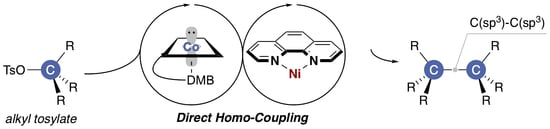
Ni/Co-Catalyzed Homo-Coupling of Alkyl Tosylates
Abstract
:1. Introduction
2. Results and Discussion
2.1. Screening of Reaction Conditions
2.2. Substrate Scope
2.3. Plausible Reaction Mechanism
3. Materials and Methods
3.1. General Information
3.2. General Procedure of the NiBr2phen/VB12-Catalyzed Homo-Coupling of Alkyl Tosylates
3.3. Product Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tasker, S.Z.; Standley, E.A.; Jamison, T.F. Recent advances in homogeneous nickel catalysis. Nature 2014, 509, 299–309. [Google Scholar] [CrossRef]
- Netherton, M.R.; Dai, C.Y.; Neuschütz, K.; Fu, G.C. Room-temperature alkyl-alkyl Suzuki cross-coupling of alkyl bromides that possess beta hydrogens. J. Am. Chem. Soc. 2001, 123, 10099–10100. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Fu, G.C. Palladium-Catalyzed Negishi Cross-Coupling Reactions of Unactivated Alkyl Iodides, Bromides, Chlorides, and Tosylates. J. Am. Chem. Soc. 2003, 125, 12527–12530. [Google Scholar] [CrossRef]
- Hadei, N.; Kantchev, E.A.B.; O’Brien, C.J.; Organ, M.G. Room-temperature Negishi cross-coupling of unactivated alkyl bromides with alkyl organozinc reagents utilizing a Pd/N-heterocyclic carbene catalyst. J. Org. Chem. 2005, 70, 8503–8507. [Google Scholar] [CrossRef] [PubMed]
- Burns, D.H.; Miller, J.D.; Chan, H.-K.; Delaney, M.O. Scope and Utility of a New Soluble Copper Catalyst [CuBr−LiSPh−LiBr−THF]: A Comparison with Other Copper Catalysts in Their Ability to Couple One Equivalent of a Grignard Reagent with an Alkyl Sulfonate. J. Am. Chem. Soc. 1997, 119, 2125–2133. [Google Scholar] [CrossRef]
- Terao, J.; Todo, H.; Begum, S.A.; Kuniyasu, H.; Kambe, N. Copper-catalyzed cross-coupling reaction of grignard reagents with primary-alkyl halides: Remarkable effect of 1-phenylpropyne. Angew. Chem. Int. Ed. 2007, 46, 2086–2089. [Google Scholar] [CrossRef] [PubMed]
- Devasagayaraj, A.; Stüdemann, T.; Knochel, P. A New Nickel-Catalyzed Cross-Coupling Reaction between sp3 Carbon Centers. Angew. Chem. Int. Ed. 1995, 34, 2723–2725. [Google Scholar] [CrossRef]
- Terao, J.; Ikumi, A.; Kuniyasu, H.; Kambe, N. Ni- or Cu-catalyzed cross-coupling reaction of alkyl fluorides with Grignard reagents. J. Am. Chem. Soc. 2003, 125, 5646–5647. [Google Scholar] [CrossRef]
- Saito, B.; Fu, G.C. Alkyl-alkyl suzuki cross-couplings of unactivated secondary alkyl halides at room temperature. J. Am. Chem. Soc. 2007, 129, 9602–9603. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Gagné, M.R. Diastereoselective Ni-Catalyzed Negishi Cross-Coupling Approach to Saturated, Fully Oxygenated C-Alkyl and C-Aryl Glycosides. J. Am. Chem. Soc. 2008, 130, 12177–12183. [Google Scholar] [CrossRef]
- Vechorkin, O.; Hu, X. Nickel-catalyzed cross-coupling of non-activated and functionalized alkyl halides with alkyl Grignard reagents. Angew. Chem. Int. Ed. 2009, 48, 2937–2940. [Google Scholar] [CrossRef] [PubMed]
- Qin, T.; Cornella, J.; Li, C.; Malins, L.R.; Edwards, J.T.; Kawamura, S.; Maxwell, B.D.; Eastgate, M.D.; Baran, P.S. A general alkyl-alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents. Science 2016, 352, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.; Choi, J.; Liu, A.T.; Slusarczyk, M.; Fu, G.C. A general, modular method for the catalytic asymmetric synthesis of alkylboronate esters. Science 2016, 354, 1265–1269. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.; Auffrant, A.; Felouat, A.; Gosmini, C. Cobalt-Catalyzed Reductive Allylation of Alkyl Halides with Allylic Acetates or Carbonates. Angew. Chem. Int. Ed. 2011, 50, 10402–10405. [Google Scholar] [CrossRef]
- Yu, X.; Yang, T.; Wang, S.; Xu, H.; Gong, H. Nickel-catalyzed reductive cross-coupling of unactivated alkyl halides. Org. Lett. 2011, 13, 2138–2141. [Google Scholar] [CrossRef]
- Dai, Y.; Wu, F.; Zang, Z.; You, H.; Gong, H. Ni-Catalyzed Reductive Allylation of Unactivated Alkyl Halides with Allylic Carbonates. Chem. Eur. J. 2012, 18, 808–812. [Google Scholar] [CrossRef]
- Xu, H.; Zhao, C.; Qian, Q.; Deng, W.; Gong, H. Nickel-catalyzed cross-coupling of unactivated alkyl halides using bis(pinacolato)diboron as reductant. Chem. Sci. 2013, 4, 4022. [Google Scholar] [CrossRef]
- Chen, H.; Jia, X.; Yu, Y.; Qian, Q.; Gong, H. Nickel-Catalyzed Reductive Allylation of Tertiary Alkyl Halides with Allylic Carbonates. Angew. Chem. Int. Ed. 2017, 129, 13283–13286. [Google Scholar] [CrossRef]
- Smith, R.T.; Zhang, X.; Rincon, J.A.; Agejas, J.; Mateos, C.; Barberis, M.; García-Cerrada, S.; de Frutos, O.; MacMillan, D.W.C. Metallaphotoredox-Catalyzed Cross-Electrophile Csp3-Csp3 Coupling of Aliphatic Bromides. J. Am. Chem. Soc. 2018, 140, 17433–17438. [Google Scholar] [CrossRef]
- Goldup, S.M.; Leigh, D.A.; McBurney, R.T.; McGonigal, P.R.; Plant, A. Ligand-assisted nickel-catalysed sp3–sp3 homocoupling of unactivated alkyl bromides and its application to the active template synthesis of rotaxanes. Chem. Sci. 2010, 1, 383. [Google Scholar] [CrossRef]
- Prinsell, M.R.; Everson, D.A.; Weix, D.J. Nickel-catalyzed, sodium iodide-promoted reductive dimerization of alkyl halides, alkyl pseudohalides, and allylic acetates. Chem. Commun. 2010, 46, 5743–5745. [Google Scholar] [CrossRef]
- Peng, Y.; Luo, L.; Yan, C.S.; Zhang, J.-J.; Wang, Y.W. Ni-Catalyzed Reductive Homocoupling of Unactivated Alkyl Bromides at Room Temperature and Its Synthetic Application. J. Org. Chem. 2013, 78, 10960–10967. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Qian, X.; Gosmini, C. Cobalt-Catalyzed Csp3−Csp3 Homocoupling. Adv. Synth. Catal. 2016, 358, 2427–2430. [Google Scholar] [CrossRef]
- Yang, C.-T.; Zhang, Z.-Q.; Liang, J.; Liu, J.-H.; Lu, X.-Y.; Chen, H.-H.; Liu, L. Copper-catalyzed cross-coupling of nonactivated secondary alkyl halides and tosylates with secondary alkyl Grignard reagents. J. Am. Chem. Soc. 2012, 134, 11124–11127. [Google Scholar] [CrossRef]
- Powell, D.A.; Fu, G.C. Nickel-Catalyzed Cross-Couplings of Organosilicon Reagents with Unactivated Secondary Alkyl Bromides. J. Am. Chem. Soc. 2004, 126, 7788–7789. [Google Scholar] [CrossRef] [PubMed]
- Powell, D.A.; Maki, T.; Fu, G.C. Stille cross-couplings of unactivated secondary alkyl halides using monoorganotin reagents. J. Am. Chem. Soc. 2005, 127, 510–511. [Google Scholar] [CrossRef] [PubMed]
- González-Bobes, F.; Fu, G.C. Amino Alcohols as Ligands for Nickel-Catalyzed Suzuki Reactions of Unactivated Alkyl Halides, Including Secondary Alkyl Chlorides, with Arylboronic Acids. J. Am. Chem. Soc. 2006, 128, 5360–5361. [Google Scholar] [CrossRef] [Green Version]
- Dudnik, A.S.; Fu, G.C. Nickel-catalyzed coupling reactions of alkyl electrophiles, including unactivated tertiary halides, to generate carbon-boron bonds. J. Am. Chem. Soc. 2012, 134, 10693–10697. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Fu, G.C. Nickel-Catalyzed Alkyl-Alkyl Cross-Couplings of Fluorinated Secondary Electrophiles: A General Approach to the Synthesis of Compounds having a Perfluoroalkyl Substituent. Angew. Chem. Int. Ed. 2015, 54, 9047–9051. [Google Scholar] [CrossRef]
- Lévêque, C.; Corcé, V.; Chenneberg, L.; Ollivier, C.; Fensterbank, L. Photoredox/Nickel Dual Catalysis for the C(sp3)–C(sp3) Cross-Coupling of Alkylsilicates with Alkyl Halides. Eur. J. Org. Chem. 2017, 2017, 2118–2121. [Google Scholar] [CrossRef]
- Lu, X.; Wang, Y.; Zhang, B.; Pi, J.-J.; Wang, X.-X.; Gong, T.-J.; Xiao, B.; Fu, Y. Nickel-Catalyzed Defluorinative Reductive Cross-Coupling of gem-Difluoroalkenes with Unactivated Secondary and Tertiary Alkyl Halides. J. Am. Chem. Soc. 2017, 139, 12632–12637. [Google Scholar] [CrossRef] [PubMed]
- Komeyama, K.; Ohata, R.; Kiguchi, S.; Osaka, I. Highly nucleophilic vitamin B12-assisted nickel-catalysed reductive coupling of aryl halides and non-activated alkyl tosylates. Chem. Commun. 2017, 53, 6401–6404. [Google Scholar] [CrossRef] [PubMed]
- Komeyama, K.; Yamahata, Y.; Osaka, I. Nickel and Nucleophilic Cobalt-Catalyzed Trideuteriomethylation of Aryl Halides Using Trideuteriomethyl p-Toluenesulfonate. Org. Lett. 2018, 20, 4375–4378. [Google Scholar] [CrossRef]
- Ito, S.; Fujiwara, Y.-I.; Nakamura, E.; Nakamura, M. Iron-Catalyzed Cross-Coupling of Alkyl Sulfonates with Arylzinc Reagents. Org. Lett. 2009, 11, 4306–4309. [Google Scholar] [CrossRef] [PubMed]
- Closson, W.D.; Wriede, P.; Bank, S. Reductive Cleavage of Toluenesulfonates with Sodium Naphthalene 1. J. Am. Chem. Soc. 1966, 88, 1581–1583. [Google Scholar] [CrossRef]
- Lipshutz, B.H.; Wilhelm, R.S.; Nugent, S.T.; Little, R.D.; Baizer, M.M. Electrochemical peak potentials of typical substrates used for coupling reactions with organocuprates: Effects of solvent. J. Org. Chem. 1983, 48, 3306–3308. [Google Scholar] [CrossRef]
- Sridhar, M.; Kumar, B.A.; Narender, R. Expedient and simple method for regeneration of alcohols from toluenesulfonates using Mg-MeOH. Tetrahedron Lett. 1998, 39, 2847–2850. [Google Scholar] [CrossRef]
- Kotsuki, H.; Kadota, I.; Ochi, M. A new expeditious synthesis of (+)-exo-brevicomin via efficient C-C bond formation of triflates. Tetrahedron Lett. 1989, 30, 3999–4000. [Google Scholar] [CrossRef]
- O’Donnel, C.J.; Burke, S.D. Selective Mesylation of Vicinal Diols: A Systematic Case Study. J. Org. Chem. 1998, 63, 8614–8616. [Google Scholar] [CrossRef]
- Bouzide, A.; Sauvé, G. Silver(I) Oxide Mediated Highly Selective Monotosylation of Symmetrical Diols. Application to the Synthesis of Polysubstituted Cyclic Ethers. Org. Lett. 2002, 4, 2329–2332. [Google Scholar] [CrossRef] [PubMed]
- Ram, M.S.; Riordan, C.G. Methyl transfer from a cobalt complex to Ni(tmc)+ yielding Ni(tmc)Me+: A model for methylcobalamin alkylation of CO dehydrogenase. J. Am. Chem. Soc. 1995, 117, 2365–2366. [Google Scholar] [CrossRef]
- Eckert, N.A.; Dougherty, W.G.; Yap, G.P.A.; Riordan, C.G. Methyl Transfer from Methylcobaloxime to (Triphos)Ni(PPh3): Relevance to the Mechanism of Acetyl Coenzyme A Synthase. J. Am. Chem. Soc. 2007, 129, 9286–9287. [Google Scholar] [CrossRef]
- Gansäuer, A.; Fleckhaus, A.; Lafont, M.A.; Okkel, A.; Kotsis, K.; Anoop, A.; Neese, F. Catalysis via Homolytic Substitutions with C−O and Ti−O Bonds: Oxidative Additions and Reductive Eliminations in Single Electron Steps. J. Am. Chem. Soc. 2009, 131, 16989–16999. [Google Scholar] [CrossRef]
- Martin, B.D.; Finke, R.G. Cobalt-carbon homolysis and bond dissociation energy studies of biological alkylcobalamins: Methylcobalamin, including a > 1015 Co-CH3 homolysis rate enhancement at 25 °C following one-electron reduction. J. Am. Chem. Soc. 1990, 112, 2419–2420. [Google Scholar] [CrossRef]
- Birke, R.L.; Huang, Q.; Spataru, T.; Gosser, D.K. Electroreduction of a Series of Alkylcobalamins: Mechanism of Stepwise Reductive Cleavage of the Co−C Bond. J. Am. Chem. Soc. 2006, 128, 1922–1936. [Google Scholar] [CrossRef]
- Bialek, M.; Cramail, H.; Deffieux, A.; Guillaume, S.M. Styrene polymerization using nickel(II) complexes as catalysts. Eur. Polym. J. 2005, 41, 2678–2684. [Google Scholar] [CrossRef]
- Panagiotopoulos, A.; Ladomenou, K.; Sun, D.; Artero, V.; Coutsolelos, A.G. Photochemical hydrogen production and cobaloximes: The influence of the cobalt axial N-ligand on the system stability. Dalton Trans. 2016, 45, 6732–6738. [Google Scholar] [CrossRef]
- Bissember, A.C.; Levina, A.; Fu, G.C. A Mild, Palladium-Catalyzed Method for the Dehydrohalogenation of Alkyl Bromides: Synthetic and Mechanistic Studies. J. Am. Chem. Soc. 2012, 134, 14232–14237. [Google Scholar] [CrossRef] [Green Version]
- Komber, H.; Müllers, S.; Lombeck, F.; Held, A.; Walter, M.; Sommer, M. Soluble and stable alternating main-chain merocyanine copolymers through quantitative spiropyran–merocyanine conversion. Polym. Chem. 2013, 5, 443–453. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |






| Entry | Ni catalysts | Yield of 2a (%) a | Conversion of 1a (%) a |
|---|---|---|---|
| 1 | NibpyCl2 | 72 | 100 |
| 2 b | NibpyCl2 | 0 | 20 |
| 3 | None | 0 | 32 |
| 4 c | NibpyCl2 | 0 | 0 |
| 5 d | NibpyCl2 | 38 | 65 |
| 6 | Ni(4,4’-(MeO2C)2bpy)Cl2 | 38 | 76 |
| 7 | Ni(4,4’-tBu2bpy)Cl2 | 18 | 67 |
| 8 | Ni(4,4’-Mes2bpy)Cl2 e | 38 | 78 |
| 9 | Ni[4,4’-(MeO)2bpy]Cl2 | 10 | 66 |
| 10 | Ni(6-Mebpy)Cl2 | 42 | 92 |
| 11 | Ni(6,6’-Me2bpy)Cl2 | 17 | 95 |
| 12 | Ni(1,10-phen)Cl2 | 75 | 100 |
| 13 | Ni(4,7-Ph2phen)Cl2 | 60 | 100 |
| 14 f | NiphenCl2 | 82 | 100 |
| 15 g | NiphenCl2 | 0 | 0 |
| 16 f | NiphenBr2 | 93 | 100 |
| 17 h | NiphenBr2 | 21–28 | 51–59 |

| Entry | Alkyl Tosylates 1 | Product 2 and Yield (%) a | |||
|---|---|---|---|---|---|
| 1 |  | 1b |  | 2b | 86 |
| 2 |  | 1c |  | 2c | 70 |
| 3 |  | 1d |  | 2d | 67 |
| 4 |  | 1e |  | 2e | 63 |
| 5 |  | 1f |  | 2f | 65 |
| 6 |  | 1g |  | 2g | 75 |
| 7 |  | 1h |  | 2h | 70 |
| 8 b |  | 1i |  | 2i | 60 |
| 9 |  | 1j |  | 2j | 73 |
| 10 |  | 1k |  | 2k | 90 [1:1] c |
| 11 d,e |  | 1l |  | 2l | 80 [1:1] c |
| 12 e, f |  | 1m |  | 2m | 65 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komeyama, K.; Tsunemitsu, R.; Michiyuki, T.; Yoshida, H.; Osaka, I. Ni/Co-Catalyzed Homo-Coupling of Alkyl Tosylates. Molecules 2019, 24, 1458. https://doi.org/10.3390/molecules24081458
Komeyama K, Tsunemitsu R, Michiyuki T, Yoshida H, Osaka I. Ni/Co-Catalyzed Homo-Coupling of Alkyl Tosylates. Molecules. 2019; 24(8):1458. https://doi.org/10.3390/molecules24081458
Chicago/Turabian StyleKomeyama, Kimihiro, Ryusuke Tsunemitsu, Takuya Michiyuki, Hiroto Yoshida, and Itaru Osaka. 2019. "Ni/Co-Catalyzed Homo-Coupling of Alkyl Tosylates" Molecules 24, no. 8: 1458. https://doi.org/10.3390/molecules24081458