Fabrication and Characterization of Piezoelectric PEO/SF/BaTiO3 Scaffolds for Cardiac Tissue Engineering
Abstract
:1. Introduction
2. Materials and Methods
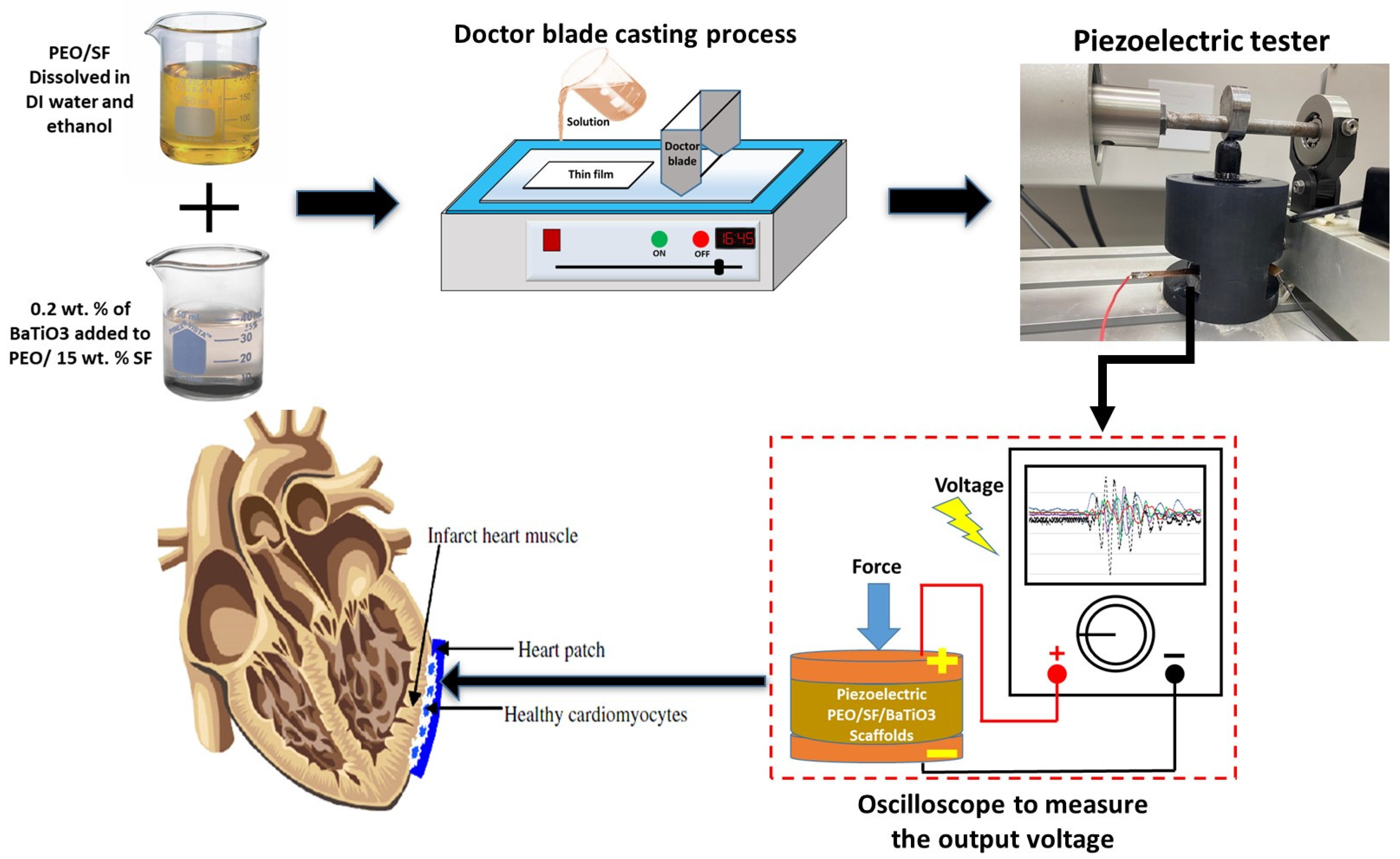
2.1. PEO/SF/BaTiO3 Scaffold Preparation
2.2. Characterization of the Thin Film
3. Results and Discussions
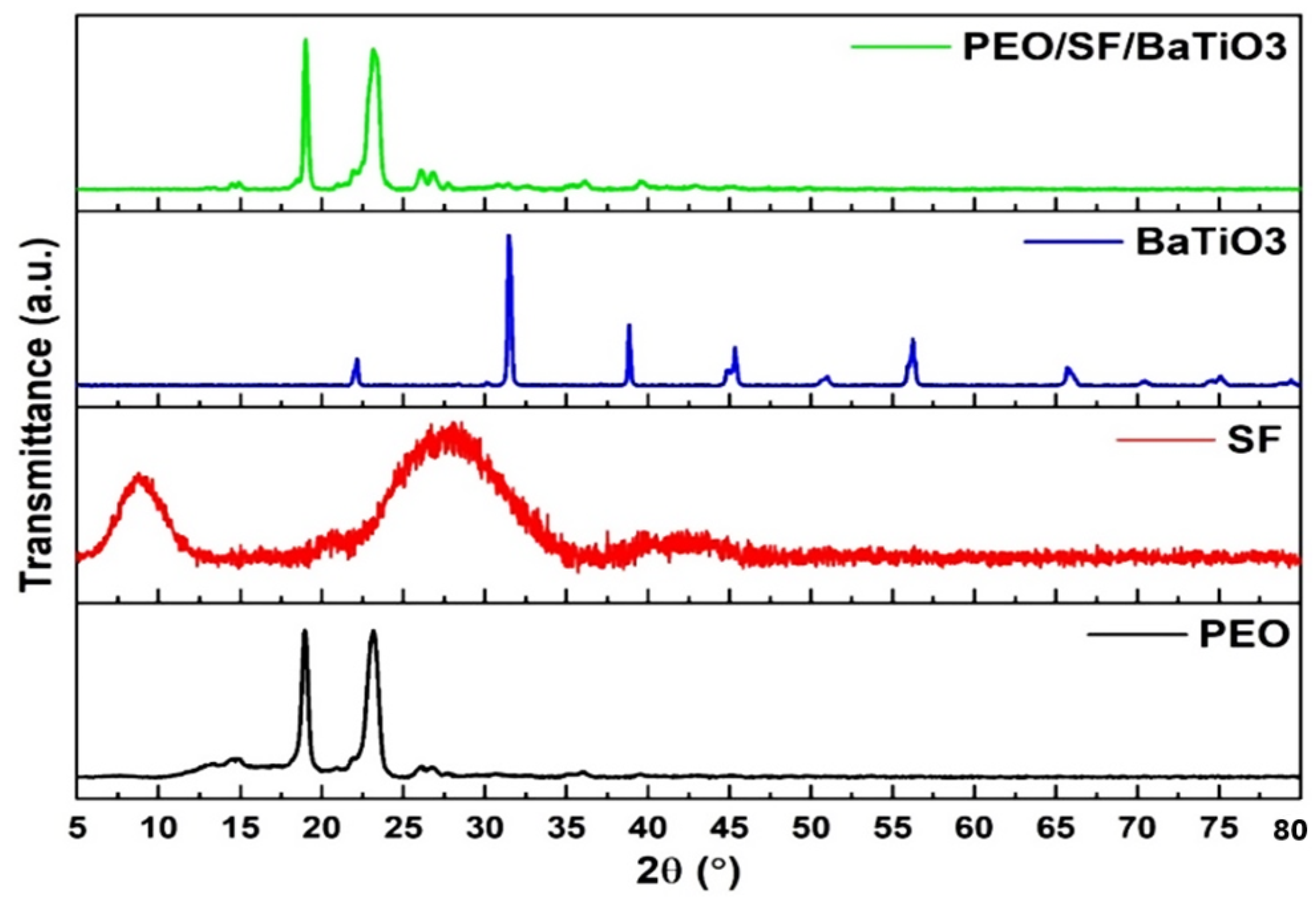
3.1. XRD, FTIR, and Raman Characterizations
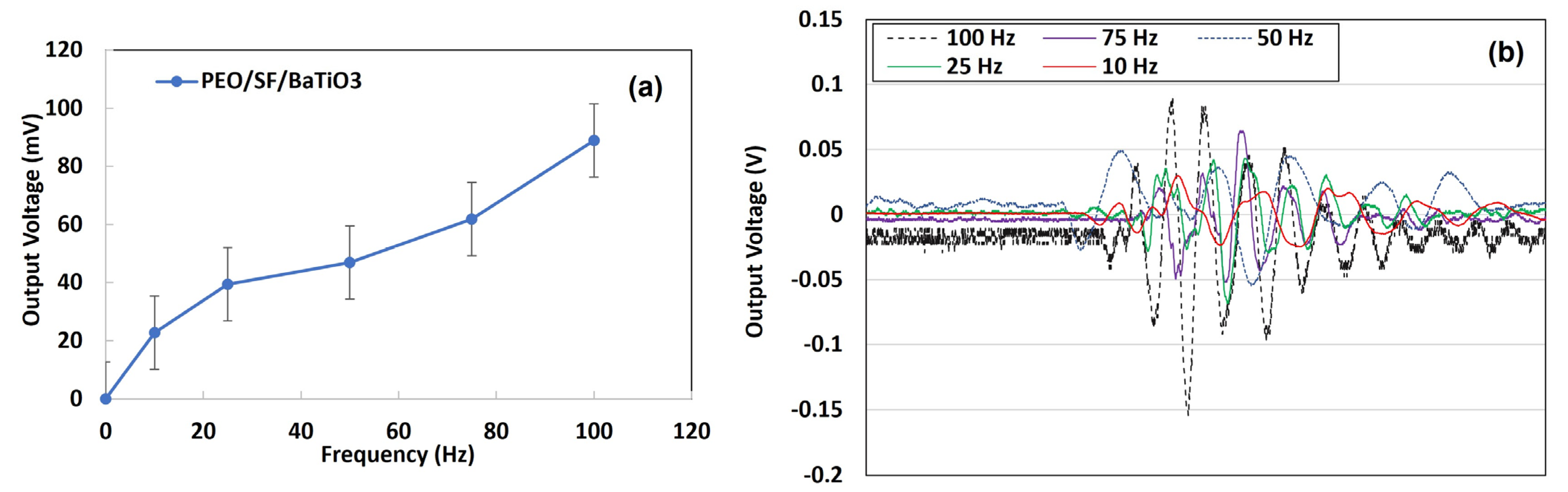
3.2. Piezoelectric Properties Evaluation
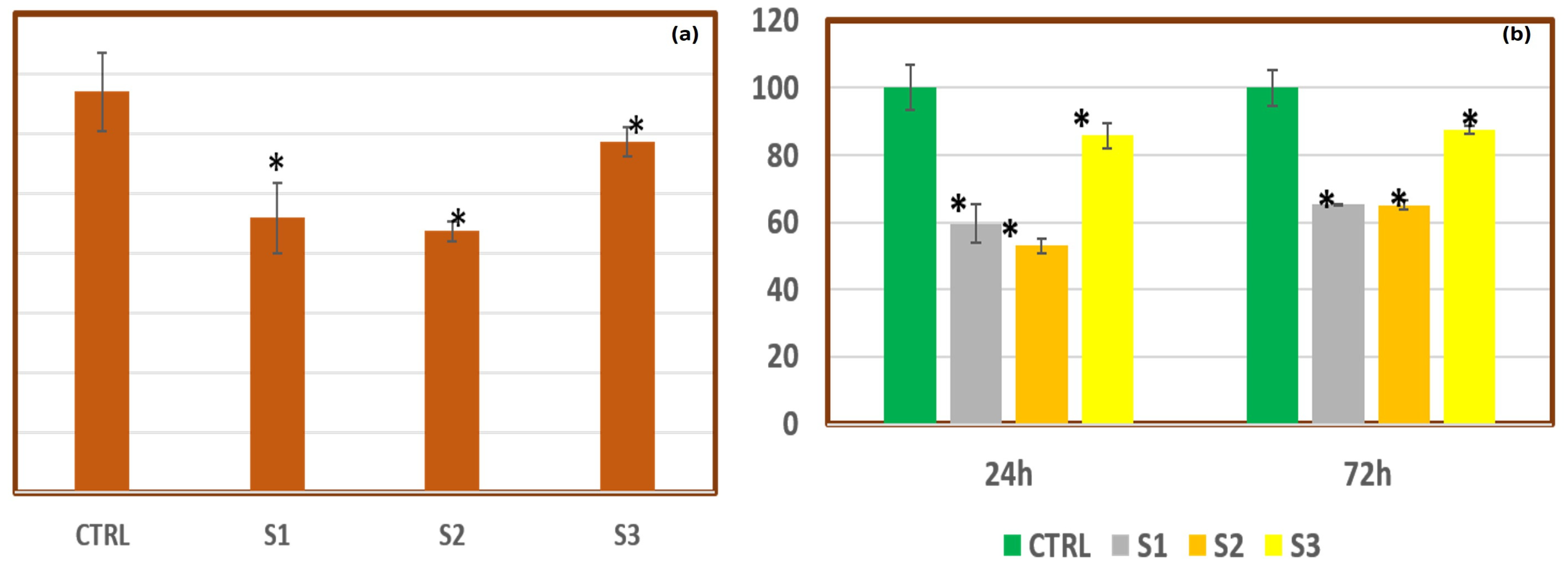
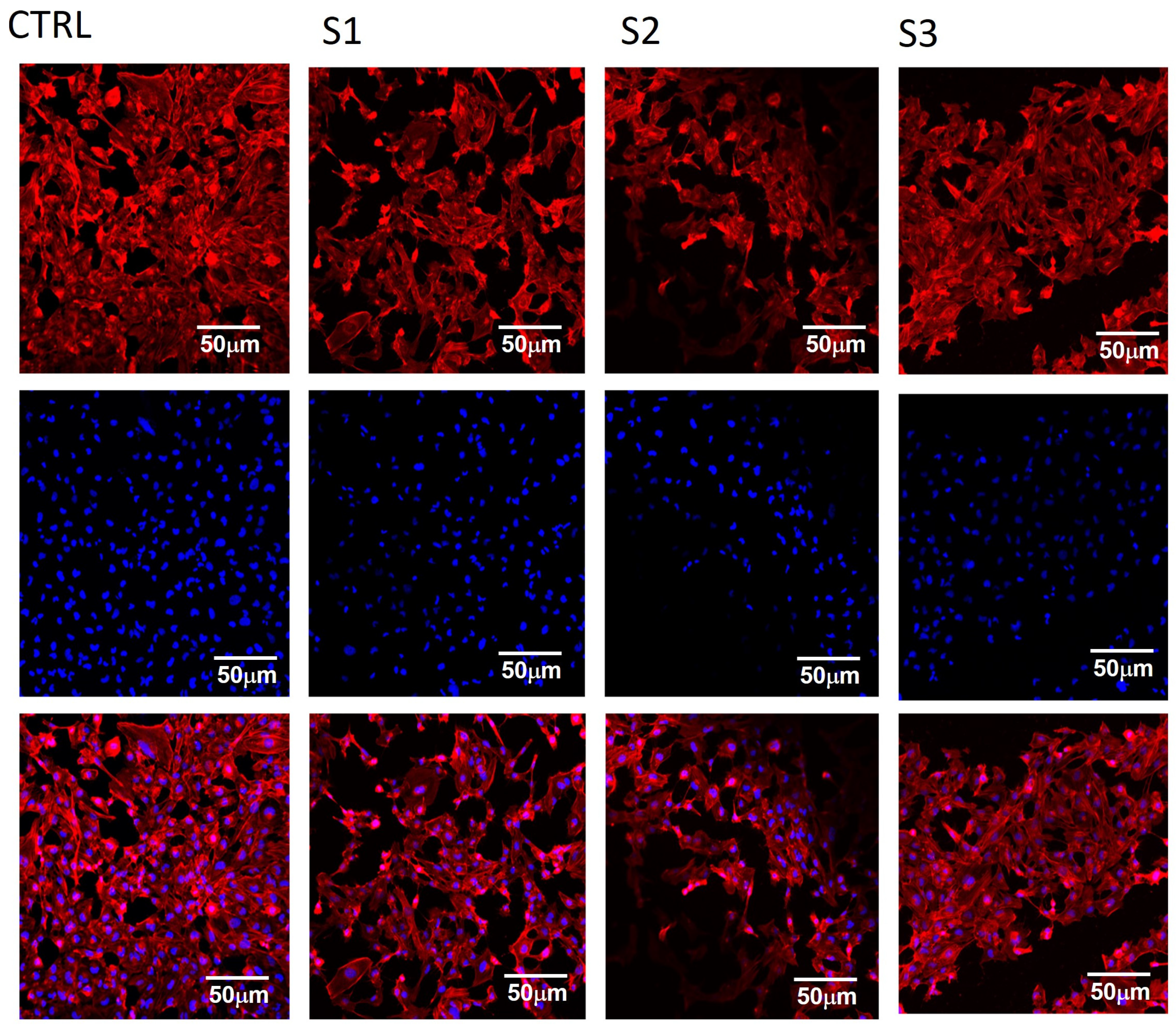
3.3. PEO/SF/BaTiO3 In Vitro Study: MTT
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Taylor, D.O.; Edwards, L.B.; Aurora, P.; Christie, J.D.; Dobbels, F.; Kirk, R.; Rahmel, A.O.; Kucheryavaya, A.Y.; Hertz, M.I. Registry of the International Society for Heart and Lung Transplantation: Twenty-fifth official adult heart transplant report. J. Heart Lung Transplant. 2008, 27, 943–956. [Google Scholar] [CrossRef] [PubMed]
- Bardy, G.H.; Lee, K.L.; Mark, D.B.; Poole, J.E.; Packer, D.L.; Boineau, R.; Domanski, M.; Troutman, C.; Anderson, J.; Johnson, G. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N. Engl. J. Med. 2005, 352, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Langone, A.J.; Helderman, J.H. Disparity between Solid-Organ Supply and Demand. N. Engl. J. Med. 2003, 349, 704. [Google Scholar] [CrossRef] [PubMed]
- Granger, C.B.; McMurray, J.J.; Yusuf, S.; Held, P.; Michelson, E.L.; Olofsson, B.; Östergren, J.; Pfeffer, M.A.; Swedberg, K. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: The CHARM-Added trial. Lancet 2003, 362, 772–776. [Google Scholar] [CrossRef] [PubMed]
- Pitt, B.; Zannad, F.; Remme, W.J.; Cody, R.; Castaigne, A.; Perez, A.; Palensky, J.; Wittes, J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 1999, 341, 709–717. [Google Scholar] [CrossRef]
- Picascia, A.; Grimaldi, V.; Zullo, A.; Infante, T.; Maiello, C.; Crudele, V.; Sessa, M.; Mancini, F.P.; Napoli, C. Experimental and clinical transplantation: Official journal of the Middle East Society for Organ Transplantation. MESOT 2012, 10, 209. [Google Scholar]
- Brautbar, A.; Ballantyne, C.M. Pharmacological strategies for lowering LDL cholesterol: Statins and beyond. Nat. Rev. Cardiol. 2011, 8, 253–265. [Google Scholar] [CrossRef]
- Brizzio, M.E.; Zapolanski, A. Reviews in cardiovascular medicine. RCM 2011, 12, S40. [Google Scholar]
- Nolan, C.J.; Damm, P.; Prentki, M. Type 2 diabetes across generations: From pathophysiology to prevention and management. Lancet 2011, 378, 169–181. [Google Scholar] [CrossRef]
- Zhao, D.; Qi, Y.; Zheng, Z.; Wang, Y.; Zhang, X.-Y.; Li, H.-J.; Liu, H.-H.; Zhang, X.-T.; Du, J.; Liu, J. Dietary factors associated with hypertension. Nat. Rev. Cardiol. 2011, 8, 456–465. [Google Scholar] [CrossRef]
- Trialists, F.T. Indications for fibrinolytic therapy in suspected acute myocardial infarction: Collaborative overview of early mortality and major morbidity results from all randomised trials of more than 1000 patients. Lancet 1994, 343, 311–322. [Google Scholar]
- Antman, E.M.; Morrow, D.A.; McCabe, C.H.; Murphy, S.A.; Ruda, M.; Sadowski, Z.; Budaj, A.; López-Sendón, J.L.; Guneri, S.; Jiang, F. Enoxaparin versus Unfractionated Heparin with Fibrinolysis for ST-Elevation Myocardial Infarction. N. Engl. J. Med. 2006, 354, 1477–1488. [Google Scholar] [CrossRef] [PubMed]
- Schömig, A.; Kastrati, A.; Dirschinger, J.; Mehilli, J.; Schricke, U.; Pache, J.; Martinoff, S.; Neumann, F.-J.; Schwaiger, M. Coronary Stenting plus Platelet Glycoprotein IIb/IIIa Blockade Compared with Tissue Plasminogen Activator in Acute Myocardial Infarction. N. Engl. J. Med. 2000, 343, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Kannel, M.W.B. Incidence and epidemiology of heart failure. Heart Fail. Rev. 2000, 5, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Swedberg, K.; Kjekshus, J. Effects of enalapril on mortality in severe congestive heart failure: Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). Am. J. Cardiol. 1988, 62, 60A–66A. [Google Scholar] [CrossRef]
- Abdal-hay, A.; Pant, H.R.; Lim, J.K. Super-hydrophilic electrospun nylon-6/hydroxyapatite membrane for bone tissue engineering. Eur. Polym. J. 2013, 49, 1314–1321. [Google Scholar] [CrossRef]
- Badrossamay, M.R.; McIlwee, H.A.; Goss, J.A.; Parker, K.K. Nanofiber Assembly by Rotary Jet-Spinning. Nano Lett. 2010, 10, 2257–2261. [Google Scholar] [CrossRef]
- Chien, K.R. Stress pathways and heart failure. Cell 1999, 98, 555–558. [Google Scholar] [CrossRef]
- Epstein, F.H.; Hunter, J.J.; Chien, K.R. Signaling pathways for cardiac hypertrophy and failure. N. Engl. J. Med. 1999, 341, 1276–1283. [Google Scholar]
- Van der Linde, D.; Konings, E.E.; Slager, M.A.; Witsenburg, M.; Helbing, W.A.; Takkenberg, J.J.; Roos-Hesselink, J.W. Birth prevalence of congenital heart disease worldwide: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2011, 58, 2241–2247. [Google Scholar] [CrossRef]
- Akins, R.E., Jr.; Rockwood, D.; Robinson, K.G.; Sandusky, D.; Rabolt, J.; Pizarro, C. Three-dimensional culture alters primary cardiac cell phenotype. Tissue Eng. Part A 2009, 16, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Battista, S.; Guarnieri, D.; Borselli, C.; Zeppetelli, S.; Borzacchiello, A.; Mayol, L.; Gerbasio, D.; Keene, D.R.; Ambrosio, L.; Netti, P.A. The effect of matrix composition of 3D constructs on embryonic stem cell differentiation. Biomaterials 2005, 26, 6194–6207. [Google Scholar] [CrossRef] [PubMed]
- Fomovsky, G.M.; Thomopoulos, S.; Holmes, J.W. Contribution of extracellular matrix to the mechanical properties of the heart. J. Mol. Cell. Cardiol. 2010, 48, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Reilly, G.C.; Engler, A.J. Intrinsic extracellular matrix properties regulate stem cell differentiation. J. Biomech. 2010, 43, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Florit, M.; Pardo, A.; Domingues, R.M.A.; Graça, A.L.; Babo, P.S.; Reis, R.L.; Gomes, M.E. Natural-Based Hydrogels for Tissue Engineering Applications. Molecules 2020, 25, 5858. [Google Scholar] [CrossRef]
- Pardo, A.; Gómez-Florit, M.; Barbosa, S.; Taboada, P.; Domingues, R.M.; Gomes, M.E. Magnetic Nanocomposite Hydrogels for Tissue Engineering: Design Concepts and Remote Actuation Strategies to Control Cell Fate. ACS Nano 2021, 15, 175–209. [Google Scholar] [CrossRef]
- Pardo, A.; Bakht, S.M.; Gomez-Florit, M.; Rial, R.; Monteiro, R.F.; Teixeira, S.P.; Taboada, P.; Reis, R.L.; Domingues, R.M.A.; Gomes, M.E. Magnetically-Assisted 3D Bioprinting of Anisotropic Tissue-Mimetic Constructs. Adv. Funct. Mater. 2022, 32, 2208940. [Google Scholar] [CrossRef]
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24, 401–416. [Google Scholar] [CrossRef]
- Gotoh, Y.; Tsukada, M.; Minoura, N. Effect of the chemical modification of the arginyl residue in Bombyx mori silk fibroin on the attachment and growth of fibroblast cells. J. Biomed. Mater. Res. 1998, 39, 351–357. [Google Scholar] [CrossRef]
- Gotoh, Y.; Tsukada, M.; Minoura, N.; Imai, Y. Synthesis of poly(ethylene glycol)-silk fibroin conjugates and surface interaction between L-929 cells and the conjugates. Biomaterials 1997, 18, 267–271. [Google Scholar] [CrossRef]
- Cai, K.; Yao, K.; Cui, Y.; Yang, Z.; Li, X.; Xie, H.; Qing, T.; Gao, L. Influence of Different Surface Modification Treatments on Poly (D, L-lactic acid) with Silk Fibroin and their Effects on the Culture of Osteoblast in vitro. Biomaterials 2002, 23, 1603–1611. [Google Scholar] [CrossRef] [PubMed]
- Fouad, H.; Khalil, K.A.; Alshammari, B.A.; Abdal-hay, A.; Abd El-salam, N.M. Development of New Bio-Composite of PEO/Silk Fibroin Blends Loaded with Piezoelectric Material. Polymers 2022, 14, 4209. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.F.; Yang, D.Z.; Xu, F.; Zhang, Z.P.; Yin, R.X.; Nie, J. Electrospun core-shell structure nanofibers from a homogeneous solution of poly(ethylene oxide)/chitosan. Macromolecules 2009, 42, 5278–5528. [Google Scholar] [CrossRef]
- Frenot, A.; Chronakis, I.S. Polymer nanofibers assembled by electrospinning. Curr. Opin. Colloid Interface Sci. 2003, 8, 64–75. [Google Scholar] [CrossRef]
- Chirakanphaisarn, N.; Thongkanluang, T.; Chiwpreechar, Y. Heart rate measurement and electrical pulse signal analysis for subjects span of 20–80 years. J. Electr. Syst. Inf. Technol. 2018, 5, 112–120. [Google Scholar] [CrossRef]
- Thiene, G.; Basso, C.; Cozza, A.; Zanatta, A. The fascinating discovery of the electrical system in the heart: A story telling. Int. J. Cardiol. 2020, 317, 81–85. [Google Scholar] [CrossRef]
- Chen, S.; Hsieh, M.H.; Li, S.H.; Wu, J.; Weisel, R.D.; Chang, Y.; Sung, H.W.; Li, R.K. A conductive cell-delivery construct as a bioengineered patch that can improve electrical propagation and synchronize cardiomyocyte contraction for heart repair. J. Control. Release 2020, 320, 73–82. [Google Scholar] [CrossRef]
- Gibbs, A.; McNamee, E.; Slade, D. Piezoelectric nanofibers as biomaterials for bone regeneration and wound healing. In Undergraduate Research in MSE; Review Article; University of Washington URSME: Seattle, WA, USA, 2020; Volume 1, Available online: https://digital.lib.washington.edu/researchworks/handle/1773/46255 (accessed on 15 March 2023).
- Dashtizad, S.; Alizadeh, P.; Yourdkhani, A. Improving piezoelectric properties of PVDF fibers by compositing with BaTiO3-Ag particles prepared by sol-gel method and photochemical reaction. J. Alloys Compd. 2021, 883, 160810. [Google Scholar] [CrossRef]
- Sencadas, V.; Garvey, C.; Mudie, S.; Kirkensgaard, J.J.; Gouadec, G.; Hauser, S. Electroactive properties of electrospun silk fibroin for energy harvesting applications. Nano Energy 2019, 66, 104106. [Google Scholar] [CrossRef]
- Sorayani Bafqi, M.S.; Sadeghi, A.H.; Latifi, M.; Bagherzadeh, R. Design and fabrication of a piezoelectric output evaluation system for sensitivity measurements of fibrous sensors and actuators. J. Ind. Text. 2021, 50, 1643–1659. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalil, A.K.A.; Fouad, H.; Abdal-hay, A.; Abd El-salam, N.M.; Abdelrazek Khalil, K. Fabrication and Characterization of Piezoelectric PEO/SF/BaTiO3 Scaffolds for Cardiac Tissue Engineering. J. Compos. Sci. 2023, 7, 200. https://doi.org/10.3390/jcs7050200
Khalil AKA, Fouad H, Abdal-hay A, Abd El-salam NM, Abdelrazek Khalil K. Fabrication and Characterization of Piezoelectric PEO/SF/BaTiO3 Scaffolds for Cardiac Tissue Engineering. Journal of Composites Science. 2023; 7(5):200. https://doi.org/10.3390/jcs7050200
Chicago/Turabian StyleKhalil, Abdelrahman K. A., Hassan Fouad, Abdalla Abdal-hay, Nasser M. Abd El-salam, and Khalil Abdelrazek Khalil. 2023. "Fabrication and Characterization of Piezoelectric PEO/SF/BaTiO3 Scaffolds for Cardiac Tissue Engineering" Journal of Composites Science 7, no. 5: 200. https://doi.org/10.3390/jcs7050200








