Citric Acid Production by Yarrowia lipolytica Yeast on Different Renewable Raw Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganisms and Chemicals
2.2. Media and Cultivation Conditions
2.3. Assays
2.4. Calculations
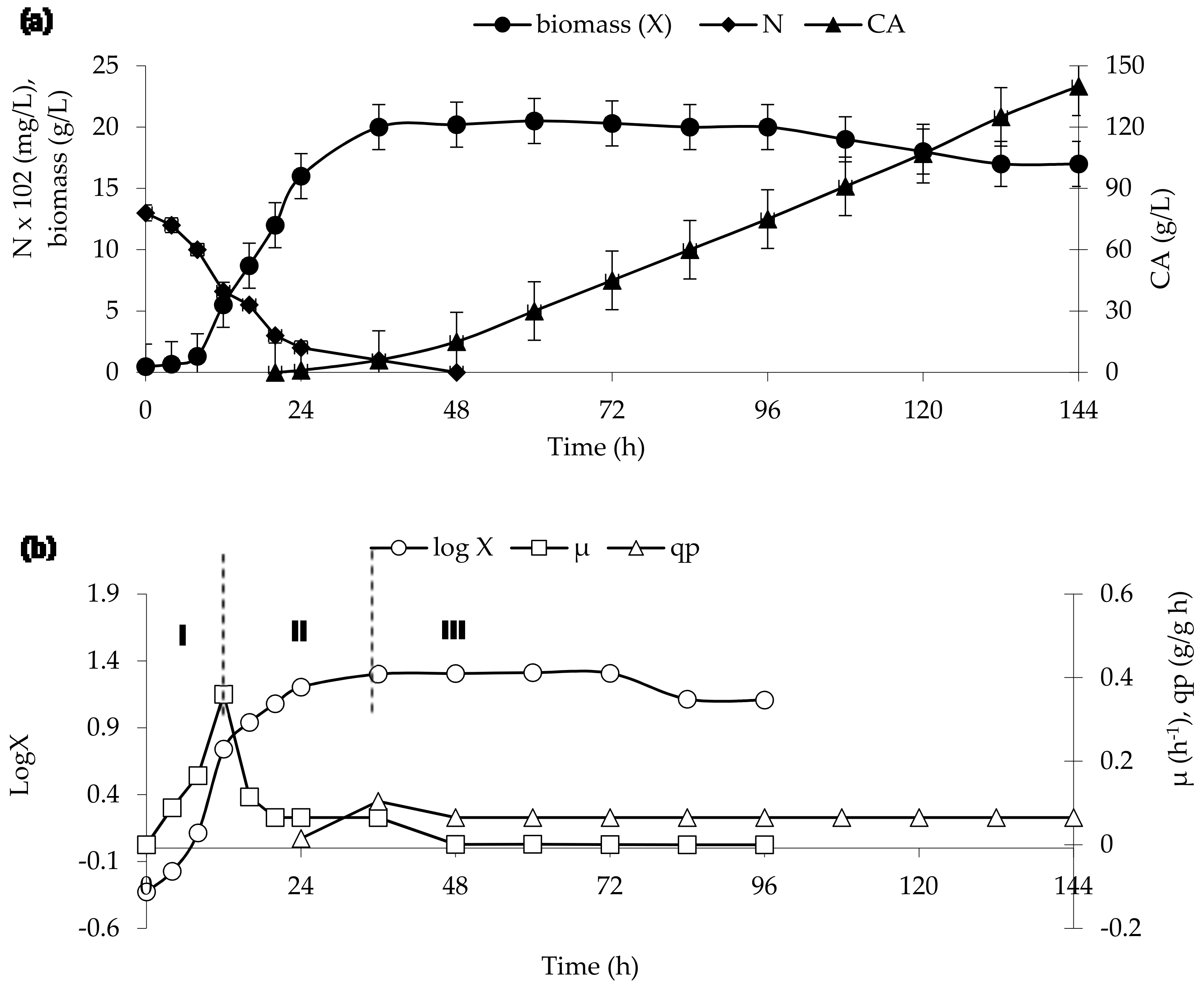
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cavallo, E.; Charreau, H.; Cerrutti, P.; Foresti, M.L. Yarrowia lipolytica: A model yeast for citric acid production. FEMS Yeast Res. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Anastassiadis, S.; Morgunov, I.G.; Kamzolova, S.V.; Finogenova, T.V. Citric acid production patent review. Recent Pat. Biotechnol. 2008, 2, 107–123. [Google Scholar] [CrossRef] [PubMed]
- Finogenova, T.V.; Morgunov, I.G.; Kamzolova, S.V.; Chernyavskaya, O.G. Organic acid production by the yeast Yarrowia lipolytica: A review of prospects. Appl. Biochem. Microbiol. 2005, 41, 418–425. [Google Scholar] [CrossRef]
- Moeller, L.; Strehlitz, B.; Aurich, A.; Zehnsdorf, A.; Bley, T. Optimization of citric acid production from glucose by Yarrowia lipolytica. Eng. Life Sci. 2007, 7, 504–511. [Google Scholar] [CrossRef]
- Tan, M.J.; Chen, X.; Wang, Y.K.; Liu, G.L.; Chi, Z.M. Enhanced citric acid production by a yeast Yarrowia lipolytica over-expressing a pyruvate carboxylase gene. Bioprocess Biosyst. Eng. 2016, 39, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.Y.; Lu, Y.; Chi, Z.; Liu, G.L.; Zhao, S.F.; Jiang, H.; Chi, Z.M. Cloning and characterization of a pyruvate carboxylase from Penicillium rubens and overexpression of the gene in the yeast Yarrowia lipolytica for enhanced citric acid production. Mar. Biotechnol. 2016, 18, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kamzolova, S.V.; Morgunov, I.G. Metabolic peculiarities of the citric acid overproduction from glucose in yeasts Yarrowia lipolytica. Bioresour. Technol. 2017, 243, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Kamzolova, S.V.; Morgunov, I.G.; Aurich, A.; Perevoznikova, O.A.; Shishkanova, N.V.; Stottmeister, U.; Finogenova, T.V. Lipase secretion and citric acid production in Yarrowia lipolytica yeast grown on animal and vegetable fat. Food Technol. Biotechnol. 2005, 43, 113–122. [Google Scholar]
- Kamzolova, S.V.; Lunina, J.N.; Morgunov, I.G. Biochemistry of citric acid production from rapeseed oil by Yarrowia lipolytica yeast. J. Am. Oil Chem. Soc. 2011, 88, 1965–1976. [Google Scholar] [CrossRef]
- Aurich, A.; Specht, R.; Müller, R.A.; Stottmeister, U.; Yovkova, V.; Otto, C.; Holz, M.; Barth, G.; Heretsch, P.; Thomas, F.A.; et al. Microbiologically Produced Carboxylic Acids Used as Building Blocks in Organic Synthesis. In Reprogramming Microbial Metabolic Pathways; Wang, X., Chen, J., Quinn, P., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 391–424. Available online: https://link.springer.com/chapter/10.1007%2F978-94-007-5055-5_19 (accessed on 28 September 2012).
- Rymowicz, W.; Rywińska, A.; Żarowska, B.; Juszczyk, P. Citric acid production from raw glycerol by acetate mutants of Yarrowia lipolytica. Chem. Pap. 2006, 60, 391–395. [Google Scholar] [CrossRef]
- Levinson, W.E.; Kurtzman, C.P.; Kuo, T.M. Characterization of Yarrowia lipolytica and related species for citric acid production from glycerol. Enzym. Microb. Technol. 2007, 41, 292–295. [Google Scholar] [CrossRef]
- Rywińska, A.; Rymowicz, W.; Źarowska, B.; Wojtatowicz, M. Biosynthesis of citric acid from glycerol by acetate mutants of Yarrowia lipolytica in fed-batch fermentation. Food Technol. Biotechnol. 2009, 47, 1–6. [Google Scholar]
- Makri, A.; Fakas, S.; Aggelis, G. Metabolic activities of biotechnological interest in Yarrowia lipolytica grown on glycerol in repeated batch cultures. Bioresour. Technol. 2010, 101, 2351–2358. [Google Scholar] [CrossRef] [PubMed]
- Morgunov, I.G.; Kamzolova, S.V.; Lunina, J.N. The citric acid production from raw glycerol by Yarrowia lipolytica yeast and its regulation. Appl. Microbiol. Biotechnol. 2013, 97, 7387–7397. [Google Scholar] [CrossRef] [PubMed]
- Rywinska, A.; Juszczyk, P.; Wojtatowicz, M.; Robak, M.; Lazar, Z.; Tomaszewska, L.; Rymowicz, W. Glycerol as a promising substrate for Yarrowia lipolytica biotechnological applications. Biomass Bioenergy 2013, 48, 148–166. [Google Scholar] [CrossRef]
- Morgunov, I.G.; Kamzolova, S.V. Physiologo-biochemical characteristics of citrate-producing yeast Yarrowia lipolytica grown on glycerol-containing waste of biodiesel industry. Appl. Microbiol. Biotechnol. 2015, 99, 6443–6450. [Google Scholar] [CrossRef] [PubMed]
- Wojtatowicz, M.; Rymowicz, W.; Kautola, H. Comparison of different strains of the yeast Yarrowia lipolytica for citric acid production from glucose hydrol. Appl. Biochem. Biotechnol. 1991, 31, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Ledesma-Amaro, R.; Lazar, Z.; Rakicka, M.; Guo, Z.; Fouchard, F.; Coq, A.C.; Nicaud, J.M. Metabolic engineering of Yarrowia lipolytica to produce chemicals and fuels from xylose. Metab. Eng. 2016, 38, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, S.; Fakas, S.; Fick, M.; Chevalot, I.; Galiotou-Panayotou, M.; Komaitis, M.; Marc, I.; Aggelis, G. Biotechnological valorisation of raw glycerol discharged after biodiesel (fatty acid methyl-esters) manufacturing process: Production of 1,3-propanediol, citric acid and single oil. Biomass Bioengergy 2008, 32, 60–71. [Google Scholar] [CrossRef]
- Rakicka, M.; Lazar, Z.; Rywinska, A.; Rymowicz, W. Efficient utilization of inulin and glycerol as fermentation substrates in erythritol and citric acid production using expressing inulinase. Chem. Pap. 2016, 70, 1452–1459. [Google Scholar] [CrossRef]
- Stephanopoulos, G. Challenges in engineering microbes for biofuels production. Science 2007, 315, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Weusthuis, R.A.; Aarts, J.M.M.J.G.; Sanders, J.P.M. From biofuel to bioproduct: Is bioethanol a suitable fermentation feedstock for synthesis of bulk chemicals? Biofuels Bioprod. Biorefin. 2011, 5, 486–494. [Google Scholar] [CrossRef]
- Finogenova, T.V.; Puntus, I.F.; Kamzolova, S.V.; Lunina, I.N.; Monastyrskaia, S.E.; Morgunov, I.G.; Boronin, A.M. Obtaining of the mutant Yarrowia lipolytica strains producing citric acid from glucose. Prikl. Biokhimiia Mikrobiol. 2008, 44, 219–224. [Google Scholar]
- Good, D.W.; Droniuk, R.; Lawford, R.G.; Fein, J.E. Isolation and characterization of a Saccharomycopsis lipolytica mutant showing increased production of citric acid from canola oil. Can. J. Microbiol. 1985, 31, 436–440. [Google Scholar] [CrossRef]
- Aurich, A.; Förster, A.; Mauesberger, S.; Barth, G.; Stottmeister, U. Citric acid production from renewable resources by Yarrowia lipolytica. Biotechnol. Adv. 2003, 21, 454–455. [Google Scholar]
- Papanikolaou, S.; Chatzifragkou, A.; Fakas, S.; Galiotou-Panayotou, M.; Komaitis, M.; Nicaud, J.M.; Aggelis, G. Biosynthesis of lipids and organic acids by Yarrowia lipolytica strains cultivated on glucose. Eur. J. Lipid Sci. Technol. 2009, 111, 1221–1232. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Galiotou-Panayotou, M.; Fakas, S.; Komaitis, M.; Aggelis, G. Citric acid production by Yarrowia lipolytica cultivated on olive-mill wastewater-based media. Bioresour. Technol. 2008, 99, 2419–2428. [Google Scholar] [CrossRef] [PubMed]
- Sarris, D.; Stoforos, N.G.; Mallouchos, A.; Kookos, I.K.; Koutinas, A.A.; Aggelis, G.; Papanikolaou, S. Production of added-value metabolites by Yarrowia lipolytica growing in olive mill wastewater-based media under aseptic and non-aseptic conditions. Eng. Life Sci. 2017, 17, 695–709. [Google Scholar] [CrossRef]
- Celińska, E.; Grajek, W. A novel multigene expression construct for modification of glycerol metabolism in Yarrowia lipolytica. Microb. Cell Fact. 2013, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- Förster, A.; Aurich, A.; Mauersberger, S.; Barth, G. Citric acid production from sucrose using a recombinant strain of the yeast Yarrowia lipolytica. Appl. Microbiol. Biotechnol. 2007, 75, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
| Substrates | Biomass (g L−1) | CA (g L−1) | ICA (g L−1) | CA:ICA | Productivity (g (L·h)−1) | YCA (g g−1) |
|---|---|---|---|---|---|---|
| Rapeseed oil | 17.0 ± 1.1 | 140.0 ± 5.0 | 5.3 ± 0.8 | 26.2:1 | 1.46 | 1.50 |
| Glucose | 18.7 ± 1.3 | 100.8 ± 9.2 | 4.9 ± 0.9 | 20.6:1 | 1.05 | 0.80 |
| Glucose-containing aspen waste | 5.6 ± 0.8 | 31.2 ± 2.1 | 7.84 ± 0.9 | 4:1 | 0.325 | 0.50 |
| Glycerol | 16.8 ± 1.1 | 87 ± 6.4 | 13 ± 1.1 | 6.7:1 | 0.906 | 0.64 |
| Glycerol waste of biodiesel industry | 20.0 ± 1.8 | 100 ± 3.4 | 15 ± 1.2 | 7.7:1 | 1.04 | 0.90 |
| Ethanol | 15.3 ± 1.4 | 106.7 ± 2.7 | 15 ± 1.4 | 7.1:1 | 1.32 | 0.87 |
| Strain | Substrate | Characteristics of Strain | CA (g L−1) | YCA (g g−1) | References |
|---|---|---|---|---|---|
| S. lipolytica NTG9 | canola oil | mutant/nitrosoguanidine | 152.3 | 1.50 | [25] |
| Y. lipolytica H181 | sunflower oil | wild type | 198.0 | 1.16 | [26] |
| Y. lipolytica H222 | glucose | wild type | 41.0 | 0.55 | [4] |
| Y. lipolytica W29l | wild type | 49.0 | 0.85 | [27] | |
| Y. lipolytica VKM Y 2373 | wild type | 80–85 | 0.70–0.75 | [7] | |
| Y. lipolytica PG86 | PYC gene expression | 95.0 | 0.75 | [5] | |
| Y. lipolytica PR32 | PYC gene expression | 111.1 | 0.93 | [6] | |
| Y. lipolytica ACA-DS 50109 | glucose + olive mill wastewaters | wild type | 28.9 | 0.53 | [28] |
| Y. lipolytica ACA-YC 5033 | wild type | 52.0 | 0.64 | [29] | |
| Y. lipolytica NRRL YB-423 | glycerol | wild type | 21.6 | 0.54 | [12] |
| Y. lipolytica ACA-DC 50109 | raw glycerol | wild type | 62.5 | 0.56 | [20] |
| Y. lipolytica NCYC 3825 | raw glycerol | multigene expression | 58.8 | 0.17 | [30] |
| Y. lipolytica NG40/UV7 | raw glycerol | mutant/nitrosoguanidine/UV | 122.2 | 0.95 | [17] |
| Y. lipolytica Wratislavia AWG7 | raw glycerol | mutant/acetate− | 139.0 | 0.70 | [13] |
| Y.lipolytica H222-S4 (p67ICL1) | sucrose | ScSUC2/ICL1 | 127–140 | 0.75–0.82 | [31] |
| Y. lipolytica XYL+ | xylose | XYL gene expression | 80.0 | 0.53 | [19] |
| Y. lipolytica Wratislavia K1 | inulin | INU1 gene expression | 105.2 | 0.53 | [21] |
| Y. lipolytica NG40/UV5 | rapeseed oil | mutant/nitrosoguanidine/UV | 140.0 | 1.5 | Present study |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morgunov, I.G.; Kamzolova, S.V.; Lunina, J.N. Citric Acid Production by Yarrowia lipolytica Yeast on Different Renewable Raw Materials. Fermentation 2018, 4, 36. https://doi.org/10.3390/fermentation4020036
Morgunov IG, Kamzolova SV, Lunina JN. Citric Acid Production by Yarrowia lipolytica Yeast on Different Renewable Raw Materials. Fermentation. 2018; 4(2):36. https://doi.org/10.3390/fermentation4020036
Chicago/Turabian StyleMorgunov, Igor G., Svetlana V. Kamzolova, and Julia N. Lunina. 2018. "Citric Acid Production by Yarrowia lipolytica Yeast on Different Renewable Raw Materials" Fermentation 4, no. 2: 36. https://doi.org/10.3390/fermentation4020036