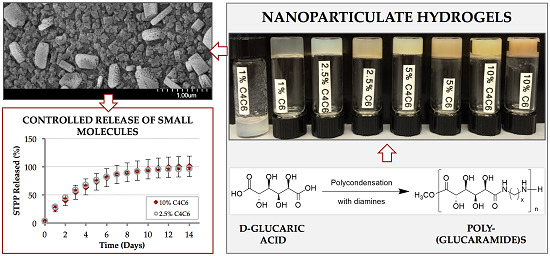
Nanoparticulate Poly(glucaramide)-Based Hydrogels for Controlled Release Applications
Abstract
:1. Introduction
2. Results and Discussion
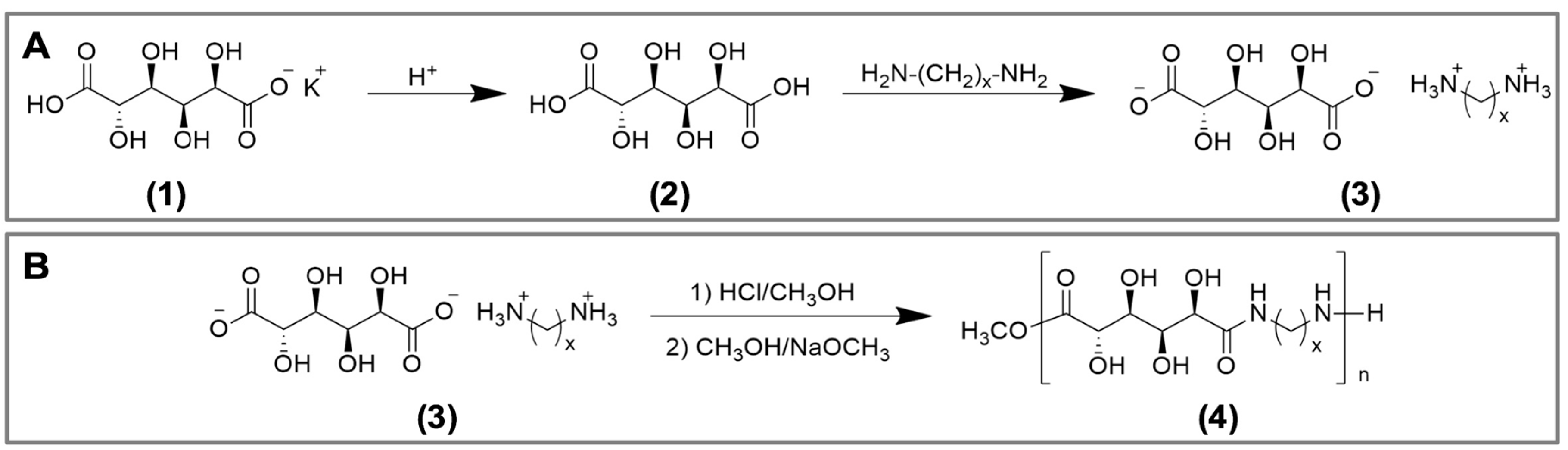
2.1. Synthesis of Poly(glucaramide)s
2.2. Preparation of Hydrogels
2.3. Characterization of Hydrogels
2.3.1. SEM Characterization
2.3.2. DLS Analyses
2.3.3. Rheological Characterization
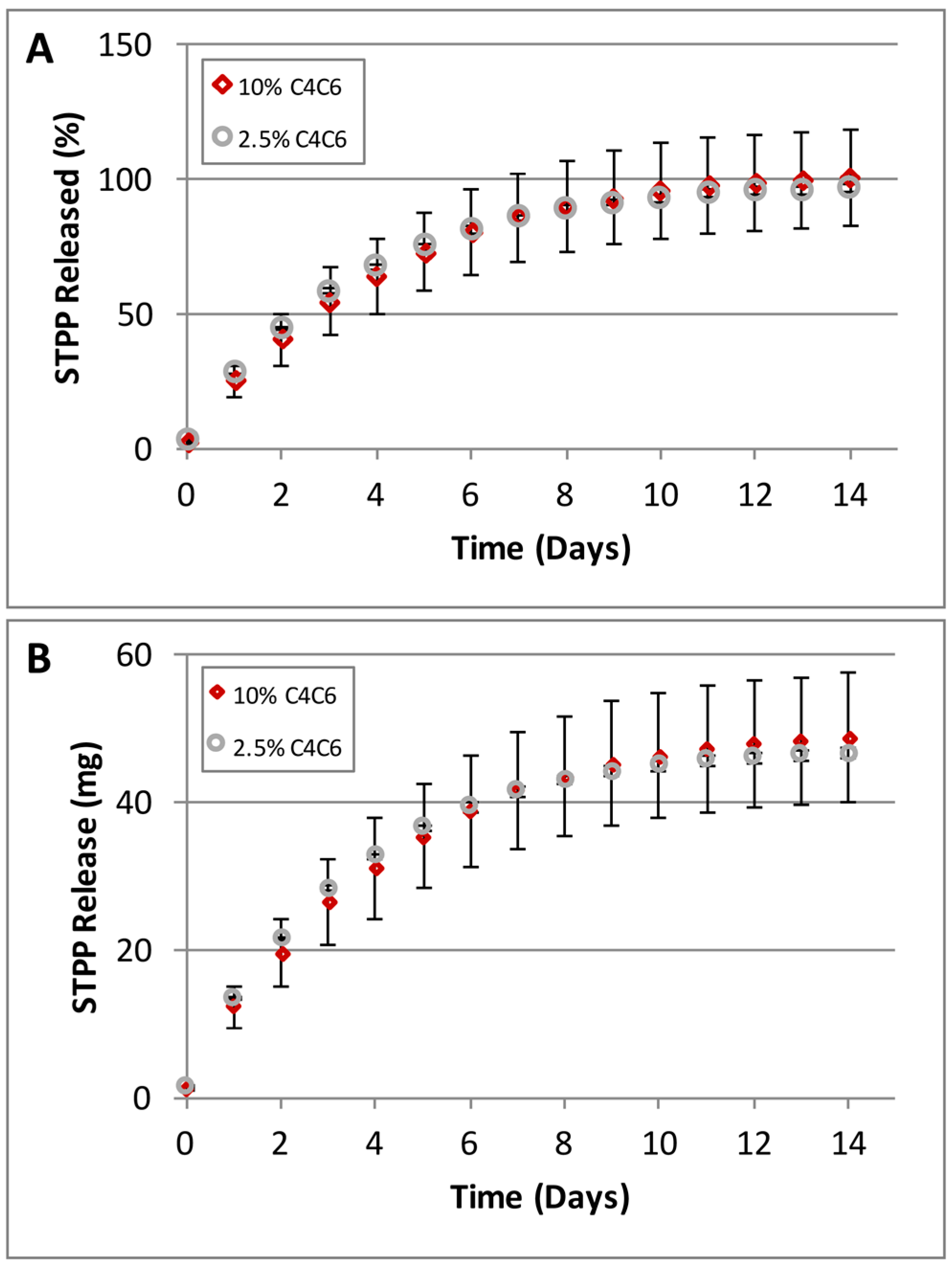
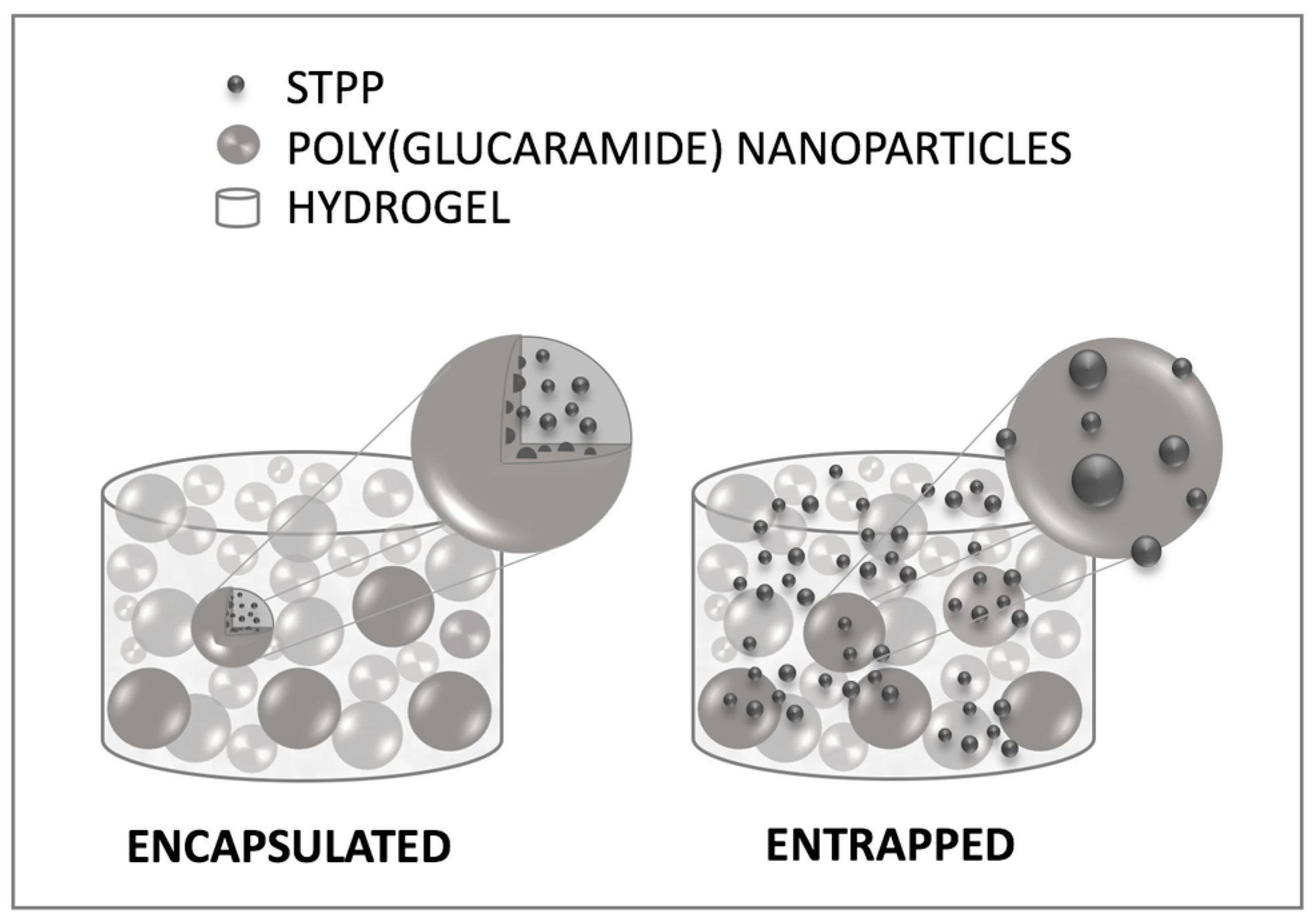
2.4. Controlled Release of STPP
3. Conclusions
4. Materials and Methods
4.1. Reagents and Materials
4.2. Synthesis of Poly(glucaramide)s
4.3. Preparation of Poly(glucaramide) Hydrogels
4.4. Hydrogel Characterization
4.4.1. Scanning Electron Microscopy (SEM)
4.4.2. Dynamic Light Scattering (DLS)
4.4.3. Rheological Characterization
4.5. Sodium Tripolyphosphate (STPP) Release Studies
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1. [Google Scholar] [CrossRef]
- Ullah, F.; Othman, M.B.; Javed, F.; Ahmad, Z.; Akil, H. Classification, processing and application of hydrogels: A review. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 57, 414–433. [Google Scholar] [CrossRef] [PubMed]
- Vashist, A.; Ahmad, S. Hydrogels in tissue engineering: Scope and applications. Curr. Pharm. Biotechnol. 2015, 16, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Zhou, J.; Shi, J.; Xu, B. Supramolecular hydrogelators and hydrogels: From soft matter to molecular biomaterials. Chem. Rev. 2015, 115, 13165–13307. [Google Scholar] [CrossRef] [PubMed]
- Highley, C.B.; Prestwich, G.D.; Burdick, J.A. Recent advances in hyaluronic acid hydrogels for biomedical applications. Curr. Opin. Biotechnol. 2016, 40, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Kyburz, K.A.; Anseth, K.S. Synthetic mimics of the extracellular matrix: How simple is complex enough? Ann. Biomed. Eng. 2015, 43, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Seliktar, D. Designing cell-compatible hydrogels for biomedical applications. Science 2012, 336, 1124–1128. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Rollett, A.; Kaplan, D.L. Silk-elastin-like protein biomaterials for the controlled delivery of therapeutics. Expert Opin. Drug Deliv. 2015, 12, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Sood, N.; Bhardwaj, A.; Mehta, S.; Mehta, A. Stimuli-responsive hydrogels in drug delivery and tissue engineering. Drug Deliv. 2016, 23, 758–780. [Google Scholar] [CrossRef] [PubMed]
- Alge, D.L.; Azagarsamy, M.A.; Donohue, D.F.; Anseth, K.S. Synthetically tractable click hydrogels for three-dimensional cell culture formed using tetrazine-norbornene chemistry. Biomacromolecules 2013, 14, 949–953. [Google Scholar] [CrossRef] [PubMed]
- Mironi-Harpaz, I.; Wang, D.Y.; Venkatraman, S.; Seliktar, D. Photopolymerization of cell-encapsulating hydrogels: Crosslinking efficiency versus cytotoxicity. Acta Biomater. 2012, 8, 1838–1848. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Park, W.H. Chemically cross-linked silk fibroin hydrogel with enhanced elastic properties, biodegradability, and biocompatibility. Int. J. Nanomed. 2016, 11, 2967–2978. [Google Scholar]
- Moon, T.S.; Dueber, J.E.; Shiue, E.; Prather, K.L.J. Use of modular, synthetic scaffolds for improved production of glucaric acid in engineered E. coli. Metab. Eng. 2010, 12, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.N.; Hash, K.; Davey, C.L.; Mills, H.; Williams, H.; Kiely, D.E. Modifications in the nitric acid oxidation of d-glucose. Carbohydr. Res. 2012, 350, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Thaburet, J.F.; Merbouh, N.; Ibert, M.; Marsais, F.; Queguiner, G. Tempo-mediated oxidation of maltodextrins and d-glucose: Effect of ph on the selectivity and sequestering ability of the resulting polycarboxylates. Carbohydr. Res. 2001, 330, 21–29. [Google Scholar] [CrossRef]
- Kiely, D.E.; Hash, K.R., Sr. Method of oxidation using nitric acid. U.S. Patent 7,692,041 B2, 6 April 2010. [Google Scholar]
- Werpy, T.; Petersen, G. Top Value Added Chemicals from Biomass: Volume I—Results of Screening for Potential Candidates from Sugars and Synthesis Gas; DOE/GO-102004-1992; U.S. Department of Energy: Oak Ridge, TN, USA, 2004.
- Kiely, D.E.; Chen, L.; Lin, T.H. Hydroxylated nylons based on unprotected esterified d-glucaric acid by simple condensation-reactions. J. Am. Chem. Soc. 1994, 116, 571–578. [Google Scholar] [CrossRef]
- Kiely, D.E.; Smith, T.N. Hydroxypolyamide Gel Forming Agents. U.S. Patent 8,623,943 B2, 7 January 2014. [Google Scholar]
- McBeath, T.M.; Lombi, E.; McLaughlin, M.J.; Bunemann, E.K. Polyphosphate-fertilizer solution stability with time, temperature, and pH. J. Plant Nutr. Soil Sci. 2007, 170, 387–391. [Google Scholar] [CrossRef]
- Perez, J.J.; Francois, N.J. Chitosan-starch beads prepared by ionotropic gelation as potential matrices for controlled release of fertilizers. Carbohydr. Polym. 2016, 148, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Tyliszczak, B.; Polaczek, J.; Pielichowski, J.; Pielichowski, K. Preparation and Properties of Biodegradable Slow-Release PAA Superabsorbent Matrixes for Phosphorus Fertilizers. Macromol. Symp. 2009, 279, 236–242. [Google Scholar] [CrossRef]
- Villalba, G.; Liu, Y.; Schroder, H.; Ayres, R.U. Global phosphorus flows in the industrial economy from a production perspective. J. Ind. Ecol. 2008, 12, 557–569. [Google Scholar] [CrossRef]
- Lampila, L.E. Functions and uses of phosphates in the seafood industry. J. Aquat. Food Prod. Technol. 1993, 1, 29–41. [Google Scholar] [CrossRef]
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of us freshwaters: Analysis of potential economic damages. Environ. Sci. Technol. 2009, 43, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Jahns, T.; Kiely, D.E. Abiotic hydrolysis of some poly-d-glucaramides and subsequent microbial utilization/degradation. J. Polym. Environ. 2006, 14, 165–169. [Google Scholar] [CrossRef]


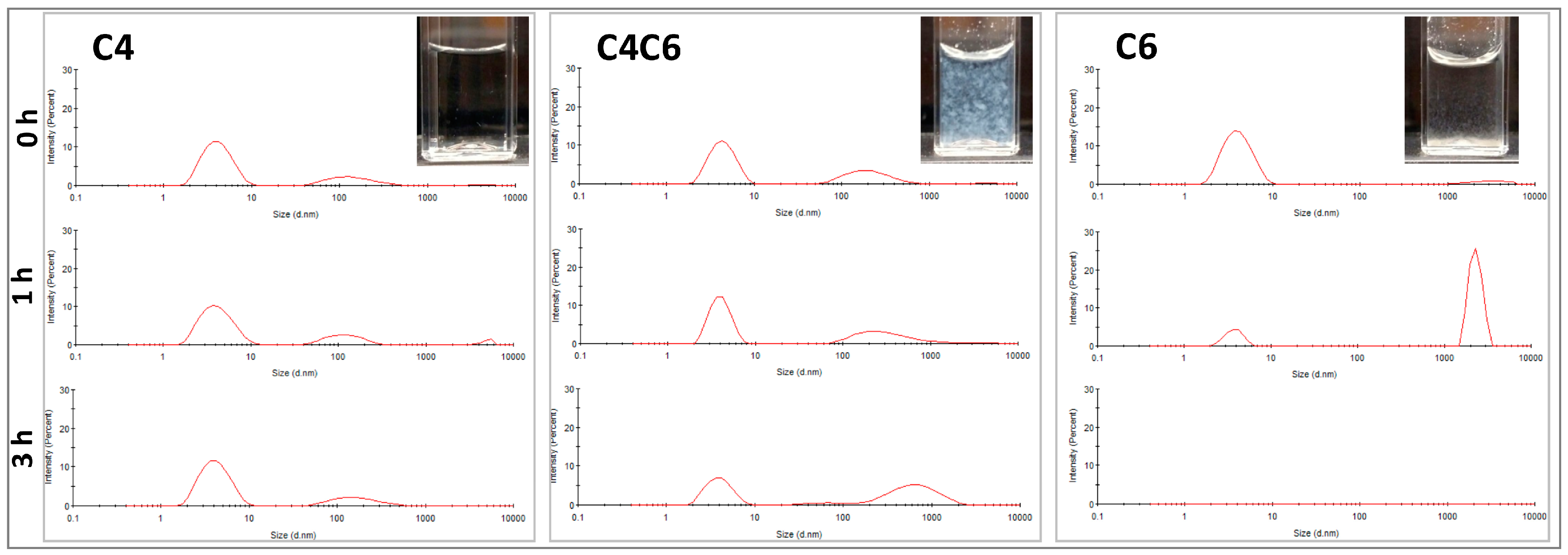




| Poly(glucaramide) | Water Solubility | Gel Formation | Minimum Gelling Concentration (MGC, % w/w) | Solvent |
|---|---|---|---|---|
| Poly(tetramethylene d-glucaramide) (C4) | Yes | No | N/A | N/A |
| Poly(tetramethylene/hexamethylene d-glucaramide) (C4C6) | Partial | Yes | 2.5% | Water |
| Poly(hexamethylene d-glucaramide) (C6) | No | Yes | 1% | Acetic acid (20% v/v) |
| Time (h) | C4 | C4C6 | C6 | |||
|---|---|---|---|---|---|---|
| Particle Size (Diameter, nm) | Intensity * (%) | Particle Size (Diameter, nm) | Intensity * (%) | Particle Size (Diameter, nm) | Intensity * (%) | |
| 0 | 4.3 ± 1.6 148.4 ± 83.8 4065 ± 1060 | 76.6 22.3 1.0 | 4.5 ± 1.4 218.1 ± 118.1 4468 ± 897.9 | 65.8 33.2 1.0 | 3.9 ± 1.6 4429 ± 921.3 | 94.8 5.2 |
| 1 | 4.3 ± 1.8 124.8 ± 55.6 5032 ± 593.8 | 75.2 21.8 3.0 | 4.1 ± 1.1 387.2 ± 361.2 3709 ± 1061 | 60.4 37.6 2.0 | 3.8 ± 0.8 2304 ± 382.2 | 18.4 81.6 |
| 3 | 4.3 ± 1.6 175.4 ± 97.4 | 78.8 21.2 | 4.1 ± 1.3 68.2 ± 28.5 700.2 ± 412.4 | 40.7 4.9 54.4 | ND | ND |
| Polymer Concentration (% w/w) | TG→L (°C) | |
|---|---|---|
| C4C6 | C6 | |
| 1 | Not tested | 85.5 ± 0.5 |
| 2.5 | 100.4 ± 0.7 | 93.0 ± 0.7 |
| 5 | 103.7 ± 0.3 | 101.0 ± 1.4 |
| 10 | 107.3 ± 0.5 | Not tested |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnston, E.R.; Smith, T.N.; Serban, M.A. Nanoparticulate Poly(glucaramide)-Based Hydrogels for Controlled Release Applications. Gels 2017, 3, 17. https://doi.org/10.3390/gels3020017
Johnston ER, Smith TN, Serban MA. Nanoparticulate Poly(glucaramide)-Based Hydrogels for Controlled Release Applications. Gels. 2017; 3(2):17. https://doi.org/10.3390/gels3020017
Chicago/Turabian StyleJohnston, Erik R., Tyler N. Smith, and Monica A. Serban. 2017. "Nanoparticulate Poly(glucaramide)-Based Hydrogels for Controlled Release Applications" Gels 3, no. 2: 17. https://doi.org/10.3390/gels3020017