Diamond-Like Carbon Nanofoam from Low-Temperature Hydrothermal Carbonization of a Sucrose/Naphthalene Precursor Solution
Abstract
:1. Introduction
2. Results
2.1. Microscopy
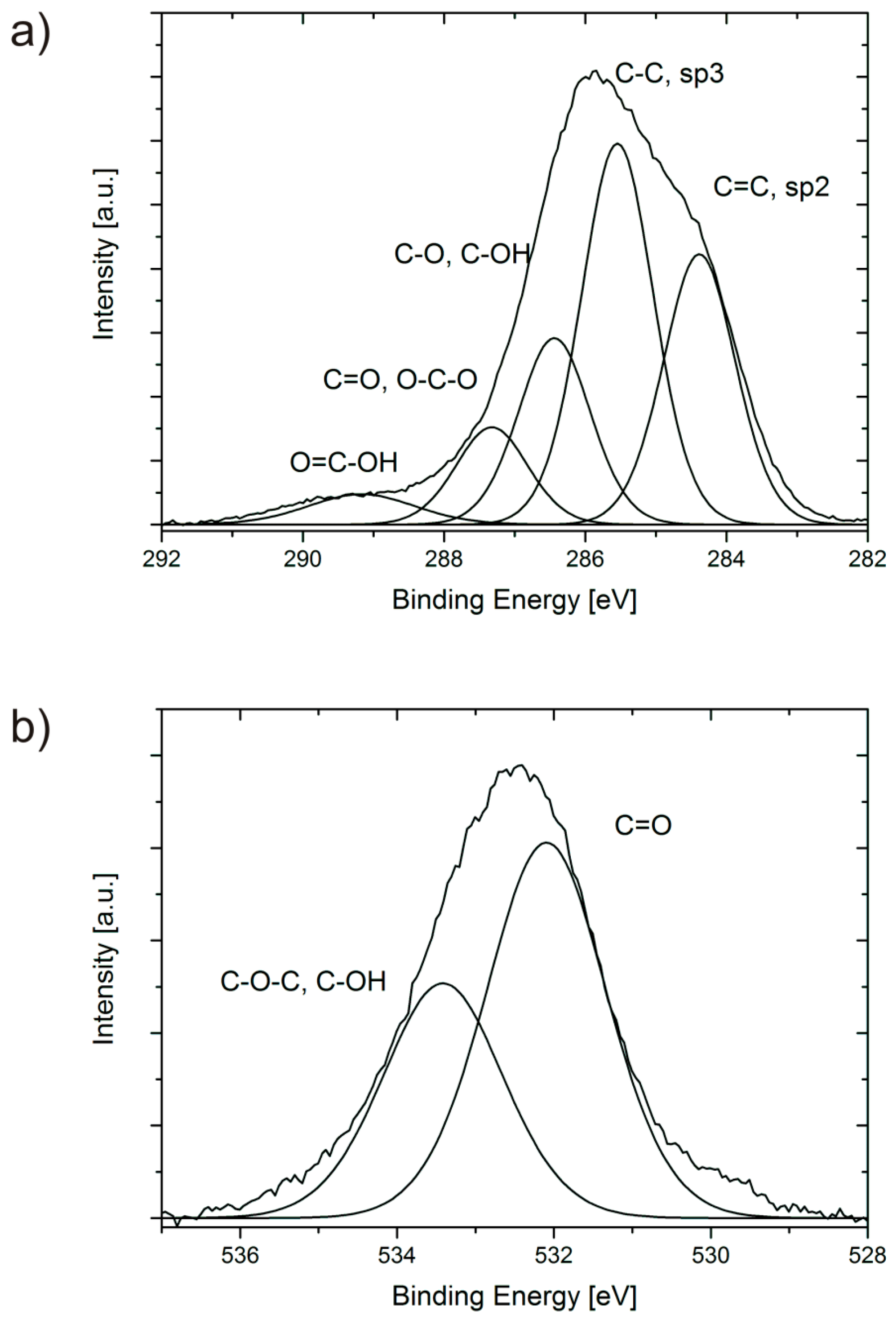
2.2. XPS
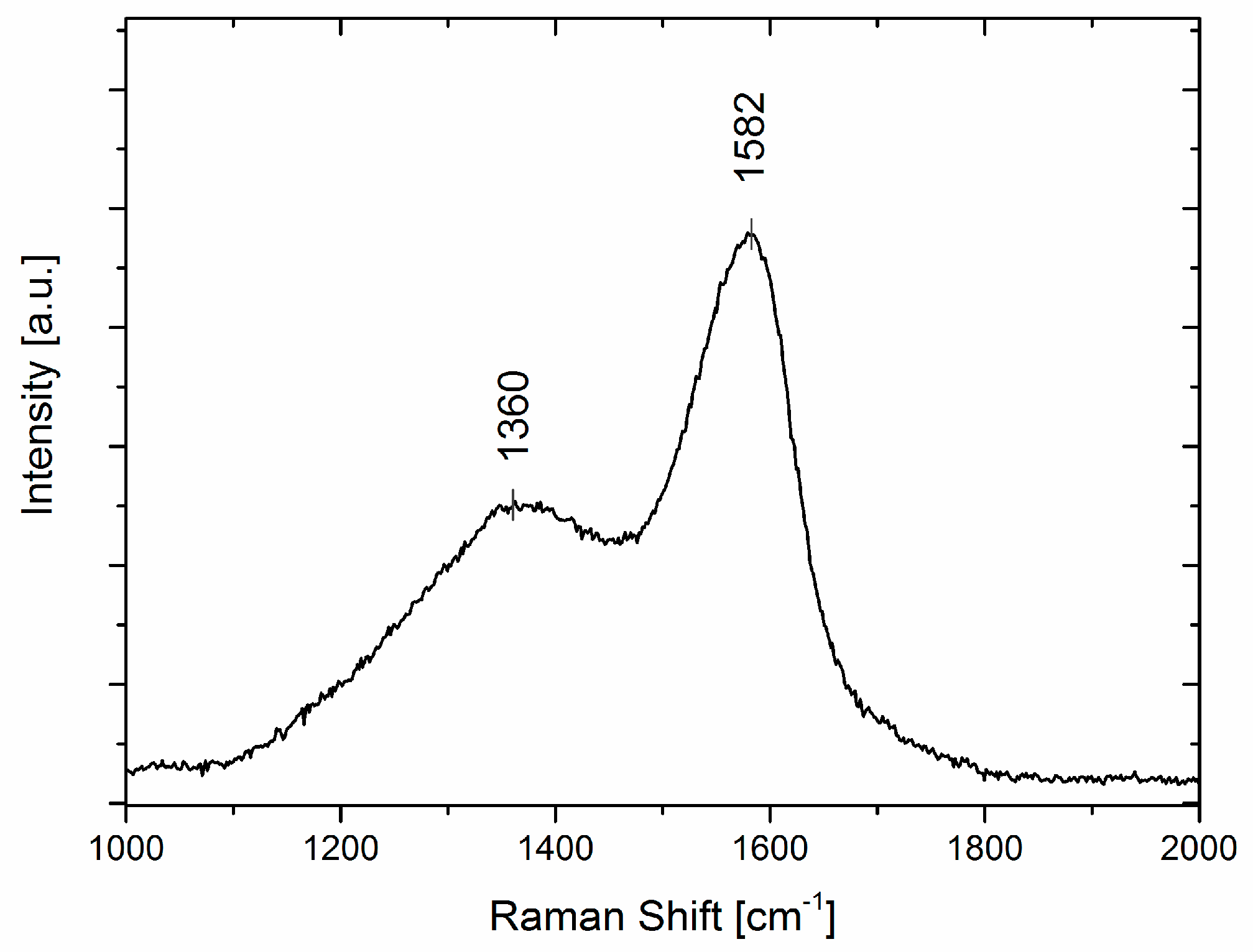
2.3. Raman Spectroscopy
3. Discussion
4. Instruments and Methods
4.1. Hydrothermal Synthesis
4.2. Helium Ion Microscopy
4.3. Raman Spectroscopy
4.4. X-ray Photoelectron Spectroscopy
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Robertson, J. Diamond-like amorphous carbon. Mater. Sci. Eng. R Rep. 2002, 37, 129–281. [Google Scholar] [CrossRef]
- Hu, B.; Wang, K.; Wu, L.H.; Yu, S.H.; Antonietti, M.; Titirici, M.M. Engineering carbon materials from the hydrothermal carbonization process of biomass. Adv Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Hata, T.; Vystavel, T.; Bronsveld, P.; DeHosson, J.; Kikuchi, H.; Nishimiya, K.; Imamura, Y. Catalytic carbonization of wood charcoal: Graphite or diamond? Carbon 2004, 42, 961–964. [Google Scholar] [CrossRef]
- Tekin, K.; Karagoz, S.; Bektas, S. A review of hydrothermal biomass processing. Renew. Sustain. Energy Rev. 2014, 40, 673–687. [Google Scholar] [CrossRef]
- Titirici, M.M.; White, R.J.; Falco, C.; Sevilla, M. Black perspectives for a green future: Hydrothermal carbons for environment protection and energy storage. Energy Environ. Sci. 2012, 5, 6796–6822. [Google Scholar] [CrossRef]
- Kruse, A.; Funke, A.; Titirici, M.M. Hydrothermal conversion of biomass to fuels and energetic materials. Curr. Opin. Chem. Biol. 2013, 17, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Titirici, M.M.; Antonietti, M.; Baccile, N. Hydrothermal carbon from biomass: A comparison of the local structure from poly- to monosaccharides and pentoses/hexoses. Green Chem. 2008, 10, 1204–1212. [Google Scholar] [CrossRef]
- Mitchell, S.T.; Frese, N.; Golzhauser, A.; Bowers, A.; Sattler, K. Ultralight carbon nanofoam from naphtalene-mediated hydrothermal sucrose carbonization. Carbon 2015, 95, 434–441. [Google Scholar] [CrossRef]
- Titirici, M.M.; Antonietti, M. Chemistry and materials options of sustainable carbon materials made by hydrothermal carbonization. Chem. Soc. Rev. 2010, 39, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, D.; Raidongia, K.; Shao, J.J.; Huang, J.X. Graphene oxide assisted hydrothermal carbonization of carbon hydrates. ACS Nano 2014, 8, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Titirici, M.M.; Thomas, A.; Yu, S.H.; Muller, J.O.; Antonietti, M. A direct synthesis of mesoporous carbons with bicontinuous pore morphology from crude plant material by hydrothermal carbonization. Chem. Mater. 2007, 19, 4205–4212. [Google Scholar] [CrossRef]
- Blinc, R.; Arcon, D.; Umek, P.; Apih, T.; Milia, F.; Rode, A.V. Carbon nanofoam as a potential hydrogen storage material. Phys. Status Solidi B Basic Solid State Phys. 2007, 244, 4308–4310. [Google Scholar] [CrossRef]
- Chervin, C.N.; Long, J.W.; Brandell, N.L.; Wallace, J.M.; Kucko, N.W.; Rolison, D.R. Redesigning air cathodes for metal–air batteries using mnox-functionalized carbon nanofoam architectures. J. Power Sources 2012, 207, 191–198. [Google Scholar] [CrossRef]
- Chervin, C.N.; Wattendorf, M.J.; Long, J.W.; Kucko, N.W.; Rolison, D.R. Carbon nanofoam-based cathodes for Li–O2 batteries: Correlation of pore solid architecture and electrochemical performance. J. Electrochem. Soc. 2013, 160, A1510–A1516. [Google Scholar] [CrossRef]
- Long, J.W.; Chervin, C.N.; Brandell, N.L.; Wallace, J.M.; Rolison, D.R. Functionalized carbon nanofoam electrode architectures as cathodes for Zn–air and Li–air batteries. Abstr. Pap. Am. Chem. Soc. 2011, 241. [Google Scholar]
- Spanakis, E.; Pervolaraki, M.; Giapintzakis, J.; Katsarakis, N.; Koudoumas, E.; Vernardou, D. Effect of gold and silver nanoislands on the electrochemical properties of carbon nanofoam. Electrochim. Acta 2013, 111, 305–313. [Google Scholar] [CrossRef]
- Boden, S.A.; Asadollahbaik, A.; Rutt, H.N.; Bagnall, D.M. Helium ion microscopy of lepidoptera scales. Scanning 2012, 34, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.T.; Nuzzo, R.G. Determining hybridization differences for amorphous-carbon from the XPS c-1s envelope. Appl. Surf. Sci. 1995, 90, 195–203. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. Chemical and structural properties of carbonaceous products obtained by hydrothermal carbonization of saccharides. Chem. A Eur. J. 2009, 15, 4195–4203. [Google Scholar] [CrossRef] [PubMed]
- Boehm, H.P. Surface oxides on carbon and their analysis: A critical assessment. Carbon 2002, 40, 145–149. [Google Scholar] [CrossRef]
- Darmstadt, H.; Roy, C.; Kaliaguine, S.; Choi, S.J.; Ryoo, R. Surface chemistry of ordered mesoporous carbons. Carbon 2002, 40, 2673–2683. [Google Scholar] [CrossRef]
- Puziy, A.M.; Poddubnaya, O.I.; Socha, R.P.; Gurgul, J.; Wisniewski, M. XPS and NMR studies of phosphoric acid activated carbons. Carbon 2008, 46, 2113–2123. [Google Scholar] [CrossRef]
- Blyth, R.I.R.; Buqa, H.; Netzer, F.P.; Ramsey, M.G.; Besenhard, J.O.; Golob, P.; Winter, M. XPS studies of graphite electrode materials for lithium ion batteries. Appl. Surf. Sci. 2000, 167, 99–106. [Google Scholar] [CrossRef]
- Cuesta, A.; Dhamelincourt, P.; Laureyns, J.; Martinezalonso, A.; Tascon, J.M.D. Raman microprobe studies on carbon materials. Carbon 1994, 32, 1523–1532. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Chu, P.K.; Li, L.H. Characterization of amorphous and nanocrystalline carbon films. Mater. Chem. Phys. 2006, 96, 253–277. [Google Scholar] [CrossRef]
- Wopenka, B.; Xu, Y.C.; Zinner, E.; Amari, S. Murchison presolar carbon grains of different density fractions: A Raman spectroscopic perspective. Geochim. Cosmochim. Acta 2013, 106, 463–489. [Google Scholar] [CrossRef]
- Schwan, J.; Ulrich, S.; Batori, V.; Ehrhardt, H.; Silva, S.R.P. Raman spectroscopy on amorphous carbon films. J. Appl. Phys. 1996, 80, 440–447. [Google Scholar] [CrossRef]
- Guiotoku, M.; Rambo, C.R.; Hotza, D. Charcoal produced from cellulosic raw materials by microwave-assisted hydrothermal carbonization. J. Therm. Anal. Calorim. 2014, 117, 269–275. [Google Scholar] [CrossRef]
- Kumar, S.; Kothari, U.; Kong, L.Z.; Lee, Y.Y.; Gupta, R.B. Hydrothermal pretreatment of switchgrass and corn stover for production of ethanol and carbon microspheres. Biomass Bioenergy 2011, 35, 956–968. [Google Scholar] [CrossRef]
- Ryu, J.; Suh, Y.W.; Suh, D.J.; Ahn, D.J. Hydrothermal preparation of carbon microspheres from mono-saccharides and phenolic compounds. Carbon 2010, 48, 1990–1998. [Google Scholar] [CrossRef]
- Li, T.; Shen, J.F.; Li, N.; Ye, M.X. Facile and novel hydrothermal preparation of functionalised carbon microspheres from glucose by using graphene sheets as a substrate. Mater. Lett. 2012, 89, 202–205. [Google Scholar] [CrossRef]
- Liu, J.; Tian, P.; Ye, J.W.; Zhou, L.; Gong, W.T.; Lin, Y.; Ning, G.L. Hydrothermal synthesis of carbon microspheres from glucose: Tuning sphere size by adding oxalic acid. Chem. Lett. 2009, 38, 948–949. [Google Scholar] [CrossRef]
- Bazargan, A.; Yan, Y.; Hui, C.W.; McKay, G. A review: Synthesis of carbon-based nano and micro materials by high temperature and high pressure. Ind. Eng. Chem. Res. 2013, 52, 12689–12702. [Google Scholar] [CrossRef]
- Serp, P.; Feurer, R.; Kalck, P.; Kihn, Y.; Faria, J.L.; Figueiredo, J.L. A chemical vapour deposition process for the production of carbon nanospheres. Carbon 2001, 39, 621–626. [Google Scholar] [CrossRef]
- Wang, P.; Wei, J.Y.; Huang, B.B.; Qin, X.Y.; Yao, S.S.; Zhang, Q.; Wang, Z.Y.; Xu, G.G.; Jing, X.Y. Synthesis and characterization of carbon spheres prepared by chemical vapour deposition. Mater. Lett. 2007, 61, 4854–4856. [Google Scholar] [CrossRef]
- Sharon, M.; Mukhopadhyay, K.; Yase, K.; Ijima, S.; Ando, Y.; Zhao, X.L. Spongy carbon nanobeads—A new material. Carbon 1998, 36, 507–511. [Google Scholar] [CrossRef]
- Hou, H.Q.; Schaper, A.K.; Weller, F.; Greiner, A. Carbon nanotubes and spheres produced by modified ferrocene pyrolysis. Chem. Mater. 2002, 14, 3990–3994. [Google Scholar] [CrossRef]
- Liu, X.Y.; Huang, B.C.; Coville, N.J. The Fe(CO)5 catalyzed pyrolysis of pentane: Carbon nanotube and carbon nanoball formation. Carbon 2002, 40, 2791–2799. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. The production of carbon materials by hydrothermal carbonization of cellulose. Carbon 2009, 47, 2281–2289. [Google Scholar] [CrossRef]
- Qi, X.H.; Lian, Y.F.; Yan, L.L.; Smith, R.L. One-step preparation of carbonaceous solid acid catalysts by hydrothermal carbonization of glucose for cellulose hydrolysis. Catal. Commun. 2014, 57, 50–54. [Google Scholar] [CrossRef]
- Li, M.; Li, W.; Liu, S.X. Hydrothermal synthesis, characterization, and KOH activation of carbon spheres from glucose. Carbohydr. Res. 2011, 346, 999–1004. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.Z.; Hu, W.B.; Dan, Y.M.; Liu, Y.L. Synthesis of carbon micro-spheres by a glucose hydrothermal method. Mater. Lett. 2008, 62, 1194–1196. [Google Scholar] [CrossRef]
- Yi, Z.H.; Liang, Y.G.; Lei, X.F.; Wang, C.W.; Sun, J.T. Low-temperature synthesis of nanosized disordered carbon spheres as an anode material for lithium ion batteries. Mater. Lett. 2007, 61, 4199–4203. [Google Scholar] [CrossRef]
- Wang, Q.; Li, H.; Chen, L.Q.; Huang, X.J. Monodispersed hard carbon spherules with uniform nanopores. Carbon 2001, 39, 2211–2214. [Google Scholar] [CrossRef]
- Zheng, M.T.; Liu, Y.L.; Xiao, Y.; Zhu, Y.; Guan, Q.; Yuan, D.S.; Zhang, J.X. An easy catalyst-free hydrothermal method to prepare monodisperse carbon microspheres on a large scale. J. Phys. Chem. C 2009, 113, 8455–8459. [Google Scholar] [CrossRef]
- Chang, J.C.; Tzeng, Y.F.; Chen, J.M.; Chiu, H.T.; Lee, C.Y. Carbon nanobeads as an anode material on high rate capability lithium ion batteries. Electrochim. Acta 2009, 54, 7066–7070. [Google Scholar] [CrossRef]
- Kohno, H.; Tatsutani, K.; Ichikawa, S. Carbon nanofoam formed by laser ablation. J. Nanosci. Nanotechnol. 2012, 12, 2844–2848. [Google Scholar] [CrossRef] [PubMed]
- Rode, A.V.; Elliman, R.G.; Gamaly, E.G.; Veinger, A.I.; Christy, A.G.; Hyde, S.T.; Luther-Davies, B. Electronic and magnetic properties of carbon nanofoam produced by high-repetition-rate laser ablation. Appl.Surf. Sci. 2002, 197, 644–649. [Google Scholar] [CrossRef]
- Rode, A.V.; Gamaly, E.G.; Christy, A.G.; Gerald, J.F.; Hyde, S.T.; Elliman, R.G.; Luther-Davies, B.; Veinger, A.I.; Androulakis, J.; Giapintzakis, J. Strong paramagnetism and possible ferromagnetism in pure carbon nanofoam produced by laser ablation. J. Magn. Magn. Mater. 2005, 290, 298–301. [Google Scholar] [CrossRef]
- Carrasco, J.A.; Prima-Garcia, H.; Romero, J.; Hernandez-Saz, J.; Molina, S.I.; Abellan, G.; Coronado, E. CVD synthesis of carbon spheres using NiFe–LDHs as catalytic precursors: Structural, electrochemical and magnetoresistive properties. J. Mater. Chem. C 2016, 4, 440–448. [Google Scholar] [CrossRef]
- Levesque, A.; Binh, V.T.; Semet, V.; Guillot, D.; Fillit, R.Y.; Brookes, M.D.; Nguyen, T.P. Monodisperse carbon nanopearls in a foam-like arrangement: A new carbon nano-compound for cold cathodes. Thin Solid Films 2004, 464, 308–314. [Google Scholar] [CrossRef]
- Ma, A.L.; Wang, X.M.; Li, T.B.; Liu, X.G.; Xu, B.S. Characteristics of carbon microspheres and study on its adsorption isotherms. Mater. Sci. Eng. A 2007, 443, 54–59. [Google Scholar]
- Mondal, K.C.; Strydom, A.M.; Tetana, Z.; Mhlanga, S.D.; Witcomb, M.J.; Havel, J.; Erasmus, R.M.; Coville, N.J. Boron-doped carbon microspheres. Mater. Chem. Phys. 2009, 114, 973–977. [Google Scholar] [CrossRef]
- Pradhan, D.; Sharon, M. Carbon nanotubes, nanofilaments and nanobeads by thermal chemical vapor deposition process. Mater. Sci. Eng. B 2002, 96, 24–28. [Google Scholar] [CrossRef]
- Shi, J.L.; Peng, H.J.; Zhu, L.; Zhu, W.C.; Zhang, Q. Template growth of porous graphene microspheres on layered double oxide catalysts and their applications in lithium-sulfur batteries. Carbon 2015, 92, 96–105. [Google Scholar] [CrossRef]
- Vieira, S.M.C.; Rego, C.A.; Birkett, P.R. Carbon spheres formed by hot filament chemical vapour deposition. Diam. Relat. Mat. 2008, 17, 100–104. [Google Scholar] [CrossRef]
- Wang, Z.L.; Kang, Z.C. Pairing of pentagonal and heptagonal carbon rings in the growth of nanosize carbon spheres synthesized by a mixed-valent oxide-catalytic carbonization process. J. Phys. Chem. 1996, 100, 17725–17731. [Google Scholar] [CrossRef]
- Wright, W.P.; Marsicano, V.D.; Keartland, J.M.; Erasmus, R.M.; Dube, S.M.A.; Coville, N.J. The electrical transport properties of nitrogen doped carbon microspheres. Mater. Chem. Phys. 2014, 147, 908–914. [Google Scholar] [CrossRef]
- Zhong, Z.Y.; Chen, H.Y.; Tang, S.B.; Ding, J.; Lin, J.Y.; Tan, K.L. Catalytic growth of carbon nanoballs with and without cobalt encapsulation. Chem. Phys. Lett. 2000, 330, 41–47. [Google Scholar] [CrossRef]
- Wohlgemuth, S.A.; Vilela, F.; Titirici, M.M.; Antonietti, M. A one-pot hydrothermal synthesis of tunable dual heteroatom-doped carbon microspheres. Green Chem. 2012, 14, 741–749. [Google Scholar] [CrossRef]
- Chen, J.Z.; Chen, Z.H.; Wang, C.H.; Li, X.D. Calcium-assisted hydrothermal carbonization of an alginate for the production of carbon microspheres with unique surface nanopores. Mater. Lett. 2012, 67, 365–368. [Google Scholar] [CrossRef]
- Qi, X.H.; Li, L.Y.; Tan, T.F.; Chen, W.T.; Smith, R.L. Adsorption of 1-butyl-3-methylimidazolium chloride ionic liquid by functional carbon microspheres from hydrothermal carbonization of cellulose. Environ. Sci. Technol. 2013, 47, 2792–2798. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L.; Shi, Z.Q.; Jin, J.; Chong, C.B.; Wang, C.Y. Properties and sodium insertion behavior of phenolic resin-based hard carbon microspheres obtained by a hydrothermal method. J. Electroanal. Chem. 2015, 755, 87–91. [Google Scholar] [CrossRef]
- Wu, Q.; Li, W.; Tan, J.; Wu, Y.J.; Liu, S.X. Hydrothermal carbonization of carboxymethylcellulose: One-pot preparation of conductive carbon microspheres and water-soluble fluorescent carbon nanodots. Chem.Eng. J. 2015, 266, 112–120. [Google Scholar] [CrossRef]
- Zheng, M.T.; Xiao, Y.; Zhang, H.R.; Dong, H.W.; Gong, X.B.; Xu, R.C.; Lei, B.F.; Liu, Y.L.; Liu, X.T. Hydrothermal synthesis and characterization of sulfur-doped carbon microspheres. Chin. J. Inorg. Chem. 2013, 29, 1391–1399. [Google Scholar]
- Zhou, X.; You, S.J.; Wang, X.H.; Gan, Y.; Zhong, Y.J.; Ren, N.Q. Hydrothermal synthesis of magnetic carbon microspheres for effective adsorption of CD(II) in water. J. Chem. Technol. Biotechnol. 2014, 89, 1051–1059. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Wang, Q.; Zhu, Y.Q.; Chen, X.Y. Nanoporous graphitic carbon materials: Systematic incorporation of p-/m-/o-nitroaniline as effective redox additives for largely improving the capacitive performance. Carbon 2016, 100, 564–577. [Google Scholar] [CrossRef]
- Dyjak, S.; Kicinski, W.; Norek, M.; Huczko, A.; Labedz, O.; Budner, S.; Polanski, M. Hierarchical, nanoporous graphenic carbon materials through an instant, self-sustaining magnesiothermic reduction. Carbon 2016, 96, 937–946. [Google Scholar] [CrossRef]
- Jafari, S.M.; Khosravi, M.; Mollazadeh, M. Nanoporous hard carbon microspheres as anode active material of lithium ion battery. Electrochim. Acta 2016, 203, 9–20. [Google Scholar] [CrossRef]
- Li, S.Y.; Wang, Y.; Wei, Y.; Zeng, J.; Shi, W.Y.; Wang, Y.W. Preparation and adsorption performance of palm fiber-based nanoporous carbon materials with high specific surface area. J. Porous Mater. 2016, 23, 1059–1064. [Google Scholar] [CrossRef]
- Shrestha, L.K.; Shrestha, R.G.; Yamauchi, Y.; Hill, J.P.; Nishimura, T.; Miyazawa, K.; Kawai, T.; Okada, S.; Wakabayashi, K.; Ariga, K. Nanoporous carbon tubes from fullerene crystals as the pi-electron carbon source. Angewa. Chem. Int. Ed. 2015, 54, 951–955. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.C.; Lawrence, G.; Balasubramanian, V.V.; Choi, G.; Choy, J.H.; Abdullah, A.M.; Elzatahry, A.; Ariga, K.; Vinu, A. Highly ordered nanoporous carbon films with tunable pore diameters and their excellent sensing properties. Chem. A Eur. J. 2015, 21, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Bandosz, T.J. Nanoporous carbons: Looking beyond their perception as adsorbents, catalyst supports and supercapacitors. Chem. Rec. 2016, 16, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, L.; Kuchta, B.; Firlej, L.; Roth, M.W.; Wexler, C. Methane adsorption in nanoporous carbon: The numerical estimation of optimal storage conditions. Mater. Res. Express 2016, 3, 5. [Google Scholar] [CrossRef]
- Rashidi, A.M.; Kazemi, D.; Izadi, N.; Pourkhalil, M.; Jorsaraei, A.; Ganji, E.; Lotfi, R. Preparation of nanoporous activated carbon and its application as nano adsorbent for CO2 storage. Korean J. Chem. Eng. 2016, 33, 616–622. [Google Scholar] [CrossRef]
- Puziy, A.M.; Poddubnaya, O.I.; Derylo-Marczewska, A.; Marczewski, A.W.; Blachnio, M.; Tsyba, M.M.; Sapsay, V.I.; Klymchuk, D.O. Kinetics of protein adsorption by nanoporous carbons with different pore size. Adsorpt. J. Int. Adsorpt. Soc. 2016, 22, 541–552. [Google Scholar] [CrossRef]
- Ghiloufi, I.; Khezami, L.; El Mir, L. Nanoporous activated carbon for fast uptake of heavy metals from aqueous solution. Desalination Water Treat. 2015, 55, 935–944. [Google Scholar] [CrossRef]
- Li, W.L.; Seredych, M.; Rodriguez-Castellon, E.; Bandosz, T.J. Metal-free nanoporous carbon as a catalyst for electrochemical reduction of CO2 to CO and CH4. Chemsuschem 2016, 9, 606–616. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Travlou, N.A.; Bashkova, S.; Rodriguez-Castellon, E.; Bandosz, T.J. Nanoporous carbons as gas sensors: Exploring the surface sensitivity. Carbon 2014, 80, 183–192. [Google Scholar] [CrossRef]
- Zhong, C.Y.; Yuan, W.Y.; Kang, Y.J.; Xie, J.L.; Hu, F.X.; Li, C.M. Biomass-derived hierarchical nanoporous carbon with rich functional groups for direct-electron-transfer-based glucose sensing (vol 3, pg 144, 2016). Chemelectrochem 2016, 3. [Google Scholar] [CrossRef]
- Bandosz, T.J.; Rodriguez-Castellon, E.; Montenegro, J.M.; Seredych, M. Photoluminescence of nanoporous carbons: Opening a new application route for old materials. Carbon 2014, 77, 651–659. [Google Scholar] [CrossRef]
- Maeland, A.J.; Skjeltorp, A.T. Hydrogen Storage in Carbon Material. U.S. Patent 6,290,753, 18 September 2001. [Google Scholar]
- Geng, Z.; Zhang, C.M.; Wang, D.B.; Zhou, X.Y.; Cai, M. Pore size effects of nanoporous carbons with ultra-high surface area on high-pressure hydrogen storage. J. Energy Chem. 2015, 24, 1–8. [Google Scholar] [CrossRef]
- Leyva-Garcia, S.; Morallon, E.; Cazorla-Amoros, D.; Beguin, F.; Lozano-Castello, D. New insights on electrochemical hydrogen storage in nanoporous carbons by in situ Raman spectroscopy. Carbon 2014, 69, 401–408. [Google Scholar] [CrossRef]
- Ting, V.P.; Ramirez-Cuesta, A.J.; Bimbo, N.; Sharpe, J.E.; Noguera-Diaz, A.; Presser, V.; Rudic, S.; Mays, T.J. Direct evidence for solid-like hydrogen in a nanoporous carbon hydrogen storage material at supercritical temperatures. ACS Nano 2015, 9, 8249–8254. [Google Scholar] [CrossRef] [PubMed]
- Nigmatullin, R.R.; Baleanu, D.; Povarova, D.; Salah, N.; Habib, S.S.; Memic, A. Raman spectra of nanodiamonds: New treatment procedure directed for improved Raman signal marker detection. Math. Probl. Eng. 2013, 11. [Google Scholar] [CrossRef]
- Dychalska, A.; Popielarski, P.; Frankow, W.; Fabisiak, K.; Paprocki, K.; Szybowicz, M. Study of CVD diamond layers with amorphous carbon admixture by Raman scattering spectroscopy. Mater. Sci. Pol. 2015, 33, 799–805. [Google Scholar] [CrossRef]
- Namba, Y.; Heidarpour, E.; Nakayama, M. Size effects appearing in the Raman-spectra of polycrystalline diamonds. J. Appl. Phys. 1992, 72, 1748–1751. [Google Scholar] [CrossRef]
- Liu, P.; Sun, Q.; Zhu, F.; Liu, K.; Jiang, K.; Liu, L.; Li, Q.; Fan, S. Measuring the work function of carbon nanotubes with thermionic method. Nano Lett. 2008, 8, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Mori, Y.; Maegawa, M.; Katagiri, G.; Ishida, H.; Ishitani, A. Raman-scattering from diamond particles. Appl. Phys. Lett. 1993, 62, 3114–3116. [Google Scholar] [CrossRef]
- Osswald, S.; Mochalin, V.N.; Havel, M.; Yushin, G.; Gogotsi, Y. Phonon confinement effects in the Raman spectrum of nanodiamond. Phys. Rev. B 2009, 80, 075419. [Google Scholar] [CrossRef]
- Shin, J.K.; Lee, C.S.; Lee, K.R.; Eun, K.Y. Effect of residual stress on the Raman-spectrum analysis of tetrahedral amorphous carbon films. Appl. Phys. Lett. 2001, 78, 631–633. [Google Scholar] [CrossRef]
- Zhou, J.H.; Sui, Z.J.; Zhu, J.; Li, P.; De, C.; Dai, Y.C.; Yuan, W.K. Characterization of surface oxygen complexes on carbon nanofibers by TPD, XPS and FT-IR. Carbon 2007, 45, 785–796. [Google Scholar] [CrossRef]
- Liu, X.; Su, D.S.; Schlogl, R. Oxidative dehydrogenation of 1-butene to butadiene over carbon nanotube catalysts. Carbon 2008, 46, 547–549. [Google Scholar] [CrossRef]
- Desimoni, E.; Casella, G.I.; Morone, A.; Salvi, A.M. XPS determination of oxygen-containing functional-groups on carbon-fiber surfaces and the cleaning of these surfaces. Surf. Interface Anal. 1990, 15, 627–634. [Google Scholar] [CrossRef]
- Fan, X.M.; Yu, C.; Yang, J.; Ling, Z.; Qiu, J.S. Hydrothermal synthesis and activation of graphene-incorporated nitrogen-rich carbon composite for high-performance supercapacitors. Carbon 2014, 70, 130–141. [Google Scholar] [CrossRef]
- Mori, Y.; Kawarada, H.; Hiraki, A. Properties of metal diamond interfaces and effects of oxygen adsorbed onto diamond surface. Appl. Phys. Lett. 1991, 58, 940–941. [Google Scholar] [CrossRef]
- Ghodbane, S.; Haensel, T.; Coffinier, Y.; Szunerits, S.; Steinmuller-Nethl, D.; Boukherroub, R.; Ahmed, S.I.U.; Schaefer, J.A. Hreels investigation of the surfaces of nanocrystalline diamond films oxidized by different processes. Langmuir 2010, 26, 18798–18805. [Google Scholar] [CrossRef] [PubMed]
- Gajewski, W.; Huth, M.; Buth, F.; Nickel, B.; Stutzmann, M.; Garrido, J.A. Photoresponse and morphology of pentacene thin films modified by oxidized and reduced diamond surfaces. Phys. Rev.B 2009, 80, 235311. [Google Scholar] [CrossRef]
- Makau, N.W.; Derry, T.E. Study of oxygen on the three low index diamond surfaces by XPS. Surf. Rev. Lett. 2003, 10, 295–301. [Google Scholar] [CrossRef]
- Speranza, G.; Torrengo, S.; Miotello, A.; Minati, L.; Bernagozzi, I.; Ferrari, M.; Dipalo, M.; Kohn, E. XPS and UPS in situ study of oxygen thermal desorption from nanocrystalline diamond surface oxidized by different process. Diam. Relat. Mat. 2011, 20, 560–563. [Google Scholar] [CrossRef]
- Takabayashi, S.; Okamoto, K.; Motoyama, H.; Nakatani, T.; Sakaue, H.; Takahagi, T. X-ray photoelectron analysis of surface functional groups on diamond-like carbon films by gas-phase chemical derivatization method. Surf. Interface Anal. 2010, 42, 77–87. [Google Scholar] [CrossRef]
- Shandilya, M.; Rai, R.; Singh, J. Review: Hydrothermal technology for smart materials. Adv. Appl. Ceram. 2016, 115, 354–376. [Google Scholar] [CrossRef]
- Funke, A.; Ziegler, F. Hydrothermal carbonization of biomass: A summary and discussion of chemical mechanisms for process engineering. Biofuels Bioprod. Biorefin.Biofpr 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Gron, L.U.; LaCroix, J.E.; Higgins, C.J.; Steelman, K.L.; Tinsley, A.S. Heck reactions in hydrothermal, sub-critical water: Water density as an important reaction variable. Tetrahedron Lett. 2001, 42, 8555–8557. [Google Scholar] [CrossRef]
- Kurniawan, A.; Effendi, C.; Ong, L.K.; Kurniawan, F.; Lin, C.X.; Angkawijaya, A.E.; Ju, Y.H.; Ismadji, S.; Zhao, X.S. Preparation of nanoporous carbon microspheres by subcritical water carbonization and electrocapacitive study. Electrochim. Acta 2013, 111, 99–107. [Google Scholar] [CrossRef]
- Economou, N.P.; Notte, J.A.; Thompson, W.B. The history and development of the helium ion microscope. Scanning 2012, 34, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Scipioni, L.; Stern, L.A.; Notte, J.; Sijbrandij, S.; Griffin, B. Helium ion microscope. Adv. Mater. Process. 2008, 166, 27–30. [Google Scholar]
- Scipioni, L.; Sanford, C.A.; Notte, J.; Thompson, B.; McVey, S. Understanding imaging modes in the helium ion microscope. J. Vac. Sci. Technol. B 2009, 27, 3250–3255. [Google Scholar] [CrossRef]
- Petrov, Y.V.; Vyvenko, O.F.; Bondarenko, A.S. Scanning helium ion microscope: Distribution of secondary electrons and ion channeling. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2010, 4, 792–795. [Google Scholar] [CrossRef]
- Sanford, C.; Notte, J.; Scipioni, L.; McVey, S.; Hill, R.; Sijbrandij, S.; Farkas, L. Advances in high resolution helium ion microscope (HIM) imaging. Microsc. Microanal. 2009, 15, 654–655. [Google Scholar] [CrossRef]
- Bell, D.C. Contrast mechanisms and image formation in helium ion microscopy. Microsc. Microanal. 2009, 15, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Tanugi, D.; Yao, N. Superior imaging resolution in scanning helium-ion microscopy: A look at beam-sample interactions. J. Appl. Phys. 2008, 104. [Google Scholar] [CrossRef]
- Fox, D.; Zhou, Y.B.; O'Neill, A.; Kumar, S.; Wang, J.J.; Coleman, J.N.; Duesberg, G.S.; Donegan, J.F.; Zhang, H.Z. Helium ion microscopy of graphene: Beam damage, image quality and edge contrast. Nanotechnology 2013, 24, 33. [Google Scholar] [CrossRef] [PubMed]
- Ohya, K.; Yamanaka, T.; Inai, K.; Ishitani, T. Comparison of secondary electron emission in helium ion microscope with gallium ion and electron microscopes. Nucl. Instrum. Methods Phys. Res. Sect B Beam Interact. Mater. At. 2009, 267, 584–589. [Google Scholar] [CrossRef]
- Ward, B.W.; Notte, J.A.; Economou, N.P. Helium ion microscope: A new tool for nanoscale microscopy and metrology. J. Vac. Sci. Technol. B 2006, 24, 2871–2874. [Google Scholar] [CrossRef]
- Sijbrandij, S.; Notte, J.; Sanford, C.; Hill, R. Analysis of subsurface beam spread and its impact on the image resolution of the helium ion microscope. J. Vac. Sci. Technol. B 2010, 28, C6F6–C6F9. [Google Scholar] [CrossRef]
- Chipara, D.M.; Chipara, A.C.; Chipara, M. Raman spectroscopy of carbonaceous materials: A concise review. Spectroscopy 2011, 26, 42–47. [Google Scholar]
- Dresselhaus, M.S.; Jorio, A.; Hofmann, M.; Dresselhaus, G.; Saito, R. Perspectives on carbon nanotubes and graphene Raman spectroscopy. Nano Lett. 2010, 10, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Retzko, I.; Unger, W.E.S. Analysis of carbon materials by X-ray photoelectron spectroscopy and X-ray absorption spectroscopy. Adv. Eng. Mater. 2003, 5, 519–522. [Google Scholar] [CrossRef]
- Lennon, D.; Lundie, D.T.; Jackson, S.D.; Kelly, G.J.; Parker, S.F. Characterization of activated carbon using X-ray photoelectron spectroscopy and inelastic neutron scattering spectroscopy. Langmuir 2002, 18, 4667–4673. [Google Scholar] [CrossRef]
- Ray, S.C.; Tetana, Z.N.; Erasmus, R.; Mathur, A.; Coville, N.J. Carbon spheres for energy applications: Raman and X-ray photoemission spectroscopy studies. Int.l J. Energy Res. 2014, 38, 444–451. [Google Scholar] [CrossRef]
- Petit, T.; Arnault, J.C.; Girard, H.A.; Sennour, M.; Bergonzo, P. Early stages of surface graphitization on nanodiamond probed by X-ray photoelectron spectroscopy. Phys. Rev. B 2011, 84, 233407. [Google Scholar] [CrossRef]
- Takabayashi, S.; Okamoto, K.; Shimada, K.; Motomitsu, K.; Motoyama, H.; Nakatani, T.; Sakaue, H.; Suzuki, H.; Takahagi, T. Chemical structural analysis of diamondlike carbon films with different electrical resistivities by X-ray photoelectron spectroscopy. Jpn. J. Appl. Phys. 2008, 47, 3376–3379. [Google Scholar] [CrossRef]
- Abbas, M.; Wu, Z.Y.; Zhong, J.; Ibrahim, K.; Fiori, A.; Orlanducci, S.; Sessa, V.; Terranova, M.L.; Davoli, I. X-ray absorption and photoelectron spectroscopy studies on graphite and single-walled carbon nanotubes: Oxygen effect. Appl. Phys. Lett. 2005, 87. [Google Scholar] [CrossRef]
- Gordeev, S.K.; Grechinskaya, A.V.; Danishevskii, A.M.; Smorgonskaya, E.A.; Shchukarev, A.V. Study of bonding in nanoporous carbon by X-ray photoelectron spectroscopy. Mol. Mater. 2000, 13, 355–360. [Google Scholar]
- Yang, D.; Velamakanni, A.; Bozoklu, G.; Park, S.; Stoller, M.; Piner, R.D.; Stankovich, S.; Jung, I.; Field, D.A.; Ventrice, C.A.; et al. Chemical analysis of graphene oxide films after heat and chemical treatments by X-ray photoelectron and micro-Raman spectroscopy. Carbon 2009, 47, 145–152. [Google Scholar] [CrossRef]
- Takahiro, K.; Terai, A.; Oizumi, S.; Kawatsura, K.; Yamamoto, S.; Naramoto, H. Amorphization of carbon materials studied by X-ray photoelectron spectroscopy. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2006, 242, 445–447. [Google Scholar] [CrossRef]
- Achour, A.; Vizireanu, S.; Dinescu, G.; Le Brizoual, L.; Djouadi, M.A.; Boujtita, M. Electrochemical anodic oxidation of nitrogen doped carbon nanowall films: X-ray photoelectron and micro-Raman spectroscopy study. Appl. Surf. Sci. 2013, 273, 49–57. [Google Scholar] [CrossRef]
- Barty, A.; Marchesini, S.; Chapman, H.N.; Cui, C.; Howells, M.R.; Shapiro, D.A.; Minor, A.M.; Spence, J.C.H.; Weierstall, U.; Ilavsky, J.; et al. Three-dimensional coherent X-ray diffraction imaging of a ceramic nanofoam: Determination of structural deformation mechanisms. Phys. Rev. Lett. 2008, 101, 055501. [Google Scholar] [CrossRef] [PubMed]
- Jablonski, A.; Tilinin, I.S.; Powell, C.J. Mean escape depth of signal photoelectrons from amorphous and polycrystalline solids. Phys. Rev. B 1996, 54, 10927–10937. [Google Scholar] [CrossRef]
- Jablonski, A. Elastic photoelectron scattering effects in the XPS analysis of stratified samples. J. Phys. D Appl. Phys. 2012, 45, 31. [Google Scholar] [CrossRef]
- Nefedov, V.I. Photoelectron elastic scattering effects in XPS. J. Electron Spectrosc. Relat. Phenom. 1999, 100, 1–15. [Google Scholar] [CrossRef]
- Jablonski, A.; Powell, C.J. Relationships between electron inelastic mean free paths, effective attenuation lengths, and mean escape depths (vol 100, pg 137, 1999). J. Electron Spectrosc. Relat. Phenom. 2000, 107, 201. [Google Scholar] [CrossRef]
- Tilinin, I.S.; Jablonski, A.; Lesiakorlowska, B. Mean escape depth of signal photoelectrons from amorphous and polycrystalline solids. Acta Phys. Pol. A 1994, 86, 845–852. [Google Scholar] [CrossRef]
- Hovis, J.S.; Hamers, R.J.; Greenlief, C.M. Preparation of clean and atomically flat germanium(001) surfaces. Surf. Sci. 1999, 440, L815–L819. [Google Scholar] [CrossRef]
- Wang, B.; Ma, Y.F.; Wu, Y.P.; Li, N.; Huang, Y.; Chen, Y.S. Direct and large scale electric arc discharge synthesis of boron and nitrogen doped single-walled carbon nanotubes and their electronic properties. Carbon 2009, 47, 2112–2115. [Google Scholar] [CrossRef]
- Reinke, P.; Francz, G.; Oelhafen, P.; Ullmann, J. Structural changes in diamond and amorphous carbon induced by low-energy ion irradiation. Phys. Rev. B 1996, 54, 7067–7073. [Google Scholar] [CrossRef]
- Onoe, J.; Nakao, A.; Takeuchi, K. XPS study of a photopolymerized c-60 film. Phys.l Rev. B 1997, 55, 10051–10056. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frese, N.; Taylor Mitchell, S.; Bowers, A.; Gölzhäuser, A.; Sattler, K. Diamond-Like Carbon Nanofoam from Low-Temperature Hydrothermal Carbonization of a Sucrose/Naphthalene Precursor Solution. C 2017, 3, 23. https://doi.org/10.3390/c3030023
Frese N, Taylor Mitchell S, Bowers A, Gölzhäuser A, Sattler K. Diamond-Like Carbon Nanofoam from Low-Temperature Hydrothermal Carbonization of a Sucrose/Naphthalene Precursor Solution. C. 2017; 3(3):23. https://doi.org/10.3390/c3030023
Chicago/Turabian StyleFrese, Natalie, Shelby Taylor Mitchell, Amanda Bowers, Armin Gölzhäuser, and Klaus Sattler. 2017. "Diamond-Like Carbon Nanofoam from Low-Temperature Hydrothermal Carbonization of a Sucrose/Naphthalene Precursor Solution" C 3, no. 3: 23. https://doi.org/10.3390/c3030023





