Left Right Patterning, Evolution and Cardiac Development
Abstract
:1. Introduction
2. Establishment of Left-Right Asymmetry in the Early Embryo
2.1. Axis Establishment in the Mouse Embryo
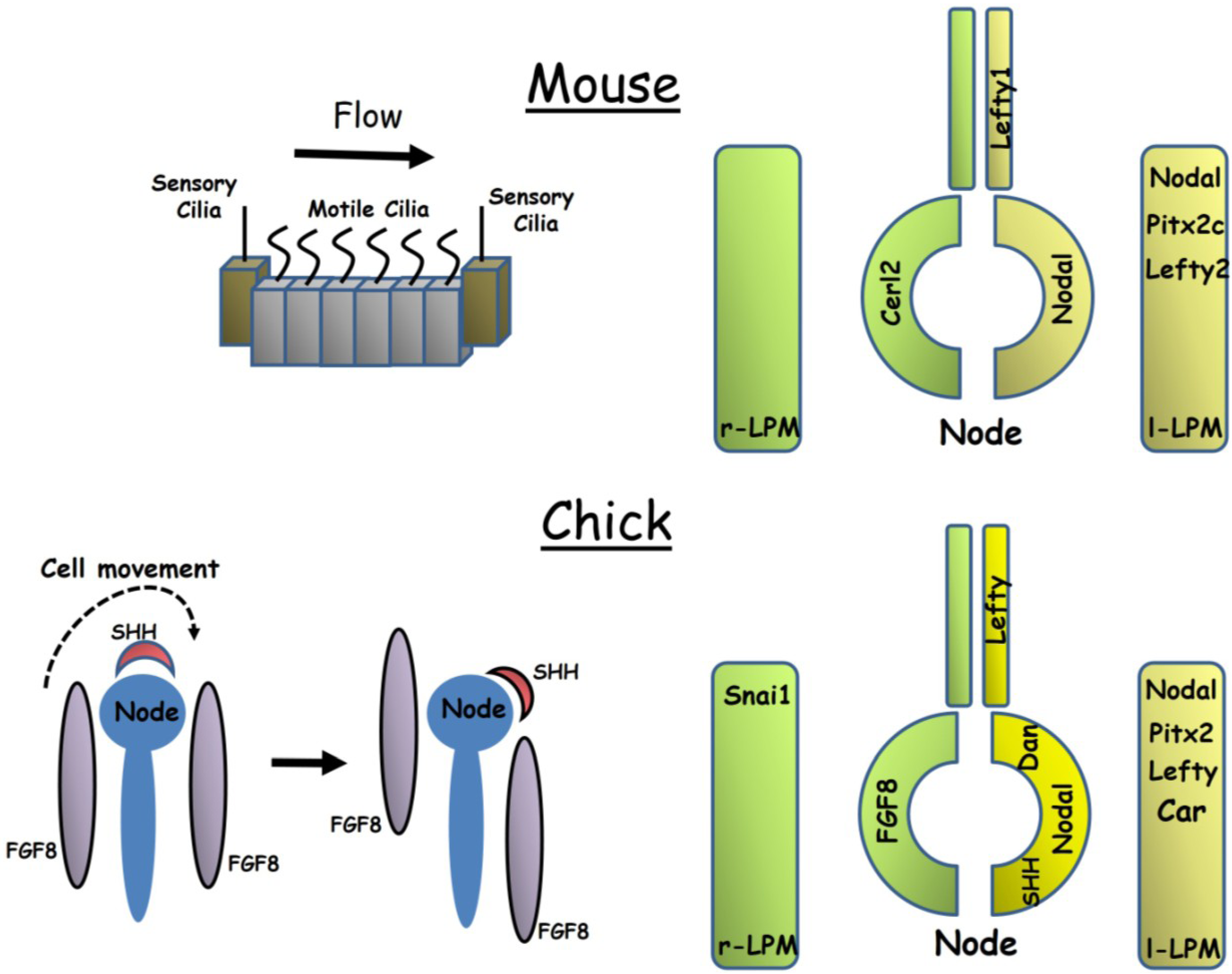
2.2. Axis Establishment in the Chick Embryo
2.3. The Evolution of Left-Right Axis Establishment

3. Cardiac Phenotypes Associated with Impaired Left-Right Patterning
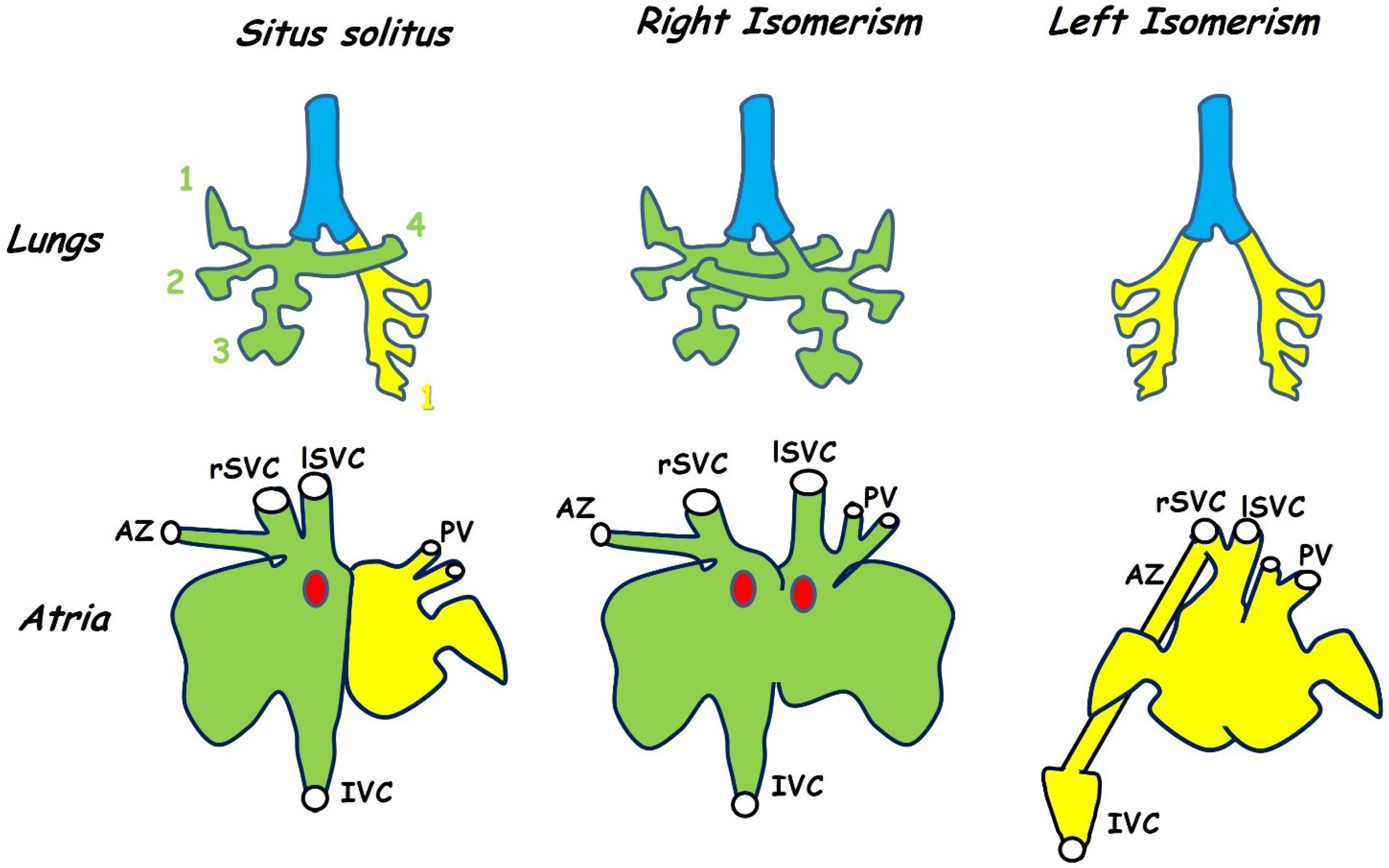
3.1. Atrial Isomerism and Venous Drainage
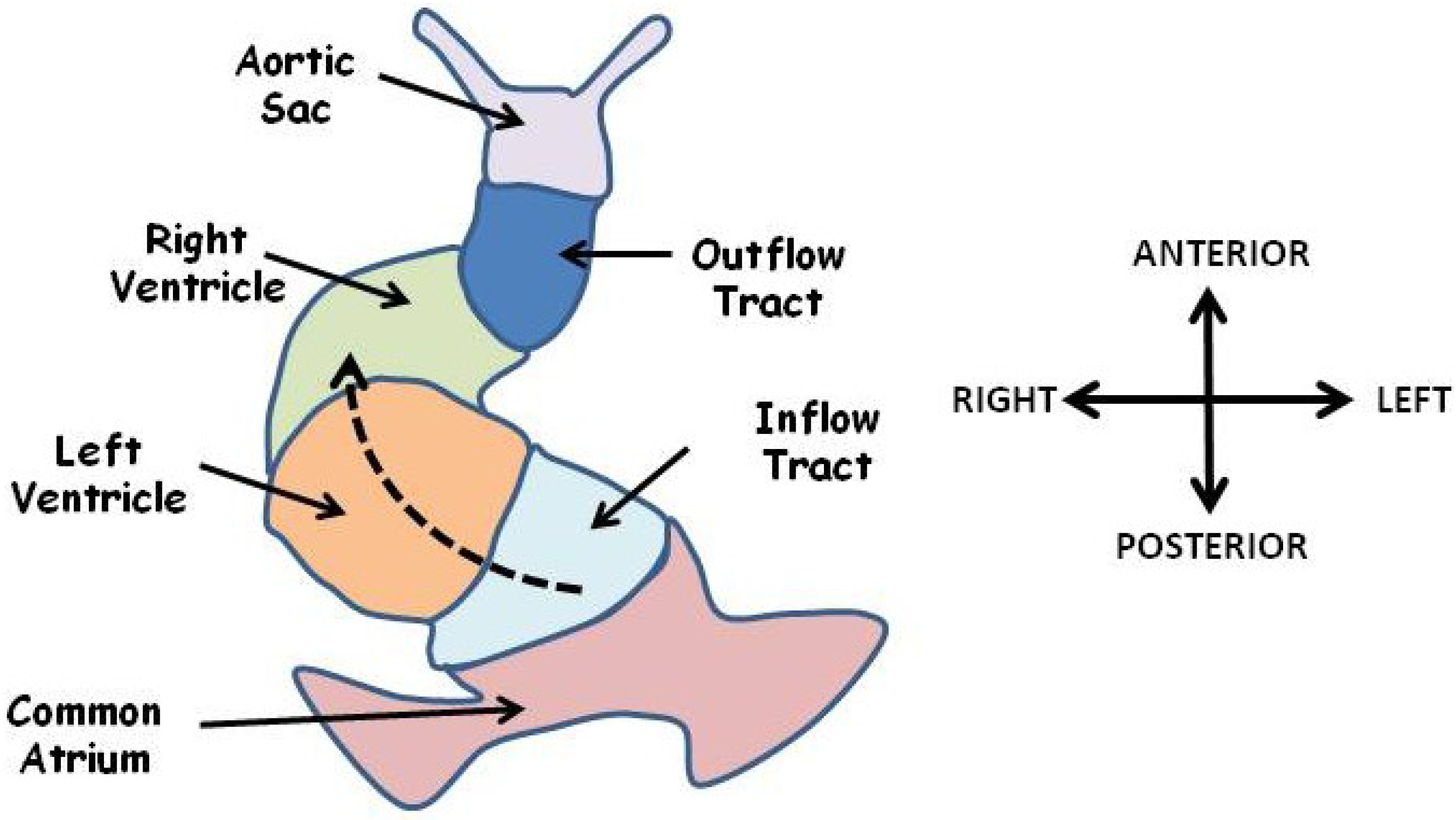
3.2. Defective Looping Morphogenesis
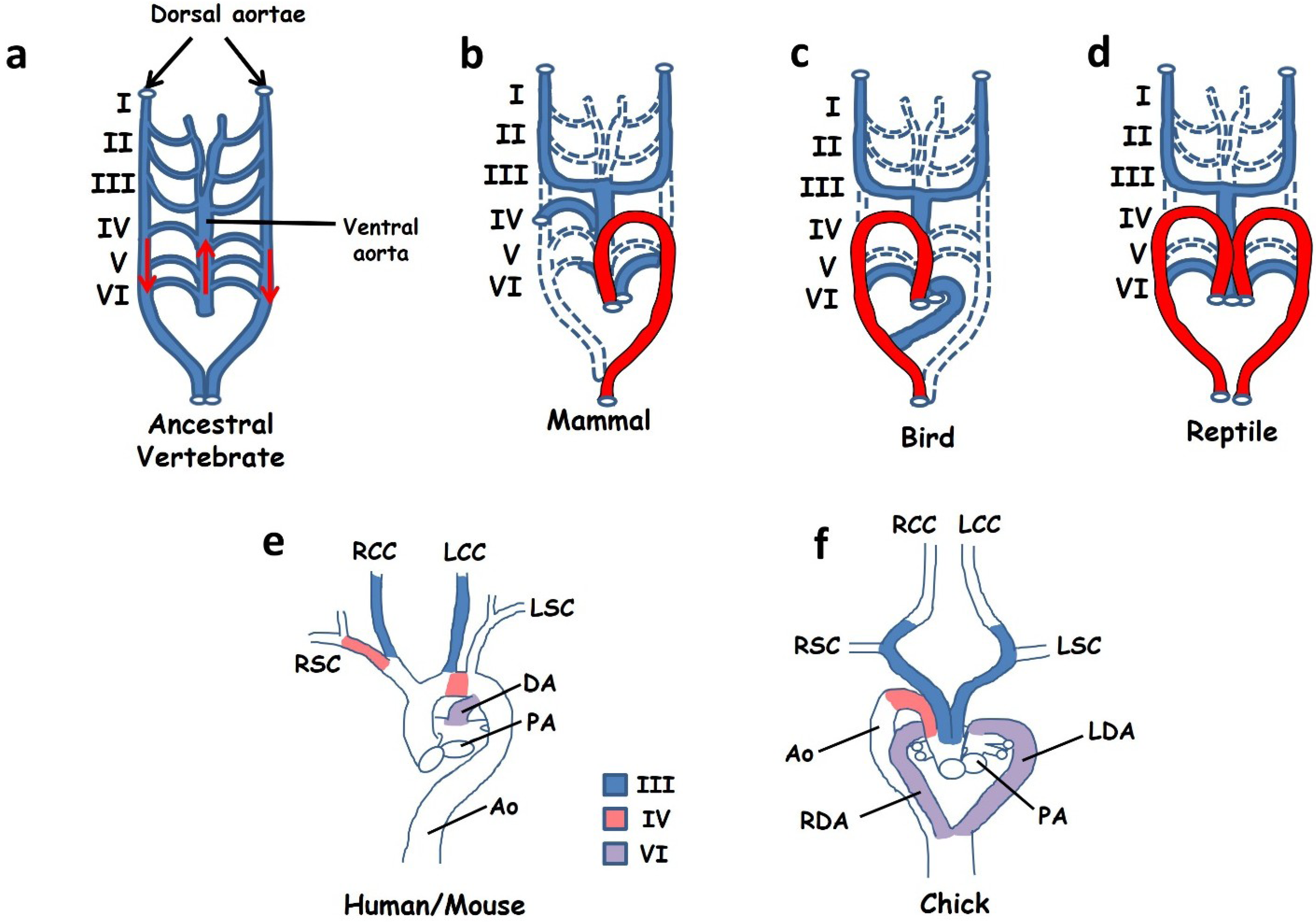
3.3. Aortic Arch Defects
4. Concluding Remarks: Left Right Patterning and Cardiac Disease
Acknowledgments
Conflicts of Interest
References
- Sutherland, M.J.; Ware, S.M. Disorders of left-right asymmetry: Heterotaxy and situs inversus. Am. J. Med. Genet. C Semin. Med. Genet. 2009, 151C, 307–317. [Google Scholar] [CrossRef]
- Zhu, L.; Belmont, J.W.; Ware, S.M. Genetics of human heterotaxias. Eur. J. Hum. Genet. 2006, 14, 17–25. [Google Scholar]
- Jacobs, J.P.; Anderson, R.H.; Weinberg, P.M.; Walters, H.L., 3rd; Tchervenkov, C.I.; Del Duca, D.; Franklin, R.C.; Aiello, V.D.; Beland, M.J.; Colan, S.D.; et al. The nomenclature, definition and classification of cardiac structures in the setting of heterotaxy. Cardiol. Young 2007, 17 (Suppl. 2), 1–28. [Google Scholar]
- Evans, S.M.; Yelon, D.; Conlon, F.L.; Kirby, M.L. Myocardial lineage development. Circ. Res. 2010, 107, 1428–1444. [Google Scholar] [CrossRef]
- Manner, J. The anatomy of cardiac looping: A step towards the understanding of the morphogenesis of several forms of congenital cardiac malformations. Clin. Anat. 2009, 22, 21–35. [Google Scholar] [CrossRef]
- Savolainen, S.M.; Foley, J.F.; Elmore, S.A. Histology atlas of the developing mouse heart with emphasis on e11.5 to e18.5. Toxicol. Pathol. 2009, 37, 395–414. [Google Scholar] [CrossRef]
- Moorman, A.; Webb, S.; Brown, N.A.; Lamers, W.; Anderson, R.H. Development of the heart: (1) Formation of the cardiac chambers and arterial trunks. Heart 2003, 89, 806–814. [Google Scholar] [CrossRef]
- Chen, C.M.; Norris, D.; Bhattacharya, S. Transcriptional control of left-right patterning in cardiac development. Pediatr. Cardiol. 2010, 31, 371–377. [Google Scholar] [CrossRef]
- Hamada, H.; Meno, C.; Watanabe, D.; Saijoh, Y. Establishment of vertebrate left-right asymmetry. Nat. Rev. Genet. 2002, 3, 103–113. [Google Scholar]
- Hirokawa, N.; Tanaka, Y.; Okada, Y. Left-right determination: Involvement of molecular motor kif3, cilia, and nodal flow. Cold Spring Harbor Perspect. Biol. 2009, 1. [Google Scholar] [CrossRef]
- Nonaka, S.; Shiratori, H.; Saijoh, Y.; Hamada, H. Determination of left-right patterning of the mouse embryo by artificial nodal flow. Nature 2002, 418, 96–99. [Google Scholar] [CrossRef]
- Collignon, J.; Varlet, I.; Robertson, E.J. Relationship between asymmetric nodal expression and the direction of embryonic turning. Nature 1996, 381, 155–158. [Google Scholar] [CrossRef]
- Lowe, L.A.; Supp, D.M.; Sampath, K.; Yokoyama, T.; Wright, C.V.; Potter, S.S.; Overbeek, P.; Kuehn, M.R. Conserved left-right asymmetry of nodal expression and alterations in murine situs inversus. Nature 1996, 381, 158–161. [Google Scholar] [CrossRef]
- Marques, S.; Borges, A.C.; Silva, A.C.; Freitas, S.; Cordenonsi, M.; Belo, J.A. The activity of the nodal antagonist cerl-2 in the mouse node is required for correct l/r body axis. Genes Dev. 2004, 18, 2342–2347. [Google Scholar] [CrossRef]
- Inacio, J.M.; Marques, S.; Nakamura, T.; Shinohara, K.; Meno, C.; Hamada, H.; Belo, J.A. The dynamic right-to-left translocation of cerl2 is involved in the regulation and termination of nodal activity in the mouse node. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Nakamura, T.; Saito, D.; Kawasumi, A.; Shinohara, K.; Asai, Y.; Takaoka, K.; Dong, F.; Takamatsu, A.; Belo, J.A.; Mochizuki, A.; et al. Fluid flow and interlinked feedback loops establish left-right asymmetric decay of cerl2 mrna. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef]
- Norris, D.P. Cilia, calcium and the basis of left-right asymmetry. BMC Biol. 2012, 10. [Google Scholar] [CrossRef]
- McGrath, J.; Somlo, S.; Makova, S.; Tian, X.; Brueckner, M. Two populations of node monocilia initiate left-right asymmetry in the mouse. Cell 2003, 114, 61–73. [Google Scholar] [CrossRef]
- Field, S.; Riley, K.L.; Grimes, D.T.; Hilton, H.; Simon, M.; Powles-Glover, N.; Siggers, P.; Bogani, D.; Greenfield, A.; Norris, D.P. Pkd1l1 establishes left-right asymmetry and physically interacts with pkd2. Development 2011, 138, 1131–1142. [Google Scholar] [CrossRef]
- Tanaka, Y.; Okada, Y.; Hirokawa, N. Fgf-induced vesicular release of sonic hedgehog and retinoic acid in leftward nodal flow is critical for left-right determination. Nature 2005, 435, 172–177. [Google Scholar] [CrossRef]
- Brennan, J.; Norris, D.P.; Robertson, E.J. Nodal activity in the node governs left-right asymmetry. Genes Dev. 2002, 16, 2339–2344. [Google Scholar] [CrossRef]
- Oki, S.; Hashimoto, R.; Okui, Y.; Shen, M.M.; Mekada, E.; Otani, H.; Saijoh, Y.; Hamada, H. Sulfated glycosaminoglycans are necessary for nodal signal transmission from the node to the left lateral plate in the mouse embryo. Development 2007, 134, 3893–3904. [Google Scholar] [CrossRef]
- Saund, R.S.; Kanai-Azuma, M.; Kanai, Y.; Kim, I.; Lucero, M.T.; Saijoh, Y. Gut endoderm is involved in the transfer of left-right asymmetry from the node to the lateral plate mesoderm in the mouse embryo. Development 2012, 139, 2426–2435. [Google Scholar] [CrossRef]
- Viotti, M.; Niu, L.; Shi, S.H.; Hadjantonakis, A.K. Role of the gut endoderm in relaying left-right patterning in mice. PLoS Biol. 2012, 10. [Google Scholar] [CrossRef]
- Norris, D.P.; Brennan, J.; Bikoff, E.K.; Robertson, E.J. The foxh1-dependent autoregulatory enhancer controls the level of nodal signals in the mouse embryo. Development 2002, 129, 3455–3468. [Google Scholar]
- Shiratori, H.; Sakuma, R.; Watanabe, M.; Hashiguchi, H.; Mochida, K.; Sakai, Y.; Nishino, J.; Saijoh, Y.; Whitman, M.; Hamada, H. Two-step regulation of left-right asymmetric expression of pitx2: Initiation by nodal signaling and maintenance by nkx2. Mol. Cell. 2001, 7, 137–149. [Google Scholar] [CrossRef]
- Saijoh, Y.; Adachi, H.; Mochida, K.; Ohishi, S.; Hirao, A.; Hamada, H. Distinct transcriptional regulatory mechanisms underlie left-right asymmetric expression of lefty-1 and lefty-2. Genes Dev. 1999, 13, 259–269. [Google Scholar] [CrossRef]
- Franco, D.; Christoffels, V.M.; Campione, M. Homeobox transcription factor pitx2: The rise of an asymmetry gene in cardiogenesis and arrhythmogenesis. Trends Cardiovasc. Med. 2014, 24, 23–31. [Google Scholar] [CrossRef]
- Ishimaru, Y.; Yoshioka, H.; Tao, H.; Thisse, B.; Thisse, C.; Wright, C.V.E.; Hamada, H.; Ohuchi, H.; Noji, S. Asymmetric expression of antivin/lefty1 in the early chick embryo. Mech Dev. 2000, 90, 115–118. [Google Scholar] [CrossRef]
- Logan, M.; Pagan-Westphal, S.M.; Smith, D.M.; Paganessi, L.; Tabin, C.J. The transcription factor pitx2 mediates situs-specific morphogenesis in response to left-right asymmetric signals. Cell 1998, 94, 307–317. [Google Scholar] [CrossRef]
- Palmer, A.R. Symmetry breaking and the evolution of development. Science 2004, 306, 828–833. [Google Scholar] [CrossRef]
- Levin, M.; Johnson, R.L.; Stern, C.D.; Kuehn, M.; Tabin, C. A molecular pathway determining left-right asymmetry in chick embryogenesis. Cell 1995, 82, 803–814. [Google Scholar] [CrossRef]
- Pagan-Westphal, S.M.; Tabin, C.J. The transfer of left-right positional information during chick embryogenesis. Cell 1998, 93, 25–35. [Google Scholar] [CrossRef]
- Boettger, T.; Wittler, L.; Kessel, M. Fgf8 functions in the specification of the right body side of the chick. Curr. Biol. 1999, 9, 277–280. [Google Scholar] [CrossRef]
- Patel, K.; Isaac, A.; Cooke, J. Nodal signalling and the roles of the transcription factors SnR and Pitx2 in vertebrate left-right asymmetry. Curr. Biol. 1999, 9, 609–612. [Google Scholar] [CrossRef]
- Tavares, A.T.; Andrade, S.; Silva, A.C.; Belo, J.A. Cerberus is a feedback inhibitor of nodal asymmetric signaling in the chick embryo. Development 2007, 134, 2051–2060. [Google Scholar] [CrossRef]
- Yu, X.; He, F.; Zhang, T.; Espinoza-Lewis, R.A.; Lin, L.; Yang, J.; Chen, Y. Cerberus functions as a bmp agonist to synergistically induce nodal expression during left-right axis determination in the chick embryo. Dev. Dyn. 2008, 237, 3613–3623. [Google Scholar] [CrossRef]
- Rodriguez Esteban, C.; Capdevila, J.; Economides, A.N.; Pascual, J.; Ortiz, A.; Izpisua Belmonte, J.C. The novel cer-like protein caronte mediates the establishment of embryonic left-right asymmetry. Nature 1999, 401, 243–251. [Google Scholar] [CrossRef]
- Katsu, K.; Tokumori, D.; Tatsumi, N.; Suzuki, A.; Yokouchi, Y. Bmp inhibition by dan in hensen’s node is a critical step for the establishment of left-right asymmetry in the chick embryo. Dev. Biol. 2012, 363, 15–26. [Google Scholar] [CrossRef]
- Manner, J. Does an equivalent of the “ventral node” exist in chick embryos? A scanning electron microscopic study. Anat. Embryol. 2001, 203, 481–490. [Google Scholar] [CrossRef]
- Dathe, V.; Gamel, A.; Manner, J.; Brand-Saberi, B.; Christ, B. Morphological left-right asymmetry of hensen’s node precedes the asymmetric expression of shh and fgf8 in the chick embryo. Anat. Embryol. 2002, 205, 343–354. [Google Scholar] [CrossRef]
- Cui, C.; Little, C.D.; Rongish, B.J. Rotation of organizer tissue contributes to left-right asymmetry. Anat. Rec. 2009, 292, 557–561. [Google Scholar] [CrossRef]
- Gros, J.; Feistel, K.; Viebahn, C.; Blum, M.; Tabin, C.J. Cell movements at hensen’s node establish left/right asymmetric gene expression in the chick. Science 2009, 324, 941–944. [Google Scholar] [CrossRef]
- Levin, M.; Thorlin, T.; Robinson, K.R.; Nogi, T.; Mercola, M. Asymmetries in h+/k+-atpase and cell membrane potentials comprise a very early step in left-right patterning. Cell 2002, 111, 77–89. [Google Scholar] [CrossRef]
- Levin, M.; Mercola, M. Gap junction-mediated transfer of left-right patterning signals in the early chick blastoderm is upstream of shh asymmetry in the node. Development 1999, 126, 4703–4714. [Google Scholar]
- Okabe, N.; Xu, B.; Burdine, R.D. Fluid dynamics in zebrafish kupffer’s vesicle. Dev. Dyn. 2008, 237, 3602–3612. [Google Scholar] [CrossRef]
- Vick, P.; Schweickert, A.; Weber, T.; Eberhardt, M.; Mencl, S.; Shcherbakov, D.; Beyer, T.; Blum, M. Flow on the right side of the gastrocoel roof plate is dispensable for symmetry breakage in the frog xenopus laevis. Dev. Biol. 2009, 331, 281–291. [Google Scholar] [CrossRef]
- Schweickert, A.; Weber, T.; Beyer, T.; Vick, P.; Bogusch, S.; Feistel, K.; Blum, M. Cilia-driven leftward flow determines laterality in xenopus. Curr. Biol. 2007, 17, 60–66. [Google Scholar] [CrossRef]
- Blum, M.; Weber, T.; Beyer, T.; Vick, P. Evolution of leftward flow. Semin. Cell Dev. Biol. 2009, 20, 464–471. [Google Scholar] [CrossRef]
- Okada, Y.; Takeda, S.; Tanaka, Y.; Izpisua Belmonte, J.C.; Hirokawa, N. Mechanism of nodal flow: A conserved symmetry breaking event in left-right axis determination. Cell 2005, 121, 633–644. [Google Scholar] [CrossRef]
- Nakamura, T.; Hamada, H. Left-right patterning: Conserved and divergent mechanisms. Development 2012, 139, 3257–3262. [Google Scholar] [CrossRef]
- Chea, H.K.; Wright, C.V.; Swalla, B.J. Nodal signaling and the evolution of deuterostome gastrulation. Dev. Dyn. 2005, 234, 269–278. [Google Scholar] [CrossRef]
- Grande, C.; Patel, N.H. Lophotrochozoa get into the game: The nodal pathway and left/right asymmetry in bilateria. Cold Spring Harbor Symp. Quant. Biol. 2009, 74, 281–287. [Google Scholar] [CrossRef]
- Namigai, E.K.; Kenny, N.J.; Shimeld, S.M. Right across the tree of life: The evolution of left-right asymmetry in the bilateria. Genesis 2014. [Google Scholar] [CrossRef]
- Vorbruggen, G.; Constien, R.; Zilian, O.; Wimmer, E.A.; Dowe, G.; Taubert, H.; Noll, M.; Jackle, H. Embryonic expression and characterization of a ptx1 homolog in drosophila. Mech. Dev. 1997, 68, 139–147. [Google Scholar] [CrossRef]
- Jin, Y.; Hoskins, R.; Horvitz, H.R. Control of type-d gabaergic neuron differentiation by C. elegans UNC-30 homeodomain protein. Nature 1994, 372, 780–783. [Google Scholar] [CrossRef]
- Grande, C.; Patel, N.H. Nodal signalling is involved in left-right asymmetry in snails. Nature 2009, 457, 1007–1011. [Google Scholar] [CrossRef]
- Duboc, V.; Rottinger, E.; Lapraz, F.; Besnardeau, L.; Lepage, T. Left-right asymmetry in the sea urchin embryo is regulated by nodal signaling on the right side. Dev. Cell. 2005, 9, 147–158. [Google Scholar] [CrossRef]
- Duboc, V.; Lepage, T. A conserved role for the nodal signaling pathway in the establishment of dorso-ventral and left-right axes in deuterostomes. J. Exp. Zool. B Mol. Dev. Evol. 2008, 310, 41–53. [Google Scholar] [CrossRef]
- Adams, D.S.; Robinson, K.R.; Fukumoto, T.; Yuan, S.; Albertson, R.C.; Yelick, P.; Kuo, L.; McSweeney, M.; Levin, M. Early, h+-v-atpase-dependent proton flux is necessary for consistent left-right patterning of non-mammalian vertebrates. Development 2006, 133, 1657–1671. [Google Scholar] [CrossRef]
- Aw, S.; Adams, D.S.; Qiu, D.; Levin, M. H,k-atpase protein localization and kir4.1 function reveal concordance of three axes during early determination of left-right asymmetry. Mech. Dev. 2008, 125, 353–372. [Google Scholar] [CrossRef]
- Ramsdell, A.F. Left-right asymmetry and congenital cardiac defects: Getting to the heart of the matter in vertebrate left-right axis determination. Dev. Biol. 2005, 288, 1–20. [Google Scholar] [CrossRef]
- Warburton, D.; Schwarz, M.; Tefft, D.; Flores-Delgado, G.; Anderson, K.D.; Cardoso, W.V. The molecular basis of lung morphogenesis. Mech. Dev. 2000, 92, 55–81. [Google Scholar] [CrossRef]
- Meno, C.; Shimono, A.; Saijoh, Y.; Yashiro, K.; Mochida, K.; Ohishi, S.; Noji, S.; Kondoh, H.; Hamada, H. Lefty-1 is required for left-right determination as a regulator of lefty-2 and nodal. Cell 1998, 94, 287–297. [Google Scholar] [CrossRef]
- Liu, C.; Liu, W.; Lu, M.F.; Brown, N.A.; Martin, J.F. Regulation of left-right asymmetry by thresholds of pitx2c activity. Development 2001, 128, 2039–2048. [Google Scholar]
- Macartney, F.J.; Zuberbuhler, J.R.; Anderson, R.H. Morphological considerations pertaining to recognition of atrial isomerism. Consequences for sequential chamber localisation. Br. Heart J. 1980, 44, 657–667. [Google Scholar] [CrossRef]
- Seo, J.W.; Brown, N.A.; Ho, S.Y.; Anderson, R.H. Abnormal laterality and congenital cardiac anomalies. Relations of visceral and cardiac morphologies in the iv/iv mouse. Circulation 1992, 86, 642–650. [Google Scholar] [CrossRef]
- Icardo, J.M.; Sanchez de Vega, M.J. Spectrum of heart malformations in mice with situs solitus, situs inversus, and associated visceral heterotaxy. Circulation 1991, 84, 2547–2558. [Google Scholar] [CrossRef]
- Hildreth, V.; Webb, S.; Chaudhry, B.; Peat, J.D.; Phillips, H.M.; Brown, N.; Anderson, R.H.; Henderson, D.J. Left cardiac isomerism in the sonic hedgehog null mouse. J. Anat. 2009, 214, 894–904. [Google Scholar] [CrossRef]
- Fasouliotis, S.J.; Achiron, R.; Kivilevitch, Z.; Yagel, S. The human fetal venous system: Normal embryologic, anatomic, and physiologic characteristics and developmental abnormalities. J. Ultrasound Med. 2002, 21, 1145–1158. [Google Scholar]
- Sharma, S.; Devine, W.; Anderson, R.H.; Zuberbuhler, J.R. Identification and analysis of left atrial isomerism. Am. J. Cardiol. 1987, 60, 1157–1160. [Google Scholar] [CrossRef]
- Kitamura, K.; Miura, H.; Miyagawa-Tomita, S.; Yanazawa, M.; Katoh-Fukui, Y.; Suzuki, R.; Ohuchi, H.; Suehiro, A.; Motegi, Y.; Nakahara, Y.; et al. Mouse pitx2 deficiency leads to anomalies of the ventral body wall, heart, extra- and periocular mesoderm and right pulmonary isomerism. Development 1999, 126, 5749–5758. [Google Scholar]
- Sadler, T.W. Langman’s Medical Embryology; Lippincott, Williams and Wilkins: Philadelphia, PA, USA, 2000. [Google Scholar]
- Rubino, M.; Van Praagh, S.; Kadoba, K.; Pessotto, R.; Van Praagh, R. Systemic and pulmonary venous connections in visceral heterotaxy with asplenia. Diagnostic and surgical considerations based on seventy-two autopsied cases. J. Thorac. Cardiovasc. Surg. 1995, 110, 641–650. [Google Scholar] [CrossRef]
- Peoples, W.M.; Moller, J.H.; Edwards, J.E. Polysplenia: A review of 146 cases. Pediatr. Cardiol. 1983, 4, 129–137. [Google Scholar] [CrossRef]
- Franco, D.; Chinchilla, A.; Aranega, A.E. Transgenic insights linking pitx2 and atrial arrhythmias. Front. Physiol. 2012, 3. [Google Scholar] [CrossRef]
- Smith, A.; Ho, S.Y.; Anderson, R.H.; Connell, M.G.; Arnold, R.; Wilkinson, J.L.; Cook, A.C. The diverse cardiac morphology seen in hearts with isomerism of the atrial appendages with reference to the disposition of the specialised conduction system. Cardiol. Young 2006, 16, 437–454. [Google Scholar] [CrossRef]
- Ammirabile, G.; Tessari, A.; Pignataro, V.; Szumska, D.; Sutera Sardo, F.; Benes, J., Jr.; Balistreri, M.; Bhattacharya, S.; Sedmera, D.; Campione, M. Pitx2 confers left morphological, molecular, and functional identity to the sinus venosus myocardium. Cardiovasc. Res. 2012, 93, 291–301. [Google Scholar] [CrossRef]
- Wang, J.; Klysik, E.; Sood, S.; Johnson, R.L.; Wehrens, X.H.; Martin, J.F. Pitx2 prevents susceptibility to atrial arrhythmias by inhibiting left-sided pacemaker specification. Proc. Nat. Acad. Sci. USA 2010, 107, 9753–9758. [Google Scholar]
- Harvey, R.P. Cardiac looping—An uneasy deal with laterality. Semin. Cell Dev. Biol. 1998, 9, 101–108. [Google Scholar]
- Manner, J. On the form problem of embryonic heart loops, its geometrical solutions, and a new biophysical concept of cardiac looping. Ann. Anat. 2013, 195, 312–323. [Google Scholar] [CrossRef]
- Baker, K.; Holtzman, N.G.; Burdine, R.D. Direct and indirect roles for nodal signaling in two axis conversions during asymmetric morphogenesis of the zebrafish heart. Proc. Nat. Acad. Sci. USA 2008, 105, 13924–13929. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhu, L.; Hu, L.; Slesnick, T.C.; Pautler, R.G.; Justice, M.J.; Belmont, J.W. Zic3 is required in the extra-cardiac perinodal region of the lateral plate mesoderm for left-right patterning and heart development. Hum. Mol. Genet. 2013, 22, 879–889. [Google Scholar] [CrossRef]
- Bamforth, S.D.; Braganca, J.; Farthing, C.R.; Schneider, J.E.; Broadbent, C.; Michell, A.C.; Clarke, K.; Neubauer, S.; Norris, D.; Brown, N.A.; et al. Cited2 controls left-right patterning and heart development through a nodal-pitx2c pathway. Nat. Genet. 2004, 36, 1189–1196. [Google Scholar] [CrossRef]
- Soshnikova, N.; Dewaele, R.; Janvier, P.; Krumlauf, R.; Duboule, D. Duplications of hox gene clusters and the emergence of vertebrates. Dev. Biol. 2013, 378, 194–199. [Google Scholar] [CrossRef]
- Lin, Q.; Schwarz, J.; Bucana, C.; Olson, E.N. Control of mouse cardiac morphogenesis and myogenesis by transcription factor mef2c. Science 1997, 276, 1404–1407. [Google Scholar] [CrossRef]
- Cai, C.L.; Liang, X.; Shi, Y.; Chu, P.H.; Pfaff, S.L.; Chen, J.; Evans, S. Isl1 identifies a cardiac progenitor population that proliferates prior to differentiation and contributes a majority of cells to the heart. Dev. Cell. 2003, 5, 877–889. [Google Scholar] [CrossRef]
- Ryan, A.K.; Blumberg, B.; Rodriguez-Esteban, C.; Yonei-Tamura, S.; Tamura, K.; Tsukui, T.; de la Pena, J.; Sabbagh, W.; Greenwald, J.; Choe, S.; et al. Pitx2 determines left-right asymmetry of internal organs in vertebrates. Nature 1998, 394, 545–551. [Google Scholar]
- Yu, X.; St Amand, T.R.; Wang, S.; Li, G.; Zhang, Y.; Hu, Y.P.; Nguyen, L.; Qiu, M.S.; Chen, Y.P. Differential expression and functional analysis of pitx2 isoforms in regulation of heart looping in the chick. Development 2001, 128, 1005–1013. [Google Scholar]
- Linask, K.K.; Yu, X.; Chen, Y.; Han, M.D. Directionality of heart looping: Effects of pitx2c misexpression on flectin asymmetry and midline structures. Dev. Biol. 2002, 246, 407–417. [Google Scholar] [CrossRef]
- Campione, M.; Steinbeisser, H.; Schweickert, A.; Deissler, K.; van Bebber, F.; Lowe, L.A.; Nowotschin, S.; Viebahn, C.; Haffter, P.; Kuehn, M.R.; et al. The homeobox gene pitx2: Mediator of asymmetric left-right signaling in vertebrate heart and gut looping. Development 1999, 126, 1225–1234. [Google Scholar]
- Gage, P.J.; Suh, H.; Camper, S.A. Dosage requirement of pitx2 for development of multiple organs. Development 1999, 126, 4643–4651. [Google Scholar]
- Shiratori, H.; Yashiro, K.; Shen, M.M.; Hamada, H. Conserved regulation and role of pitx2 in situs-specific morphogenesis of visceral organs. Development 2006, 133, 3015–3025. [Google Scholar] [CrossRef]
- Lowe, L.A.; Yamada, S.; Kuehn, M.R. Genetic dissection of nodal function in patterning the mouse embryo. Development 2001, 128, 1831–1843. [Google Scholar]
- Yan, Y.T.; Gritsman, K.; Ding, J.; Burdine, R.D.; Corrales, J.D.; Price, S.M.; Talbot, W.S.; Schier, A.F.; Shen, M.M. Conserved requirement for egf-cfc genes in vertebrate left-right axis formation. Genes Dev. 1999, 13, 2527–2537. [Google Scholar] [CrossRef]
- Liu, C.; Liu, W.; Palie, J.; Lu, M.F.; Brown, N.A.; Martin, J.F. Pitx2c patterns anterior myocardium and aortic arch vessels and is required for local cell movement into atrioventricular cushions. Development 2002, 129, 5081–5091. [Google Scholar]
- Bamforth, S.D.; Braganca, J.; Eloranta, J.J.; Murdoch, J.N.; Marques, F.I.; Kranc, K.R.; Farza, H.; Henderson, D.J.; Hurst, H.C.; Bhattacharya, S. Cardiac malformations, adrenal agenesis, neural crest defects and exencephaly in mice lacking cited2, a new tfap2 co-activator. Nat. Genet. 2001, 29, 469–474. [Google Scholar] [CrossRef]
- Muster, A.J.; Idriss, R.F.; Backer, C.L. The left-sided aortic arch in humans, viewed as the end-result of natural selection during vertebrate evolution. Cardiol. Young 2001, 11, 111–122. [Google Scholar] [CrossRef]
- Waldo, K.; Kirby, M.L. Development of the great arteries. In Living Morphogenesis of the Heart; De la Cruz, M.V., Markwald, R.R., Eds.; Birkhauser: Boston, MA, USA, 1998; pp. 187–217. [Google Scholar]
- Kingsley, J.S. Outline of Comparative Anatomy of Vertebrates; The Blakiston Company: Philadelphia, PA, USA, 1920. [Google Scholar]
- Bamforth, S.D.; Chaudhry, B.; Bennett, M.; Wilson, R.; Mohun, T.J.; Van Mierop, L.H.; Henderson, D.J.; Anderson, R.H. Clarification of the identity of the mammalian fifth pharyngeal arch artery. Clin. Anat. 2013, 26, 173–182. [Google Scholar] [CrossRef]
- Jacobs, K.; Goy, S.K.; Dzialowski, E.M. Morphology of the embryonic and hatchling american alligator ductus arteriosi and implications for embryonic cardiovascular shunting. J. Morphol. 2012, 273, 186–194. [Google Scholar] [CrossRef]
- Paffett-Lugassy, N.; Singh, R.; Nevis, K.R.; Guner-Ataman, B.; O’Loughlin, E.; Jahangiri, L.; Harvey, R.P.; Burns, C.G.; Burns, C.E. Heart field origin of great vessel precursors relies on nkx2.5-mediated vasculogenesis. Nat. Cell. Biol. 2013, 15, 1362–1369. [Google Scholar] [CrossRef]
- Oh, S.P.; Li, E. The signaling pathway mediated by the type iib activin receptor controls axial patterning and lateral asymmetry in the mouse. Genes Dev. 1997, 11, 1812–1826. [Google Scholar] [CrossRef]
- Yoshioka, H.; Meno, C.; Koshiba, K.; Sugihara, M.; Itoh, H.; Ishimaru, Y.; Inoue, T.; Ohuchi, H.; Semina, E.V.; Murray, J.C.; et al. Pitx2, a bicoid-type homeobox gene, is involved in a lefty-signaling pathway in determination of left-right asymmetry. Cell 1998, 94, 299–305. [Google Scholar] [CrossRef]
- Schweickert, A.; Campione, M.; Steinbeisser, H.; Blum, M. Pitx2 isoforms: Involvement of pitx2c but not pitx2a or pitx2b in vertebrate left-right asymmetry. Mech. Dev. 2000, 90, 41–51. [Google Scholar] [CrossRef]
- Meyers, E.N.; Martin, G.R. Differences in left-right axis pathways in mouse and chick: Functions of fgf8 and shh. Science 1999, 285, 403–406. [Google Scholar] [CrossRef]
- Schneider, A.; Mijalski, T.; Schlange, T.; Dai, W.; Overbeek, P.; Arnold, H.H.; Brand, T. The homeobox gene nkx3.2 is a target of left-right signalling and is expressed on opposite sides in chick and mouse embryos. Curr. Biol. 1999, 9, 911–914. [Google Scholar] [CrossRef]
- Brown, C.B.; Baldwin, H.S. Neural crest contribution to the cardiovascular system. Adv. Exp. Med. Biol. 2006, 589, 134–154. [Google Scholar] [CrossRef]
- Yashiro, K.; Shiratori, H.; Hamada, H. Haemodynamics determined by a genetic programme govern asymmetric development of the aortic arch. Nature 2007, 450, 285–288. [Google Scholar] [CrossRef]
- Unolt, M.; Putotto, C.; Silvestri, L.M.; Marino, D.; Scarabotti, A.; Valerio, M.; Caiaro, A.; Versacci, P.; Marino, B. Transposition of great arteries: New insights into the pathogenesis. Front. Pediatr. 2013, 1. [Google Scholar] [CrossRef]
- MacDonald, S.T.; Bamforth, S.D.; Chen, C.M.; Farthing, C.R.; Franklyn, A.; Broadbent, C.; Schneider, J.E.; Saga, Y.; Lewandoski, M.; Bhattacharya, S. Epiblastic cited2 deficiency results in cardiac phenotypic heterogeneity and provides a mechanism for haploinsufficiency. Cardiovasc. Res. 2008, 79, 448–457. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dykes, I.M. Left Right Patterning, Evolution and Cardiac Development. J. Cardiovasc. Dev. Dis. 2014, 1, 52-72. https://doi.org/10.3390/jcdd1010052
Dykes IM. Left Right Patterning, Evolution and Cardiac Development. Journal of Cardiovascular Development and Disease. 2014; 1(1):52-72. https://doi.org/10.3390/jcdd1010052
Chicago/Turabian StyleDykes, Iain M. 2014. "Left Right Patterning, Evolution and Cardiac Development" Journal of Cardiovascular Development and Disease 1, no. 1: 52-72. https://doi.org/10.3390/jcdd1010052



