To define the canonical ensemble at equilibrium we need to consider the system of interest in contact with a heat reservoir. This reservoir can exchange energy with the system but is sufficiently large that its thermodynamic state is not effected, so at equilibrium the details of the thermal contact are not believed to be important. Away from equilibrium the details of the thermal contact are central. We are not only interested in exchanging energy to equilibrate a system of interest, but also to consider reservoirs that actively couple to the system to measure the temperature, as intended in the zeroth law.
In this section we examine a very simple microscopic model for thermal coupling to a reservoir which can be used in contact with either, an equilibrium system, or to supply and remove heat from a system to maintain it a nonequilibrium steady state. As this microscopic model couples mechanically to the system it may be modified to measure the temperature. In all that follows we will consider this microscopic model and no systematic attempts to improve the model will be made.
3.1. Equilibrium
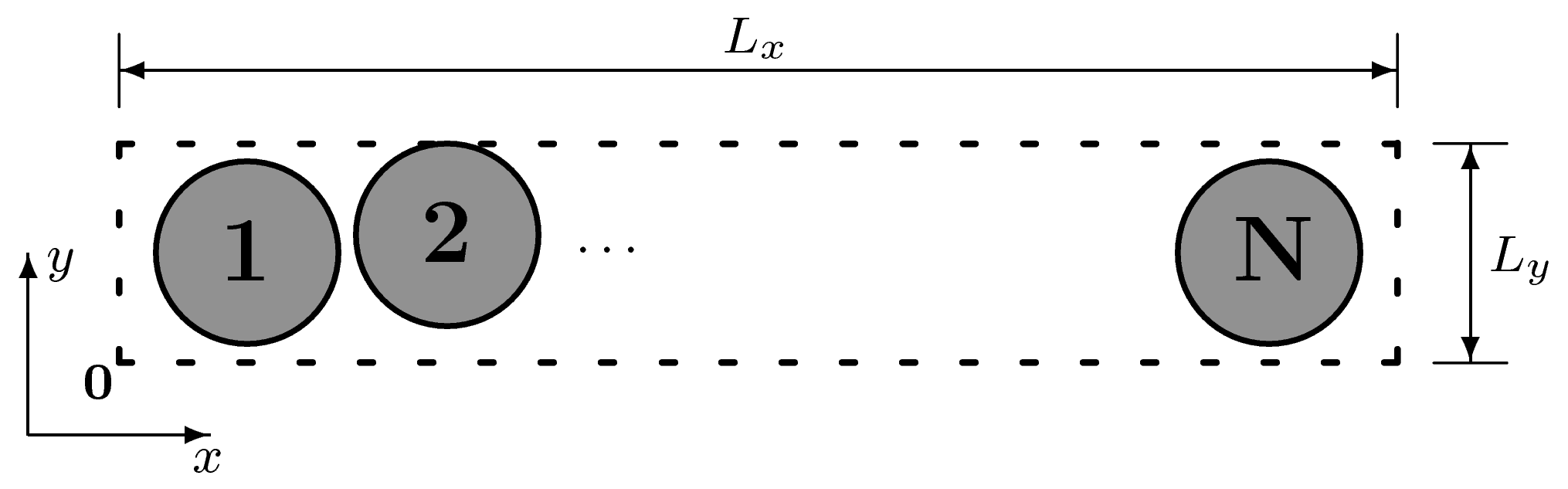
The quasi-one-dimensional (QOD) system introduced recently [
16] can be modified to interact with an idealized
heat reservoir in a deterministic and reversible way [
17], to study heat conduction in low dimensional systems [
21] and the Lyapunov mode structure. Similar systems have been used recently to study heat flow [
19,
20]. This system contains hard disks in a narrow channel that does not allow the disks to change their (left to right) order, see
Figure 1.
Figure 1.
Schematic of an N hard-disk quasi-one-dimensional (QOD)system. The height is sufficiently small that the disks cannot pass one another. We choose the coordinate origin to be located at the bottom left corner of the system, and system boundaries are denoted by dashed lines. The boundaries at are hard walls and those at periodic (that is, a HP quasi-one-dimensional system).
Figure 1.
Schematic of an N hard-disk quasi-one-dimensional (QOD)system. The height is sufficiently small that the disks cannot pass one another. We choose the coordinate origin to be located at the bottom left corner of the system, and system boundaries are denoted by dashed lines. The boundaries at are hard walls and those at periodic (that is, a HP quasi-one-dimensional system).
The equations of motion connecting the QOD system to the two reservoirs at
define the
thermal contact. When a particle collides with a reservoir wall the normal component of the momentum of the particle is changed by
is the value of the reservoir momentum for the
reservoir (either
or
). If
there is no interaction with the reservoir, and if
the incoming momentum is completely replaced by the reservoir momentum. Generally we use a value of
which provides an effective mixing of the incoming momentum with the reservoir momentum, and a density of 0.8. The value of
and
varies with the number of particles
N as
. For a boundary temperature of
the reservoir momentum is given by
. The temperature profile is determined from the average components of the kinetic temperature of each particle (the average of equation 8 ). As the QOD system is narrow enough to prevent particles interchanging their positions, the order of the particles remains fixed. Therefore, the temperature profiles are shown as functions of the particle number, rather than particle position. Also both
x and
y components of the temperature are presented together to show the level of local thermal equilibration.
Figure 2 shows both components of the temperature profile for a QOD system of 100 disks with reservoirs of temperature
on each side.
Figure 2.
The temperature profile and , and inter-disk heat current for a QOD system of 100 hard-disks. Notice that the temperature increases near the two reservoirs with the temperature in the centre is about 20% smaller than the nominal reservoir temperature . This is termed the contact resistance. There is a very small residual heat current to the left. There is also a noticeable difference between the x and y temperatures of the particles closest to the two boundaries. This arises because the boundary collisions treat x and y components of momentum differently.
Figure 2.
The temperature profile and , and inter-disk heat current for a QOD system of 100 hard-disks. Notice that the temperature increases near the two reservoirs with the temperature in the centre is about 20% smaller than the nominal reservoir temperature . This is termed the contact resistance. There is a very small residual heat current to the left. There is also a noticeable difference between the x and y temperatures of the particles closest to the two boundaries. This arises because the boundary collisions treat x and y components of momentum differently.
The change in temperature near the contact with the reservoir has been observed before and has been termed the Kapitza resistance [
21,
22]. Here the centre of this equilibrium system is about 20% colder than the two reservoirs. Numerical calculation of the
-distribution for the particle in the centre of the system gives a Gaussian distribution with the same temperature as the centre of the profile, as shown in
Figure 2. We observe that to a good approximation, the two halves of this Gaussian propagate to the left and the right unchanged. Thus the distribution of incident momentum
at each reservoir is Gaussian with the same temperature as the centre of the system. As the system is intended to be at equilibrium with the two reservoirs it is steady when the net energy flow through the boundaries is zero. Thus if
p is the momentum before collision with the wall and
is the momentum after collision then
. Using the collision rule in equation (9) and taking the wall momentum as
, we require
The incoming half Gaussian has a mean of
and a variance of
, so inserting these into equation (10), setting
, and solving for the variance of the incoming Gaussian
gives
For
the temperature is
which is in good agreement with the temperature at the middle of the system.
As the incoming momentum distribution is half Gaussian, using the collision rule in equation (9), we can estimate the outgoing distribution for particle 1 as the weighted convolution of the reflected half Gaussian and the delta function reservoir momentum distribution. Recollisions of particle 1 and 2 will modify this approximate distribution, but for our purposes this will be sufficient. This out-going distribution for particle 1 interacts with the in-coming distribution of particle 2 to generate the out-going distribution for particle 2. We assume that the inputs to this process are two independent random variables so the out-going distribution is the convolution of the two input distributions. Thus for particle 2 the outgoing distribution is the convolution of a half Gaussian and a delta function, with its maximum at and half the variance.
To generate approximate distributions for successive particles 3, 4 and 5 etc. we again assume that we can again convolve the out-going distribution of particle
i with the incoming distribution of particle
to get the out-going distribution of particle
. With each convolution the maximum of the distribution moves to
and the variance becomes
. The contribution to the variance from the outgoing distribution of the
particle is
Notice that in the limit as
,
so the distribution again matches the full Gaussian distribution in the centre of the system. The temperature is then
In a numerical calculation the kinetic energy transferred from particle
i to particle
in each collision can be calculated from
where
and
. The time average of this quantity gives the average energy current flowing from particle
i to
. Notice that when the total momentum of the two colliding particles is zero, no energy is transferred. The transfer of energy is correlated with fluctuations of the pair momentum away from zero.
3.2. Nonequilibrium
Figure 3 shows the temperature profile for a nonequilibrium QOD system in an external temperature gradient
and
. The contact resistance is again evident at both boundaries, both the
hot boundary and the
cold boundary. The mechanism that produced this effect in the equilibrium case occurs again, but now modified by the temperature gradient. The incoming distributions will be Gaussian but with a variance that changes slowly with position. This is due to the
local equilibration of the energy between
x and
y coordinates of the same particle as evident in
Figure 3, leading to the
x and
y components of the temperature of each particle being approximately equal.
Figure 3.
The temperature profile and inter-disk heat current for a nonequilibrium QOD system of 100 hard-disks with and . Notice that the contact resistance is evident at both reservoirs. Due to the temperature difference there is a heat current of to the right.
Figure 3.
The temperature profile and inter-disk heat current for a nonequilibrium QOD system of 100 hard-disks with and . Notice that the contact resistance is evident at both reservoirs. Due to the temperature difference there is a heat current of to the right.
The process is clear when we look at the distributions of
for the first eight particles on the left-hand side of the system in
Figure 4. These are the particles closest to the hot reservoir. The incoming distributions are again approximately Gaussian, and the out-going distributions show the same behaviour as the equilibrium system. For particle 1, the distribution is changed by the collision process at the boundary, but for particle 2 this diminishes and each successive particle distribution diminishes the effect of the wall even more, until at particle 8 the whole distribution looks almost completely Gaussian again. While the momentum distributions cannot be exactly Gaussian because this nonequilibrium system supports an energy current, the deviations from Gaussian are at best only subtle.
Figure 4.
The probability distribution for the
x component of the momentum for particles 1 to 8 in the nonequilibrium QOD system shown in
figure (3). These are the particles nearest the hot reservoir. The incoming distributions (the negative part of the
distribution) are very close to Gaussian for each particle, while the outgoing distributions show the same qualitative behaviour as the outgoing distributions in the equilibrium case. There is a strong effect in the outgoing distribution of particle 1, and that effect successively diminishes in particles 2, 3, etc. as the distribution slowly returns to its approximately Gaussian shape.
Figure 4.
The probability distribution for the
x component of the momentum for particles 1 to 8 in the nonequilibrium QOD system shown in
figure (3). These are the particles nearest the hot reservoir. The incoming distributions (the negative part of the
distribution) are very close to Gaussian for each particle, while the outgoing distributions show the same qualitative behaviour as the outgoing distributions in the equilibrium case. There is a strong effect in the outgoing distribution of particle 1, and that effect successively diminishes in particles 2, 3, etc. as the distribution slowly returns to its approximately Gaussian shape.
3.3. Stochastic Models
Eckmann and Young [
18] have developed Hamiltonian and stochastic models for heat transport in low dimensional systems. The Hamiltonian models consist of energy storage devices which couple to each other through the motion of tracer particles that carry the energy. Although these models principally store energy as rotational kinetic energy, they are similar in many ways to the QOD system that we consider. The Lorentz gas can be considered to be the model obtained by taking two disks on a torus and expanding one disk to twice its diameter and shrinking the other to a point. In this way the interaction between the scatterer and point is the same as the interaction between the two disks. In a similar way, expanding all the even numbered disks and contracting all the old numbered disks in the QOD system, leaves the interaction between neighbouring disks the same. The transformed QOD system then resembles an array of scatterers (the even numbered disks) with a tracer particle between each pair (the odd number disks), except that the QOD system does not lead to equally spaced scatterers. Therefore it is reasonable to suppose that the QOD system may be in the same universality class as those considered by Eckmann and Young and their result for the temperature profile of the system may be valid. That is
where
and
are fixed the temperatures at the left and right ends, and
x goes from 0 to 1 as we move from the left end to the right end. The constant
α depends on the nature of the coupling, and if the energy is purely kinetic a value of
is expected. The model is based on two very reasonable assumptions: that energy is shared in tracer-scatterer collisions, and that the speed of the tracer particles is proportional to some power of their kinetic energy. Therefore we would expect the speed and collision frequency to be highest near the hot reservoir and lowest near the cold reservoir. The rate at which a tracer does a cycle of interaction (interacting with both its neighbours) is the inverse of the time it takes to do two collisions. In a molecular dynamics simulation of the QOD system in
Figure 5 we observe exactly the opposite effect. The collision frequency is greatest near the cold reservoir and smallest near the hot reservoir.
In
Figure 6 we present the temperature profile calculated in the simulation. Clearly the temperature profile must decay from a value of
at the left-hand boundary to a value of
at the right-hand boundary. From the results of Eckmann and Young we expect a value of
, whereas the observed temperature profile in
Figure 6 implies a value of
. Further, even fitting the observed temperature profile to the function given in equation (15) with
α unconstrained gives a very poor fit. The assumption that is made about the collision rate in the stochastic model, although reasonable, is not correct for this system. One possible reason for this discrepancy may be the fact that the temperature gradient in the QOD system produces a density gradient where the hot region is less dense than the cold region. This would lead the QOD system to give tracer-scatterer system non-equally spaced scatterers.