Expression of Kir2.1 Inward Rectifying Potassium Channels in Optic Nerve Glia: Evidence for Heteromeric Association with Kir4.1 and Kir5.1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Optic Nerve Explant Cultures
2.2. Optic Nerve Explant Cultures
2.3. Immunocytochemistry
2.4. Image Capture and Analysis
2.5. Western Blot and Co-Immunoprecipitation
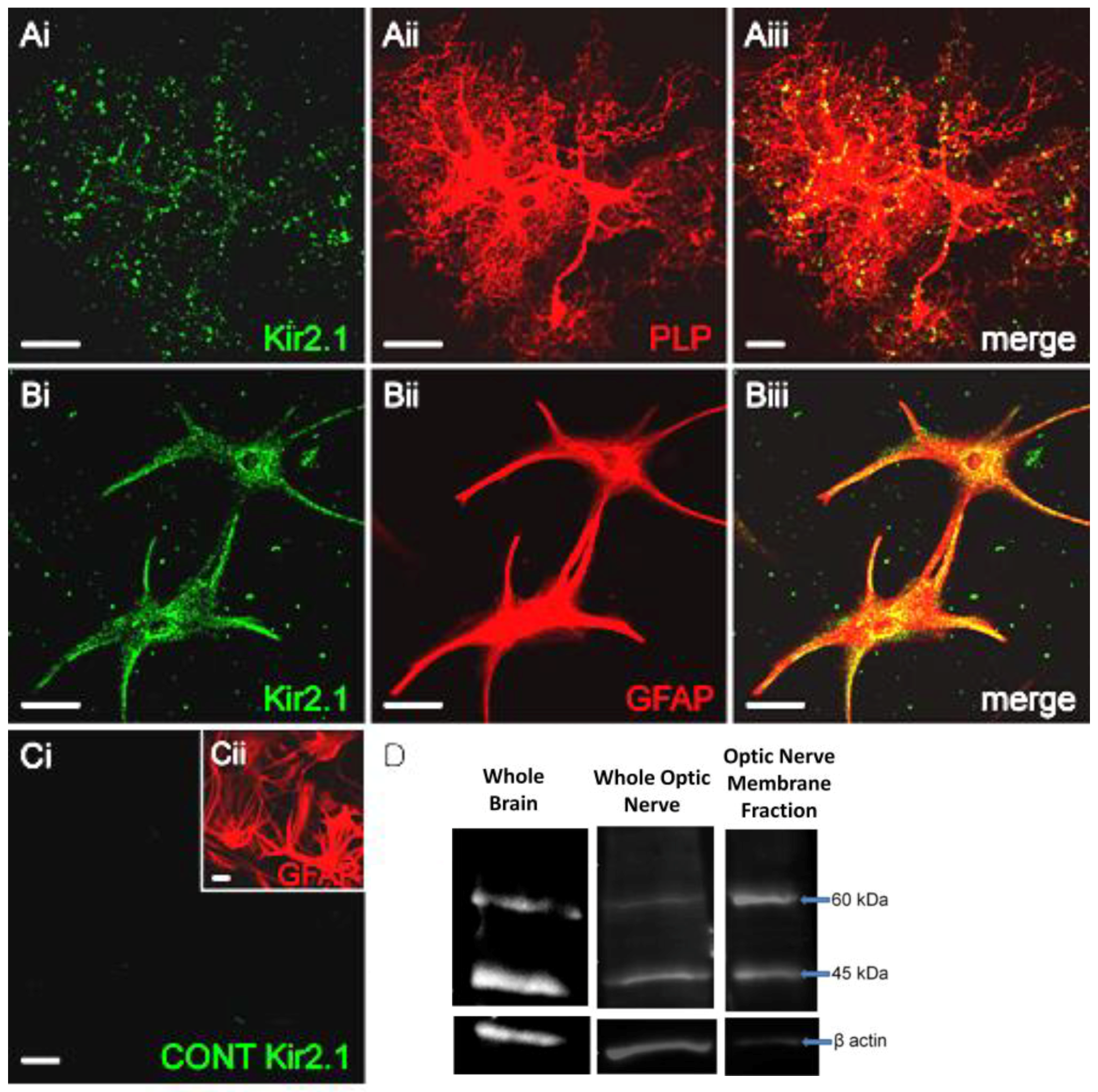
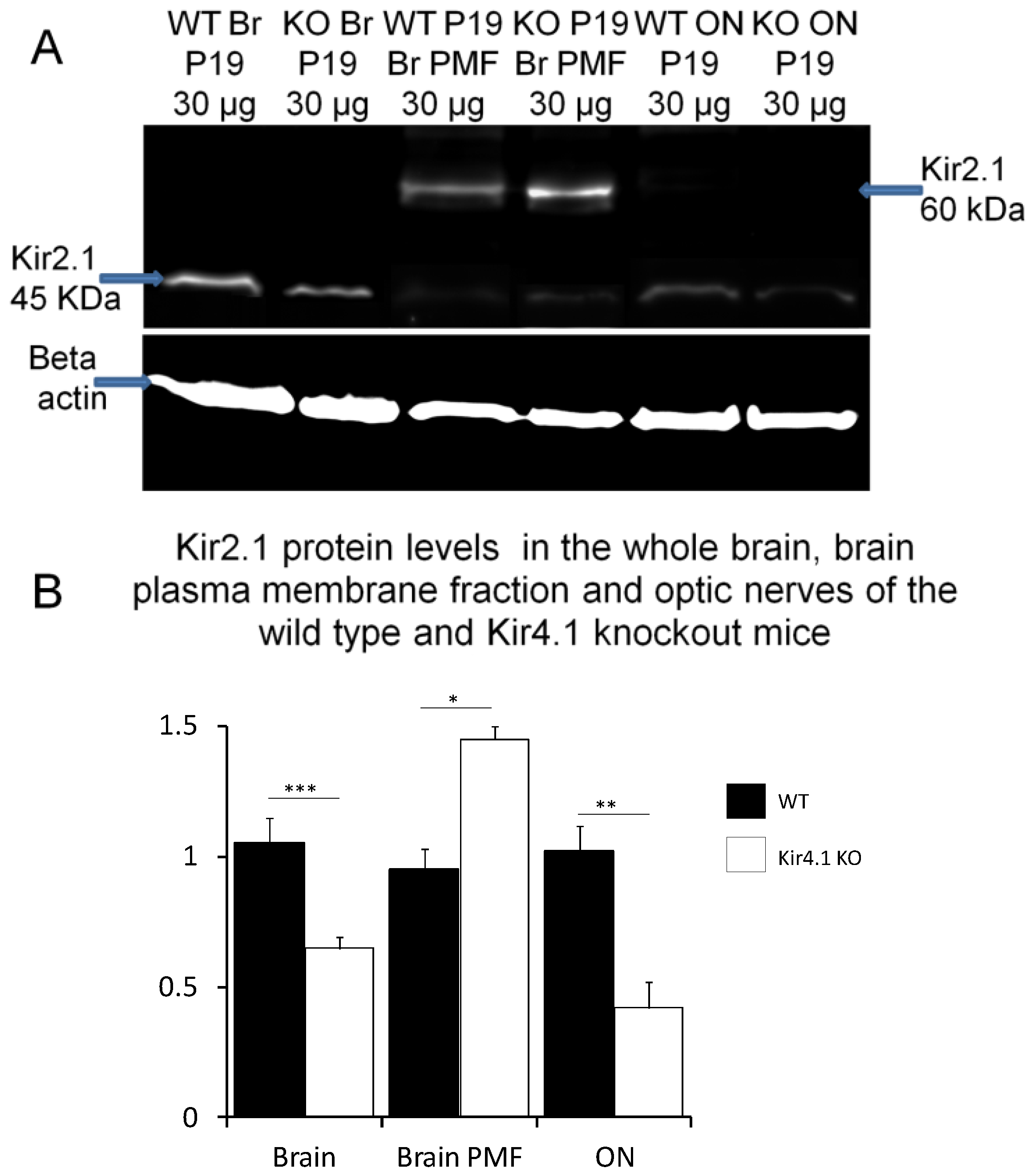
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Butt, A.M.; Kalsi, A. Inwardly rectifying potassium channels (Kir) in central nervous system glia: A special role for Kir4.1 in glial functions. J. Cell. Mol. Med. 2006, 10, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Kofuji, P.; Connors, N.C. Molecular substrates of potassium spatial buffering in glial cells. Mol. Neurobiol. 2003, 28, 195–208. [Google Scholar] [CrossRef]
- Poopalasundaram, S.; Knott, C.; Shamotienko, O.G.; Foran, P.G.; Dolly, J.O.; Ghiani, C.A.; Gallo, V.; Wilkin, G.P. Glial heterogeneity in expression of the inwardly rectifying K+ channel, Kir4.1, in adult rat CNS. Glia 2000, 30, 362–372. [Google Scholar] [CrossRef]
- Kalsi, A.S.; Greenwood, K.; Wilkin, G.; Butt, A.M. Kir4.1 expression by astrocytes and oligodendrocytes in CNS white matter: A developmental study in the rat optic nerve. J. Anat. 2004, 204, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Brasko, C.; Hawkins, V.; De La Rocha, I.C.; Butt, A.M. Expression of Kir4.1 and Kir5.1 inwardly rectifying potassium channels in oligodendrocytes, the myelinating cells of the CNS. Brain Struct. Funct. 2017, 222, 41–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, X.; Taniguchi, K.; Kofuji, P. Heterogeneity of Kir4.1 channel expression in glia revealed by mouse transgenesis. Glia 2009, 57, 1706–1715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Howe, M.W.; Feig, S.L.; Osting, S.M.; Haberly, L.B. Cellular and subcellular localization of Kir2.1 subunits in neurons and glia in piriform cortex with implications for K+ spatial buffering. J. Comp. Neurol. 2008, 506, 877–893. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Cho, S.H.; Park, K.; Yi, J.; Yoo, S.J.; Shin, K.S. Expression of Kir2.1 channels in astrocytes under pathophysiological conditions. Mol. Cells 2008, 25, 124–130. [Google Scholar] [PubMed]
- Schröder, W.; Seifert, G.; Hüttmann, K.; Hinterkeuser, S.; Steinhäuser, C. AMPA receptor-mediated modulation of inward rectifier K+ channels in astrocytes of mouse hippocampus. Mol. Cell. Neurosci. 2002, 19, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Stonehouse, A.H.; Pringle, J.H.; Norman, R.I.; Stanfield, P.R.; Conley, E.C.; Brammar, W.J. Characterisation of Kir2.0 proteins in the rat cerebellum and hippocampus by polyclonal antibodies. Histochem. Cell Biol. 1999, 112, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Prüss, H.; Derst, C.; Lommel, R.; Veh, R.W. Differential distribution of individual subunits of strongly inwardly rectifying potassium channels (Kir2 family) in rat brain. Mol. Brain Res. 2005, 139, 63–79. [Google Scholar] [CrossRef] [PubMed]
- Kofuji, P.; Biedermann, B.; Siddharthan, V.; Raap, M.; Iandiev, I.; Milenkovic, I.; Thomzig, A.; Veh, R.W.; Bringmann, A.; Reichenbach, A. Kir potassium channel subunit expression in retinal glial cells: Implications for spatial potassium buffering. Glia 2002, 39, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Hibino, H.; Inanobe, A.; Furutani, K.; Murakami, S.; Findlay, I.A.N.; Kurachi, Y. Inwardly rectifying potassium channels: Their structure, function, and physiological roles. Physiol. Rev. 2010, 90, 291–366. [Google Scholar] [CrossRef] [PubMed]
- Ulbricht, E.; Pannicke, T.; Hollborn, M.; Raap, M.; Goczalik, I.; Iandiev, I.; Härtig, W.; Uhlmann, S.; Wiedemann, P.; Reichenbach, A.; et al. Proliferative gliosis causes mislocation and inactivation of inwardly rectifying K+ (Kir) channels in rabbit retinal glial cells. Exp. Eye Res. 2008, 86, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Hibino, H.; Fujita, A.; Iwai, K.; Yamada, M.; Kurachi, Y. Differential assembly of inwardly rectifying K+ channel subunits, Kir4.1 and Kir5.1, in brain astrocytes. J. Biol. Chem. 2004, 279, 44065–44073. [Google Scholar] [CrossRef] [PubMed]
- Ishii, M.; Fujita, A.; Iwai, K.; Kusaka, S.; Higashi, K.; Inanobe, A.; Hibino, H.; Kurachi, Y. Differential expression and distribution of Kir5.1 and Kir4.1 inwardly rectifying K+ channels in retina. Am. J. Physiol. Cell Physiol. 2003, 285, C260–C267. [Google Scholar] [CrossRef] [PubMed]
- Lichter-Konecki, U.; Mangin, J.M.; Gordish-dressman, H.; Hoffman, E.P.; Gallo, V. Gene expression profiling of astrocytes from hyperammonemic mice reveals altered pathways for water and potassium homeostasis in vivo. Glia 2008, 56, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Mulkey, D.K.; Wenker, I.C. Astrocyte chemoreceptors: Mechanisms of H+ sensing by astrocytes in the retrotrapezoid nucleus and their possible contribution to respiratory drive. Exp. Physiol. 2011, 96, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Puissant, M.M.; Mouradian, G.C., Jr.; Liu, P.; Hodges, M.R. Identifying candidate genes that underlie cellular pH sensitivity in serotonin neurons using transcriptomics: A potential role for Kir5.1 channels. Front. Cell. Neurosci. 2017, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Raap, M.; Biedermann, B.; Braun, P.; Milenkovic, I.; Skatchkov, S.N.; Bringmann, A.; Reichenbach, A. Diversity of Kir channel subunit mRNA expressed by retinal glial cells of the guinea-pig. Neuroreport 2002, 13, 1037–1040. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, L.; Srivastava, R.; Kalluri, S.R.; Böttinger, S.; Herwerth, M.; Carassiti, D.; Srivastava, B.; Gempt, J.; Schlegel, J.; Kuhlmann, T.; Korn, T. Differential loss of KIR4.1 immunoreactivity in multiple sclerosis lesions. Ann. Neurol. 2014, 75, 810–828. [Google Scholar] [CrossRef] [PubMed]
- Pessia, M.; Imbrici, P.; D’Adamo, M.C.; Salvatore, L.; Tucker, S.J. Differential pH sensitivity of Kir4.1 and Kir4.2 potassium channels and their modulation by heteropolymerisation with Kir5.1. J. Physiol. 2001, 532, 359–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konstas, A.A.; Korbmacher, C.; Tucker, S.J. Identification of domains that control the heteromeric assembly of Kir5.1/Kir4.0 potassium channels. Am. J. Physiol. Cell Physiol. 2003, 284, C910–C917. [Google Scholar] [CrossRef] [PubMed]
- Kofuji, P.; Ceelen, P.; Zahs, K.R.; Surbeck, L.W.; Lester, H.A.; Newman, E.A. Genetic inactivation of an inwardly rectifying potassium channel (Kir4.1 subunit) in mice: Phenotypic impact in retina. J. Neurosci. 2000, 20, 5733–5740. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, K.; Butt, A.M. Evidence that perinatal and adult NG2-glia are not conventional oligodendrocyte progenitors and do not depend on axons for their survival. Mol. Cell. Neurosci. 2003, 23, 544–558. [Google Scholar] [CrossRef]
- Barlow, A.L.; MacLeod, A.; Noppen, S.; Sanderson, J.; Guérin, C.J. Colocalization analysis in fluorescence micrographs: Verification of a more accurate calculation of Pearson’s correlation coefficient. Microsc. Microanal. 2010, 16, 710–724. [Google Scholar] [CrossRef] [PubMed]
- Lorincz, A.; Nusser, Z. Specificity of immunoreactions: The importance of testing specificity in each method. J. Neurosci. 2008, 28, 9083–9086. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, V.; Butt, A. TASK-1 channels in oligodendrocytes: A role in ischemia mediated disruption. Neurobiol. Dis. 2013, 55, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Neusch, C.; Rozengurt, N.; Jacobs, R.E.; Lester, H.A.; Kofuji, P. Kir4.1 potassium channel subunit is crucial for oligodendrocyte development and in vivo myelination. J. Neurosci. 2013, 21, 5429–5438. [Google Scholar] [CrossRef]
- Bay, V.; Butt, A.M. Relationship between glial potassium regulation and axon excitability: A role for glial Kir4.1 channels. Glia 2003, 60, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Derst, C.; Karschin, C.; Wischmeyer, E.; Hirsch, J.R.; Preisig-Müller, R.; Rajan, S.; Engel, H.; Grzeschik, K.; Daut, J.; Karschin, A. Genetic and functional linkage of Kir5.1 and Kir2.1 channel subunits. FEBS Lett. 2001, 491, 305–311. [Google Scholar] [CrossRef] [Green Version]
- Mi, H.; Deerinck, T.J.; Jones, M.; Ellisman, M.H.; Schwarz, T.L. Inwardly rectifying K+ channels that may participate in K+ buffering are localized in microvilli of Schwann cells. J. Neurosci. 1996, 16, 2421–2429. [Google Scholar] [CrossRef] [PubMed]
- Fakler, B.; Schultz, J.H.; Yang, J.; Schulte, U.; Brandle, U.; Zenner, H.P.; Jan, L.Y.; Ruppersberg, J.P. Identification of a titratable lysine residue that determines sensitivity of kidney potassium channels (ROMK) to intracellular pH. EMBO J. 1996, 15, 4093–4099. [Google Scholar] [PubMed]
- Seifert, G.; Hüttmann, K.; Binder, D.K.; Hartmann, C.; Wyczynski, A.; Neusch, C.; Steinhäuser, C. Analysis of astroglial K+ channel expression in the developing hippocampus reveals a predominant role of the Kir4.1 subunit. J. Neurosci. 2009, 29, 7474–7488. [Google Scholar] [CrossRef] [PubMed]
- Heuser, K.; Eid, T.; Lauritzen, F.; Thoren, A.E.; Vindedal, G.F.; TaubÃll, E.; Gjerstad, L.; Spencer, D.D.; Ottersen, O.P.; Nagelhus, E.A.; et al. Loss of perivascular Kir4.1 potassium channels in the sclerotic hippocampus of patients with mesial temporal lobe epilepsy. J. Neuropathol. Exp. Neurol. 2012, 71, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Djukic, B.; Casper, K.B.; Philpot, B.D.; Chin, L.-S.; McCarthy, K.D. Conditional knock-out of Kir4.1 leads to glial membrane depolarization, inhibition of potassium and glutamate uptake, and enhanced short-term synaptic potentiation. J. Neurosci. 2007, 27, 11354–11365. [Google Scholar] [CrossRef] [PubMed]
- Neusch, C.; Papadopoulos, N.; Muller, M.; Maletzki, I.; Winter, S.M.; Hirrlinger, J.; Handschuh, M.; Bahr, M.; Richter, D.W.; Kirchhoff, F.; et al. Lack of the Kir4.1 channel subunit abolishes K+ buffering properties of astrocytes in the ventral respiratory group: Impact on extracellular K+ regulation. J. Neurophysiol. 2006, 95, 1843–1852. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brasko, C.; Butt, A.M. Expression of Kir2.1 Inward Rectifying Potassium Channels in Optic Nerve Glia: Evidence for Heteromeric Association with Kir4.1 and Kir5.1. Neuroglia 2018, 1, 176-187. https://doi.org/10.3390/neuroglia1010012
Brasko C, Butt AM. Expression of Kir2.1 Inward Rectifying Potassium Channels in Optic Nerve Glia: Evidence for Heteromeric Association with Kir4.1 and Kir5.1. Neuroglia. 2018; 1(1):176-187. https://doi.org/10.3390/neuroglia1010012
Chicago/Turabian StyleBrasko, Csilla, and Arthur M. Butt. 2018. "Expression of Kir2.1 Inward Rectifying Potassium Channels in Optic Nerve Glia: Evidence for Heteromeric Association with Kir4.1 and Kir5.1" Neuroglia 1, no. 1: 176-187. https://doi.org/10.3390/neuroglia1010012





