The Impact of Antimalarial Use on the Emergence and Transmission of Plasmodium falciparum Resistance: A Scoping Review of Mathematical Models
Abstract
:1. Introduction
Study Objective
2. Review Methodology
2.1. Step One: Identifying the Research Question
- How have mathematical modeling methods been used to assess the impact of antimalarial treatment in the human population on the emergence and transmission of P. falciparum resistance?
- What risk factors have been identified by mathematical models, of the use of antimalarial treatment in the human population on the emergence and transmission of P. falciparum resistance?
- What other factors have been identified to influence this relationship, through the use of mathematical modeling?
2.2. Step Two: Identifying Relevant Studies
2.3. Step Three: Study Selection
2.3.1. Inclusion Criteria
- Article must be written in English.
- Publishing date until the end of August 2016.
- The analysis section must contain a mathematical modeling-based approach.
- Results must be provided.
- The human malaria species P. falciparum must be modeled.
- The human host must be studied in the model, with outputs relevant to the human population provided. Other populations, such as the P. falciparum parasite or female Anopheles mosquito vector may also be included.
- The model must explore the effect of an antimalarial agent on the emergence and/or transmission (spread) of antimalarial resistance in P. falciparum.
- Full text must be available.
2.3.2. Exclusion Criteria
- Mathematical model not defined in the article.
- Human malaria species: P. vivax, P. malariae, P. knowlesi.
- Results section does not discuss the dynamics in regards to the human population.
- No full text available (e.g., conference abstract, embargoed).
2.4. Stage Four: Charting the Data
2.5. Stage Five: Collating, Summarising, and Reporting the Results
3. Results
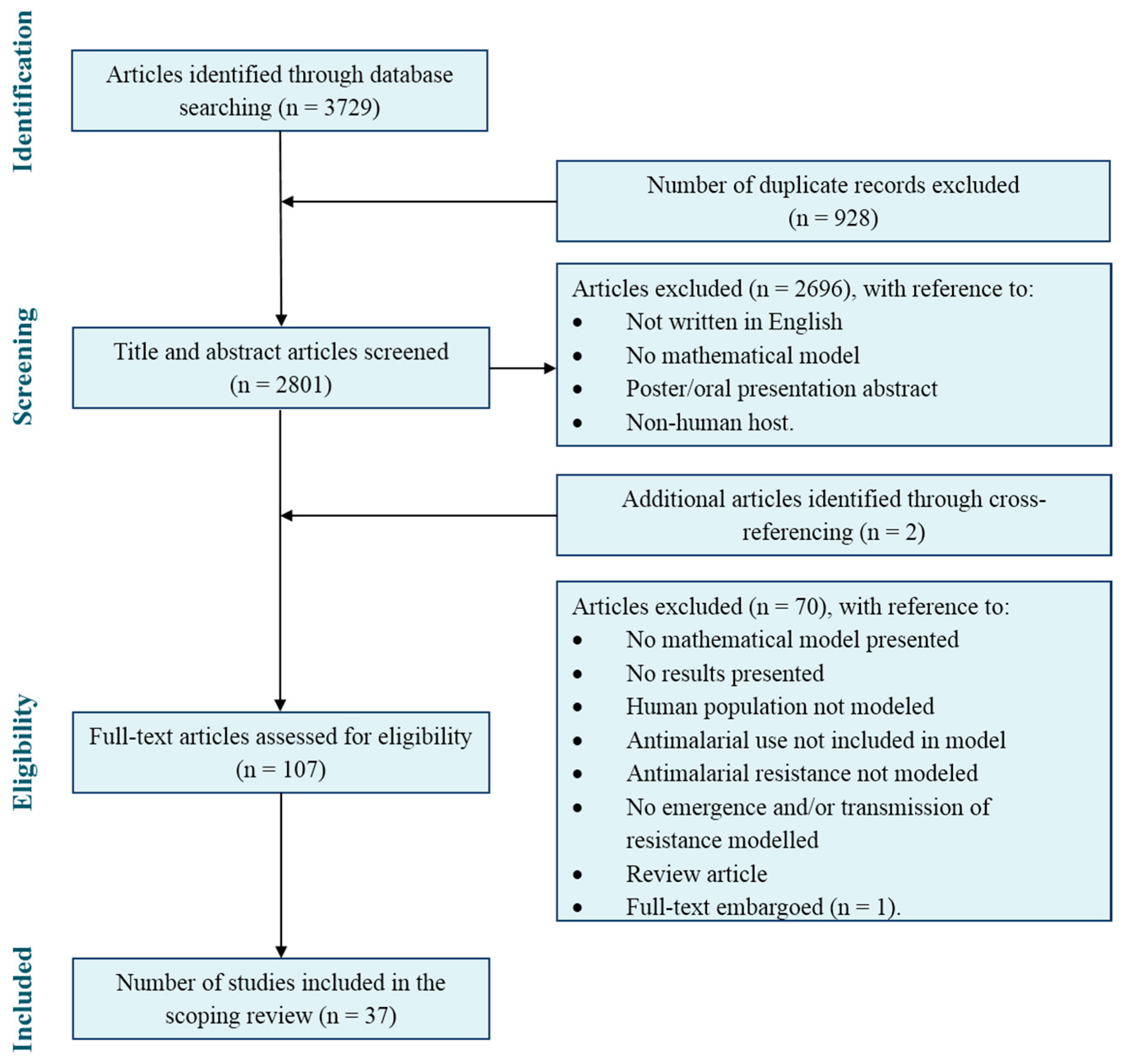
3.1. Study Selection
3.2. Mathematical Modelling Methods
3.2.1. Model Descriptions
3.2.2. Antimalarial Resistance
3.2.3. Antimalarial Treatment
3.2.4. Potential Influencing Factors
3.3. Risk Factors for the Emergence of Antimalarial Resistance Identified by Mathematical Models
3.3.1. Drug Selection Pressure
3.3.2. Plasmodium falciparum
3.3.3. Host Immunity
3.3.4. Transmission Intensity
3.3.5. Vector Control
3.4. Risk Factors for the Transmission of Antimalarial Resistance Identified by Mathematical Models
- An increase in presumptive antimalarial use [37];
- An increase in the infectious periods of hosts [28];
- A decrease in the recovery rate of nonimmune humans infected by resistant parasites [27];
- A decrease of within-host competition between drug-resistant and drug-sensitive P. falciparum parasites [32];
- An increase in the lifespan of resistant-infected mosquitoes [27];
- An increase in mosquito diffusion [27];
- An increase in the number of sporozoites injected from an infected female Anopheles mosquito to susceptible human per blood meal [34]; and
- A decrease in the use of transmission blockers (e.g., bednets) [36].
3.4.1. Drug Selection Pressure
3.4.2. Plasmodium falciparum
3.4.3. Host Immunity
3.4.4. Transmission Intensity
3.4.5. Vector Control
3.5. Risk Factors for the Emergence and Transmission of Antimalarial Resistance Identified by Mathematical Models
- Longer drug half-lives [43];
- Residual drug concentrations [45];
- An increased rate of parasite mutation [43];
- An increased relative fitness of resistant P. falciparum parasites compared to their drug-sensitive counterparts [44];
- A decrease in transmission intensity [47]; and
- The decreased use of transmission blockers (i.e., bednets) [49].
3.5.1. Drug Selection Pressure
3.5.2. Plasmodium falciparum
3.5.3. Transmission Intensity
3.5.4. Vector Control
4. Discussion
4.1. Mathematical Models
4.2. Risk Factors for the Emergence and Transmission of Resistance
4.3. Review Limitations
4.4.Implications for Further Research
4.4.1. Drug Selection Pressure
4.4.2. Influencing Factors
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. World Malaria Report 2016; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- White, N.J.; Pongtavornpinyo, W. The de novo selection of drug-resistant malaria parasites. Proc. Biol. Sci. 2003, 270, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Valderramos, S.; Valderramos, J.; Musset, L.; Purcell, L.; Mercereau-Puijalon, O.; Legrand, E.; Fidock, D.A. Identification of a mutant PfCRT-mediated chloroquine tolerance phenotype in Plasmodium falciparum. PLoS Pathog. 2010, 6, e1000887. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Report on Antimalarial Drug Efficacy and Drug Resistance: 2000–2010; Drug Resistance and Containment Unit, Global Malaria Programme, World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- White, N.J. Antimalarial drug resistance. J. Clin. Investig. 2004, 113, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- O’Meara, W.P.; Smith, D.L.; McKenzie, F.E. Potential impact of intermittent preventive treatment (IPT) on spread of drug-resistant malaria. PLoS Med. 2006, 3, e141. [Google Scholar]
- Hastings, I.M.; Paget-McNicol, S.; Saul, A. Can mutation and selection explain virulence in human P. falciparum infections? Malar. J. 2004, 3, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pongtavornpinyo, W.; Hastings, I.M.; Dondorp, A.; White, L.J.; Saralamba, S.; Day, N.P.; White, N.J.; Boni, M.F. Probability of emergence of antimalarial resistance in different stages of the parasite life cycle. Evol. Appl. 2009, 2, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Walliker, D. Malaria. In Molecular Epidemiology of Infectious Diseases; Thompson, R.A., Ed.; Arnold: London, UK, 2000; pp. 93–112. [Google Scholar]
- Walliker, D.; Hunt, P.; Babiker, H. Fitness of drug-resistant malaria parasites. Acta. Trop. 2005, 94, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Sarkar, R.R.; Sinha, S. Mathematical models of malaria—A review. Malar. J. 2011, 10, 202. [Google Scholar] [CrossRef] [PubMed]
- White, L.J.; Maude, R.J.; Pongtavornpinyo, W.; Saralamba, S.; Aguas, R.; Van Effelterre, T.; Day, N.P.J.; White, N.J. The role of simple mathematical models in malaria elimination strategy design. Malar. J. 2009, 8, 212. [Google Scholar] [CrossRef] [PubMed]
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef] [Green Version]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping studies: Advancing the methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Colquhoun, H.L.; Levac, D.; O’Brien, K.K.; Straus, S.; Tricco, A.C.; Perrier, L.; Kastner, M.; Moher, D. Scoping reviews: Time for clarity in definition, methods, and reporting. J. Clin. Epidemiol. 2014, 67, e1291–e1294. [Google Scholar] [CrossRef] [PubMed]
- Covidence Systematic Review Software. Available online: www.covidence.org (accessed on 9 October 2017).
- Cross, A.P.; Singer, B. Modelling the development and resistance of Plasmodium falciparum to anti-malarial drugs. Trans. R. Soc. Trop. Med. Hyg. 1991, 85, 349–355. [Google Scholar] [CrossRef]
- Curtis, C.F.; Otoo, L.N. A simple model of the build-up of resistance to mixtures of anti-malarial drugs. Trans. R. Soc. Trop. Med. Hyg. 1986, 80, 889–892. [Google Scholar] [CrossRef]
- Gatton, M.L.; Hogarth, W.; Saul, A. Time of treatment influences the appearance of drug-resistant parasites in Plasmodium falciparum infections. Parasitology 2001, 123, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Hastings, I.M. A model for the origins and spread of drug-resistant malaria. Parasitology 1997, 115, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Janssen, M.A.; Martens, W.J.M. Modeling malaria as a complex adaptive system. Artif. Life 1997, 3, 213–236. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Escalante, A.A.; Schneider, K.A. A population genetic model for the initial spread of partially resistant malaria parasites under anti-malarial combination therapy and weak intrahost competition. PLoS ONE 2014, 9, e101601. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.A.; Watkins, E.R.; Price, R.N.; Aarons, L.; Kyle, D.E.; White, N.J. Mefloquine pharmacokinetic-pharmacodynamic models: Implications for dosing and resistance. Antimicrob. Agents Chemother. 2000, 44, 3414–3424. [Google Scholar] [CrossRef] [PubMed]
- Watkins, W.M.; Sibley, C.H.; Hastings, I.M. The search for effective and sustainable treatments for Plasmodium falciparum malaria in Africa: A model of the selection of resistance by antifolate drugs and their combinations. Am. J. Trop. Med. Hyg. 2005, 72, 163–173. [Google Scholar] [PubMed]
- Antao, T.; Hastings, I.M. Environmental, pharmacological and genetic influences on the spread of drug-resistant malaria. Proc. R. Soc. B 2011, 278, 1705–1712. [Google Scholar] [CrossRef] [PubMed]
- Artzy-Randrup, Y.; Alonso, D.; Pascual, M. Transmission intensity and drug resistance in malaria population dynamics: Implications for climate change. PLoS ONE 2010, 5, e13588. [Google Scholar] [CrossRef] [PubMed]
- Bacaer, N.; Sokhna, C. A reaction-diffusion system modeling the spread of resistance to an antimalarial drug. Math. Biosci. Eng. 2005, 2, 227–238. [Google Scholar] [PubMed]
- Chiyaka, C.; Garira, W.; Dube, S. Effects of treatment and drug resistance on the transmission dynamics of malaria in endemic areas. Theor. Popul. Biol. 2009, 75, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Gatton, M.L.; Cheng, Q. Plasmodium falciparum infection dynamics and transmission potential following treatment with sulfadoxine-pyrimethamine. J. Antimicrob. Chemother. 2006, 58, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Hastings, I.M. Complex dynamics and stability of resistance to antimalarial drugs. Parasitology 2006, 132, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.Y.; Smith, D.L.; Boni, M.F.; Laxminarayan, R. Clinically immune hosts as a refuge for drug-sensitive malaria parasites. Malar. J. 2008, 7, 67. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.Y.; Smith, D.L.; Laxminarayan, R.; Levin, S. Superinfection and the evolution of resistance to antimalarial drugs. Proc. R. Soc. B 2012, 279, 3834–3842. [Google Scholar] [CrossRef] [PubMed]
- Koella, J.; Antia, R. Epidemiological models for the spread of anti-malarial resistance. Malar J. 2003, 2, 3. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, M.J.; Hastings, I.M. The evolution of multiple drug resistance in malaria parasites. Trans. R. Soc. Trop. Med. Hyg. 1998, 92, 188–195. [Google Scholar] [CrossRef]
- Maude, R.J.; Socheat, D.; Nguon, C.; Saroth, P.; Dara, P.; Li, G.; Song, J.; Yeung, S.; Dondorp, A.M.; Day, N.P.; et al. Optimising strategies for Plasmodium falciparum malaria elimination in Cambodia: Primaquine, mass drug administration and artemisinin resistance. PLoS ONE 2012, 7, e37166. [Google Scholar] [CrossRef] [PubMed]
- Maude, R.J.; Pontavornpinyo, W.; Saralamba, S.; Aguas, R.; Yeung, S.; Dondorp, A.M.; Day, N,P.; White, N.J.; White, L.J. The last man standing is the most resistant: Eliminating artemisinin-resistant malaria in Cambodia. Malar. J. 2009, 8, 31. [Google Scholar]
- Pongtavornpinyo, W.; Yeung, S.; Hastings, I.M.; Dondorp, A.M.; Day, N.P.J.; White, N.J. Spread of anti-malarial drug resistance: Mathematical model with implications for ACT drug policies. Malar. J. 2008, 7, 299. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.A.; Kim, Y. An analytical model for genetic hitchhiking in the evolution of antimalarial drug resistance. Theor. Popul. Biol. 2010, 78, 93–108. [Google Scholar] [CrossRef] [PubMed]
- Slater, H.C.; Griffin, J.T.; Ghani, A.C.; Okell, L.C. Assessing the potential impact of artemisinin and partner drug resistance in sub-Saharan Africa. Malar. J. 2016, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Tasman, H.; Soewono, E.; Sidarto, K.A. A model for transmission of partial resistance to anti-malarial drugs. Math. Biosci. Eng. 2009, 6, 649–661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teboh-Ewungkem, M.I.; Mohammed-Awel, J.; Baliraine, F.N.; Duke-Sylvester, S.M. The effect of intermittent preventive treatment on anti-malarial drug resistance spread in areas with population movement. Malar. J. 2014, 13, 428. [Google Scholar] [CrossRef] [PubMed]
- Winter, K.; Hastings, I.M. Development, evaluation, and application of an in silico model for antimalarial drug treatment and failure. Antimicrob. Agents Chemother. 2011, 55, 3380–3392. [Google Scholar] [CrossRef] [PubMed]
- An, L.T.T.; Jäger, W. A quantitative model of population dynamics in malaria with drug treatment. J. Math. Biol. 2014, 69, 659–685. [Google Scholar] [CrossRef] [PubMed]
- Esteva, L.; Gumel, A.B.; de León, C.V. Qualitative study of transmission dynamics of drug-resistant malaria. Math. Comput. Model. 2009, 50, 611–630. [Google Scholar] [CrossRef]
- Hastings, I.M.; Watkins, W.M.; White, N.J. The evolution of drug-resistant malaria: The role of drug elimination half-life. Philoso. Trans. R. Soc. B 2002, 357, 505–519. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.Y. The impact of heterogeneous transmission on the establishment and spread of antimalarial drug resistance. J. Theor. Biol. 2014, 340, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Legros, M.; Bonhoeffer, S. A combined within-host and between-hosts modelling framework for the evolution of resistance to antimalarial drugs. J. R. Soc. Interface 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, M.J. Drug resistance models for malaria. Acta. Trop. 2005, 94, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Maude, R.J.; Nguon, C.; Dondorp, A.M.; White, L.J.; White, N.J. The diminishing returns of atovaquone-proguanil for elimination of Plasmodium falciparum malaria: Modelling mass drug administration and treatment. Malar. J. 2014, 13, 380. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.L.; Klein, E.Y.; McKenzie, F.E.; Laxminarayan, R. Prospective strategies to delay the evolution of anti-malarial drug resistance: Weighing the uncertainty. Malar. J. 2010, 9, 217. [Google Scholar] [CrossRef] [PubMed]
- Tchuenche, J.M.; Chiyaka, C.; Chan, C.; Matthews, A.; Mayer, G. A mathematical model for antimalarial drug resistance. Math. Med. Biol. 2011, 28, 335–355. [Google Scholar] [CrossRef] [PubMed]
- Bloland, P.B.; Williams, H.A.; National Research Council; Committee on Population. Malaria Control during Mass Population Movements and Natural Disasters; The National Academies Press: Washington, DC, USA, 2003. [Google Scholar]
- WHO. Guidelines for the Treatment of Malaria, 3rd edition; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Bassat, Q.; Tanner, M.; Guerin, P.J.; Stricker, K.; Hamed, K. Combating poor quality antimalarial medicines: A call to action. Malar. J. 2016, 15, 302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ambroise-Thomas, P. The tragedy caused by fake antimalarial drugs. Mediterr. J. Hematol. Infect. Dis. 2012, 4, e2012027. [Google Scholar] [CrossRef] [PubMed]
- Newton, P.; Green, M.; Fernandez, F. Impact of poor quality-quality medicines in the ‘developing’ world. Trends Pharmacol. Sci. 2009, 31, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Newton, P.N.; Caillet, C.; Guerin, P.J. A link between poor quality antimalarials and malaria drug resistance? Expert Rev. Anti Infect. Ther. 2016, 14, 531–533. [Google Scholar] [CrossRef] [PubMed]
- Newton, P.N.; Green, M.D.; Fernandez, F.; Day, N.P.; White, N.J. Counterfeit anti-infective drugs. Lancet Infect. Dis. 2006, 6, 602–613. [Google Scholar] [CrossRef]
- Harper, J.; Morris, J.; Satchwell, G.; Stevens, P.; Taylor, D.; Tremblay, M. Coincidence or Crisis? Prescription Medicine Counterfeiting; Pitts, P.J., Ed.; The Stockholm Network: London, UK, 2006. [Google Scholar]
- Newton, P.N.; Fernandez, F.M.; Plancon, A.; Mildenhall, D.C.; Green, M.D.; Ziyong, L.; Christophel, E.M.; Phanouvong, S.; Howells, S.; McIntosh, E.; et al. A collaborative epidemiological investigation into the criminal fake artesunate trade in South East Asia. PLoS Med. 2008, 5, e32. [Google Scholar] [CrossRef] [PubMed]
| Model Feature | Frequency [Reference(s)] | ||
|---|---|---|---|
| Emergence (Nine Articles) | Transmission (19 Articles) | Both (Nine Articles) | |
| Model Description | |||
| Model type: | |||
| Deterministic | 6 [8,18,19,20,23,24] | 13 [6,25,26,27,28,31,33,35,38,39,40,41,42] | 6 [43,44,45,48,49,51] |
| Stochastic | 3 [17,21,22] | 5 [29,30,32,34,37] | 1 [46] |
| Both | 1 [36] | 2 [47,50] | |
| Scope of model: | |||
| Applied | 2 [17,23] | 4 [35,36,39,42] | 3 [43,49,51] |
| Theoretical | 7 [8,18,19,20,21,22,24] | 15 [6,25,26,27,28,29,30,31,32,33,34,37,38,40,41] | 6 [44,45,46,47,48,50] |
| Populations modeled: | |||
| Human | 8 [6,30,31,35,36,37,39,41] | 3 [48,49,50] | |
| Human & mosquito | 2 [17,18] | 6 [26,27,28,33,34,40] | 4 [44,46,47,51] |
| Human & plasmodia | 5 [8,19,20,23,24] | 3 [25,29,42] | 1 [45] |
| Human, mosquito & plasmodia | 2 [21,22] | 2 [32,38] | 1 [43] |
| Transparency and reproducibility of model: | |||
| Assumptions | 9 [8,17,18,19,20,21,22,23,24] | 19 [6,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42] | 8 [43,44,45,46,47,48,49,50,51] |
| Equations provided | 7 [8,19,20,21,22,23,24] | 17 [6,26,27,28,30,31,32,33,34,35,36,37,38,39,40,41,42] | 8 [43,44,45,46,47,48,50,51] |
| Model flowchart provided | 6 [8,17,18,19,21,22] | 18 [6,26,27,28,29,31,32,33,34,35,36,37,38,39,40,41] | 5 [43,46,48,49,51] |
| Model validation | 2 [17,23] | 5 [26,27,29,35,42] | 2 [43,45] |
| Parameters provided | 8 [8,17,19,20,21,22,23,24] | 16 [6,26,27,28,30,31,32,34,35,36,37,38,39,40,41,42] | 8 [43,44,45,46,47,49,50,51] |
| Sensitivity analysis | 2 [19,21] | 5 [26,29,30,36,37] | 2 [47,50] |
| Antimalarial Resistance | |||
| Monotherapy | |||
| Artemisinin | 1 [8] | 3 [35,39,42] | 1 [49] |
| Artesunate | 3 [6,35,36] | 1 [43] | |
| Atovaquone | 1 [49] | ||
| Chloroproguanil-dapsone | 1 [41] | ||
| Chloroquine | 1 [42] | 1 [43] | |
| Lumefantrine | 1 [42] | ||
| Mefloquine | 1 [23] | 1 [42] | |
| Piperaquine | 5 [6,35,36,39,42] | ||
| Pyrimethamine | 1 [17] | ||
| Quinine | 1 [43] | ||
| Not specified | 2 [18,22] | 3 [25,27,34] | |
| Combination therapy | |||
| Artemisinin-based combination therapy (ACT) | 1 [39] | 1 [50] | |
| Sulfadoxine-pyrimethamine | 1 [24] | 4 [25,29,39,41] | 2 [43,45] |
| Not specified | 2 [18,22] | 2 [25,34] | |
| Partner-drug resistance (not specified) | 2 [8,24] | 3 [34,35,37] | |
| Resistance type not specified | 2 [19,20] | 8 [26,28,30,31,32,33,38,40] | 5 [44,46,47,48,51] |
| Degree of resistance specified (partial, full) | 3 [21,23,24] | 5 [6,29,39,40,41] | 3 [45,46,51] |
| Drug Selection Pressure | |||
| Antimalarial treatment: | |||
| Monotherapies | |||
| Artemisinin | 1 [8] | ||
| Artesunate | 1 [36] | 2 [43,49] | |
| Chloroproguanil-dapsone | 1 [24] | 2 [6,41] | |
| Chloroquine | 1 [42] | 1 [43] | |
| Lumefantrine | 1 [42] | ||
| Mefloquine | 1 [23] | 1 [42] | |
| Piperaquine | 1 [42] | ||
| Pyrimethamine | 1 [17] | ||
| Quinine | 1 [43] | ||
| Not specified | 2 [18,22] | 3 [25,27,34] | |
| Combination therapies | |||
| Artemether-lumefantrine | 1 [42] | ||
| Artemisinin-based combination therapy (ACT) | 1 [8] | 1 [37] | 2 [49,50] |
| Artemisinin-piperaquine | 1 [35] | ||
| Artemisinin-piperaquine + primaquine | 2 [35,36] | 1 [49] | |
| Artesunate + piperaquine | 1 [36] | ||
| Artesunate + mefloquine | 1 [42] | ||
| Artesunate + chloroquine | 1 [42] | ||
| Artesunate-lumefantrine | 1 [42] | ||
| Atovaquone + progunail | 1 [36] | 1 [49] | |
| Atovaquone + progunail + primaquine | 1 [36] | 1 [49] | |
| Chloroproguanil-dapsone + artesunate | 1 [24] | ||
| Dihydroartemisinin + piperaquine | 2 [39,42] | ||
| Sulfadoxine-pyrimethamine | 1 [24] | 3 [6,29,41] | 2 [43,45] |
| Sulfadoxine-pyrimethamine + amodiaquine | 1 [24] | ||
| Sulfadoxine-pyrimethamine + artesunate | 1 [24] | 1 [39] | 1 [45] |
| Chloroproguanil-dapsone + artesunate | 1 [45] | ||
| Not specified | 2 [18,22] | 2 [25,34] | |
| Treatment not specified | 3 [19,20,21] | 8 [26,28,30,31,32,33,38,40] | 5 [44,46,47,48,51] |
| Antimalarial treatment strategies: | |||
| Intermittent-preventive treatment (IPT) | 3 [6,39,41] | 1 [49] | |
| Mass drug administration (MDA) | 2 [35,36] | 1 [49] | |
| Mass screening and treatment (MSAT) | 1 [36] | ||
| Antimalarial properties and duration of treatment: | |||
| Full/partial treatment duration | 1 [25] | ||
| Half-life/decay of concentration with time | 2 [22,23] | 3 [6,41,42] | 2 [43,45] |
| High/low dose | 2 [22,23] | 1 [49] | |
| Residual levels | 2 [8,23] | 1 [41] | |
| Levels of drug efficacy | 1 [17] | ||
| Parasite growth restriction following treatment | 1 [22] | ||
| Patient compliance | 1 [37] | ||
| Protection from reinfection | 1 [24] | ||
| Transmissibility following treatment | 1 [22] | ||
| Potential Influencing Factors | |||
| Plasmodium falciparum: | |||
| Asexual parasite density | 3 [17,22,23] | 1 [29] | |
| Epistasis | 1 [25] | ||
| Frequency of mutation | 1 [25] | ||
| Gametocyte parasite density | 3 [17,22,24] | 1 [29] | |
| Genetic recombination | 3 [18,20,22] | 2 [25,34] | |
| Inbreeding and/or random mating | 3 [25,34,38] | ||
| Infectivity/transmissibility following treatment | 1 [17] | 4 [28,29,31,34] | |
| Parasite fitness | 4 [19,20,21,22] | 12 [6,25,26,27,30,31,32,33,34,38,40,41] | 6 [44,46,47,48,50,51] |
| Multiplicity of infection (MOI) | 1 [32] | ||
| Mutation rate | 1 [22] | ||
| Natural selection | 1 [22] | 3 [26,30,32] | 1 [46] |
| Host immunity: | |||
| Acquired/clinical immunity or host age-dependent | 4 [8,17,21,23] | 13 [6,25,26,27,28,31,33,35,36,39,40,41,42] | 3 [43,46,49] |
| Immune response | 1 [19] | 1 [47] | |
| Generalized/strain specific immunity | 1 [20] | 1 [47] | |
| Symptomatic and/or asymptomatic infection | 1 [22] | 7 [6,29,31,35,36,39,41] | |
| Transmission intensity | 3 [8,21,24] | 11 [6,25,26,31,32,36,37,38,39,41] | 1 [45] |
| Female Anopheles mosquito: | |||
| Competition for blood meal | 1 [43] | ||
| Entomological inoculation rate (EIR) | 5 [31,32,37,41,42] | ||
| Fitness of mosquitoes to produce offspring | 1 [21] | ||
| Insecticide resistance | 1 [21] | ||
| Population size dependent on climatic factors | 2 [17,21] | 1 [26] | |
| Sporozoite measure (count/rate) | 1 [34] | ||
| Transmission blockers: | 1 [45] | ||
| Insecticidal bednets | 3 [35,36,39] | 1 [49] | |
| Transmission potential | 1 [21] | 1 [26] | |
| Vectorial capacity | 4 [26,31,32,37] | ||
| Treatment Scenario | Emergence | Transmission | Emergence and Transmission |
|---|---|---|---|
| Contributing to population treatment coverage | |||
| IPT use | X | ||
| MDA | X | ||
| MSAT | X | X | |
| Self-medication | X | X | X |
| Contributing to residual drug concentrations | |||
| Drug efficacy | X | X | |
| Drug quality (falsified, substandard and degraded) | X | X | X |
| Full/partial treatment and patient compliance | X | X | |
| High/low dose | X | ||
| Percentage API | X | X | X |
| Residual/subtherapeutic API | X | ||
| Self-medication | X | X | X |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brock, A.R.; Gibbs, C.A.; Ross, J.V.; Esterman, A. The Impact of Antimalarial Use on the Emergence and Transmission of Plasmodium falciparum Resistance: A Scoping Review of Mathematical Models. Trop. Med. Infect. Dis. 2017, 2, 54. https://doi.org/10.3390/tropicalmed2040054
Brock AR, Gibbs CA, Ross JV, Esterman A. The Impact of Antimalarial Use on the Emergence and Transmission of Plasmodium falciparum Resistance: A Scoping Review of Mathematical Models. Tropical Medicine and Infectious Disease. 2017; 2(4):54. https://doi.org/10.3390/tropicalmed2040054
Chicago/Turabian StyleBrock, Aleisha R., Carole A. Gibbs, Joshua V. Ross, and Adrian Esterman. 2017. "The Impact of Antimalarial Use on the Emergence and Transmission of Plasmodium falciparum Resistance: A Scoping Review of Mathematical Models" Tropical Medicine and Infectious Disease 2, no. 4: 54. https://doi.org/10.3390/tropicalmed2040054
APA StyleBrock, A. R., Gibbs, C. A., Ross, J. V., & Esterman, A. (2017). The Impact of Antimalarial Use on the Emergence and Transmission of Plasmodium falciparum Resistance: A Scoping Review of Mathematical Models. Tropical Medicine and Infectious Disease, 2(4), 54. https://doi.org/10.3390/tropicalmed2040054





