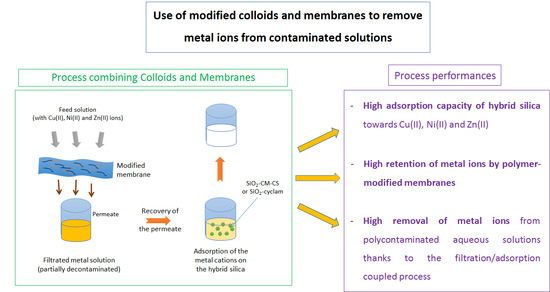
Use of Modified Colloids and Membranes to Remove Metal Ions from Contaminated Solutions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
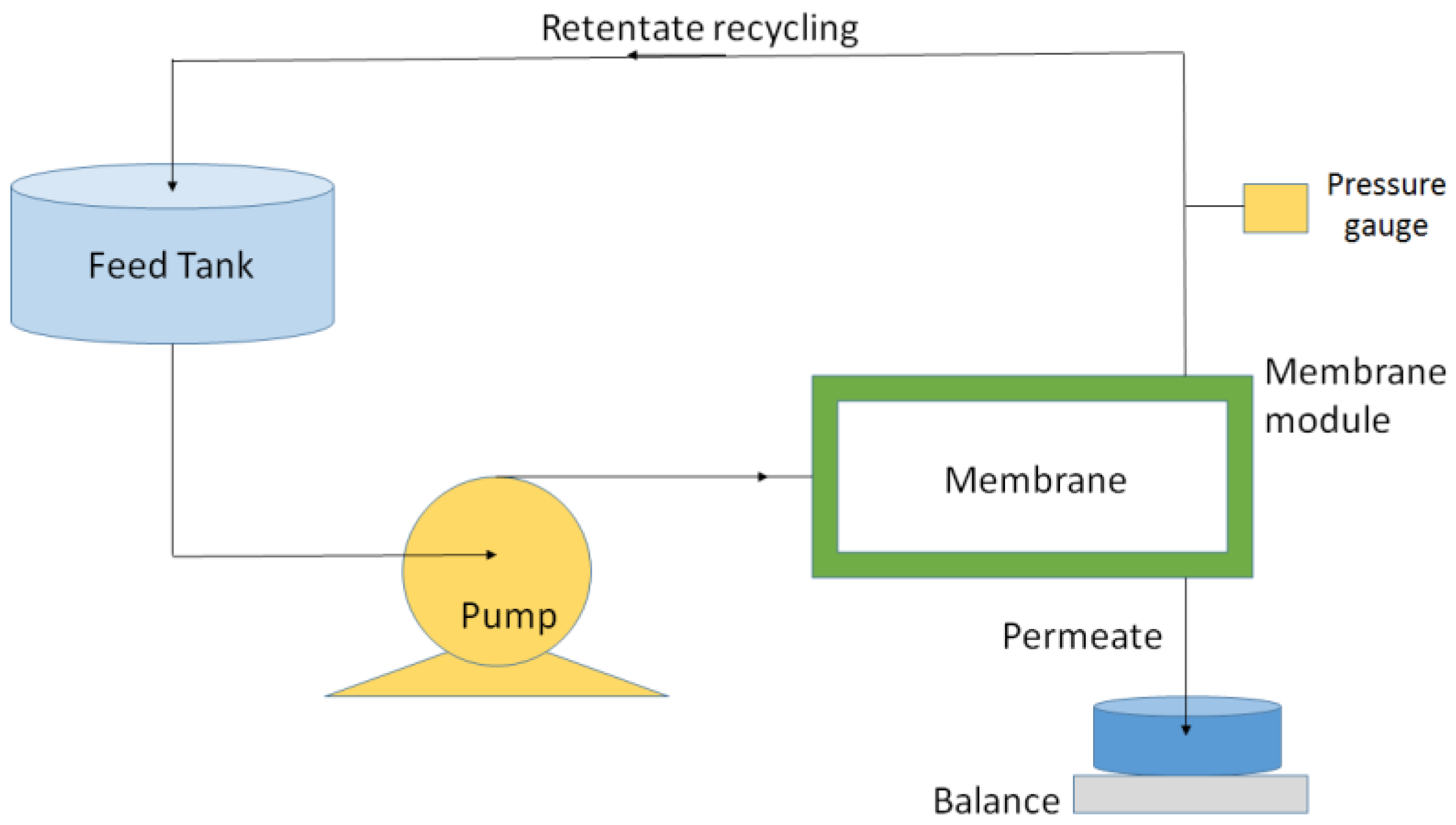
2.2. Membranes and Filtration Setup
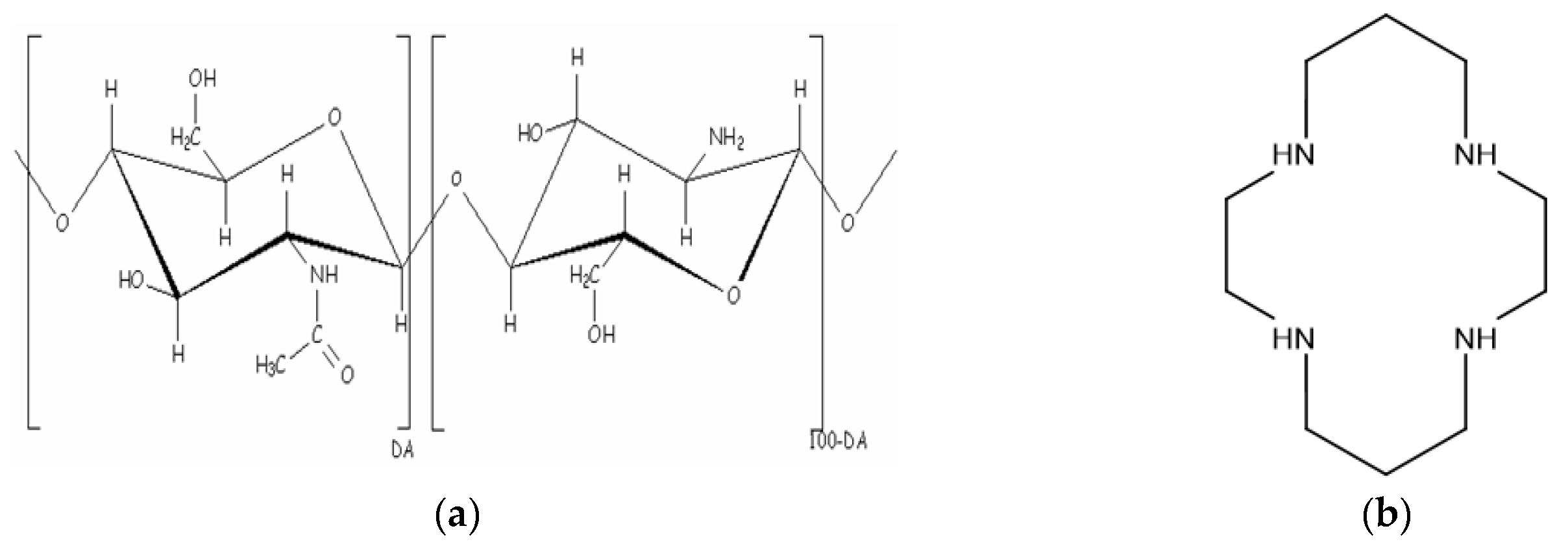
2.3. Preparation of the SiO2-CMCS Hybrid Materials
2.4. Preparation of the SiO2-Cyclam Hybrid Materials
2.5. Adsorption Experiments
2.6. Analytic Methods
3. Results
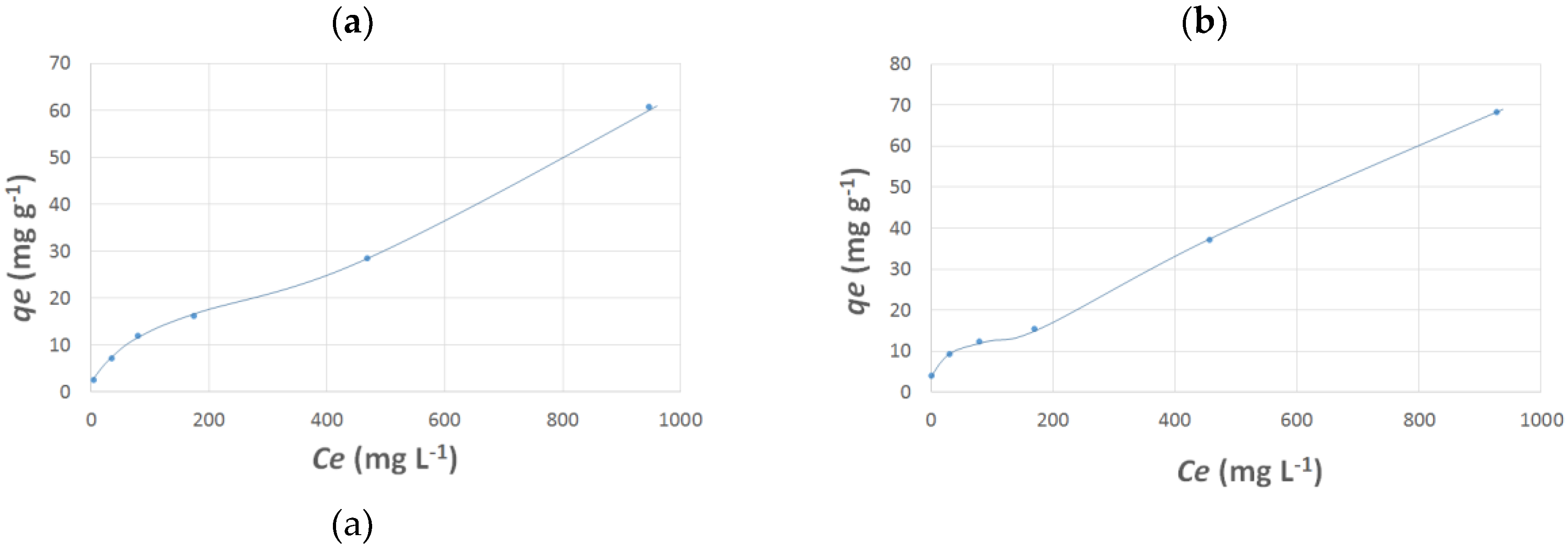
3.1. Adsorption of the Metal Ions by SiO2-CMCS and SiO2-Cyclam Adsorbents
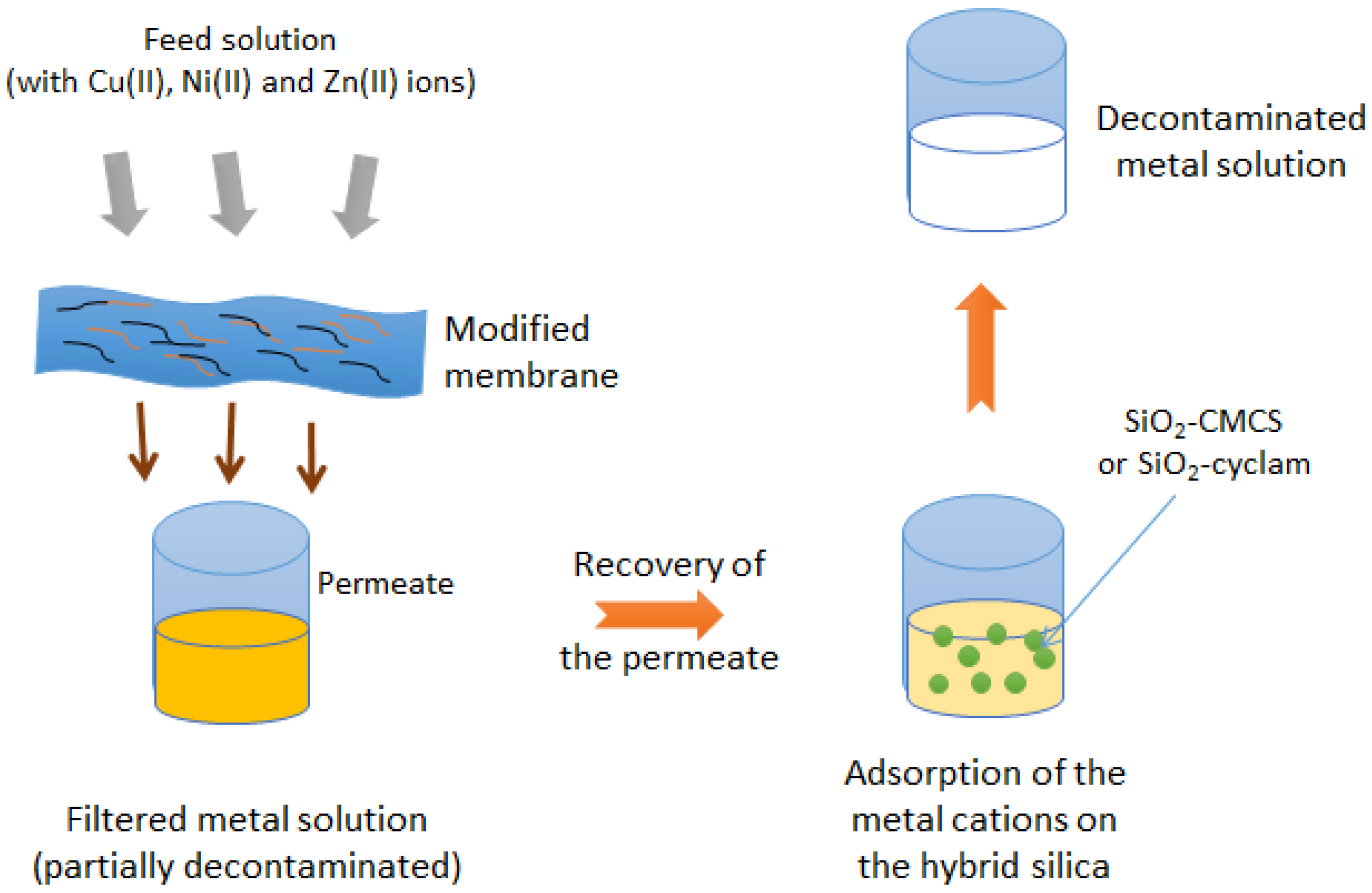
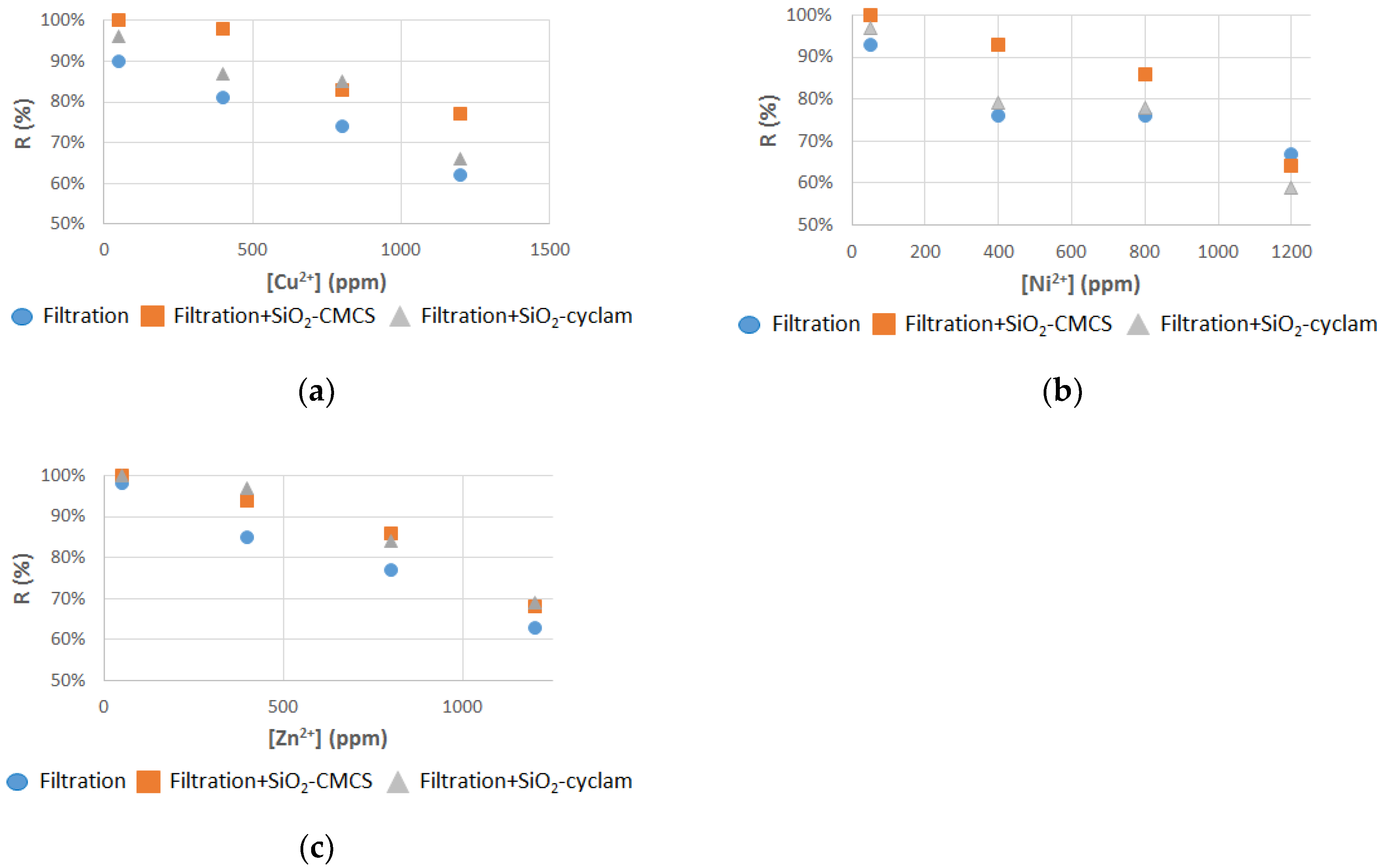
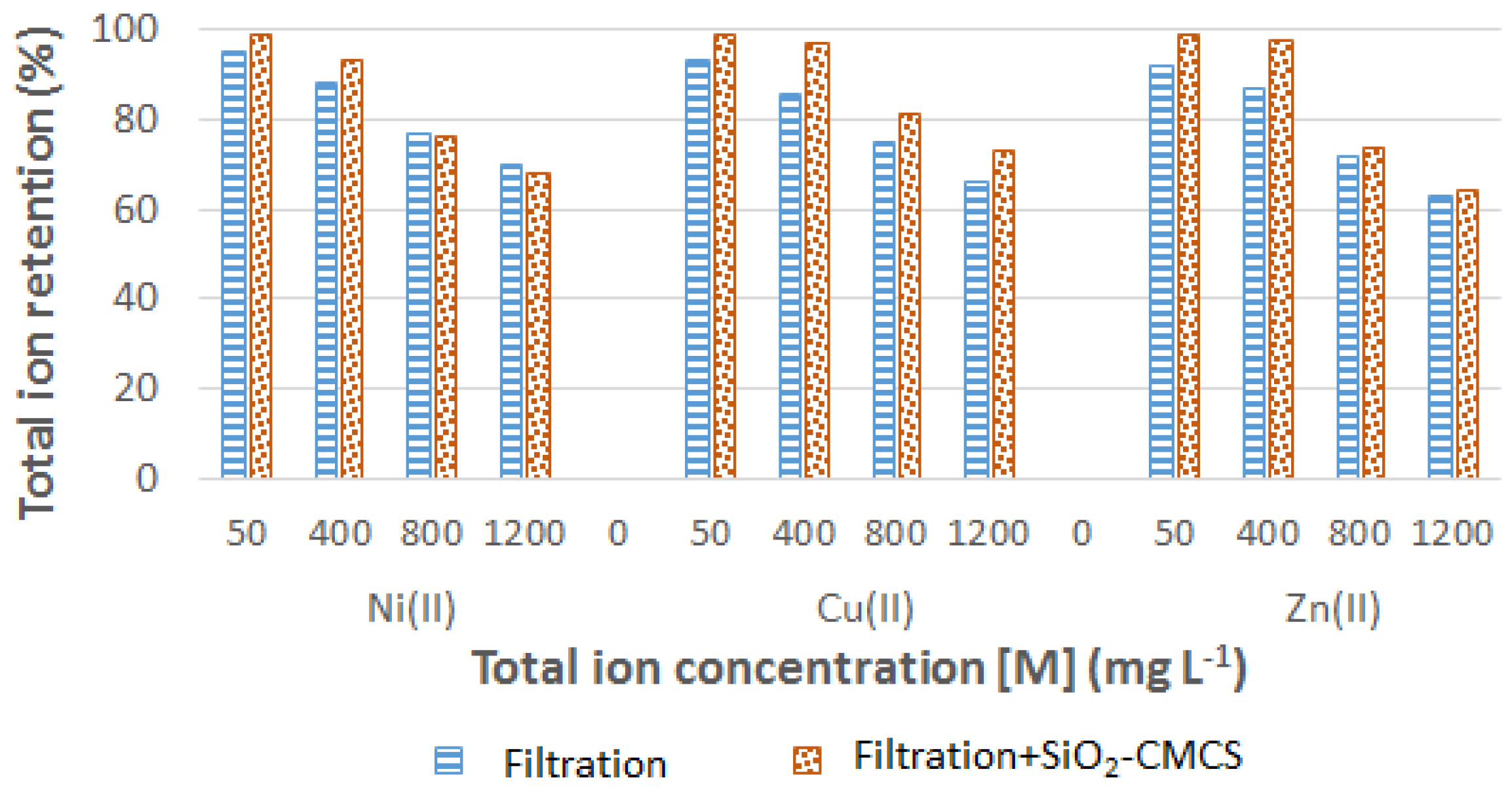
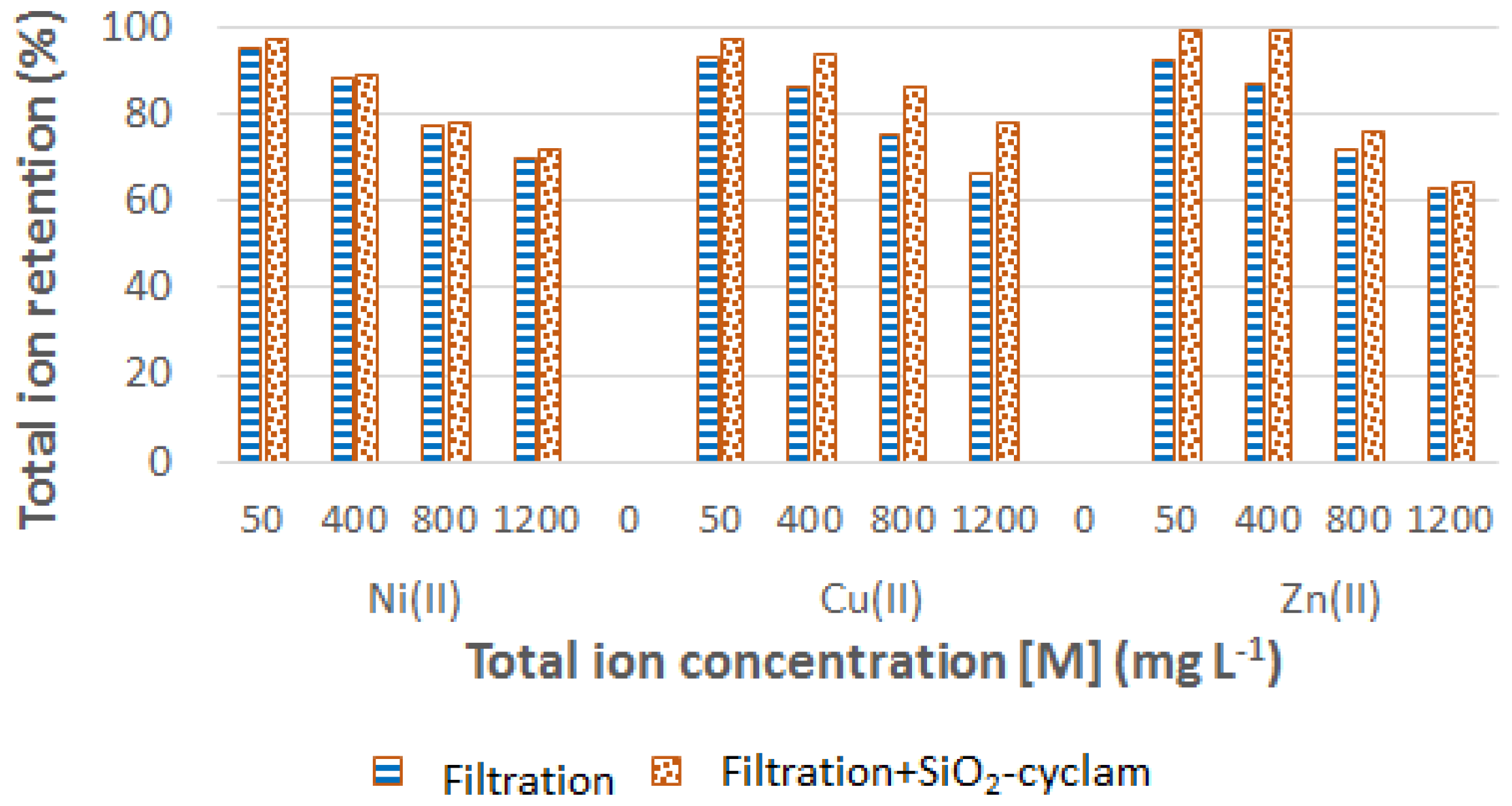
3.2. Filtration–Adsorption Combined Process
3.2.1. Single Component Solutions
3.2.2. Multicomponent Solutions
4. Discussion
4.1. Comparison with Other Works
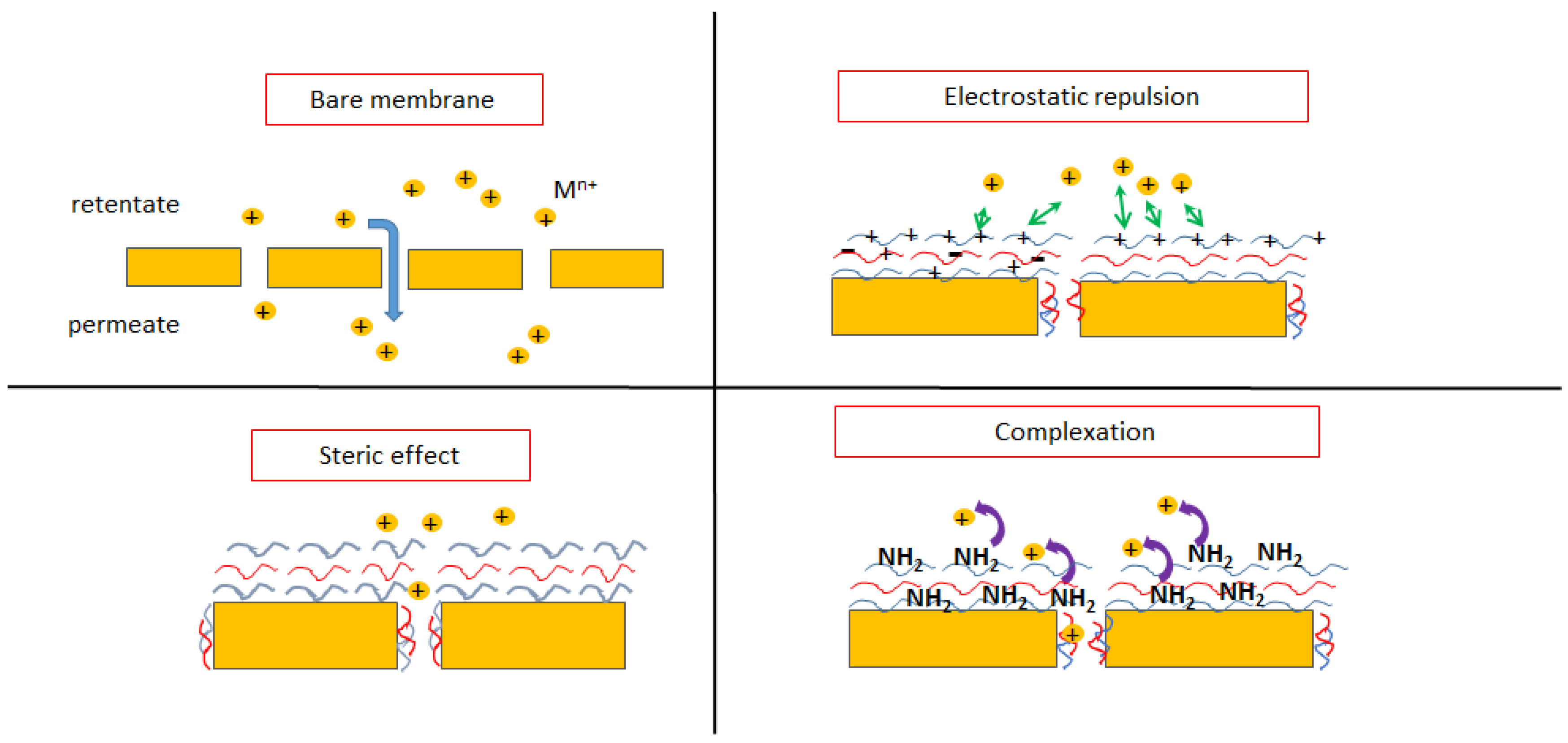
4.2. Interaction Mechanism of Metal Ions with the Adsorbents
4.3. Interaction Mechanism of Metal Ions with the Membranes
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Brandt, M.J.; Johnson, K.M.; Elphinston, A.J.; Ratnayaka, D.D. Twort’s Water Supply, 7th ed.; Elsevier: Amsterdam, The Netherlands, 2017; 932p, ISBN 9780081022559. [Google Scholar]
- Clemens, S.; Ma, J.F. Toxic heavy metal and metalloid accumulation in crop plants and foods. Annu. Rev. Plant Biol. 2016, 67, 489–512. [Google Scholar] [CrossRef] [PubMed]
- Nriagu, J.O. History of global metal pollution. Science 1996, 272, 223–224. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, Z. Industrial water pollution, water environment treatment, and health risks in China. Environ. Pollut. 2016, 218, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Jarup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Moiseenko, T.I.; Megorskii, V.V.; Gashkina, N.A.; Kudryavtseva, L.P. Water Pollution Effect on Population Health in an Industrial Northern Region. Water. Res. 2010, 37, 194–203. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Mohsen-Nia, M.; Montazeri, P.; Modarress, H. Removal of Cu2+ and Ni2+ from wastewater with a chelating agent and reverse osmosis processes. Desalination 2007, 217, 276–281. [Google Scholar] [CrossRef]
- Escobar, I.C.; Van der Bruggen, C. Modern Applications in Membrane Science and Technology; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2011; Volume 1078. [Google Scholar]
- Abu Qdais, H.; Moussa, H. Removal of heavy metals from wastewater by membrane processes: A comparative study. Desalination 2004, 164, 105–110. [Google Scholar] [CrossRef]
- Gherasim, C.V.; Hanckova, K.; Palarcik, J.; Mikulasek, P. Investigation of cobalt (II) retention from aqueous solutions by a polyamide nanofiltration membrane. J. Membr. Sci. 2015, 490, 46–56. [Google Scholar] [CrossRef]
- Basaran, G.; Kavak, D.; Dizge, N.; Asci, Y.; Solener, M.; Ozbey, B. Comparative study of the removal of nickel(II) and chromium(VI) heavy metals from metal plating wastewater by two nanofiltration membranes. Desal. Water Treat. 2016, 57, 21870–21880. [Google Scholar] [CrossRef]
- Ng, L.Y.; Mohammad, A.W.; Ng, C.Y. A review on nanofiltration membrane fabrication and modification using polyelectrolytes: Effective ways to develop membrane selective barriers and rejection capability. Adv. Colloid Interface Sci. 2013, 197–198, 85–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yao, Y.; Yue, Z. Preparation of polyelectrolyte multilayer films consisting of sulfonated poly(ether ketone) alternating with selected anionic layers. J. Membr. Sci. 2009, 337, 200–207. [Google Scholar] [CrossRef]
- Park, J.; Park, J.; Kim, S.H.; Cho, J.; Bang, J. Desalination membranes from pH-controlled and thermally-crosslinked layer-by-layer assembled multilayers. J. Mater. Chem. 2010, 20, 2085–2091. [Google Scholar] [CrossRef]
- Fadhillah, F.; Zaidi, S.M.J.; Khan, Z.; Khaled, M.M.; Rahman, F.; Hammond, P.T. Development of polyelectrolyte multilayer thin film composite membrane for water desalination application. Desalination 2013, 318, 19–24. [Google Scholar] [CrossRef]
- Magnenet, C.; Jurin, F.E.; Lakard, S.; Buron, C.C.; Lakard, B. Polyelectrolyte modification of ultrafiltration membrane for removal of copper ions. Coll. Surf. A 2013, 435, 170–177. [Google Scholar] [CrossRef]
- Magnenet, C.; Lakard, S.; Buron, C.C.; Lakard, B. Functionalization of organic membranes by polyelectrolyte multilayer assemblies: Application to the removal of copper ions from aqueous solutions. J. Colloid Interface Sci. 2012, 376, 202–208. [Google Scholar] [CrossRef]
- Mokhter, M.A.; Lakard, S.; Magnenet, C.; Euvrard, M.; Lakard, B. Preparation of polyelectrolyte-modified membranes for heavy metal ions removal. Environ. Technol. 2017, 38, 2476–2485. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.P.; Geng, C.L.; Guo, H.X.; Du, Z.; Zhang, G.J.; Ji, S.L. Synthesis of positively charged polyelectrolyte multilayer membranes for removal of divalent metal ions. J. Mater. Res. 2013, 28, 1449–1457. [Google Scholar] [CrossRef]
- Kochan, J.; Wintgens, T.; Wong, J.E.; Melin, T. Polyelectrolyte-modified polyethersulfone ultrafiltration membranes for wastewater treatment applications. Desal. Water Treat. 2009, 9, 175–180. [Google Scholar] [CrossRef]
- Corapcioglu, M.O.; Huang, C.P. The adsorption of heavy metals onto hydrous activated carbon. Water Res. 1987, 21, 1031–1044. [Google Scholar] [CrossRef]
- Kang, K.C.; Kim, S.S.; Choi, J.W.; Kwon, S.H. Sorption of Cu2+ and Cd2+ onto acid- and base- pretreated granular activated carbon and activated carbon fiber samples. J. Ind. Eng. Chem. 2008, 14, 131–135. [Google Scholar] [CrossRef]
- Kim, D.S. The removal by crab shell of mixed heavy metal ions in aqueous solution. Bioresour. Technol. 2003, 87, 355–357. [Google Scholar] [CrossRef]
- Vold, I.M.N.; Varum, K.M.; Guibal, E.; Smidsrod, O. Binding of ions to chitosan—Selectivity studies. Carbohydr. Polym. 2003, 54, 471–477. [Google Scholar] [CrossRef]
- Guibal, E.; Vincent, T.; Navarro, R. Metal ion biosorption on chitosan for the synthesis of advanced materials. J. Mater. Sci. 2014, 49, 5505–5518. [Google Scholar] [CrossRef]
- Benhamou, A.; Baudu, M.; Derriche, Z.; Basly, J.P. Aqueous heavy metals removal on amine-functionalized Si-MCM-41 and Si-MCM-48. J. Hazard. Mater. 2009, 171, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Aguado, J.; Arsuaga, J.M.; Arencibia, A.; Lindo, M.; Gascon, V. Aqueous heavy metals removal by adsorption on amine-functionalized mesoporous silica. J. Hazard. Mater. 2009, 163, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Koong, L.F.; Lam, K.F.; Barford, J.; McKay, G. A comparative study on selective adsorption of metal ions using aminated adsorbents. J. Colloid Interface Sci. 2013, 395, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Alauzun, J.; Besson, E.; Mehdi, A.; Corriu, R.J.P. Reversible covalent chemistry of CO2: An opportunity for nano-structured hybrid organic-inorganic materials. Chem. Mater. 2008, 20, 503–513. [Google Scholar] [CrossRef]
- Munoz, S.V.; Martinez, M.S.; Torres, M.G.; Alcala, S.P.; Quintanilla, F.; Rodriguez-Canto, A.; Rodriguez, J.R. Adsorption and Removal of Cadmium Ions from Simulated Wastewater Using Commercial Hydrophilic and Hydrophobic Silica Nanoparticles: A Comparison with Sol–gel Particles. Water Air Soil Pollut. 2014, 225, 2165–2176. [Google Scholar] [CrossRef]
- Singhon, R.; Husson, J.; Knorr, M.; Lakard, B.; Euvrard, M. Adsorption of Ni(II) ions on colloidal hybrid organic-inorganic silica composites. Coll. Surf. B 2012, 93, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Singhon, R.; Husson, J.; Knorr, M.; Euvrard, M. Preparation of Silica-Supported Biosorbents for Copper(II) Removal. J. Disp. Sci. Technol. 2011, 32, 1735–1741. [Google Scholar] [CrossRef]
- Kavakli, C.; Ali Tuncel, S.; Salih, B. Selectivity of cyclam modified poly(p-chloromethyl styrene-ethyleneglycol dimethacrylate) microbeads for Cu(II), Ni(II), Co.(II) and Zn(II). Sep. Purif. Technol. 2005, 45, 32–40. [Google Scholar] [CrossRef]
- Dubois, G.; Reyé, C.; Corriu, J.P.; Brandes, S.; Denat, F.; Guilard, R. Coordination chemistry in the solid: Study of the incorporation of Cu(II) into cyclam-containing hybrid materials. Angew. Chem. 2001, 40, 1087–1090. [Google Scholar] [CrossRef]
- Katsou, E.; Malamis, S.; Kosanovic, T.; Souma, K.; Haralambous, K.J. Application of adsorption and ultrafiltration processes for the pre-treatment of several industrial wastewater streams. Water Air Soil Pollut. 2012, 223, 5519–5534. [Google Scholar] [CrossRef]
- Malamis, S.; Katsou, E.; Haralambous, K.J. Study of Ni(II), Cu(II), Pb(II) and Zn(II) removal using sludges and minerals followed by UF/MF. Water Air Soil Pollut. 2011, 218, 81–92. [Google Scholar] [CrossRef]
- Malamis, S.; Katsou, E.; Kosanovic, T.; Haralambous, K.J. Combined adsorption and ultrafiltration processes employed for the removal of pollutants from metal plating wastewater. Sep. Sci. Technol. 2012, 47, 983–996. [Google Scholar] [CrossRef]
- Malamis, S.; Katsou, E.; Haralambous, K.J. Evaluation of the efficiency of a combined adsorption-ultrafiltration system for the removal of heavy metals, color, and organic matter from textile wastewater. Sep. Sci. Technol. 2011, 46, 920–932. [Google Scholar] [CrossRef]
- Decher, G. Fuzzy nanoassemblies: Toward layered polymeric multicomposites. Nature 1997, 277, 1232–1237. [Google Scholar] [CrossRef]
- Dirieh Egueh, A.N.; Lakard, B.; Fievet, P.; Lakard, S.; Buron, C. Charge properties of membranes modified by multilayer polyelectrolyte adsorption. J. Colloid Interface Sci. 2010, 344, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Zhao, Y.; Shen, Q.; Wang, D.; Xu, D.; et al. The effect of carboxymethyl chitosan on the precipitation of calcium carbonate. J. Cryst. Growth 2004, 261, 571–576. [Google Scholar] [CrossRef]
- Aden, M.; Ubol, R.N.; Knorr, M.; Husson, J.; Euvrard, M. Efficent removal of nickel(II) salts from aqueous solution using carboxymethylchitosan-coated silica particles as adsorbent. Carbohydr. Polym. 2017, 173, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Makowska-Janusik, M.; Kassiba, A.; Errien, N.; Mehdi, A.J. Mesoporous Silica Functionalized by Cyclam–Metal Groups: Spectroscopic Studies and Numerical Modeling. J. Inorg. Organomet. Polym. 2010, 20, 761–773. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Alqadami, A.A.; Naushad, M.; Abdalla, M.A.; Ahamad, T.; AlOthman, Z.A.; Alshehri, S.M.; Ghfar, A.A. Efficient removal of toxic metal ions from wastewater recyclable nanocomposite: A study of adsorption parameters and interaction mechanism. J. Clean. Prod. 2017, 156, 426–436. [Google Scholar] [CrossRef]
- Ringot, D.; Lerzy, B.; Chaplain, K.; Bonhoure, J.P.; Auclair, E.; Larondelle, Y. In vitro biosorption of ochratoxin A on the yeast industry by-products: Comparison of isotherm models. Bioresour. Technol. 2007, 98, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Van Erp, T.S.; Martens, J.A. A standardization for BET fitting of adsorption isotherms. Microporous Mesoporous Mater. 2011, 145, 188–193. [Google Scholar] [CrossRef]
- Shim, J.W.; Park, S.J.; Ryu, S.K. Effect of modification with HNO3 and NaOH on metal adsorption by pitch-based activated carbon fibers. Carbon 2001, 39, 1635–1642. [Google Scholar] [CrossRef]
- Monser, L.; Adhoum, N. Modified activated carbon for the removal of copper, zinc, chromium and cyanide from wastewater. Sep. Purif. Technol. 2002, 26, 137–146. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbent for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- O’Connell, D.W.; Birkinshaw, C.; O’Dwyer, T.F. Heavy metal adsorbents prepared from the modification of cellulose: A review. Bioresour. Technol. 2008, 99, 6709–6724. [Google Scholar] [CrossRef] [PubMed]
- Algarra, M.; Jimenez, M.V.; Rodriguez-Castellon, E.; Jimenez-Lopez, A.; Jimenez-Jimenez, J. Heavy metals removal from electroplating wastewater by aminopropyl-Si MCM-41. Chemosphere 2005, 59, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Kim, Y.; Lee, H.; Yi, J. Synthesis of functionalized porous silica via templating method as heavy metal ion adsorbents: The introduction of surface hydrophilicity onto the surface of adsorbents. Microporous Mesoporous Mater. 2001, 50, 77–90. [Google Scholar] [CrossRef]
- Jiang, Y.; Gao, Q.; Yu, H.; Chen, J.R.; Deng, F. Intensively competitive adsorption for heavy metal ions by PAMAM-SBA-15 and EDTA-PAMAM-SBA-15 inorganic-organic hybrid materials. Microporous Mesoporous Mater. 2007, 103, 316–324. [Google Scholar] [CrossRef]
- Ngah, W.S.W.; Isa, I.M. Comparison study of copper ion adsorption on chitosan, Dowex A-1, and Zerolit 225. J. Appl. Polym. Sci. 1998, 67, 1067–1070. [Google Scholar] [CrossRef]
- McKay, G.; Blair, H.S.; Findon, A. Equilibrium studies for the sorption of metal-ions onto chitosan. Ind. J. Chem. 1989, 28, 356–360. [Google Scholar]
- Escoda, A.; Euvrard, M.; Lakard, S.; Husson, J.; Mohamed, A.S.; Knorr, M. Ultrafiltration-assisted retention of Cu(II) ions by adsorption on chitosan-functionalized colloidal silica particles. Sep. Purif. Technol. 2013, 118, 25–32. [Google Scholar] [CrossRef]
- Dambies, L.; Guimon, C.; Yiacoumi, E.; Guibal, E. Characterization of metal ion interactions with chitosan by X-ray photoelectron spectroscopy. J. Collid Interface Sci. 2001, 177, 203–214. [Google Scholar] [CrossRef]
- Juang, R.S.; Shao, H.J. Effect of pH on competitive adsorption of Cu(II), Ni(II), and Zn(II) from water onto chitosan beads. Adsorption 2002, 8, 71–78. [Google Scholar] [CrossRef]
- Mureseanu, M.; Reiss, A.; Stefanescu, I.; David, E.; Parvulescu, V.; Renard, G.; Hulea, V. Modified SBA-15 mesoporous silica for heavy metal ions remediation. Chemosphere 2008, 73, 1499–1504. [Google Scholar] [CrossRef] [PubMed]
- Shriver, D.F.; Atkins, P.W. Inorganic Chemistry, 3rd ed.; Oxford University Press: Oxford, UK, 1999; 763p, ISBN 0198503318. [Google Scholar]
- Monteiro, O.A.C., Jr.; Airoldi, C. Some thermodynamic data on copper-chitin and copper-chitosan biopolymer interactions. J. Collid Interface Sci. 1999, 212, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Martell, A.E.; Smith, R.M. Critical Stability Constants, 1st ed.; Plenum Press: New York, NY, USA, 1974; Volume 4, 276p, ISBN 0306352141. [Google Scholar]
- Noble, R.D. Generalized microscopic mechanism of facilitated transport in fixed site carrier membranes. J. Membr. Sci. 1992, 75, 121–129. [Google Scholar] [CrossRef]
- Sheng, C.; Wijeratne, S.; Cheng, S.; Baker, G.L.; Bruening, M.L. Facilitated ion transport through polyelectrolyte multilayer films containing metal-binding ligands. J. Membr. Sci. 2014, 459, 169–176. [Google Scholar] [CrossRef]
- Bessbousse, H.; Verchère, J.F.; Lebrun, L. Characterisation of metal-complexing membranes prepared by the semi-interpenetrating polymer networks technique. Application to the removal of heavy metal ions from aqueous solutions. Chem. Eng. J. 2012, 187, 16–28. [Google Scholar] [CrossRef]
- Toutianoush, A.; Schnepf, J.; El Hashani, A.; Tieke, B. Selective ion transport and complexation in layer-by-layer assemblies made of p-sulfanoto calix[n]arenes and cationic polyelectrolytes. Adv. Func. Mater. 2005, 15, 700–708. [Google Scholar] [CrossRef]
| Metal Ion | SiO2-Cyclam Hybrid Material | SiO2-CMCS Hybrid Material |
|---|---|---|
| Cu(II) | qm = 61 mg g−1 | qm = 68 mg g−1 |
| Ni(II) | qm = 50 mg g−1 | qm = 61 mg g−1 |
| Zn(II) | qm = 26 mg g−1 | qm = 45 mg g−1 |
| Adsorbent | qm (mg g−1) | References |
|---|---|---|
| SiO2-cyclam | 61 | This work |
| SiO2-CMCS | 68 | This work |
| Activated carbon | 9–38 | [49,50] |
| Zeolite | 1–25 | [51] |
| Clays | 3–11 | [51] |
| Modified cellulose | 19–75 | [52] |
| Aminopropyl-MCM-41 | 31 | [53] |
| Mercaptopropyl-silica | 13 | [54] |
| EDTA-modified SBA-15 | 13 | [55] |
| Chitosan | 4–222 | [56,57] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mokhter, M.A.; Magnenet, C.; Lakard, S.; Euvrard, M.; Aden, M.; Clément, S.; Mehdi, A.; Lakard, B. Use of Modified Colloids and Membranes to Remove Metal Ions from Contaminated Solutions. Colloids Interfaces 2018, 2, 19. https://doi.org/10.3390/colloids2020019
Mokhter MA, Magnenet C, Lakard S, Euvrard M, Aden M, Clément S, Mehdi A, Lakard B. Use of Modified Colloids and Membranes to Remove Metal Ions from Contaminated Solutions. Colloids and Interfaces. 2018; 2(2):19. https://doi.org/10.3390/colloids2020019
Chicago/Turabian StyleMokhter, Mohd Akmali, Claire Magnenet, Sophie Lakard, Myriam Euvrard, Moumin Aden, Sébastien Clément, Ahmad Mehdi, and Boris Lakard. 2018. "Use of Modified Colloids and Membranes to Remove Metal Ions from Contaminated Solutions" Colloids and Interfaces 2, no. 2: 19. https://doi.org/10.3390/colloids2020019