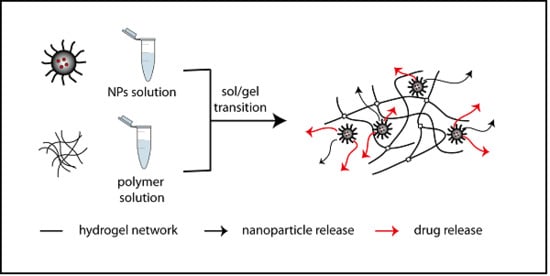
Hydrogel-Nanoparticles Composite System for Controlled Drug Delivery
Abstract
:1. Introduction
2. Results and Discussion
2.1. AC-NPs Composite Systems Synthesis
2.2. Rhodamine Release
2.3. NPs Release from AC6 Hydrogel
2.4. NPs Diffusivity Evaluation
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. PEG-b-PLA Synthesis
4.3. PEG-b-PLA NPs Synthesis and Drug Loading
4.4. AC-NPs Synthesis
4.5. Rhodamine B Release from AC Hydrogels and AC-NPs Composite System
4.6. NPs Release from AC Hydrogels and Mathematical Modeling
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Symbols
| CS | drug concentration in solution | g mL−1 |
| CG | drug concentration within hydrogel | g mL−1 |
| CG,0 | drug concentration within hydrogel at time t = 0 | g mL−1 |
| D | diffusion coefficient | m2 s−1 |
| kC | mass transfer coefficient | m s−1 |
| mG,0 | drug mass present inside matrix | g |
| r | hydrogel radius | cm |
| Sexc | exchange interfacial surface | cm2 |
| Sh | Sherwood number | - |
| t | time | s |
| VG | volume of hydrogel in hydrated state | cm3 |
| VS | volume of solution | cm3 |
References
- Mitragotri, S.; Lahann, J. Materials for drug delivery: Innovative solutions to address complex biological hurdles. Adv. Mater. 2012, 24, 3717–3723. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A. Historical perspective on advanced drug delivery: How engineering design and mathematical modeling helped the field mature. Adv. Drug Deliv. Rev. 2013, 65, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, Z.; Sahraei, R.; Ghaemy, M. Preparation of spherical porous hydrogel beads based on ion-crosslinked gum tragacanth and graphene oxide: Study of drug delivery behavior. Carbohydr. Polym. 2018, 194, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.F.; Correia, I.J.; Silva, A.S.; Mano, J.F. Biomaterials for drug delivery patches. Eur. J. Pharm. Sci. 2018, 118, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Mathaes, R.; Winter, G.; Besheer, A.; Engert, J. Non-spherical micro- and nanoparticles: Fabrication, characterization and drug delivery applications. Expert Opin. Drug Deliv. 2015, 12, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Simovic, S.; Barnes, T.J.; Tan, A.; Prestidge, C.A. Assembling nanoparticle coatings to improve the drug delivery performance of lipid based colloids. Nanoscale 2012, 4, 1220–1230. [Google Scholar] [CrossRef] [PubMed]
- Dimatteo, R.; Darling, N.J.; Segura, T. In situ forming injectable hydrogels for drug delivery and wound repair. Adv. Drug Deliv. Rev. 2018, 127, 167–184. [Google Scholar] [CrossRef] [PubMed]
- He, Z.W.; Wang, B.Y.; Hu, C.M.; Zhao, J.N. An overview of hydrogel-based intra-articular drug delivery for the treatment of osteoarthritis. Colloid Surf. B 2017, 154, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Perale, G.; Rossi, F.; Santoro, M.; Peviani, M.; Papa, S.; Llupi, D.; Torriani, P.; Micotti, E.; Previdi, S.; Cervo, L.; et al. Multiple drug delivery hydrogel system for spinal cord injury repair strategies. J. Control. Release 2012, 159, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; McKeon, R.J.; Bellamkonda, R.V. Sustained delivery of thermostabilized chabc enhances axonal sprouting and functional recovery after spinal cord injury. Proc. Natl. Acad. Sci. USA 2010, 107, 3340–3345. [Google Scholar] [CrossRef] [PubMed]
- Mokarram, N.; Merchant, A.; Mukhatyar, V.; Patel, G.; Bellamkonda, R.V. Effect of modulating macrophage phenotype on peripheral nerve repair. Biomaterials 2012, 33, 8793–8801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in regenerative medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Koehler, J.; Brandl, F.P.; Goepferich, A.M. Hydrogel wound dressings for bioactive treatment of acute and chronic wounds. Eur. Polym. J. 2018, 100, 1–11. [Google Scholar] [CrossRef]
- Spang, M.T.; Christman, K.L. Extracellular matrix hydrogel therapies: In vivo applications and development. Acta Biomater. 2018, 68, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Qi, C.; Liu, J.; Jin, Y.; Xu, L.M.; Wang, G.B.; Wang, Z.; Wang, L. Photo-crosslinkable, injectable sericin hydrogel as 3d biomimetic extracellular matrix for minimally invasive repairing cartilage. Biomaterials 2018, 163, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Ciuffreda, M.C.; Malpasso, G.; Chokoza, C.; Bezuidenhout, D.; Goetsch, K.P.; Mura, M.; Pisano, F.; Davies, N.H.; Gnecchi, M. Synthetic extracellular matrix mimic hydrogel improves efficacy of mesenchymal stromal cell therapy for ischemic cardiomyopathy. Acta Biomater. 2018, 70, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for biomedical applications: Their characteristics and the mechanisms behind them. Gels 2017, 3, 6. [Google Scholar] [CrossRef]
- Gurikov, P.; Smirnova, I. Non-conventional methods for gelation of alginate. Gels 2018, 4, 14. [Google Scholar] [CrossRef]
- Niu, Y.M.; Zhu, J.H.; Li, Y.; Shi, H.H.; Gong, Y.X.; Li, R.; Huo, Q.; Ma, T.; Liu, Y. Size shrinkable drug delivery nanosystems and priming the tumor microenvironment for deep intratumoral penetration of nanoparticles. J. Control. Release 2018, 277, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Barman, S.R.; Nain, A.; Jain, S.; Punjabi, N.; Mukherji, S.; Satija, J. Dendrimer as a multifunctional capping agent for metal nanoparticles for use in bioimaging, drug delivery and sensor applications. J. Mater. Chem. B 2018, 6, 2368–2384. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, J.; Yuan, K.J.; Zhang, Z.G.; Zhang, X.W.; Fang, X.M. A multi-controlled drug delivery system based on magnetic mesoporous fe3o4 nanopaticles and a phase change material for cancer thermo-chemotherapy. Nanotechnology 2017, 28, 405101. [Google Scholar] [CrossRef] [PubMed]
- Papa, S.; Rossi, F.; Ferrari, R.; Mariani, A.; de Paola, M.; Caron, I.; Fiordaliso, F.; Bisighini, C.; Sammali, E.; Colombo, C.; et al. Selective nanovector mediated treatment of activated proinflammatory microglia/macrophages in spinal cord injury. ACS Nano 2013, 7, 9881–9895. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Borm, P.J.A. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef]
- Xin, Y.; Yin, M.; Zhao, L.; Meng, F.; Luo, L. Recent progress on nanoparticle-based drug delivery systems for cancer therapy. Cancer Biol. Med. 2017, 14, 228–241. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wu, D.Q.; Mutschler, M.A.; Chu, C.C. Cationic hybrid hydrogels from amino-acid-based poly(ester amide): Fabrication, characterization, and biological properties. Adv. Funct. Mater. 2012, 22, 3815–3823. [Google Scholar] [CrossRef]
- Doggui, S.; Sahni, J.K.; Arseneault, M.; Dao, L.; Ramassamy, C. Neuronal uptake and neuroprotective effect of curcumin-loaded PLGA nanoparticles on the human SK-n-SH cell line. J. Alzheimers Dis. 2012, 30, 377–392. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.M.; Simon, J.K.; Baker, J.R. Applications of nanotechnology for immunology. Nat. Rev. Immunol. 2013, 13, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Perale, G.; Papa, S.; Forloni, G.; Veglianese, P. Current options for drug delivery to the spinal cord. Expert Opin. Drug Deliv. 2013, 10, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, M. Macrophage recognition of crystals and nanoparticles. Front. Immunol. 2018, 9, 103. [Google Scholar]
- Weissleder, R.; Nahrendorf, M.; Pittet, M.J. Imaging macrophages with nanoparticles. Nat. Mater. 2014, 13, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Alexis, F.; Pridgen, E.; Molnar, L.K.; Farokhzad, O.C. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol. Pharm. 2008, 4, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Al Zaki, A.; Hui, J.Z.; Higbee, E.; Tsourkas, A. Biodistribution, clearance, and toxicology of polymeric micelles loaded with 0.9 or 5 nm gold nanoparticles. J. Biomed. Nanotechnol. 2015, 11, 1836–1846. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Ferrari, R.; Papa, S.; Moscatelli, D.; Casalini, T.; Forloni, G.; Perale, G.; Veglianese, P. Tunable hydrogel—Nanoparticles release system for sustained combination therapies in the spinal cord. Colloid Surf. B 2013, 108, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Ferrari, R.; Castiglione, F.; Mele, A.; Perale, G.; Moscatelli, D. Polymer hydrogel functionalized with biodegradable nanoparticles as composite system for controlled drug delivery. Nanotechnology 2015, 26, 015602. [Google Scholar] [CrossRef] [PubMed]
- Papa, S.; Vismara, I.; Mariani, A.; Barilani, M.; Rimondo, S.; de Paola, M.; Panini, N.; Erba, E.; Mauri, E.; Rossi, F.; et al. Mesenchymal stem cells encapsulated into biomimetic hydrogel scaffold gradually release ccl2 chemokine in situ preserving cytoarchitecture and promoting functional recovery in spinal cord injury. J. Control. Release 2018, 278, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Lohleijer, B.G.G.; Pratt, R.C.; Leibfarth, F.; Logan, J.W.; Long, D.A.; Dove, A.P.; Nederberg, F.; Choi, J.; Wade, C.; Waymouth, R.M.; et al. Guanidine and amidine organocatalysts for ring-opening polymerization of cyclic esters. Macromolecules 2006, 39, 8574–8583. [Google Scholar] [CrossRef]
- Rossi, F.; Perale, G.; Masi, M. Biological buffered saline solution as solvent in agar–carbomer hydrogel synthesis. Chem. Pap. 2010, 64, 573–578. [Google Scholar] [CrossRef]
- Vulic, K.; Shoichet, M.S. Tunable growth factor delivery from injectable hydrogels for tissue engineering. J. Am. Chem. Soc. 2012, 134, 882–885. [Google Scholar] [CrossRef] [PubMed]
- Ossipov, D.; Kootala, S.; Yi, Z.; Yang, X.; Hilborn, J. Orthogonal chemoselective assembly of hyaluronic acid networks and nanogels for drug delivery. Macromolecules 2013, 46, 4105–4113. [Google Scholar] [CrossRef]
- Anguilar-Lopez, Y.A.; Villafuerte-Robles, L. Functional performance of chitosan/carbopol 974p nf matrices in captopril tablets. J. Pharm. 2016, 2016, 3240290. [Google Scholar]
- Rossi, F.; Perale, G.; Storti, G.; Masi, M. A library of tunable agarose carbomer-based hydrogels for tissue engineering applications: The role of cross-linkers. J. Appl. Polym. Sci. 2012, 123, 2211–2221. [Google Scholar] [CrossRef]
- Appel, E.A.; Tibbitt, M.W.; Webber, M.J.; Mattix, B.A.; Veiseh, O.; Langer, R. Self-assembled hydrogels utilizing polymer–nanoparticle interactions. Nat. Commun. 2015, 6, 6295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Appel, E.A.; Tibbitt, M.W.; Greer, J.M.; Fenton, O.S.; Kreuels, K.; Anderson, D.G.; Langer, R. Exploiting electrostatic interactions in polymer−nanoparticle hydrogels. ACS Macro Lett. 2015, 4, 848–852. [Google Scholar] [CrossRef]
- Gupta, D.; Tator, C.H.; Shoichet, M.S. Fast-gelling injectable blend of hyaluronan and methylcellulose for intrathecal, localized delivery to the injured spinal cord. Biomaterials 2006, 27, 2370–2379. [Google Scholar] [CrossRef] [PubMed]
- Kwon, B.K.; Sekhon, L.H.; Fehlings, M.G. Emerging repair, regeneration, and translational research advances for spinal cord injury. Spine 2010, 35, S263–S270. [Google Scholar] [CrossRef] [PubMed]
- Schoener, C.A.; Hutson, H.N.; Peppas, N.A. Ph-responsive hydrogels with dispersed hydrophobic nanoparticles for the delivery of hydrophobic therapeutic agents. Polym. Int. 2012, 61, 874–879. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.; Marchetti, P.; Rossi, F.; Perale, G.; Castiglione, F.; Mele, A.; Masi, M. Smart approach to evaluate drug diffusivity in injectable agar-carbomer hydrogels for drug delivery. J. Phys. Chem. B 2011, 115, 2503–2510. [Google Scholar] [CrossRef] [PubMed]
- Stanwick, J.C.; Baumann, M.D.; Shoichet, M.S. Enhanced neurotrophin-3 bioactivity and release from a nanoparticle-loaded composite hydrogel. J. Control. Release 2012, 160, 666–675. [Google Scholar] [CrossRef] [PubMed]





| Diameter (nm) | PDI (–) | ζ−Potential (mV) | |
|---|---|---|---|
| NPs_SDS | 75.4 | 0.12 | −21.5 |
| NPs_CC | 81.2 | 0.15 | 28.5 |
| D 1 (cm2/s) | ||
|---|---|---|
| pH = 7.4 | pH = 5 | |
| AC6-NPs_SDS | 2.99 ± 0.15 | - |
| AC6-NPs_CC | - | - |
| AC1-NPs_SDS | 2.25 ± 0.18 | 1.34 ± 0.12 |
| AC1-NPs_CC | 1.16 ± 0.1 | 1.51 ± 0.16 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mauri, E.; Negri, A.; Rebellato, E.; Masi, M.; Perale, G.; Rossi, F. Hydrogel-Nanoparticles Composite System for Controlled Drug Delivery. Gels 2018, 4, 74. https://doi.org/10.3390/gels4030074
Mauri E, Negri A, Rebellato E, Masi M, Perale G, Rossi F. Hydrogel-Nanoparticles Composite System for Controlled Drug Delivery. Gels. 2018; 4(3):74. https://doi.org/10.3390/gels4030074
Chicago/Turabian StyleMauri, Emanuele, Anna Negri, Erica Rebellato, Maurizio Masi, Giuseppe Perale, and Filippo Rossi. 2018. "Hydrogel-Nanoparticles Composite System for Controlled Drug Delivery" Gels 4, no. 3: 74. https://doi.org/10.3390/gels4030074