Antinociceptive Effect of Intrathecal Microencapsulated Human Pheochromocytoma Cell in a Rat Model of Bone Cancer Pain
Abstract
:1. Introduction
2. Results and Discussion
2.1. Bone Destruction Evaluation by Radiograph
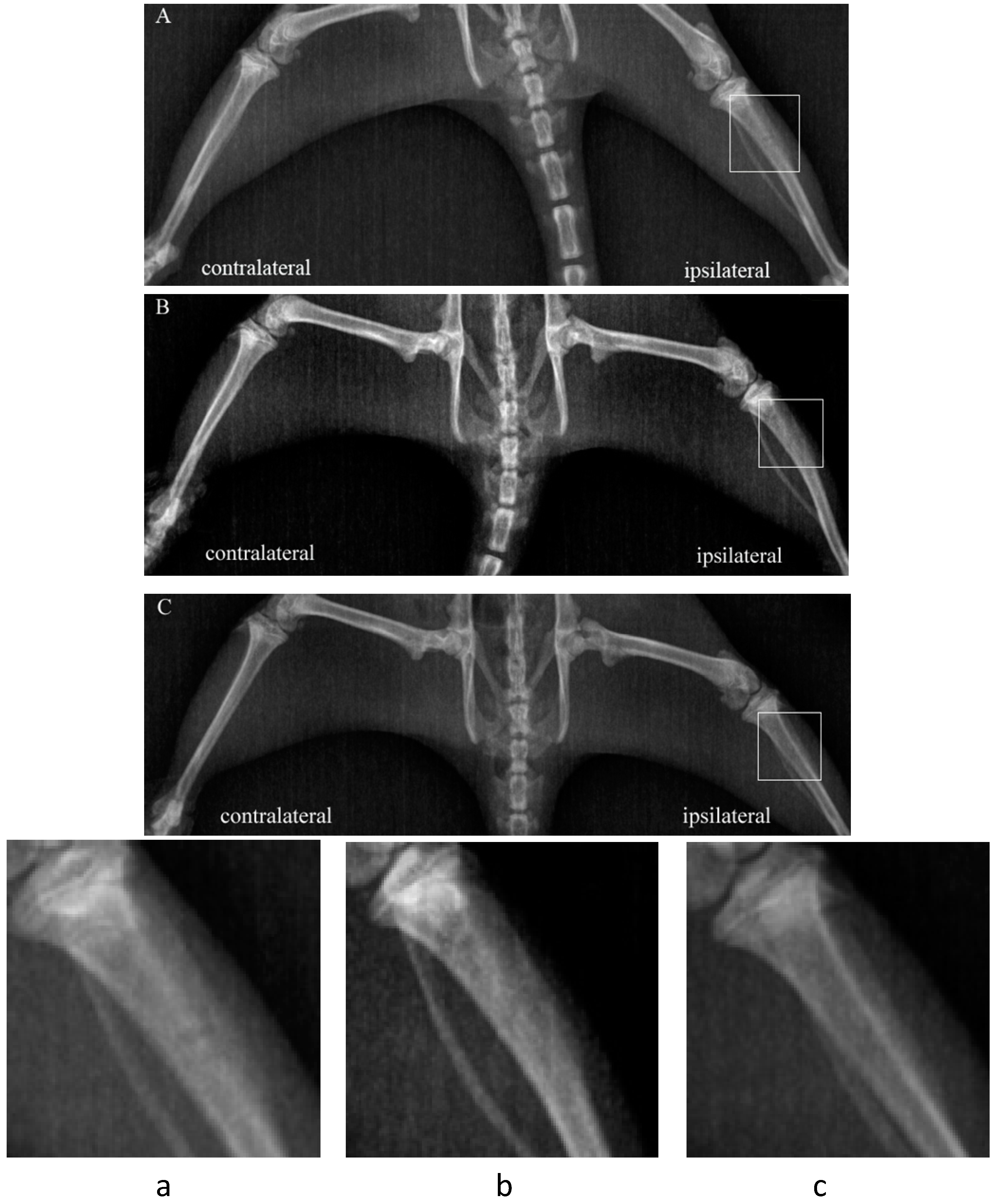
2.2. Time-Course of Mechanical Hyperalgesia Induced by Walker 256 Inoculation

2.3. Secretion of Micro-HPC (Microencapsulated Human Pheochromocytoma Cell) in Vivo and in Vitro
| Time (Pre- and Post-Microencapsulation of HPC) | Met-Enkephalin (pg/µL) | Norepinephrine (ng/µL) |
|---|---|---|
| Pre-microencapsulation | 3.14 ± 1.05 | 3.62 ± 0.61 |
| Day2 (post-microencapsulation) | 3.23 ± 0.79 | 3.59 ± 0.43 |
| Day4 (post-microencapsulation) | 3.09 ± 0.63 | 3.65 ± 0.95 |
| Day6 (post-microencapsulation) | 3.19 ± 0.48 | 3.54 ± 0.37 |
| Group | Met-Enkephalin (pg/µL) | Norepinephrine (ng/µL) |
|---|---|---|
| CIP + empty capsule | 6.50 + 0.21 | 2.85 + 0.15 |
| CIP + micro-HPC | 8.93 + 0.65 * | 5.54 + 0.73 * |
2.4. Intrathecal Injection of Micro-HPC Attenuated Mechanical Allodynia Induced by Walker 256 Inoculation
2.5. Antagonizing Opioid Signaling Reversed the Analgesic Effect of Micro-HPC on Mechanical Hyperalgesia

2.6. Discussion
3. Experimental Section
3.1. Animals
3.2. Cells
3.3. Microencapsulation
3.4. Bone Cancer Model and Radiology
3.5. Mechanical Hyperalgesia Test
3.6. Thermal Hyperalgesia
3.7. Secretion Study of Cells in Vitro and in Vivo
3.8. Antagonist Study
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Banning, A.; Sjogren, P.; Henriksen, H. Pain causes in 200 patients referred to a multidisciplinary cancer pain clinic. Pain 1991, 45, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.E. Skeletal complications of malignancy. Cancer 1997, 80, 1588–1594. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.E. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin. Cancer Res. 2006, 12, 6243–6249. [Google Scholar] [CrossRef]
- Sehgal, N.; Colson, J.; Smith, H.S. Chronic pain treatment with opioid analgesics: Benefits vs. harms of long-term therapy. Expert Rev. Neurother. 2013, 13, 1201–1220. [Google Scholar] [CrossRef] [PubMed]
- Abramowitz, L.; Beziaud, N.; Labreze, L.; Giardina, V.; Causse, C.; Chuberre, B.; Allaert, F.A.; Perrot, S. Prevalence and impact of constipation and bowel dysfunction induced by strong opioids: A cross-sectional survey of 520 patients with cancer pain: DYONISOS study. J. Med. Econ. 2013, 16, 1423–1433. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, L.F.; Costa-Pereira, A.; Mendonca, L.; Dias, C.C.; Castro-Lopes, J.M. A population-based study on chronic pain and the use of opioids in Portugal. Pain 2013, 154, 2844–2852. [Google Scholar] [CrossRef] [PubMed]
- Lazorthes, Y.; Sagen, J.; Sallerin, B.; Tkaczuk, J.; Duplan, H.; Sol, J.C.; Tafani, M.; Bes, J.C. Human chromaffin cell graft into the CSF for cancer pain management: A prospective phase II clinical study. Pain 2000, 87, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Ginzburg, R.; Seltzer, Z. Subarachnoid spinal cord transplantation of adrenal medulla suppresses chronic neuropathic painbehavior in rats. Brain Res. 1990, 523, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Lazorthes, Y.; Bes, J.C.; Sagen, J.; Tafani, M.; Tkaczuk, J.; Sallerin, B.; Nahri, I.; Verdie, J.C.; Ohayon, E.; Caratero, C.; et al. Transplantation of human chromaffin cells for control of intractable cancer pain. Acta Neurochir. Suppl. 1995, 64, 97–100. [Google Scholar] [PubMed]
- Sagen, J.; Wang, H.; Tresco, P.A.; Aebischer, P. Transplants of immunologically isolated xenogeneic chromaffin cells provide a long-term source of pain-reducing neuroactive substances. J. Neurosci. 1993, 13, 2415–2423. [Google Scholar] [PubMed]
- Hama, A.T.; Sagen, J. Reduced pain-related behavior by adrenal medullary transplants in rats with experimental painful peripheral neuropathy. Pain 1993, 52, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Ma, C.; Li, G.; Mai, M.; Wu, Y. Intrathecal implantation of microencapsulated PC12 cells reduces cold allodynia in a rat model of neuropathic pain. Artif. Organs 2011, 35, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Pappas, G.D.; Lazorthes, Y.; Bes, J.C.; Tafani, M.; Winnie, A.P. Relief of intractable cancer pain by human chromaffin cell transplants: Experience at two medical centers. Neurol. Res. 1997, 19, 71–77. [Google Scholar] [PubMed]
- Pellizzari, E.H.; Barontini, M.; Figuerola, M.L.; Cigorraga, S.B.; Levin, G. Possible autocrine enkephalin regulation of catecholamine release in human pheochromocytoma cells. Life Sci. 2008, 83, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Tkaczuk, J.; Bes, J.C.; Duplan, H.; Sallerin, B.; Tafani, M.; Charlet, J.P.; Abbal, M.; Lazorthes, Y.; Ohayon, E. Intrathecal grafting of unencapsulated adrenal medullary tissue can bring CD4 T lymphocytes into CSF: A potentially deleterious event for the graft. Cell Transpl. 2000, 9, 79–91. [Google Scholar]
- Wang, H.; Sagen, J. Optimization of adrenal medullary allograft conditions for pain alleviation. J. Neural Transpl. Plast. 1994, 5, 49–64. [Google Scholar] [CrossRef]
- Duvivier-Kali, V.F.; Omer, A.; Parent, R.J.; O’Neil, J.J.; Weir, G.C. Complete protection of islets against allorejection and autoimmunity by a simple barium-alginate membrane. Diabetes 2001, 50, 1698–1705. [Google Scholar] [CrossRef] [PubMed]
- Acarregui, A.; Pedraz, J.L.; Blanco, F.J.; Hernandez, R.M.; Orive, G. Hydrogel-based scaffolds for enclosing encapsulated therapeutic cells. Biomacromolecules 2013, 14, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Chayosumrit, M.; Tuch, B.; Sidhu, K. Alginate microcapsule for propagation and directed differentiation of hESCs to definitive endoderm. Biomaterials 2010, 31, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.; Wang, Y.; Li, S.; Wang, W.; Gu, R.; Guo, X.; Nan, X.; Ma, X.; Pei, X. Treatment of acute hepatic failure in mice by transplantation of mixed microencapsulation of rat hepatocytes and transgenic human fetal liver stromal cells. Tissue Eng. Part C Methods 2010, 16, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Murua, A.; Orive, G.; Hernandez, R.M.; Pedraz, J.L. Cryopreservation based on freezing protocols for the long-term storage of microencapsulated myoblasts. Biomaterials 2009, 30, 3495–3501. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.X.; Liu, B.; Wang, L.; Ren, K.; Qiao, J.T.; Berman, B.M.; Lao, L. Spinal glial activation in a new rat model of bone cancer pain produced by prostate cancer cell inoculation of the tibia. Pain 2005, 118, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Song, Y.; Song, C.; Xu, P.; Song, C. Successful xenotransplantation of microencapsulated newborn pig parathyroid cells in the treatment of hypoparathyroidism in rats. Chin. Med. J. Engl. 2003, 116, 1161–1165. [Google Scholar] [PubMed]
- Tam, S.K.; de Haan, B.J.; Faas, M.M.; Halle, J.P.; Yahia, L.; de Vos, P. Adsorption of human immunoglobulin to implantable alginate-poly-L-lysine microcapsules: Effect of microcapsule composition. J. Biomed. Mater Res. A 2009, 89, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.F.; Liu, R.; Li, B.G.; Lou, J.R.; Dai, K.R.; Tang, T.T. Biologic effect and immunoisolating behavior of BMP-2 gene-transfected bone marrow-derived mesenchymal stem cells in APA microcapsules. Biochem. Biophys. Res. Commun. 2007, 362, 923–927. [Google Scholar] [CrossRef] [PubMed]
- Orive, G.; Ponce, S.; Hernandez, R.M.; Gascon, A.R.; Igartua, M.; Pedraz, J.L. Biocompatibility of microcapsules for cell immobilization elaborated with different type of alginates. Biomaterials 2002, 23, 3825–3831. [Google Scholar] [CrossRef] [PubMed]
- Murua, A.; de Castro, M.; Orive, G.; Hernandez, R.M.; Pedraz, J.L. In vitro characterization and in vivo functionality of erythropoietin-secreting cells immobilized in alginate-poly-l-lysine-alginate microcapsules. Biomacromolecules 2007, 8, 3302–3307. [Google Scholar] [CrossRef] [PubMed]
- Leblond, F.A.; Tessier, J.; Halle, J.P. Quantitative method for the evaluation of biomicrocapsule resistance to mechanical stress. Biomaterials 1996, 17, 2097–2102. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.; Baek, W.Y.; Chung, S.H.; Shin, N.; Kim, H.R.; Lee, S.A. Cultured human chromaffin cells grafted in spinal subarachnoid space relieves allodynia in a pain rat model. Korean J. Anesthesiol. 2011, 60, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Jozan, S.; Aziza, J.; Châtelin, S.; Evra, C.; Courtade-Saïdi, M.; Parant, O.; Sol, J.C.; Zhou, H.; Lazorthes, Y. Human fetal chromaffin cells: A potential tool for cell pain therapy. Exp. Neurol. 2007, 205, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Gulwadi, A.G.; Hoane, M.R.; Saydoff, J.A.; Frydel, B.R.; Lindner, M.D. No detectable analgesic effects in the formalin test even with one million bovine adrenal chromaffin cells. Pain 2002, 99, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Lindner, M.D.; Francis, J.M.; Plone, M.A.; McDermott, P.E.; Frydel, B.R.; Emerich, D.F.; Saydoff, J.A. The analgesic potential of intraventricular polymer-encapsulated adrenal chromaffin cells in a rodent model of chronic neuropathic pain. Exp. Clin. Psychopharmacol. 2000, 8, 524–538. [Google Scholar] [CrossRef] [PubMed]
- Budai, D.; Fields, H.L. Endogenous opioid peptides acting at mu-opioid receptors in the dorsal horn contribute to midbrain modulation of spinal nociceptive neurons. J. Neurophysiol. 1998, 79, 677–687. [Google Scholar] [PubMed]
- Chen, W.; Song, B.; Lao, L.; Perez, O.A.; Kim, W.; Marvizon, J.C. Comparing analgesia and mu-opioid receptor internalization produced by intrathecal enkephalin: Requirement for peptidase inhibition. Neuropharmacology 2007, 53, 664–676. [Google Scholar] [CrossRef] [PubMed]
- Takemori, A.E.; Portoghese, P.S. Enkephalin antinociception in mice is mediated by delta 1- and delta 2-opioid receptors in the brain and spinal cord, respectively. Eur. J. Pharmacol. 1993, 242, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Pertovaara, A. Noradrenergic pain modulation. Prog. Neurobiol. 2006, 80, 53–83. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.D. Pheochromocytoma of the Adrenal gland Scaled Score (PASS) to separate benign from malignant neoplasms: A clinicopathologic and immunophenotypic study of 100 cases. Am. J. Surg. Pathol. 2002, 26, 551–566. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Jeon, Y.H.; Jin, G.C.; Lim, J.O.; Baek, W.Y. Immunoisolated chromaffin cells implanted into the subarachnoid space of rats reduce cold allodynia in a model of neuropathic pain: a novel application of microencapsulation technology. Artif. Organs 2004, 28, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.R.; Gao, X.F.; Wu, J.X.; Lu, Z.J.; Huang, Z.X.; Li, X.Q.; He, C.; Yu, W.F. Bilateral downregulation of Nav1.8 in dorsal root ganglia of rats with bone cancer pain induced by inoculation with Walker 256 breast tumor cells. BMC Cancer 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 1994, 53, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, K.; Dubner, R.; Brown, F.; Flores, C.; Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988, 32, 77–88. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, X.; Li, G.; Wu, S.; Zhang, B.; Wan, Q.; Yu, D.; Zhou, R.; Ma, C. Antinociceptive Effect of Intrathecal Microencapsulated Human Pheochromocytoma Cell in a Rat Model of Bone Cancer Pain. Int. J. Mol. Sci. 2014, 15, 12135-12148. https://doi.org/10.3390/ijms150712135
Li X, Li G, Wu S, Zhang B, Wan Q, Yu D, Zhou R, Ma C. Antinociceptive Effect of Intrathecal Microencapsulated Human Pheochromocytoma Cell in a Rat Model of Bone Cancer Pain. International Journal of Molecular Sciences. 2014; 15(7):12135-12148. https://doi.org/10.3390/ijms150712135
Chicago/Turabian StyleLi, Xiao, Guoqi Li, Shaoling Wu, Baiyu Zhang, Qing Wan, Ding Yu, Ruijun Zhou, and Chao Ma. 2014. "Antinociceptive Effect of Intrathecal Microencapsulated Human Pheochromocytoma Cell in a Rat Model of Bone Cancer Pain" International Journal of Molecular Sciences 15, no. 7: 12135-12148. https://doi.org/10.3390/ijms150712135





