH2CO), 3.40-3.82 (m, 2H, CH2N), 7.07-7.19 (m, 3H, Ar), 10.32 (bs, 1H, NH). | (d6-DMSO) 13.81 (2’-CH3), 19.92 (3’-CH3), 30.98 (C-5), 44.69 (C-6), 124.68 (Ar-C-6’), 125.94 (Ar-C-5’), 128.63 (C-4’), 134.01 (Ar-C-2’), 137.38 (Ar-C-3’), 140.79 (Ar-C-1’), 151.70, 151.78 (C-2), 170.54, 170.63 (C-4). | 3191.75 (NH), 1719.25 (C=O), 1689.35 (C=O). | |||
| 12b | (d6-DMSO) 2.21 (s, 6H, 3’4’-CH3Ar), 2.72 (t, J = 6.7 Hz, 2H, CH2CO), 3.76 (t, J = 6.7 Hz, 2H, CH2N), 7.00-7.18 (m, 3H, Ar), 10.26 (bs, 1H, NH). | (d6-DMSO) 18.75 (4’-CH3), 19.28 (3’-CH3), 31.03 (C-5), 44.66 (C-6), 122.60 (Ar-C-6’), 126.39 (Ar-C-2’), 129.45 (Ar-C-5’), 133.86 (Ar-C-4’), 136.37 (Ar-C-3’), 139.68 (Ar-C-1’), 152.04 (C-2), 170.41, 170.50 (C-4). | 3221.93 (NH), 1737.02 (C=O), 1689.56 (C=O). | |||
| 12c | (d6-DMSO) 2.26 (s, 6H, 3’,5’-CH3Ar), 2.67 (t, J = 6.7 Hz, 3H, CH2CO), 3.76 (t, J = 6.7 Hz, 2H, CH2N), 6.87-6.93 (m, 3H, Ar), 10.32 (s, 1H, NH). | (d6-DMSO) 20.73 (3’,5’-CH3), 31.01 (C-5), 44.59 (C-6), 122.98 (Ar-C-2’, Ar-C-6’), 127.26 (C-4’), 137.64 (Ar-C-3’, Ar-C-5’), 141.86 (Ar-C-1’), 151.98 (C-2), 170.50, (C-4). | 3207.49 (NH), 1727.61 (C=O), 1690.82 (C=O). | |||
| 13a | (d6-DMSO) 2.07 (s, 3H, 2’-CH3Ar), 2.27 (s, 3H, 3-CH3Ar), 2.78-2.92 (m, 2H, CH2CO), 3.66-4.00 (m, 2H, CH2N), 7.05-7.16 (m, 3H, Ar), 11.22 (bs, 1H, NH). | (d6-DMSO) 13.79 (2’-CH3), 19.26 (3’-CH3), 30.20 (C-5), 47.91 (C-6), 124.46 (Ar-C-6’), 126.27 (Ar-C-5’), 129.09 (Ar-C-4’), 133.08 (Ar-C-2’), 137.56 (Ar-C-3’), 143.78 (Ar-C-1’), 166.74, 166.84 (C-4), 178.44, 178.57 (C-2). | 3184.28 (NH), 1702.01 (C=O), 1213.70 (>N−CS−N< ). | |||
| 13b | (d6-DMSO) 2.20 (s, 6H, 3’,4’-CH3Ar), 2.78 (t, J = 6.9 Hz, 2H, CH2CO), 3.85 (t, J = 6.9 Hz, 2H, CH2N), 7.00-7.22 (m, 3H, Ar), 11.13 (bs, 1H, NH). | (d6-DMSO) 18.88 (4’-CH3), 19.26 (3’-CH3), 30.32, 31.02 (C-5), 48.85 (C-6), 124.03 (Ar-C-6’), 127.53 (Ar-C-2’), 129.85 (Ar-C-5’), 135.47 (Ar-C-4’), 136.89 (Ar-C-3’), 142.80 (Ar-C-1’), 166.74, 166.83 (C-4), 179.14(C-2). | 3176.11 (NH), 1717.41 (C=O), 1209.38 (>N−CS−N< ). | |||
| 13c | (d6-DMSO) 2.28 (s, 6H, 3’,5’-CH3Ar), 2.78 (t, J = 6.9 Hz, 2H, CH2CO), 3.85 (t, J = 6.9 Hz, 2H, CH2N), 6.85-7.00 (m, 3H, Ar), 11.20 (bs, 1H, NH). | (d6-DMSO) 20.67 (3’,5’-CH3), 30.32 (C-5), 48.76 (C-6), 124.32 (Ar-C-2’, Ar-C-6’), 128.80 (Ar-C-4’), 138.16 (Ar-C-3’, Ar-C-5’), 144.94 (Ar-C-1’), 166.84 (C-4), 179.00 (C-2). | 3186.44 (NH), 1736.13 (C=O), 1219.63 (>N−CS−N< ). | |||
| 14a | (d6-DMSO) 1.12, 1.15 (2d, J = 6.8 Hz, 3H, 5-CH3), 2.04, 2.08 (2s, 3H, 2’-CH3Ar), 2.26 (s, 3H, 3’-CH3Ar), 2.80-2.99 (m, 1H, CHCO), 3.38-3.64 (m, 2H, CH2N), 7.05-7.20 (m, 3H, Ar), 10.29, 10.30 (2bs, 1H, NH). | (d6-DMSO) 12.01, 12.65 (5-CH3), 13.84 (2’-CH3), 19.86 19.95 (3’-CH3), 34.80 (C-5), 51.01, 51.10 (C-6), 124.18, 125.14 (Ar-C-6’), 125.13, 126.08 (Ar-C-5’), 128.51, 128.76 (Ar-C-4’), 133.87, 134.22 (Ar-C-2’), 137.34, 137.38 (Ar-C-3’), 140.67 (Ar-C-1’), 151.83, 151.92, 152.65 (C-2), 173.08, 173.17, 173.27, 173.36 (C-4). | 3244.38 (NH), 1732.98 (C=O), 1694.97 (C=O). | |||
| 14b | (CDCl3) 1.31 (d, J = 7.0 Hz, 3H, 5-CH3), 2.26 (s, 6H, 3’,4’-CH3Ar), 2.73-3.00 (m, 1H, CH), 3.51-4.05 (m, 2H, CH2N), 7.03-7.40 (m, 3H, Ar), 7.81 (s, 1H, NH). | – | – | |||
| 14c | (d6-DMSO) 1.12 (d, J = 7.0 Hz, 3H, 5-CH3), 2.26 (s, 6H, 3’,5’-CH3Ar), 2.74-2.92 (m, 3H, CH2CO), 3.50-3.75 (m, 2H, CH2N), 6.86, 6.93 (2s, 3H, Ar), 10.30 (bs, 1H, NH). | (d6-DMSO) 12.16 (5-CH3), 20.74 (3’,5’-CH3), 34.89 (C-5), 50.88 (C-6), 122.88 (Ar-C-2’, Ar-C-6’), 127.22 (Ar-C-4’), 137.66 (Ar-C-3’, Ar-C-5’), 141.81 (Ar-C-1’), 151.97 (C-2), 173.17, (C-4). | 3201.75 (NH), 1724.34 (C=O), 1682.58 (C=O). | |||
| 15a | (d6-DMSO) 1.15 (d, J = 6.9 Hz. 3H, 5-CH3), 2.05, 2.09 (2s, 3H, 2’-CH3Ar), 2.26, 2.27 (2s, 3H, 3’-CH3Ar), 2.98-3.20 (m, 1H, CHCO), 3.65-3.90 (m, 2H, CH2N), 7.00-7.16 (m, 3H, Ar), 11.19, 11.23 (2s, 1H, NH). | (d6-DMSO) 11.86, 12.05 (5-CH3), 13.85 (2’-CH3), 19.78, 19.86 (3’-CH3), 33.93, 34.06 (C-5), 53.77, 53.84 (C-6), 124.16, 124,72 (Ar-C-6’), 126.20, 126.43 (Ar-C-5’), 129.04, 129.15 (Ar-C-4’), 133.04, 133.24 (Ar-C-2’), 137.56 (Ar-C-3’), 143.65 (Ar-C-1’), 169.62, 169.71, 169.84, 169.93 (C-4), 178.31, 178.44, 178.66 (C-2). | 3177.51 (NH), 1701.97 (C=O), 1210.30 (>N−CS−N< ). | |||
| 15b | (d6-DMSO) 1.16 (d, J = 6.9 Hz, 3H, 5-CH3), 2.27 (s, 6H, 3’,4’-CH3Ar), 2.91-3.07 (m, 1H, CHCO), 3.72-3.91 (m, 2H, CH2N), 7.00-7.26 (m, 3H, Ar), 11.24 (s, 1H, NH). | (d6-DMSO) 11.95 (5-CH3), 18.89 (4’-CH3), 19.26 (3’-CH3), 34.17 (C-5), 54.67 (C-6), 123.99 (C-6’), 127.46 (Ar-C-2’), 129.85 (Ar-C-5’), 135.46 (Ar-C-4’), 136.93 (Ar-C-3’), 142.70 (Ar-C-1’), 169.84 (C-4), 179.02 (C-2). | 3174.29 (NH), 1703.01 (C=O), 1211.74 (>N−CS−N< ). | |||
| 15c | (d6-DMSO) 1.14 (d, J = +6.9 Hz, 3H, 5-CH3), 2.33 (s, 6H, 3’,5’-CH3Ar), 2.88-3.17 (m, 3H, CHCO), 3.69-3.99 (m, 2H, CH2N), 6.85-7.00 (2s, 3H, Ar), 11.20 (s, 1H, NH). | (d6-DMSO) 11.92, 14.74 (5-CH3), 20.67, 20.74 (3’,5’-CH3), 34.17 (C-5), 54.60 (C-6), 124.28,124.87 (Ar-C-2’, Ar-C-6’), 128.77, 129.38 (C-4’), 138.18, 139.06 (Ar-C-3’, Ar-C-5’), 144.84 (Ar-C-1’), 169.83 (C-4), 178.93 (C-2). | 3252.42 (NH), 1730.95 (C=O) 1207.11 (>N−CS−N< ). | |||
| 16a | (d6-DMSO) 0.99, 1.17 (2d, J = 5.0 Hz, J = 5.1 Hz, 6-CH3), 2.04, 2.09 (2s, 3H, 2’-CH3Ar), 2.27 (s, 3H, 3’-CH3Ar), 2.40-3.15 (m, 2H, CH2CO), 3.60-4.10 (m, 1H, CHN), 6.95-7.20 (m, 3H, Ar), 10.37 (2bs, 1H, NH). | (d6-DMSO) 13.78, 14.50 (2’-CH3), 18.17, 18.70 (6-CH3), 19.96 19.98 (3’-CH3), 37.64, 38.16 (C-5), 50.18, 51.23 (C-6), 125.24, 125,43 (Ar-C-6’), 125.99, 127.26 (A-C-5’), 128.62, 128.89 (Ar-C-4’), 134.20,135.42 (Ar-C-2’), 137.16, 137,76 (Ar-C-3’), 138,81, 139.16 (Ar-C-1’), 151.16, 151.37 (C-2), 169, 94 (C-4). | 3185.92 (NH), 1714.33 (C=O), 1678.05 (C=O). | |||
| 16c | (d6-DMSO) 1.16 (d, J = 6.6 Hz, 3H, 6-CH3), 2.27 (s, 6H, 3’,5’-CH3Ar), 2.37-3.15 (m, 3H, CH2CO), 3.90-4.10 (m, 1H, CHN), 6.93 (s, 3H, Ar), 10.35 (bs, 1H, NH). | (d6-DMSO) 18.48 (6-CH3), 20.69 (3’,5’-CH3), 37.76 (C-5), 51.31 (C-6), 125.02 (Ar-C-2’, Ar-C-6’), 128.10 (Ar-C-4’), 137.91 (Ar-C-3’, Ar-C-5’), 140.51 (Ar-C-1’), 151.29 (C-2), 169.95 (C-4). | 3205.99 (NH), 1728.28 (C=O), 1690.29 (C=O). | |||
| 17a | (d6-DMSO) 1.01, 1.18 (2d, J = 6.7 Hz, 3H, 6-CH3), 2.07 (s, 3H, 3’-CH3Ar), 2.26, 2.28 (2s, 3H, 2’-CH3Ar), 2.50-3.25 (m, 2H, CH2CO), 3.80-4.25 (m, 1H, CHN), 6.99-7.20 (m, 3H, Ar), 11.27, 11.32 (2s, 1H, NH). | (d6-DMSO) 13.78, 15.00 (2’-CH3), 17.52 18.01 (6-CH3), 19.88 (3’-CH3), 36.90, 37.37, 37.62 (C-5), 53.69, 55.12 (C-6), 124.73, 125.60 (Ar-C-6’), 126.41, 126.50 (Ar-C-5’), 129.00, 129.20 (Ar-C-4’), 133.18, 134.54 (Ar-C-2’), 137.32, 137.86 (Ar-C-3’), 142.01, 142.15 (Ar-C-1’), 166.35, 166.51 (C-4), 177.92, 178.05 (C-2). | 3221.61 (NH), 1702.49 (C=O), 1209.98 (>N−CS−N< ). | |||
| 17c | (d6-DMSO) 1.23 (d, J = 6.7 Hz, 3H, 6-CH3), 2.34 (s, 6H, 3’,5’-CH3Ar), 2.45-3.30 (m, 3H, CH2CO), 3.96-4.12 (m, 1H, CHN), 6.96, 7.04 (2s, 3H, Ar), 11.29 (s, 1H, NH. | (d6-DMSO) 17.63 (6-CH3), 20.66 (3’,5’-CH3), 36.90 (C-5), 55.18 (C-6), 125.01, 125.39 (Ar-C-2’, Ar-C-6’), 129.07 (Ar-C-4’), 138.20 (Ar-C-3’, Ar-C-5’), 143.62 (Ar-C-1’), 166.34, 166.43 (C-4), 177.96 (C-2). | 3219.94 (NH), 1734.70 (C=O), 1220.85 (>N−CS−N< ). | |||
| 18a | (CDCl3) 1.24 (d, J = 6.8 Hz. 3H, 5-CH3), 1.39 (t, J = 7.1 Hz, 3H, COOCH2CH3), 1.96, 1.98 (2s, 3H, 2-CH3), 2.19, 2.25 (2s, 3H, 2’-CH3Ar), 2.33, 2.34 (2s, 3H, 3’-CH3), 2.60-2.82 (m, 1H, CHCO), 3.42-3.70 (m, 2H, CH2N), 4.30 (k, J = 7.1 Hz, COOCH2CH3), 6.90-7.30 (m, 3H, Ar). | (CDCl3) 12.88, 13.19 (5-CH3), 14.41 (COOCH2CH3), 14.21, 14.25 (2’-CH3), 19.17, 19.33 (2-CH3), 20.36, 20.43 (3’-CH3), 60.49 (COOCH2CH3), 106.02 (C-3), 38.40, 38.49 (C-5), 57.08, 57.28 (C-6), 124,44, 125.15 (Ar-C-6’), 127.00, 127.08 (Ar-C-5’), 130.11, 130.17 (Ar-C-4’), 133.64, 133.70 (Ar-C-2’), 139.20, 139.25 (Ar-C-3’), 142.75, 142.81 (Ar-C-1’), 163.47, 163.61 (C-2), 167.99, 168.03 (COOCH2CH3), 190.25, 190.36 (C-4). | 1663.80 (C=O), 1249.25 (C−O−). | |||
| 18c | (CDCl3) 1.18 (d, J = 6.9 Hz. 3H, 5-CH3), 1.34 (t, J = 7.1 Hz, 3H, COOCH2CH3), 2.00 (s, 3H, 2-CH3), 2.34 (s, 6H, 3’,5’-Ar), 2.54-2.73 (m, 1H, CHCO), 3.53-3.83 (m, 3H, CH2N), 4.28 (k, J = 7.1 Hz, 2H, COOCH2CH3), 6.77, 6.99 (2s, 3H, Ar). | (CDCl3) 12.99 (5-CH3), 14.39 (COOCH2CH3), 19.74 (2-CH3), 21.20 (3’,5’-CH3), 38.75 (C-5), 58.06, (C-6), 60.53 (COOCH2CH3), 106.70 (C-3), 124.11 (Ar-C-2’, C-6’), 129.75 (C-4’), 139.87 (C-3’,C-5’), 144.05 (Ar-C-1’), 162.54 (C-2), 168.06 (COOCH2CH3), 190.52 (C-4). | 1726.31 (C=O), 1258.74 (C−O−). | |||
| Compounds | M. p., °C; (Solv.*) | Molecular Formula | Elemental analysis data (Calculated / Found) % | Yield, % | ||
|---|---|---|---|---|---|---|
| C | H | N | ||||
| 2a | 168-195 | C14H19NO4·HCl | 55.71 / 55.48 | 6.69 / 6.58 | 4.64 / 4.32 | 62.7 |
| 2b | 194-195 | C14H19NO4·HCl | 55.71 / 55.41 | 6.69 / 6.42 | 4.64 / 4.79 | 54.9 |
| 3a | 101-102 | C11H15NO2 | 68.37 / 68.24 | 7.82 / 7.68 | 7.25 / 7.14 | 25.7 |
| 3b | 151-158 | C11H15NO2·HCl | 57.51 / 57.29 | 7.03 / 7.31 | 6.10 / 6.34 | 26.7 |
| 3c | 186-187 | C11H15NO2·HCl | 57.51 / 57.28 | 7.03 / 7.29 | 6.10 / 6.26 | 37.5 |
| 4a | 100-101 | C12H17NO2 | 69.54 / 69.67 | 8.27 / 8.35 | 6.76 / 6.92 | 38.0 |
| 4b | 185-186 | C12H17NO2·HCl | 59.14 / 59.02 | 7.46 / 7.64 | 5.75 / 5.49 | 34.1 |
| 4c | 129-130 | C12H17NO2 | 69.54 / 69.22 | 8.27 / 8.11 | 6.76 / 6.36 | 58.3 |
| 5a | 166-167 | C12H17NO2·HCl | 59.14 / 58.98 | 7.46 / 7.31 | 5.75 / 5.36 | 66.7 |
| 5c | 170-172 | C12H17NO2·HCl | 59.14 / 58.96 | 7.46 / 7.28 | 5.75 / 5.61 | 69.6 |
| 8b | 184-186 | C13H18N2O3 | 62.38 / 62.16 | 7.25 / 7.38 | 11.19 / 11.32 | 75.4 |
| 12a | 204-206 | C12H14N2O2 | 66.04 / 65.89 | 6.47 / 6.64 | 12.84 / 12.66 | 57.6 |
| 12b | 179-181 | C12H14N2O2 | 66.04 / 66.24 | 6.47 / 6.31 | 12.84 / 12.61 | 41.4 |
| 12c | 179-180 | C12H14N2O2 | 66.04 / 66.31 | 6.47 / 6.67 | 12.84 / 12.65 | 29.4 |
| 13a | 242-244 | C12H14N2OS | 61.51 / 61.32 | 6.02 / 5.84 | 11.96 / 11.72 | 51.3 |
| 13b | 247-249 | C12H14N2OS | 61.51 / 61.44 | 6.02 / 5.87 | 11.96 / 11.79 | 50.9 |
| 13c | 263-264 | C12H14N2OS | 61.51 / 61.39 | 6.02 / 5.79 | 11.96 / 11.75 | 60.5 |
| 14a | 196-197 | C13H16N2O2 | 67.22 / 67.05 | 6.94 / 6.77 | 12.06 / 12.27 | 60.3 |
| 14b | 135-136 | C13H16N2O2 | 67.22 / 67.41 | 6.94 / 6.78 | 12.06 / 12.20 | 78.3 |
| 14c | 155-156 | C13H16N2O2 | 67.22 / 67.38 | 6.94 / 6.81 | 12.06 / 12.34 | 33.2 |
| 15a | 187-188 | C13H16N2OS | 62.87 / 62.69 | 6.49 / 6.65 | 11.28 / 11.47 | 39.5 |
| 15b | 188-190 | C13H16N2OS | 62.87 / 62.64 | 6.49 / 6.35 | 11.28 / 11.41 | 38.7 |
| 15c | 206-208 | C13H16N2OS | 62.87 / 62.69 | 6.49 / 6.62 | 11.28 / 11.05 | 47.8 |
| 16a | 214 -216 | C13H16N2O2 | 67.22 / 67.32 | 6.94 / 6.75 | 12.06 / 12.32 | 31.4 |
| 16c | 181-182 | C13H16N2O2 | 67.22 / 67.07 | 6.94 / 6.72 | 12.06 / 12.28 | 38.4 |
| 17a | 224 -226 | C13H16N2OS | 62.87 / 62.45 | 6.49 / 6.58 | 11.28 / 11.11 | 26.6 |
| 17c | 254-255 | C13H16N2OS | 62.87 / 62.45 | 6.49 / 6.31 | 11.28 / 11.40 | 38.5 |
| 18a | 109-110 | C18H23NO3 | 71.73 / 71.55 | 7.69 / 7.49 | 4.65 / 4.41 | 17.0 |
| 18c | 111-112 | C18H23NO3 | 71.73 / 71.46 | 7.69 / 7.82 | 4.65 / 4.47 | 6.6 |
References
- Baltrusis, R.; Mickevicius, V.; Bylinskaite, J.; Zolotojabko, R.; Liepins, E. Synthesis and structure of 6-methyl-1-(2-R-phenyl)dihydro-2,4(1H,3H)pyrimidinedione derivatives. Khim. Geterotsikl. Soedin. 1990, 8, 1096–1106. [Google Scholar]
- Mickevicius, V.; Baltrusis, R.; Bylinskaite, J.; Liepins, E.; Zolotojabko, R. Synthesis and structure of 5-methyl-1-(2-R- phenyl)-dihydro-2,4(1H,3H)pyrimidinediones and its 2-thio analogues. Khim. Geterotsikl. Soedin. 1991, 9, 1240–1247. [Google Scholar]
- Kalinowski, H. O.; Berger, S.; Braun, S. 13C-NMR-Spektroskopie; Georg Thieme Verlag: Stuttgart, New York, 1984; pp. 97-99, 141, 228, 283-286. [Google Scholar]
- Beresneviciute, K.; Beresnevicius, Z.; Mikulskiene, G.; Kihlberg, J.; Broddefalk, J. 13C-NMR study of dihydropyrimidinedione and dihydropyrimidine-2-thione derivatives. J. Magn. Reson. Chem. 1997, 35, 553–555. [Google Scholar]
- Duddeck, H.; Dietrich, W.; Tóth, G. Structure Elucidation by Modern NMR; Steinkopff: Darmstadt; Springer: New York, 1998; pp. 10-14, 23-29. [Google Scholar]
- Sample Availability: Available from the authors with the exception of 6-11.
© 2005 by MDPI (http:www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Vaickelioniene, R.; Mickevicius, V.; Mikulskiene, G. Synthesis and Cyclizations of N-(2,3-, 3,4- and 3,5-Dimethylphenyl)-β-alanines. Molecules 2005, 10, 407-416. https://doi.org/10.3390/10020407
Vaickelioniene R, Mickevicius V, Mikulskiene G. Synthesis and Cyclizations of N-(2,3-, 3,4- and 3,5-Dimethylphenyl)-β-alanines. Molecules. 2005; 10(2):407-416. https://doi.org/10.3390/10020407
Chicago/Turabian StyleVaickelioniene, Rita, Vytautas Mickevicius, and Gema Mikulskiene. 2005. "Synthesis and Cyclizations of N-(2,3-, 3,4- and 3,5-Dimethylphenyl)-β-alanines" Molecules 10, no. 2: 407-416. https://doi.org/10.3390/10020407




