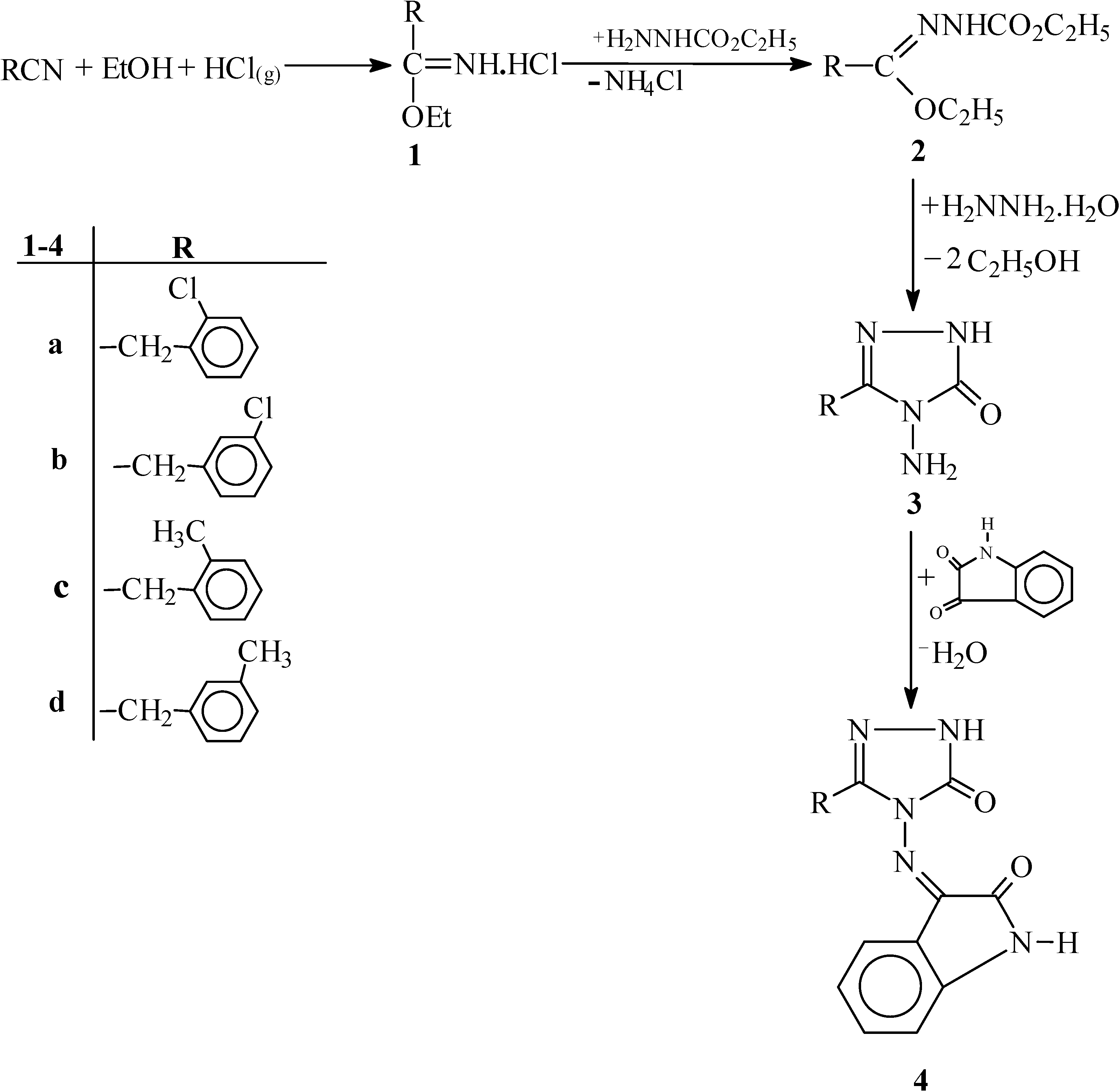Experimental
General
Melting points were determined in open capillary tubes on a Büchi oil heated melting point apparatus and are uncorrected. The IR spectra were recorded for KBr pellets on Perkin-Elmer 1600 FTIR spectrophotometer. 1H-NMR and 13C-MR spectra were recorded on a Varian 200A spectrometer (solvent DMSO-d6, TMS as internal standard). Elemental analyses were performed on a Carlo Erba 1106 CHN analyzer. Starting materials were obtained from Fluka or Aldrich.
Synthesis of Iminoester Hydrochlorides 1a-d.
These were synthesized using the reported method [
25]. To an ice-cooled solution of the appropriate nitrile (1 mole) in absolute alcohol (1.1 moles), dry hydrogen chloride was added until 1.1 moles had been absorbed. The resulting solution was then allowed to stand at 0 ºC in the refrigerator for 12 hours, after which cold absolute ether was added and the obtained crystals were filtered off immediately, washed with cold absolute ether and dried in a dessicator. The following compounds were thus prepared:
Ethyl imido-o-chlorophenylacetate hydrochloride (1a): m.p. 95-96 ºC; IR ῡ (cm-1): 2982, 2878, 812 (NH2+), 1624 (C=N), 753 (1,2-disubstituted benzene ring).
Ethyl imido-m-chlorophenylacetate hydrochloride (1b): m.p. 82-84 ºC; IR ῡ (cm-1): 2989, 2939, 823 (NH2+), 1649 (C=N), 868, 791, 684 (1,3-disubstituted benzene ring).
Ethyl imido-o-methylphenylacetate hydrochloride (1c): m.p. 80-81 ºC; IR ῡ (cm-1): 2981, 2928, 823 (NH2+), 1648 (C=N), 747 (1,2-disubstituted benzene ring).
Ethyl imido-m-methylphenylacetate hydrochloride (1d): m.p. 113-114 ºC; IR ῡ (cm-1): 2994, 2862, 811 (NH2+), 1653 (C=N), 867, 768, 714 (1,3-disubstituted benzene ring).
General Procedure for Preparation of Ethyl Substituted Formate Ethoxycarbonyl hydrazones 2a-d.
In a stoppered flask equipped with a magnetic stirrer, the corresponding ethyl imidocarboxylate hydrochloride (1a-d, 0.01 mol) was dissolved in absolute ethanol (50 mL) with ice-bath cooling and ethyl carbazate (0.01 mol) dissolved in absolute ethanol (20 mL) was then added to this solution. After stirring for 6 hr in ice-bath, the mixture was filtered to remove the ammonium chloride which separated from the solution and the filtrate was evaporated at 30-35ºC under reduced pressure. The solid residue, after drying in a dessicator, was recrystallized from petroleum ether to yield compounds 2a-d.
Ethyl o-chlorophenylacetate ethoxycarbonyl hydrazone (2a): Prepared from 1a, yield 78%; m.p. 54-55 ºC; IR ῡ (cm-1): 3274 (N-H), 1721 (C=O), 1648 (C=N), 751 (1,2-disubstituted benzene ring); 1H-NMR, δ (ppm): 1.18 (t, 3H, CH3), 1.28 (t, 3H, CH3), 3.71 (s, 2H, CH2), 3.96 (q, 2H, CH2), 4.24 (q, 2H, CH2), 6.58-7.24 (m, 4H, Ar-H), 8.18 (s, 1H, NH); Calcd. (%) for C13H17N2O3Cl (285): C, 54.78; H, 6.02; N, 9.84; found (%): C, 54.92; H, 6.01, N, 10.00.
Ethyl m-chlorophenylacetate ethoxycarbonyl hydrazone (2b): Prepared from 1b, 74%; m.p. 47-48 ºC; IR ῡ (cm-1): 3247 (N-H), 1709 (C=O), 1648 (C=N), 863, 770, 683 (1,3-disubstituted benzene ring); 1H-NMR, δ (ppm): 1.19 (t, 3H, CH3), 1.24 (t, 3H, CH3), 3.64 (s, 2H, CH2), 3.91(q, 2H, CH2), 4.11 (q, 2H, CH2), 6.80-7.11 (m, 4H, Ar-H), 8.23 (s, 1H, NH); Calcd. (%) for C13H17N2O3Cl (285): C, 54.78; H, 6.02; N, 9.84; found (%): C, 54.48; H, 6.18, N, 10.27.
Ethyl o-methylphenylacetate ethoxycarbonyl hydrazone (2c): Prepared from 1c, yield 80%; m.p. 66-67 ºC; IR ῡ (cm-1): 3207 (N-H), 1702 (C=O), 1657 (C=N), 747 (1,2-disubstituted benzene ring); 1H-NMR, δ (ppm): 1.20 (t, 3H, CH3), 1.32 (t, 3H, CH3), 2.30 (s, 3H, CH3), 3.64 (s, 2H, CH2), 4.00 (q, 2H, CH2), 4.24 (q, 2H, CH2), 6.96-7.30 (m, 4H, Ar-H); Calcd. (%) for C14H20N2O3 (264): C, 63.56; H, 7.63; N, 10.60; found (%): C, 63.68; H, 7.89, N, 10.86.
Ethyl m-methylphenylacetate ethoxycarbonyl hydrazone (2d): Prepared from 1d, yield 86%; m.p. 59-60 ºC; IR ῡ (cm-1): 3257 (N-H), 1710 (C=O), 1648 (C=N), 873, 755, 694 (1,3-disubstituted benzene ring); 1H-NMR, δ (ppm): 1.21 (t, 3H, CH3), 1.28 (t, 3H, CH3), 2.28 (s, 3H, CH3), 3.60 (s, 2H, CH2), 4.08 (q, 2H, CH2), 4.20 (q, 2H, CH2), 6.96-7.54 (m, 4H, Ar-H), 8.46 (s, 1H, NH); Calcd. (%) for C14H20N2O3 (264): C, 63.56; H, 7.63; N, 10.60; found (%): C, 63.65; H, 7.89, N, 10.58.
General Procedure for the Preparation of 3-Substituted-4-amino-4,5-dihydro-1H-1,2,4-triazole-5-ones 3a-d.
Compound 2 (0.01 mol) was refluxed for 5 hr with a solution of hydrazine hydrate (1.25 mL) in water (60 mL). The solution was crystallized by cooling to obtain the crude product (3a-d). The solid material thus obtained was filtered off and recrystallized from ethanol.
3-o-Chlorobenzyl-4-amino-4,5-dihydro-1H-1,2,4-triazole-5-one (3a): Prepared from 2a, yield 88%; m.p. 164-165 ºC; IR ῡ (cm-1): 3338, 3217 (NH2, NH), 1720 (C=O), 1633 (C=N), 749 (1,2-disubstituted benzene ring); 1H-NMR, δ (ppm): 4.10 (s, 2H, CH2), 5.28 (s, 2H, NH2), 7.25 (s, 4H, Ar-H), 11.90 (s, 1H, NH); 13C-NMR, δ (ppm): 28.80, 124.63, 126.84, 130.13, 132.86, 136.90, 138.63, 145.10, 153.14;. Calcd. (%) for C9H9N4OCl (225): C, 48.08; H, 4.04; N, 24.94; found (%): C, 48.40; H, 4.54, N, 24.68.
3-m-Chlorobenzyl-4-amino-4,5-dihydro-1H-1,2,4-triazole-5-one (3b):Prepared from 2b, yield 83%; m.p. 171-172 ºC; IR ῡ (cm-1): 3324, 3224 (NH2, NH), 1730 (C=O), 1638 (C=N), 864, 793, 719 (1,3-disubstituted benzene ring); 1H-NMR, δ (ppm): 4.00 (s, 2H, CH2), 5.18 (s, 2H, NH2), 7.16 (s, 4H, Ar-H), 11.10 (s, 1H, NH); 13C-NMR, δ (ppm): 29.62, 125.54, 127.84, 131.14, 133.62, 136.95, 137.44, 146.93, 154.11; Calcd. (%) for C9H9N4OCl (225): C, 48.08; H, 4.04; N, 24.94; found (%): C, 48.34; H, 4.63, N, 24.85.
3-o-Methylbenzyl-4-amino-4,5-dihydro-1H-1,2,4-triazole-5-one (3c): Prepared from 2c, yield 91%; m.p. 190-191 ºC; IR ῡ (cm-1): 3327, 3221 (NH2, NH), 1725 (C=O), 1635 (C=N), 736 (1,2-disubstituted benzene ring); 1H NMR, δ (ppm): 2.38 (s, 3H, CH3), 4.10 (s, 2H, CH2), 5.28 (s, 2H, NH2), 7.32 (s, 4H, Ar-H), 11.60 (s, 1H, NH); 13C-NMR, δ (ppm): 20.12, 29.76, 124.86, 125.18, 129.26, 130.18, 134.10, 135.19, 146.13, 153.81; Calcd. (%) for C10H12N4O (204): C, 58.76; H, 5.92; N, 27.42; found (%): C, 58.86; H, 6.04, N, 27.31.
3-m-Methylbenzyl-4-amino-4,5-dihydro-1H-1,2,4-triazole-5-one (3d): Prepared from 2d, yield 87%; m.p.156-157 ºC; IR ῡ (cm-1): 3322, 3222 (NH2, NH), 1731 (C=O), 1642 (C=N), 822, 751, 705 (1,3-disubstituted benzene ring); 1H-NMR, δ (ppm): 2.38 (s, 3H, CH3), 4.16 (s, 2H, CH2), 5.24 (s, 2H, NH2), 7.24 (s, 4H, Ar-H), 11.46 (s, 1H, NH); 13C-NMR, δ (ppm): 19.08, 28.12, 125.70, 126.56, 129.01, 129.78, 134.27, 136.17, 147.28, 154.00; Calcd. (%) for C10H12N4O (204): C, 58.76; H, 5.92; N, 27.42; found (%): C, 58.69; H, 6.12, N, 27.96.
General Procedure for the Preparation of Isatin-3-imines 4a-d.
Equimolar quantities (0.01 mol) of isatin and the corresponding amino compound 3a-d were dissolved in warm ethanol (40 mL) containing glacial acetic acid (0.5 mL). The reaction mixture was refluxed for 4 hr and then kept at room temperature overnight. The resulting solid was washed with ethanol, dried and recrystallized from ethanol-chloroform to afford compounds 4a-d.
3-[3’-(4’’-o-Chlorobenzyl-4’,5’-dihydro-1’H-1’,2’,4’-triazol-5’-on-4’-yl]-iminoisatin (4a): Prepared from 3a, yield 72%; m.p. 252-253 ºC; IR ῡ (cm-1): 3403, 3190 (N-H), 1754,1730 (C=O), 1684, 1613 (C=N), 1463 (C=C); 1H-NMR, δ (ppm): 4.18 (s, 2H, CH2), 7.18-7.64 (m, 8H, Ar-H), 11.22 (s, 1H, N-H), 12.40 (s, 1H, N-H); 13C-NMR, δ (ppm): 29.26, 113.42, 115.80, 121.18, 125.50, 126.18, 128.88, 129.85, 131.60, 133.36, 134.81, 137.18, 144.81, 145.83, 146.12, 156.18, 161.86 ; Calcd. (%) for C17H12N5O2Cl (354): C, 67.66; H, 3.42; N, 19.79; found (%): C, 57.18; H, 3.52, N, 19.64.
3-[3’-(4’’-m-Chlorobenzyl)-4’,5’-dihydro-1’H-1’,2’,4’-triazol-5’-on-4’-yl]-iminoisatin (4b): Prepared from 3b, yield 68%; m.p. 266-267 ºC; IR ῡ (cm-1): 3164, 3086 (N-H), 1730, 1708 (C=O), 1684, 1613 (C=N), 1464 (C=C); 1H-NMR, δ (ppm): 4.14 (s, 2H, CH2), 6.96-7.32 (m, 6H, Ar-H), 7.40-7.68 (s, 2H, Ar-H), 11.28 (s, 1H, N-H), 12.36 (s, 1H, N-H); 13C-NMR, δ (ppm): 28.31, 112.41, 116.42, 120.00, 124.81, 127.51, 128.12, 129.24, 131.18, 132.41, 135.40, 136.02, 145.10, 145.91, 146.83, 157.16, 162.15; Calcd. (%) for C17H12N5O2Cl (354): C, 57.66; H, 3.42; N, 19.79; found (%): C, 57.89; H, 3.48, N, 19.69.
3-[3’-(4’’-o-Methylbenzyl)-4’,5’-dihydro-1’H-1’,2’,4’-triazol-5’-on-4’-yl]-iminoisatin (4c): Prepared from 3c, yield 74%; m.p. 269-270 ºC; IR ῡ (cm-1): 3194, 3088 (N-H), 1746, 1706 (C=O), 1610, 1588 (C=N), 1467 (C=C); 1H-NMR, δ (ppm): 2.44 (s, 3H, CH3), 4.08 (s, 2H, CH2), 6.92-7.66 (m, 8H, Ar-H), 11.12 (s, 1H, N-H), 12.54 (s, 1H, N-H); 13C-NMR, δ (ppm): 19.10, 29.33, 111.16, 115.73, 122.41, 125.74, 127.10, 127.90, 129.37, 130.04, 133.33, 135.54, 136.53, 145.70, 146.10, 148.54, 158.04, 163.31; Calcd. (%) for C18H15N5O2 (333): C, 64.80; H, 4.54; N, 21.00; found (%): C, 64.96; H, 4.66, N, 21.44.
3-[3’-(4’’-m-Methylbenzyl)-4’,5’-dihydro-1’H-1’,2’,4’-triazol-5’-on-4’-yl]-iminoisatin (4d): Prepared from 3d, yield 75 %; m.p. 272-273 ºC; IR õ (cm-1): 3177, 3084 (N-H), 1748, 1707 (C=O), 1609, 1583 (C=N), 1466 (C=C); 1H-NMR δ (ppm): 2.41 (s, 3H, CH3), 4.12 (s, 2H, CH2), 7.00-7.47 (m, 8H, Ar-H), 11.20 (s, 1H, N-H), 12.32 (s, 1H, N-H); 13C-NMR, δ (ppm): 19.04, 28.96, 112.86, 114.61, 121.14, 124.81, 127.00, 127.50, 128.14, 131.14, 132.15, 135.20, 135.41, 144.62, 146.54, 147.51, 157.60, 162.94; Calcd. (%) for C18H15N5O2 (333): C, 64.80; H, 4.54; N, 21.00; found (%): C, 64.56; H, 4.63, N, 21.21.