A Controlled Antibiotic Release System for the Development of Single-Application Otitis Externa Therapeutics
Abstract
:1. Introduction
2. Results
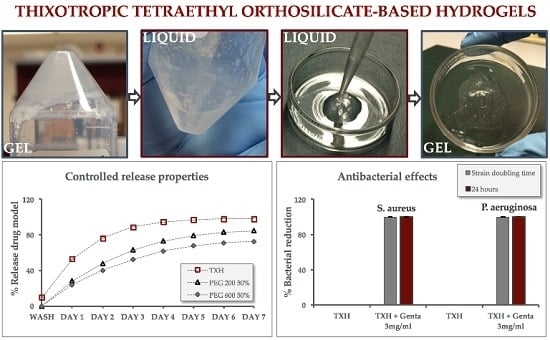
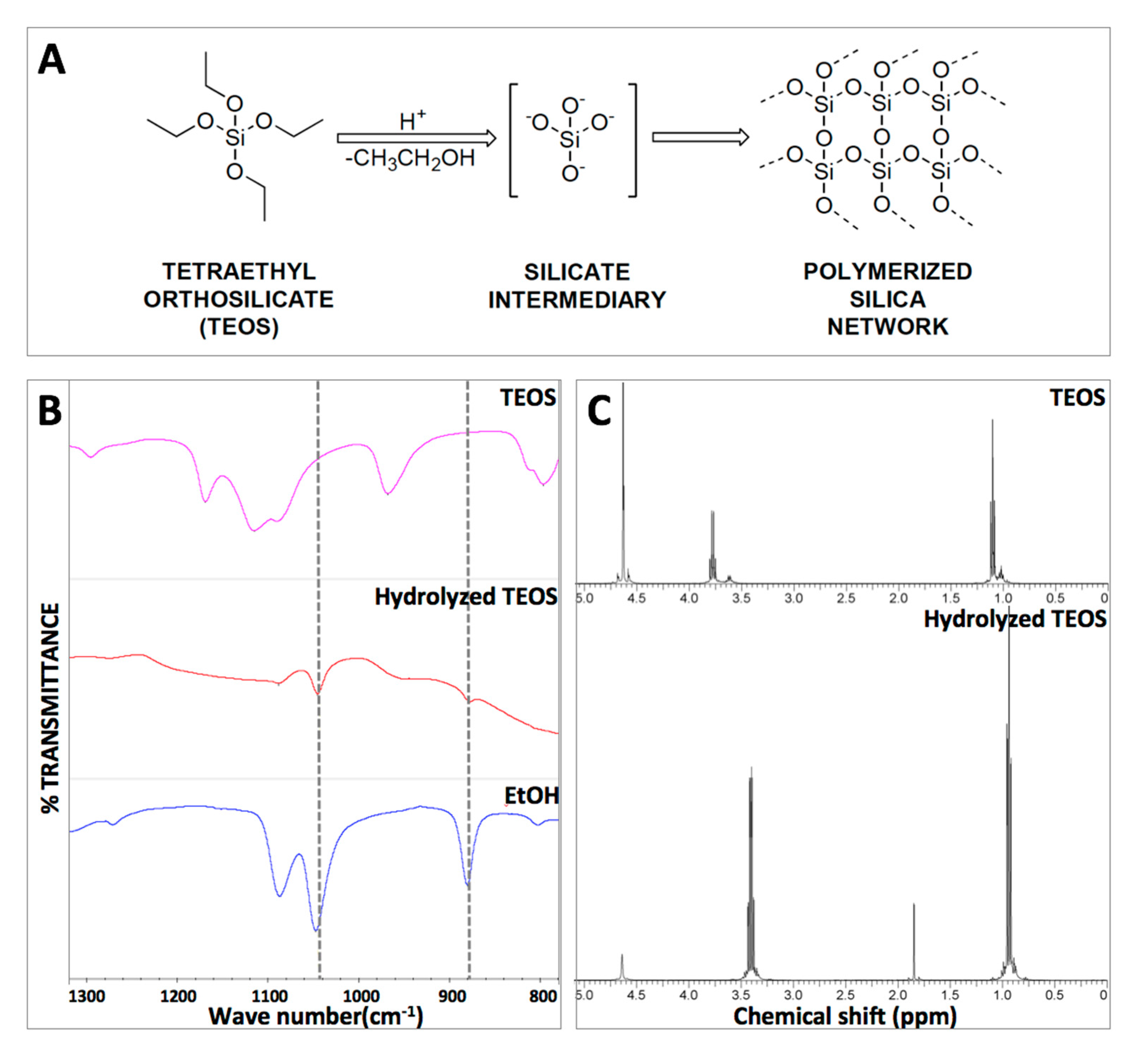
2.1. Hydrogel Formation
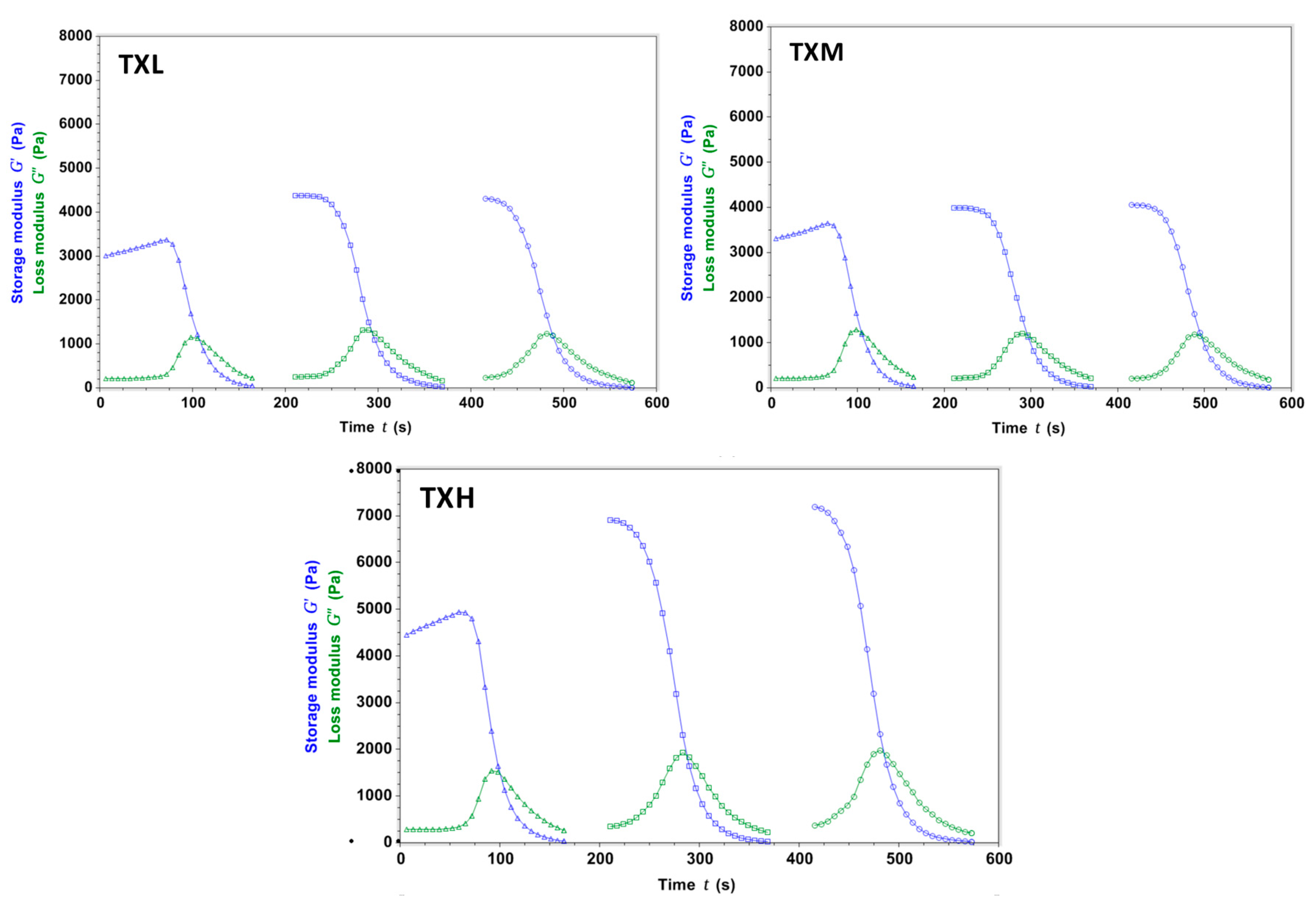
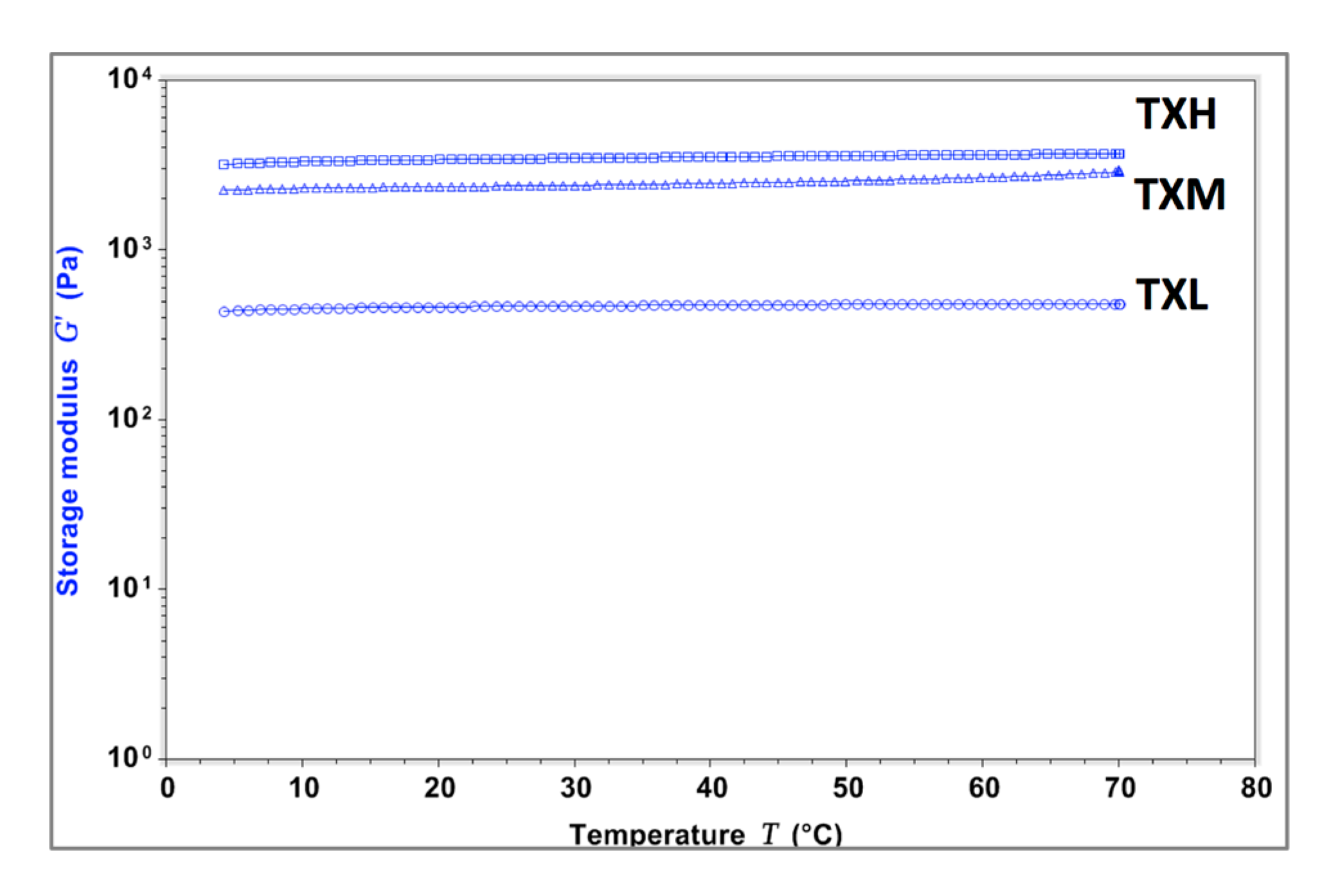
2.2. Hydrogel Rheology
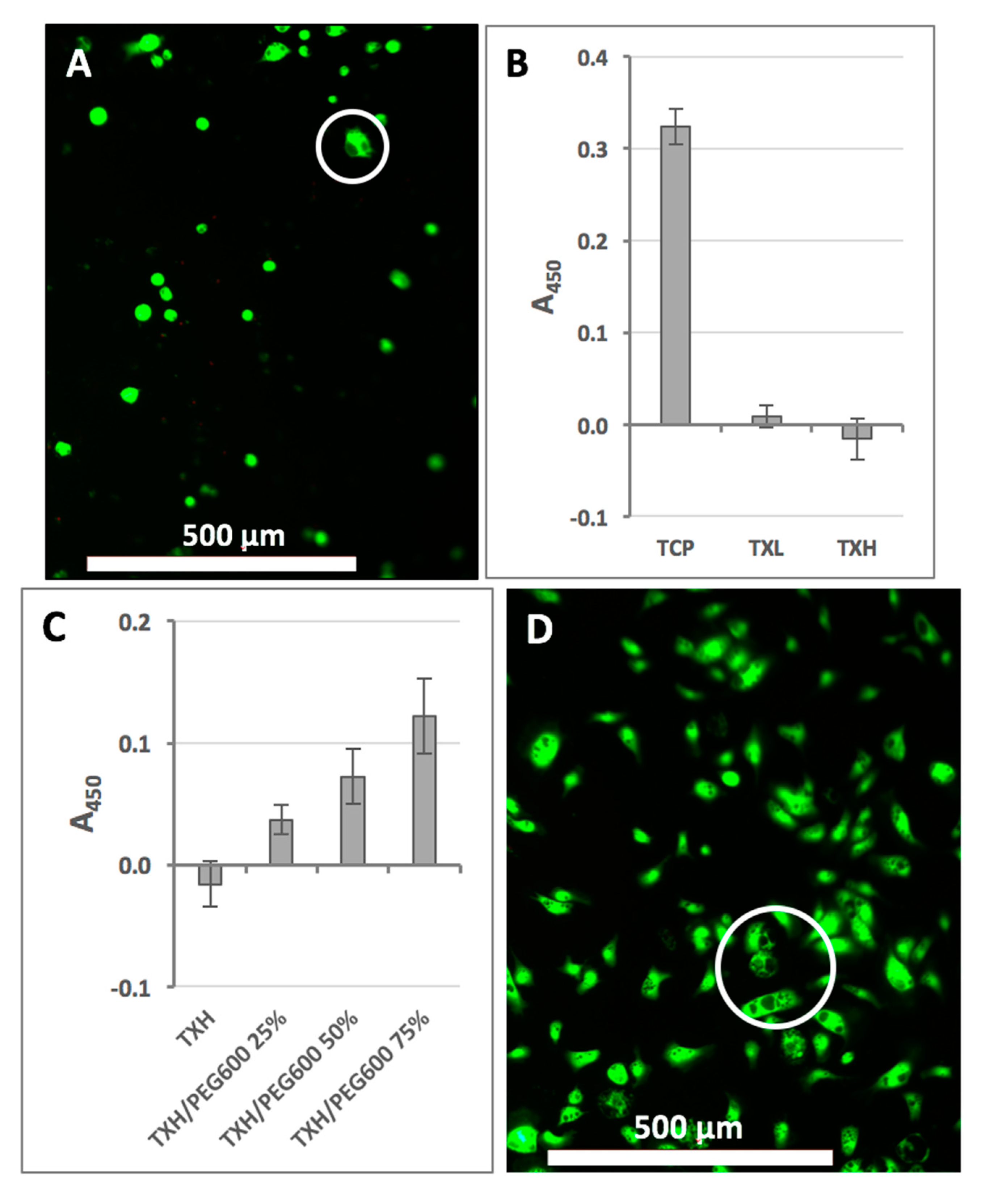
2.3. Cellular Effects of Hydrogels
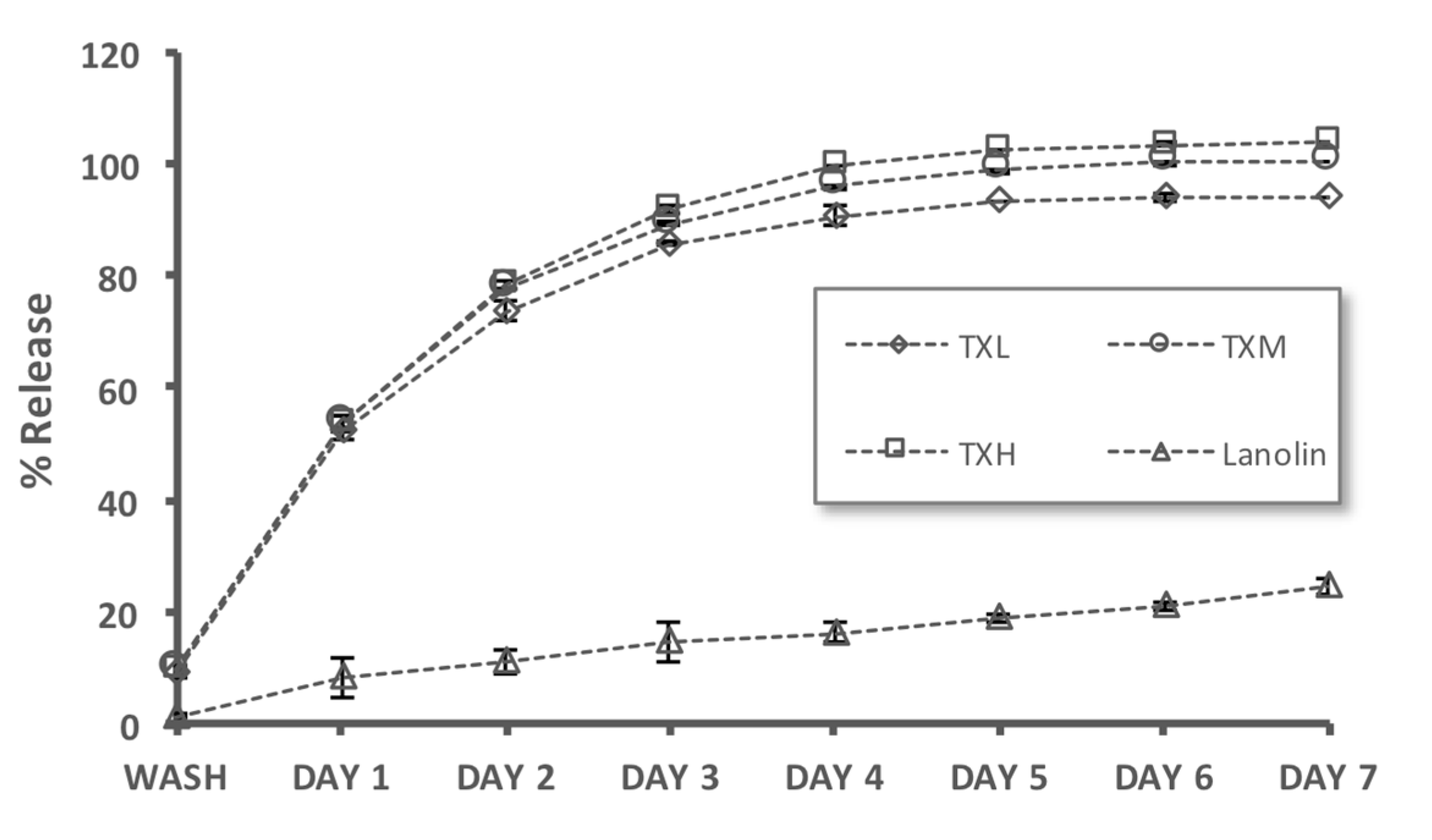
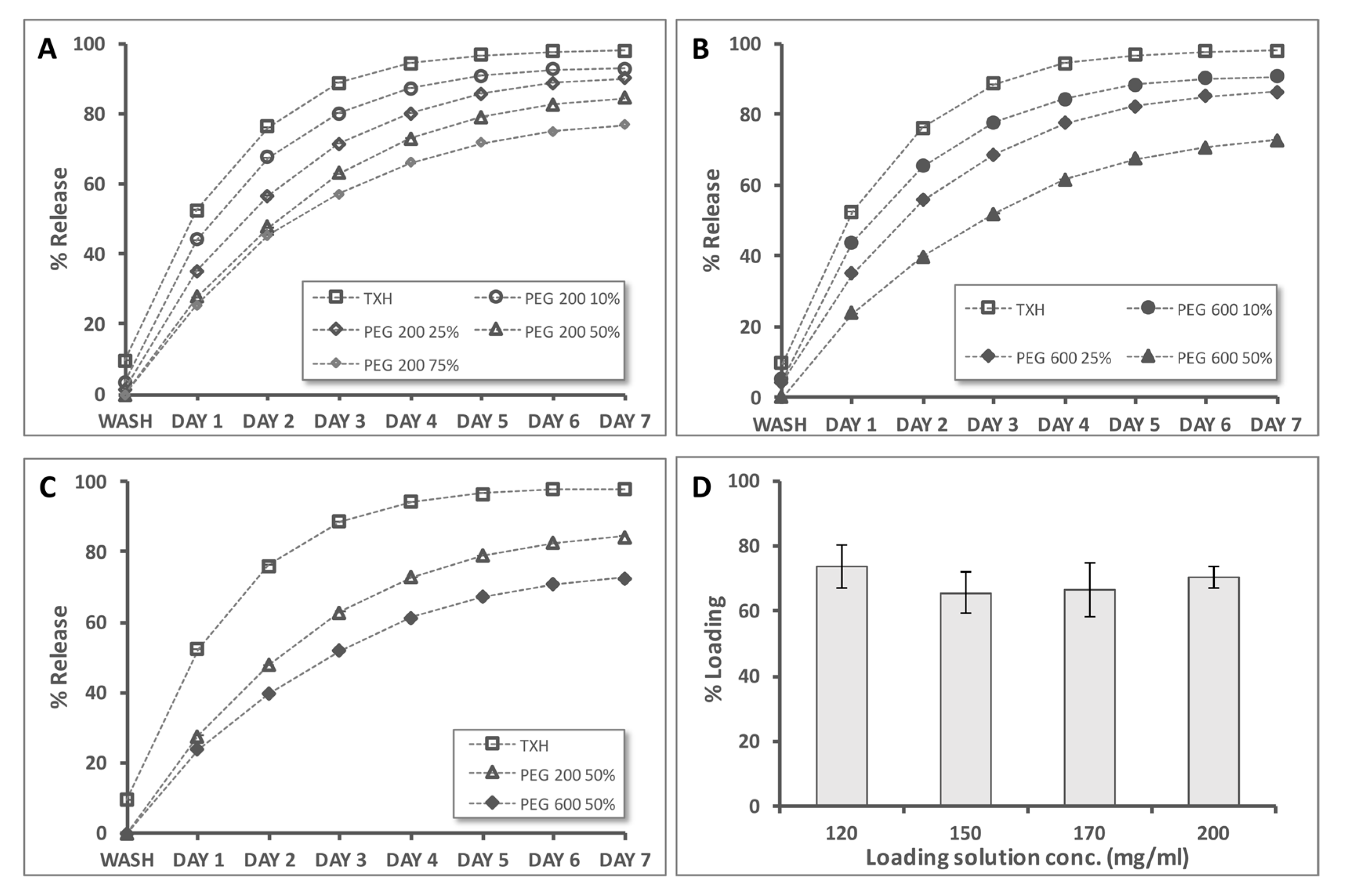
2.4. Controlled Release Evaluation
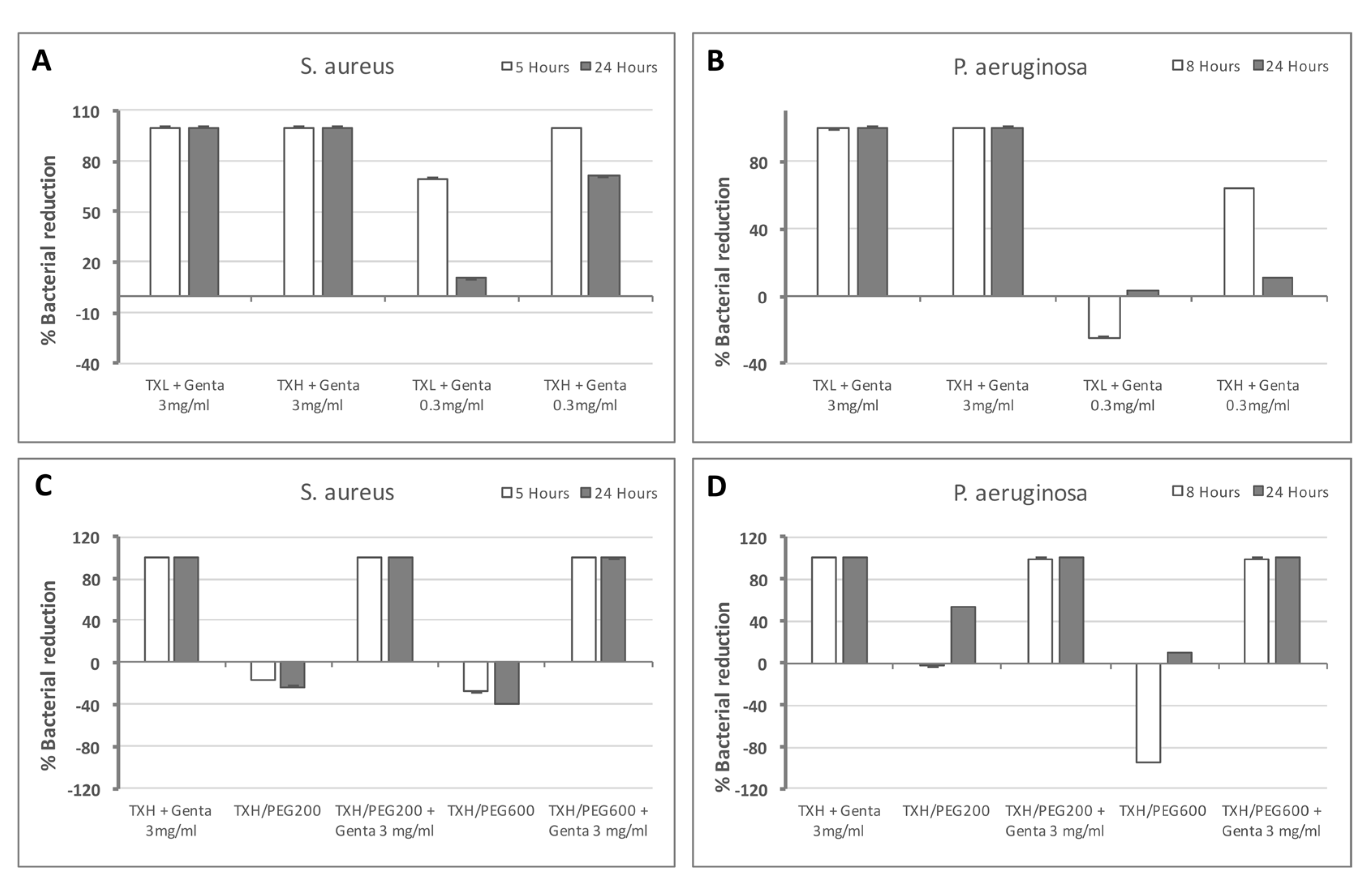
2.5. Antibacterial Activity
2.6. Hydrogel Dehydration Rates
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Materials
5.2. Analytical Instrumentation
5.3. Gel Formation
5.4. Controlled Release
5.5. Rheological Characterization
5.6. Cell Assays
5.7. Bacterial Assays
5.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Institutes of Health; U.S. Department of Health and Human Services. NICDC Fact Sheet: Ear Infections in Children; National Institutes of Health, U.S. Department of Health and Human Services: Bethesda, MD, USA, 2013.
- Centers for Disease Control and Prevention. Ear infection; Centers for Disease Control and Prevention: Bethesda, MD, USA, 2017.
- National Institutes of Health; Biological Sciences Curriculum Study. Information about Hearing, Communication, and Understanding; National Institutes of Health, Biological Sciences Curriculum Study: Bethesda, MD, USA, 2007.
- Kozin, E.D.; Sethi, R.K.; Remenschneider, A.K.; Kaplan, A.B.; Del Portal, D.A.; Gray, S.T.; Shrime, M.G.; Lee, D.J. Epidemiology of otologic diagnoses in united states emergency departments. Laryngoscope 2015, 125, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Schilder, A.G.; Chonmaitree, T.; Cripps, A.W.; Rosenfeld, R.M.; Casselbrant, M.L.; Haggard, M.P.; Venekamp, R.P. Otitis media. Nat. Rev. Dis. Prim. 2016, 2, 16063. [Google Scholar] [CrossRef] [PubMed]
- Rovers, M.M.; Schilder, A.G.; Zielhuis, G.A.; Rosenfeld, R.M. Otitis media. Lancet 2004, 363, 465–473. [Google Scholar] [CrossRef]
- National Institutes of Health; National Institute on Deafness and Other Communication Dissorders. Quick Statistics About Hearing; National Institutes of Health, National Institute on Deafness and Other Communication Dissorders: Bethesda, MD, USA, 2016.
- Lee, H.; Kim, J.; Nguyen, V. Ear infections: Otitis externa and otitis media. Prim. Care 2013, 40, 671–686. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, Y.; Wang, Q.; Li, Z.; Zhou, Y.; Zhang, Y.; Li, S. Protein-bearing cubosomes prepared by liquid precursor dilution: Inner ear delivery and pharmacokinetic study following intratympanic administration. J. Biomed. Nanotechnol. 2013, 9, 1784–1793. [Google Scholar] [CrossRef] [PubMed]
- Ward, J.A.; Sidell, D.R.; Nassar, M.; Reece, A.L.; Choo, D.I. Safety of cidofovir by intratympanic delivery technique. Antivir. Ther 2014, 19, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Wipperman, J. Otitis externa. Prim. Care 2014, 41, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Estimated Burden of Acute Otitis Externa—United States, 2003–2007; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2011.
- Hui, C.P. Canadian Paediatric Society; Infectious Diseases Immunization Committee. Acute otitis externa. Paediatr. Child Health 2013, 18, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, R.M.; Brown, L.; Cannon, C.R.; Dolor, R.J.; Ganiats, T.G.; Hannley, M.; Kokemueller, P.; Marcy, S.M.; Roland, P.S.; Shiffman, R.N.; et al. Clinical practice guideline: Acute otitis externa. Otolaryngol. Head Neck Surg. 2006, 134, S4–S23. [Google Scholar] [CrossRef] [PubMed]
- Sander, R. Otitis externa: A practical guide to treatment and prevention. Am. Fam. Physician 2001, 63, 927–937. [Google Scholar]
- England, R.J.; Homer, J.J.; Jasser, P.; Wilde, A.D. Accuracy of patient self-medication with topical eardrops. J. Laryngol. Otol. 2000, 114, 24–25. [Google Scholar] [CrossRef] [PubMed]
- Agius, A.M.; Reid, A.P.; Hamilton, C. Patient compliance with short-term topical aural antibiotic therapy. Clin. Otolaryngol. Allied Sci. 1994, 19, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Serban, M.A.; Liu, Y.; Prestwich, G.D. Effects of extracellular matrix analogues on primary human fibroblast behavior. Acta Biomater. 2008, 4, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.V.; Tan, A.S. Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT): Subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in mtt reduction. Arch. Biochem. Biophys. 1993, 303, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Serban, M.A. Translational biomaterials-the journey from the bench to the market-think ‘product’. Curr. Opin. Biotechnol. 2016, 40, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Stegemann, S.; Ternik, R.L.; Onder, G.; Khan, M.A.; van Riet-Nales, D.A. Defining patient centric pharmaceutical drug product design. AAPS J. 2016, 18, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Alwi, R.; Telenkov, S.; Mandelis, A.; Leshuk, T.; Gu, F.; Oladepo, S.; Michaelian, K. Silica-coated super paramagnetic iron oxide nanoparticles (spion) as biocompatible contrast agent in biomedical photoacoustics. Biomed. Opt. Express 2012, 3, 2500–2509. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.C.; Lee, J.H.; Lee, J.; Kim, H.Y.; Park, J.Y.; Cho, J.; Lee, J.; Han, D.W. Subtle cytotoxicity and genotoxicity differences in superparamagnetic iron oxide nanoparticles coated with various functional groups. Int. J. Nanomed. 2011, 6, 3219–3231. [Google Scholar]
- Jokinen, M.; Jalonen, H.; Forsback, A.P. Silica hydrogel composite. U.S. Patent 2016/0136088 A1, 19 May 2016. [Google Scholar]
- Ab Rahman, I.; Padavettan, V. Synthesis of silica nanoparticles by sol-gel: Size-dependent properties, surface modification, and applications in silica-polymer nanocomposites—A review. J. Nanomater. 2012, 2012, 132424. [Google Scholar] [CrossRef]
- Iler, R.K. The Chemistry of Silica: Solubility, Polymerization, Colloid and Surface Properties, and Biochemistry; Wiley: New York, NY, USA, 1979; Volume XXIV, p. 866. [Google Scholar]


| Hydrogel | Designation | Formulation |
|---|---|---|
| H2O:hTEOS (v/v) | ||
| Thixogel—low density SiO2 network | TXL | 1:1 |
| Thixogel—medium density SiO2 network | TXM | 1:1.5 |
| Thixogel—high density SiO2 network | TXH | 1:2 |
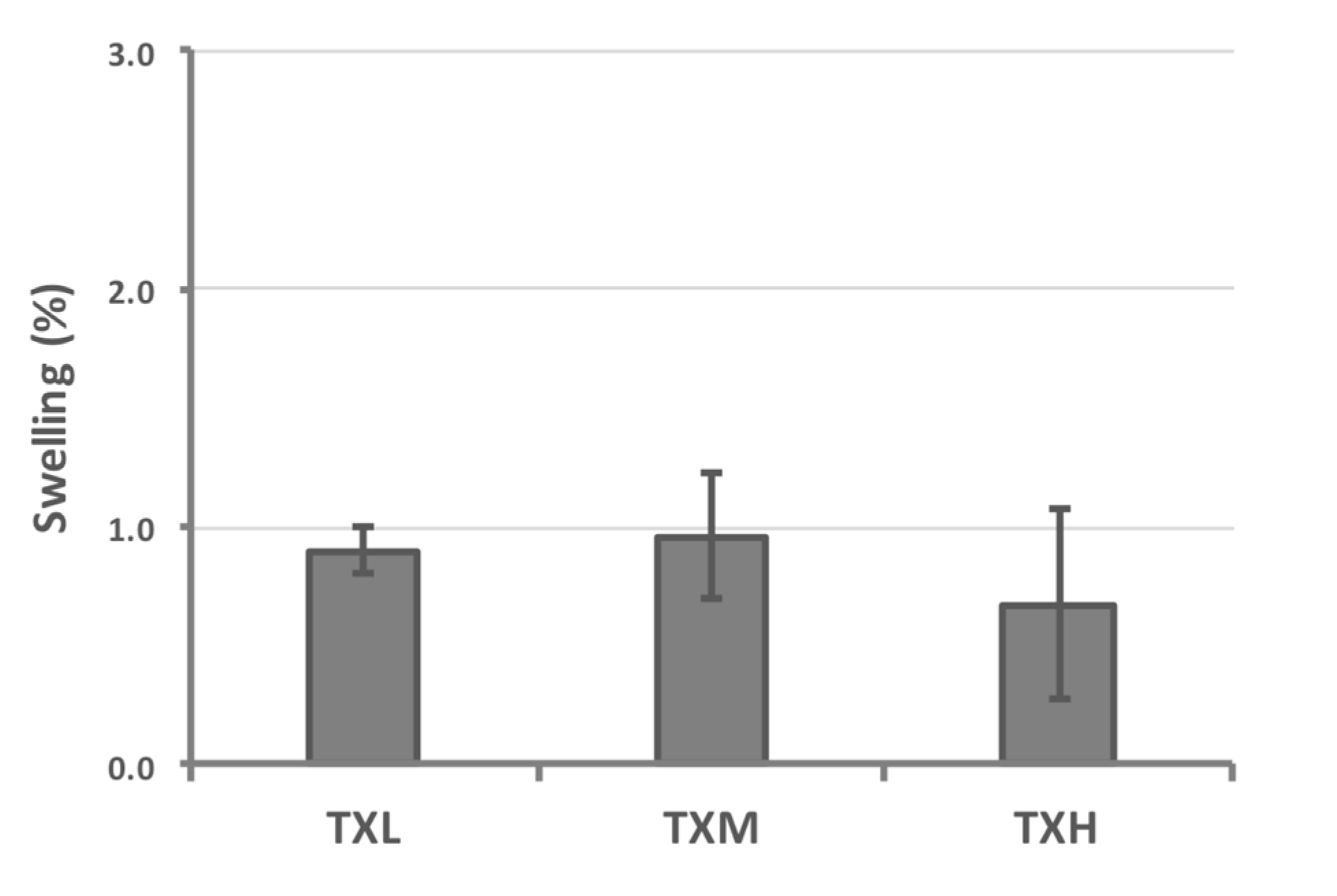
| Hydrogel | Volume Increase Due to Swelling (%) | Standard Deviation (%) |
|---|---|---|
| TXL | 0.90 | 0.10 |
| TXM | 0.96 | 0.26 |
| TXH | 0.67 | 0.40 |
| Hydrogel | Dry Weight/100 μg Hydrogel (in μg) | Standard Deviation (μg) |
|---|---|---|
| TXL | 1.198 | 0.030 |
| TXM | 1.196 | 0.005 |
| TXH | 1.238 | 0.021 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serban, B.A.; Stipe, K.T.; Alverson, J.B.; Johnston, E.R.; Priestley, N.D.; Serban, M.A. A Controlled Antibiotic Release System for the Development of Single-Application Otitis Externa Therapeutics. Gels 2017, 3, 19. https://doi.org/10.3390/gels3020019
Serban BA, Stipe KT, Alverson JB, Johnston ER, Priestley ND, Serban MA. A Controlled Antibiotic Release System for the Development of Single-Application Otitis Externa Therapeutics. Gels. 2017; 3(2):19. https://doi.org/10.3390/gels3020019
Chicago/Turabian StyleSerban, Bogdan A., Kristian T. Stipe, Jeremy B. Alverson, Erik R. Johnston, Nigel D. Priestley, and Monica A. Serban. 2017. "A Controlled Antibiotic Release System for the Development of Single-Application Otitis Externa Therapeutics" Gels 3, no. 2: 19. https://doi.org/10.3390/gels3020019