3D Cell Migration Studies for Chemotaxis on Microfluidic-Based Chips: A Comparison between Cardiac and Dermal Fibroblasts
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Fabrication of Microfluidic Devices
2.3. Migration Assay
2.4. Immunofluorescence
3. Results and Discussion
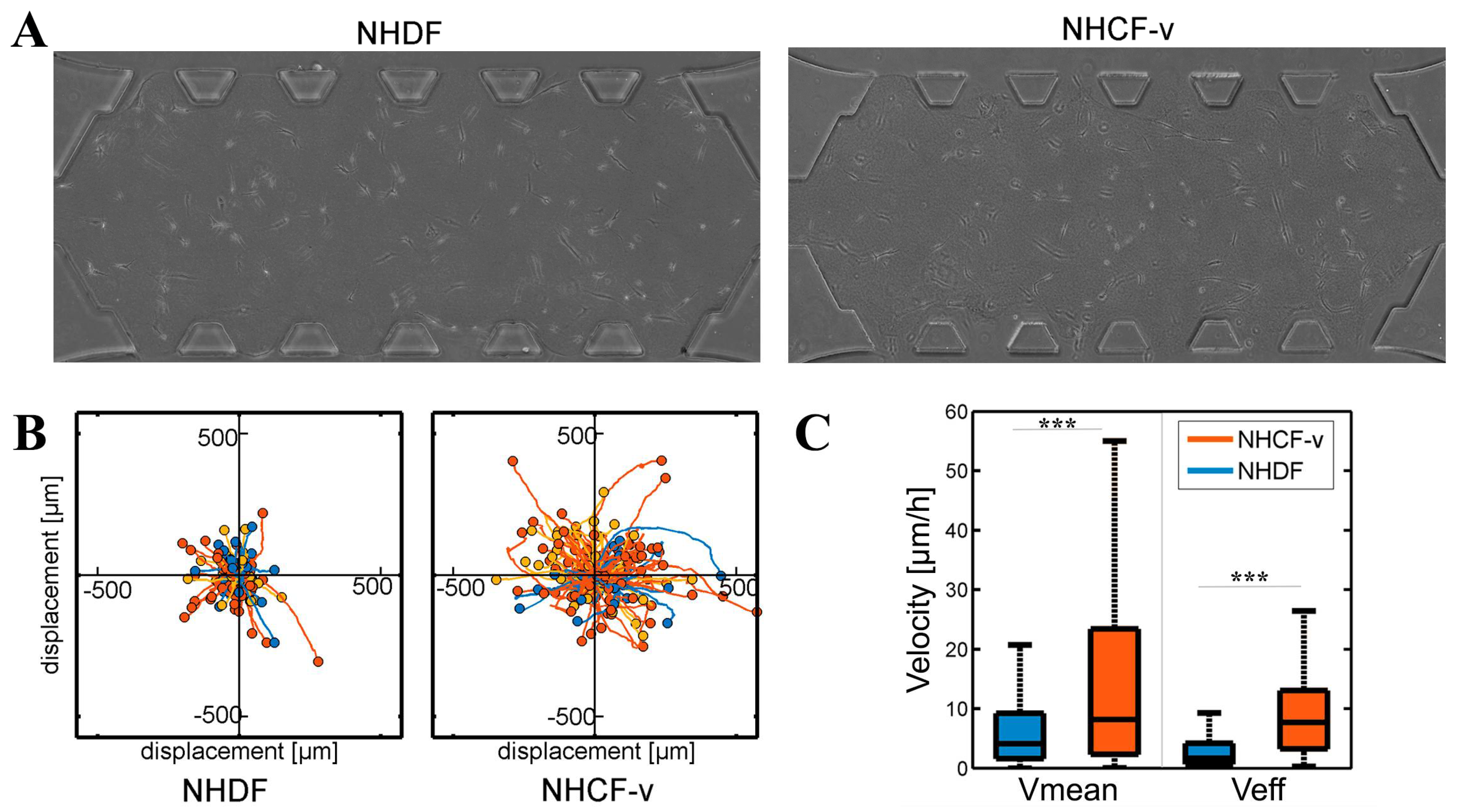
3.1. Cardiac Fibroblasts Migrate at Higher Speeds and a More Directional Pattern than Dermal Fibroblasts
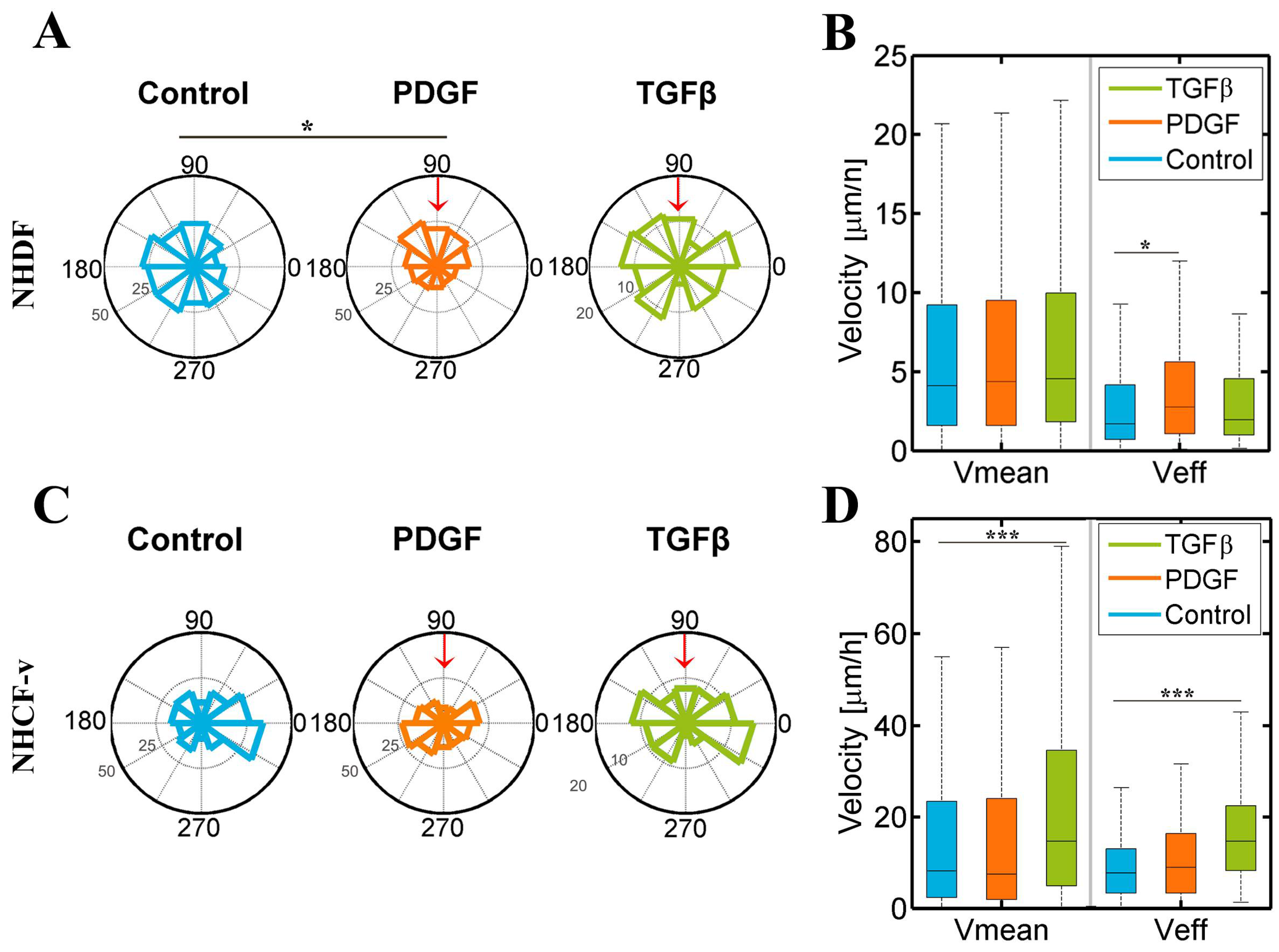
3.2. Dermal Fibroblasts Are Chemoattracted by PDGF-BB, Whereas Cardiac Fibroblasts Respond to TGF-β1, Increasing Their Mean and Effective Velocities
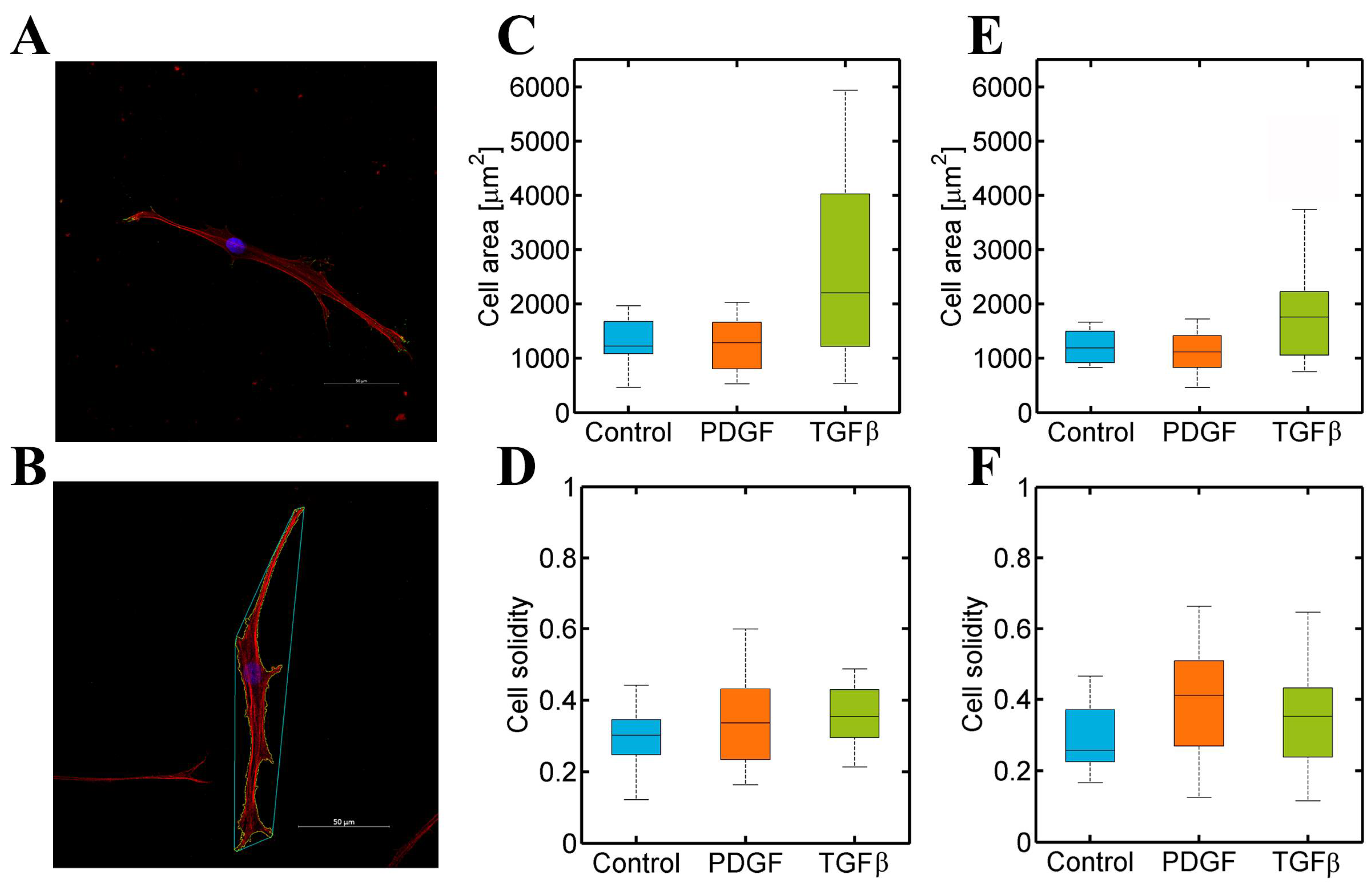
3.3. PDGF-BB and TGF-β1 Do Not Modify Dermal or Cardiac Fibroblast Morphology
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| 2D | two dimensions |
| 3D | three dimensions |
| BSA | bovine serum albumin |
| DAPI | 4′,6-diamino-2-fenilindol |
| FBS | fetal bovine serum |
| GA-1000 | gentaminicin sulfate/Amphotericin |
| NHCF-v | normal human cardio fibroblasts from ventricles |
| NHDF | normal human dermal fibroblasts |
| PBS | phosphate-buffered saline |
| PDGF-BB | platelet derived growth factor BB |
| PDL | poly-d-lysine |
| PDMS | polydimethylsiloxane |
| rhFGF-B | Fibroblast Growth Factor basic human recombinant |
| TGF-β1 | transforming growth factor beta 1 |
References
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, P.; Walter, K. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002; ISBN 0-8153-3218-1. [Google Scholar]
- Zeisberg, E.; Kalluri, R. Origins of cardiac Fibroblasts. Circ. Res. 2010, 107, 1304–1312. [Google Scholar] [CrossRef] [PubMed]
- Docherty, G.; Morales, A.; Lopez, J.; Pérez, F. The transition of epithelial cells to fibromioblasts. Mechanisms involved and its possible relationship with renal fibrosis. Nefrologia 2007, 27, 681–688. [Google Scholar] [PubMed]
- Leask, A. TGFβ, cardiac fibroblasts, and the fibrotic response. Cardiovasc. Res. 2007, 74, 207–212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrientos, S.; Stojadinovic, O.; Golinko, M.; Brem, H.; Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008, 16, 585–601. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhao, T.; Huang, V.; Chen, Y.; Ahokas, R.; Sun, Y. Platelet-Derived Growth Factor Involvement in Myocardial Remodeling following Infarction. J. Mol. Cell Cardiol. 2011, 51, 830–868. [Google Scholar] [CrossRef] [PubMed]
- Penn, J.; Grobbelaar, A.; Rolfe, K. The role of the TGF-β family in wound healing, burns and scarring: A review. Int. J. Burns Trauma 2012, 2, 18–28. [Google Scholar] [PubMed]
- Wound Healing and Growth Factors. Available online: http://emedicine.medscape.com/article/1298196-overview#a3 (accessed on 4 October 2015).
- Lindner, D.; Zietsch, C.; Tank, J.; Sossalla, S.; Fluschnik, N.; Hinrichs, S.; Maier, L.; Poller, W.; Blankenberg, S.; Schultheiss, H.; Tschöpe, C.; Westermann, D. Cardiac fibroblasts support cardiac inflammation in heart failure. Basic Res. Cardiol. 2014, 109, 428. [Google Scholar] [CrossRef] [PubMed]
- Midgley, A.; Rogers, M.; Hallett, M.; Claytonn, A.; Bowen, T.; Phillips, A.; Steadman, R. Transforming Growth Factor-β1 (TGF-β1)-stimulated Fibroblast to Myofibroblast Differentiation Is Mediated by Hyaluronan (HA)-facilitated Epidermal Growth Factor Receptor (EGFR) and CD44 Co-localization in Lipid Rafts. J. Biol. Chem. 2013, 288, 14824–14838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, P.-H.; Giri, A.; Sun, S. X.; Wirtz, D. Three-dimensional cell migration does not follow a random walk. Proc. Natl. Acad. Sci. USA 2014, 111, 3949–3954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sackmann, E.; Fulton, A.; Beebe, D. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Halldorsson, S.; Lucumi, E.; Gómez-Sjöberg, R.; Fleming, R. Advantages and challenges of microfluidic cell culture in polydimethysiloxane devices. Biosens. Bioelectron. 2015, 63, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Movilla, N.; Borau, C.; Valero, C.; García-Aznar, J. Degradation of extracellular matrix regulates osteoblast migration: A microfluidic-based study. Bone 2017, 107, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Del Amo, C.; Borau, C.; Movilla, N.; Asín, J.; García-Aznar, J. Quantifying 3D chemotaxis in microfluidic-based chips with step gradients of collagen hydrogel concentrations. Integr. Biol. 2017, 9, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Arotzena, O.; Mendoza, G.; Cóndor, M.; Rüberg, T.; García-Aznar, J. Inducing chemotactic and haptotactic cues in microfluidic devices for three-dimensional in vitro assays. Biomicrofluidics 2014, 8, 064211. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Han, S.; Jeon, J.; Yamamoto, K.; Zervantonakis, I.; Sudo, R.; Kamm, R.; Chung, S. Microfluidic assay for simultaneous culture of multiple cell types on surfaces or within hydrogels. Nat. Protoc. 2012, 7, 1247–1259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mark, D.; Haeberle, S.; Roth, G.; von Stetten, F.; Zengerle, R. Microfluidic lab-on-a-chip platforms: requirements, characteristics and applications. Chem. Soc. Rev. 2010, 39, 1153–1182. [Google Scholar] [CrossRef] [PubMed]
- Frantz, C.; Stewart, K.; Weaver, V. The extracellular matrix at a glance. J. Cell Sci. 2010, 123, 4195–4200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, M.; Whisler, J.; Fröse, J.; Yu, C.; Shin, Y.; Kamm, R. On-chip human microvasculature assay for visualization and quantification of tumor cell extravasation dynamics. Nat. Protoc. 2017, 12, 865–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sigel, A.; Centrella, M.; Eghbali-Webb, M. Regulation of proliferative response of cardiac fibroblasts by transforming growth factor-beta 1. J. Mol. Cell Cardiol. 1996, 28, 1921–1929. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Arotzena, O.; Borau, C.; Movilla, N.; Vicente-Manzanares, M.; García-Aznar, J. Fibroblast Migration in 3D is Controlled by Haptotaxis in a Non-muscle Myosin II-Dependent Manner. Ann. Biomed. Eng. 2015, 43, 3025–3039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Amo, C.; Olivares, O.; Cóndor, M.; Blanco, A.; Santolaria, J.; Asín, J.; Borau, C.; García-Aznar, J. Matrix architecture plays a pivotal role in 3D osteoblast migration: The effect of interstitial fluid flow. J. Mech. Behav. Biomed. Mater. 2018, 83, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Rebouças JS, J.D.S.; Santos-Magalhães, N.S.; Formiga, F.R. Cardiac Regeneration using Growth Factors: Advances and Challenges. Arg. Bras Cardiol 2016, 107, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.; Minasian, R.; Caterson, E. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv. Wound Care 2016, 5, 119–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeller, P.; Skalak, T.; Ponce, A.; Prince, R. In vivo chemotactic properties and spatial expression of PDGF in developing mesenteric microvascular networks. Am. J. Physiol. Hear. Circ. Physiol. 2001, 280, H2116–H2125. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Rodríguez, S.; Tomás-González, E.; García-Aznar, J.M. 3D Cell Migration Studies for Chemotaxis on Microfluidic-Based Chips: A Comparison between Cardiac and Dermal Fibroblasts. Bioengineering 2018, 5, 45. https://doi.org/10.3390/bioengineering5020045
Pérez-Rodríguez S, Tomás-González E, García-Aznar JM. 3D Cell Migration Studies for Chemotaxis on Microfluidic-Based Chips: A Comparison between Cardiac and Dermal Fibroblasts. Bioengineering. 2018; 5(2):45. https://doi.org/10.3390/bioengineering5020045
Chicago/Turabian StylePérez-Rodríguez, Sandra, Esther Tomás-González, and José Manuel García-Aznar. 2018. "3D Cell Migration Studies for Chemotaxis on Microfluidic-Based Chips: A Comparison between Cardiac and Dermal Fibroblasts" Bioengineering 5, no. 2: 45. https://doi.org/10.3390/bioengineering5020045





