Study of Hydrogen Interactions with Nd2Fe17 and Nd2Fe14B by Means of Calorimetric Method
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
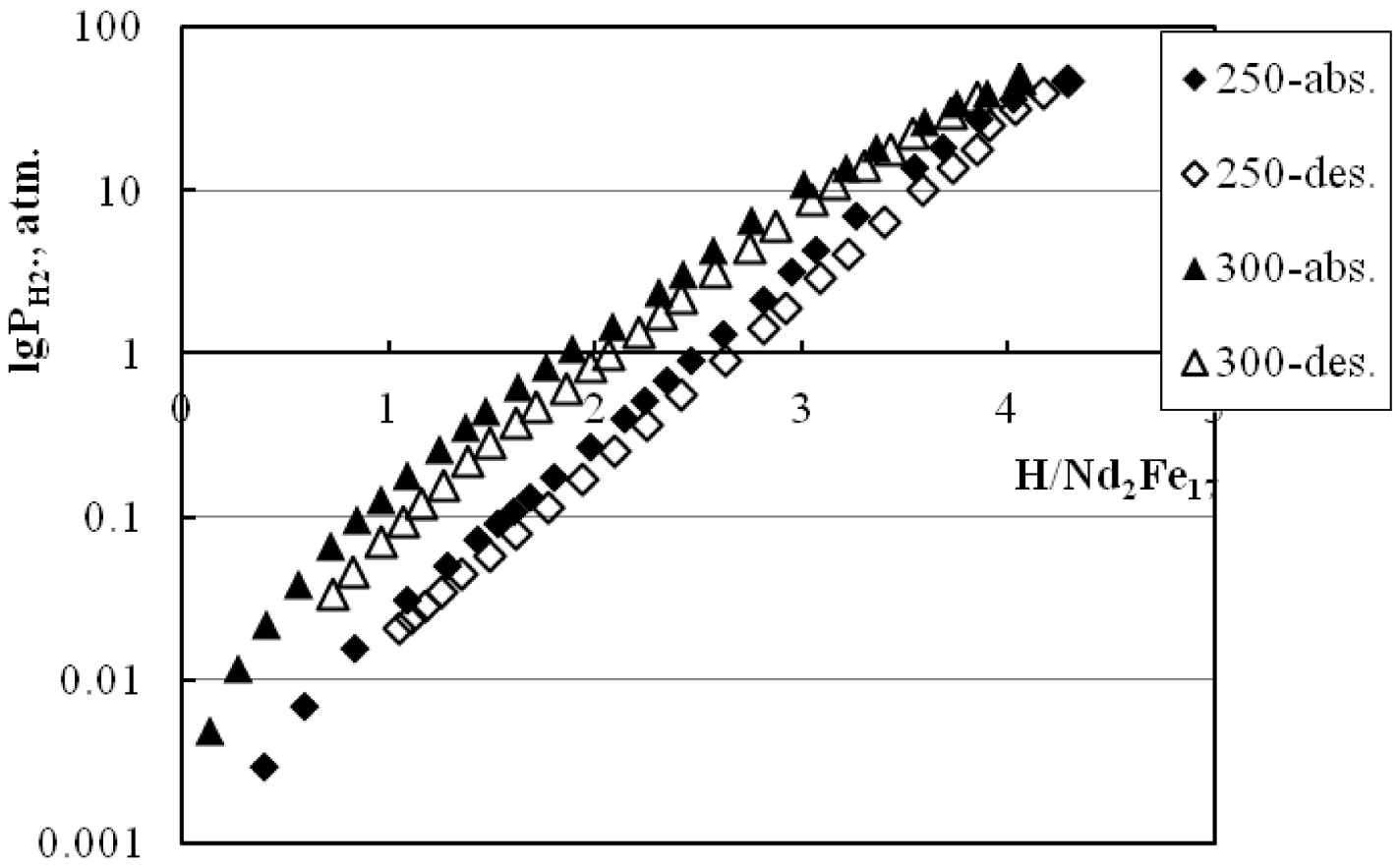
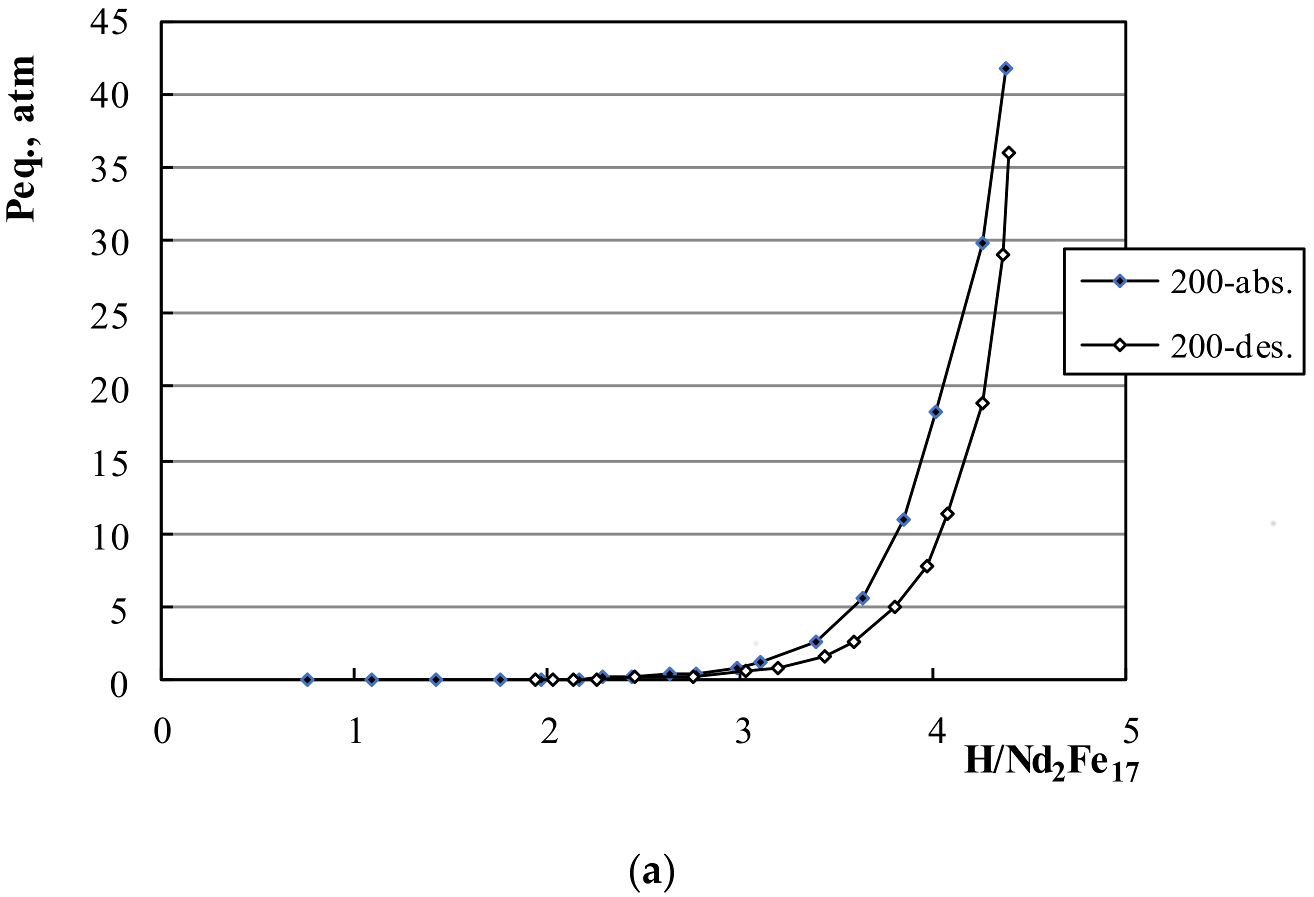
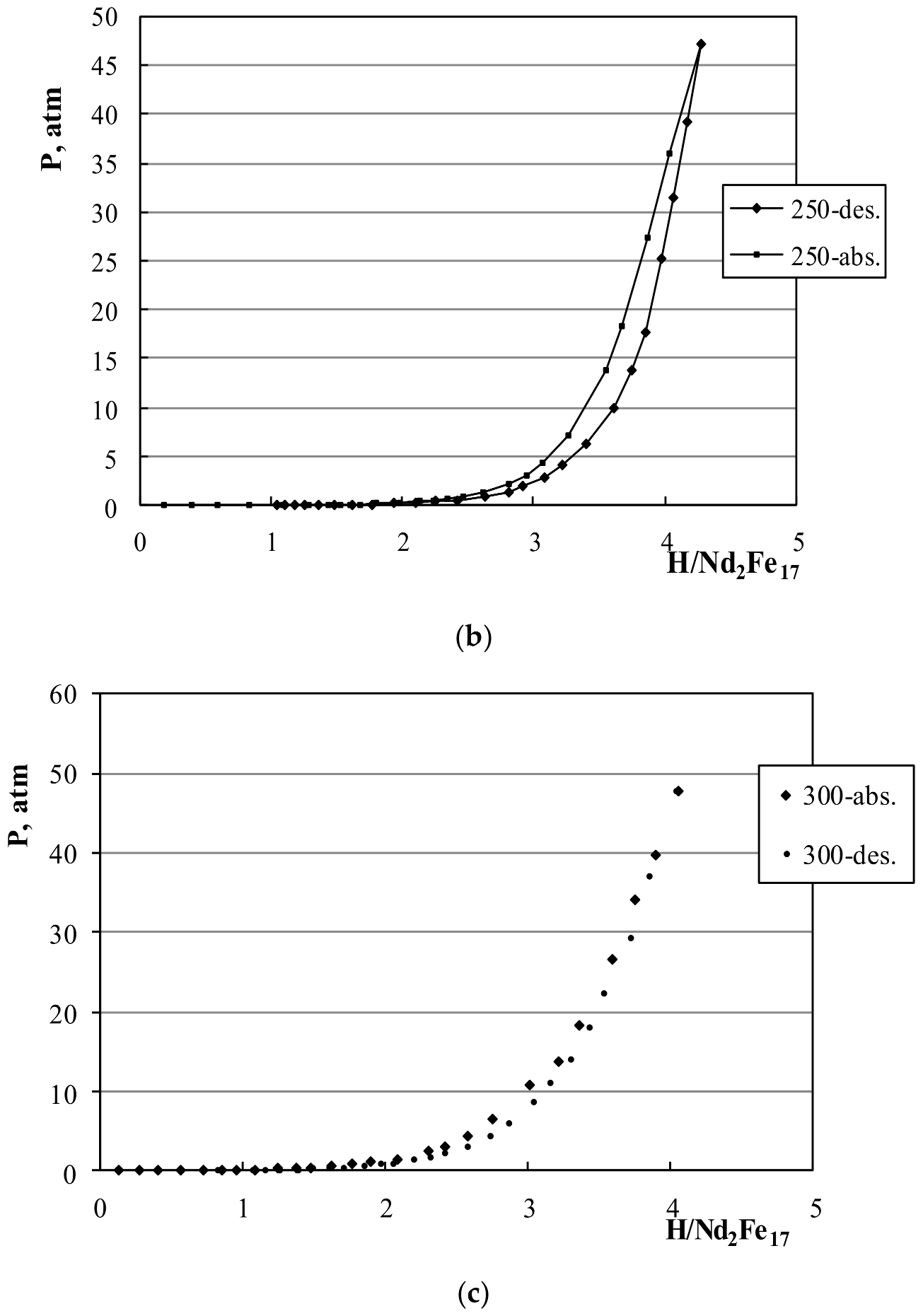
3.1. P-C Measurements
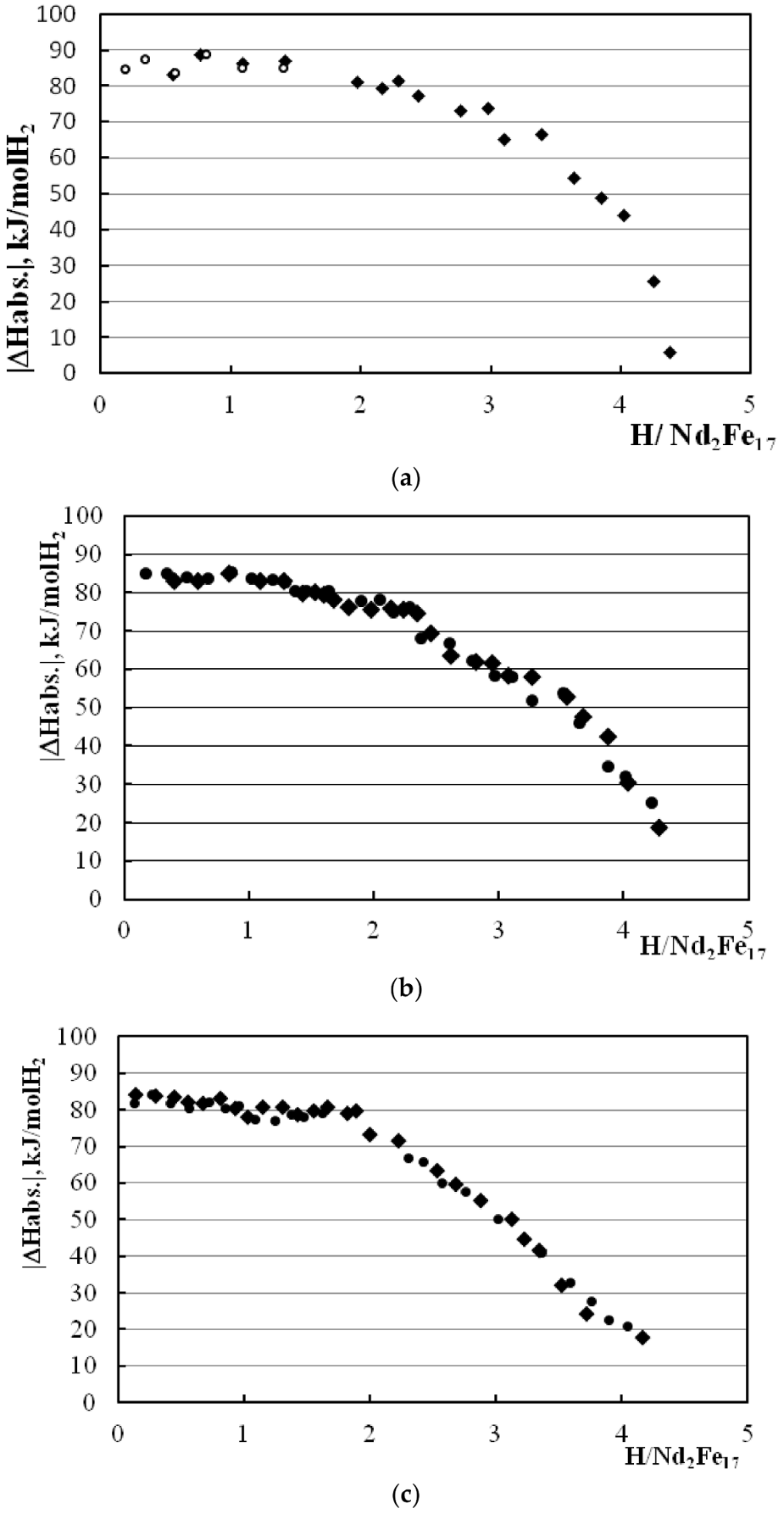
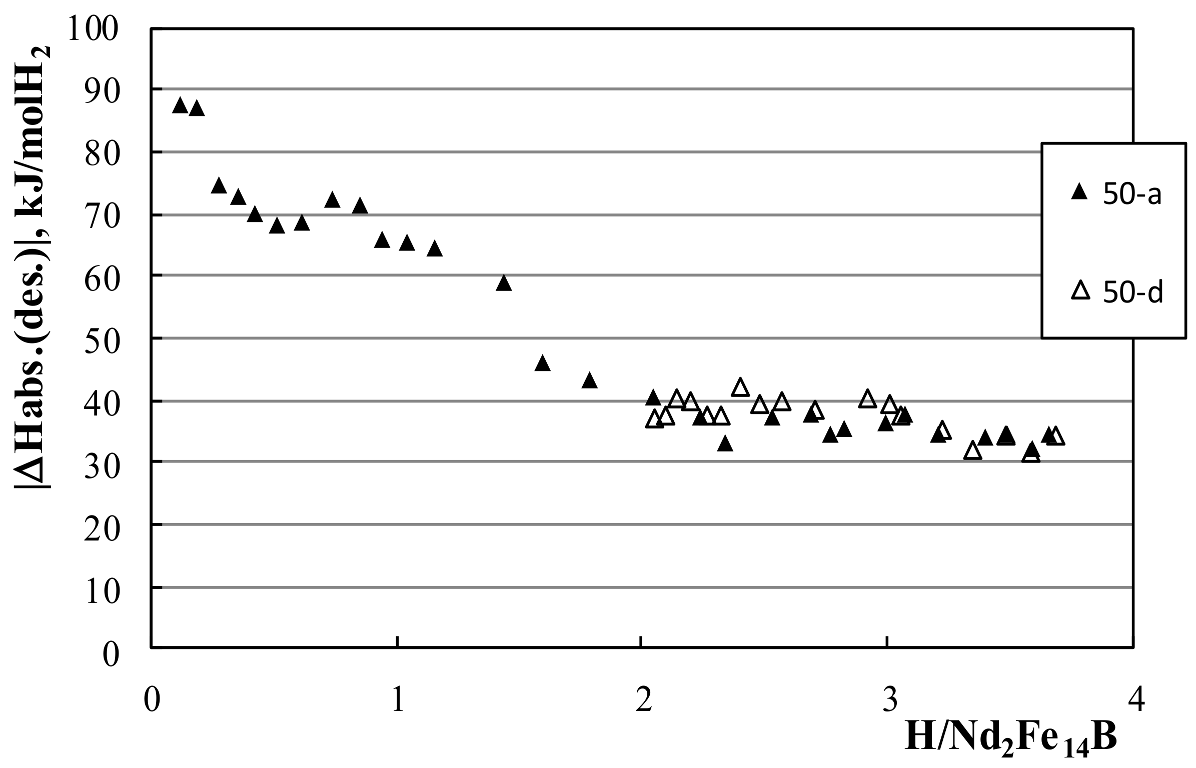
3.2. Calorimetric Results
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Isnard, O.; Miraglia, S.; Guillot, M.; Fruchart, D.; Buschow, K.H.J. High field magnetization measurements of Sm2Fe17, Sm2Fe17CX and Sm2Fe17CXH5-X. J. Appl. Phys. 1994, 76, 6035–6937. [Google Scholar] [CrossRef]
- Isnard, O.; Miraglia, S.; Soubeyroux, J.L.; Fruchart, D.; L’Héntier, P. A structural analysis and some magnetic properties of the R2Fe17HX series. J. Magn. Magn. Mater. 1994, 137, 151–156. [Google Scholar] [CrossRef]
- Cristodoulou, C.N.; Takeshita, T. Preparation, structural and magnetic properties and stability of interstitial Sm2Fe17—Carbonitrohydrides. J. Alloys Compd. 1993, 198, 1–24. [Google Scholar] [CrossRef]
- Cuevas, F.; Isnard, O.; Villeroy, B. An investigation of hydrogen desorption from Nd2Fe17HX and Dy2Fe17HX compounds by differential scanning calorimetry. Thermochim. Acta 2013, 561, 14–18. [Google Scholar] [CrossRef]
- Koeninger, V.; Koike, U.; Kamata, K.; Matsumura, Y.; Noguchi, T.; Kurino, T.; Kaneko, H.; Yanagisawa, T.; Uchida, H.H.; Uchida, H. Hydrogen absorption characteristics of Sm2Fe17 alloy. J. Phys. Chem. 1993, 181, 299–304. [Google Scholar]
- Kuji, T.; Uchda, H.; Kinoshta, K.; Yamamuro, M.; Komatsu, A. Dual site occupancy of hydrogen in Sm2Fe17. J. Alloys Compd. 2002, 330–332, 187–201. [Google Scholar] [CrossRef]
- Ram, S.; Fecht, H.J.; Haldar, S.; Ramachandraro, P.; Banerjee, H.D. Calorimetric study of the desorption of the interstitial hydrogen atoms in ferromagnetic Nd2Fe14BHX (X~5) microcrystals. Phys. Rev. B Condens. Matter 1997, 56, 726–737. [Google Scholar] [CrossRef]
- Anikina, E.Y.; Verbetsky, V.N.; Savchenko, A.G.; Menushenkov, V.P.; Shchetinin, I.V. Investigation of hydrogen interaction with intermetallic compound Nd2Fe17 by means of calorimetric method. Inorg. Mater. 2017, 53, 8–11. [Google Scholar] [CrossRef]
- Anikina, E.Y.; Verbetsky, V.N.; Savchenko, A.G.; Menushenkov, V.P.; Shchetinin, I.V. Investigation of Hydrogen Interaction with Magnetic Materials of Nd–Fe–B Type by Calorimetry Method. Inorg. Mater. Appl. Res. 2016, 7, 497–501. [Google Scholar] [CrossRef]
- Anikina, E.Y.; Verbetsky, V.N.; Savchenko, A.G.; Menushenkov, V.P.; Shchetinin, I.V. Calorimetric study of hydrogen interaction with Sm2Fe17. J. Alloys Compd. 2015, 645, S257–S260. [Google Scholar] [CrossRef]
- Sagawa, M.M.; Fujirama, S.; Togawa, M.; Yamamoto, H.; Matsuura, Y. New materials for permanent magnets on a base of Nd and Fe. J. Appl. Phys. 1984, 55, 2083–2087. [Google Scholar] [CrossRef]
- Oesterreicher, H. Structure and magnetic properties of Nd2Fe14BH2.7. J. Phys. Status Solidi 1984, 85, K61–K64. [Google Scholar] [CrossRef]
- Givord, D.; Li, H.S.; Moreau, J.M. Magnetic properties and crystal structure of Nd2Fe14B. Solid State Commun. 1984, 50, 497–499. [Google Scholar] [CrossRef]
- Herbst, J.F.; Croat, J.J.; Pinkerton, E.F.; Yelon, W.B. Relationships between crystal structure and magnetic properties in Nd2Fe14B. J. Phys. Rev. B 1984, 29, 4176–4178. [Google Scholar] [CrossRef]
- Buschow, K.H.J.; De Mooij, D.B.; Daams, J.L.C.; van Noort, H.M. Phase relationships, magnetic and crystallographic properties of Nd-Fe-B alloys. J. Less Common Met. 1986, 115, 357–366. [Google Scholar] [CrossRef]
- Anikina, E.Y.; Verbetsky, V.N. Calorimetric investigation of the hydrogen interaction with Ti0.9Zr0.1Mn1.1V0.1. J. Alloys Compd. 2002, 330–332, 45–47. [Google Scholar] [CrossRef]
- Calvet, E.; Prat, H. Microcalorimétrie: Applications physico-chimiques et biologiques. J. Am. Chem. Soc. 1957, 79, 3614–3615. [Google Scholar]
- Boureau, G.; Kleppa, O. Significance of thermal effects associated with solid-gas reaction in the Tian-Calvet calorimeter. J. Chem. Thermodym. 1977, 9, 543–548. [Google Scholar] [CrossRef]
- Currier, L.A.; Svehla, G. Nomenclature for the presentation of results of chemical analysis. (IUPAC Recommendations). J. Pure Appl. Chem. 1994, 66, 595–608. [Google Scholar] [CrossRef]
- Rupp, B.; Wiesinger, G. Magnetic and structural properties of a novel ternary hydride Nd2Fe17HX (0 < X < 5). J. Magn. Magn. Mater. 1988, 71, 269–278. [Google Scholar]
- Isnard, O.; Miraglia, S.; Fruchart, D.; Akiba, E.; Nomura, K. Hydrogen absorption in R2Fe17 alloys (R = rare earth metals) thermodynamics, structural and magnetic properties. J. Alloys Compd. 1997, 231, 150–155. [Google Scholar] [CrossRef]
- Grant, D.M.; Murray, J.J.; Post, M.L. The Thermodynamics of the System Zr(Fe0.75 Cr0.25)2-H2 Using Heat Conduction Calorimetry. J. Solid State Chem. 1990, 87, 415–422. [Google Scholar] [CrossRef]
- Sirotina, R.A.; Umerenko, E.A.; Verbetsky, V.N. Calorimetric Study of Interaction in the ZrCrFe-H2 System. Inorg. Mater. 1996, 32, 629–632. [Google Scholar]
- Ivanova, T.V.; Verbetsky, V.N. Calorimetric investigation of the hydrogen interaction with ZrCrFe1.2. J. Alloys Compd. 1999, 293–295, 34–37. [Google Scholar] [CrossRef]
- Anikina, E.Y.; Verbetsky, V.N. Investigation of the hydrogen interaction with Ti0.9Zr0.1Mn1.3V0.7 by means of the calorimetric method. Int. J. Hydrog. Energy 2016, 41, 11520–11525. [Google Scholar] [CrossRef]
- Isnard, O.; Soubeyroux, J.L.; Miraglia, S.; Fruchart, D.; Garcia, L.M.; Bartolome, J. Neutron powder diffraction study of the desorption of deuterium in R2Fe17D~X. Phys. B 1992, 180–181, 620–631. [Google Scholar]
- Isnard, O.; Miraglia, S.; Soubeyroux, J.L.; Fruchart, D. Neutron powder diffraction study of R2Fe17HX compounds with R = Pr and Nd. Solid State Commun. 1992, 81, 13–19. [Google Scholar] [CrossRef]
| T, °C | H/IMC | ∆Habs. ±δ, kJ/mol H2 | H/IMC | ∆Hdes. ±δ, kJ/mol H2 | Ref. |
|---|---|---|---|---|---|
| 300 | 0–2.0 | −80.5 ± 0.9 | 0.7–1.9 | 81.8 ± 1.3 | This work |
| 250 | 0–1.3 | −84.2 ± 0.3 | 0.8–2.0 | 80.0 ± 0.2 | This work |
| 250 | 1.4–1.8 | −79.8 ± 1.3 | This work | ||
| 200 | 0–2.0 | −85.95 ± 0.65 | [8] | ||
| 200 | 2.0–2.7 | −80.64 ± 1.0 | 1.9–2.7 | 76.48 ± 0.85 | [8] |
| T, °C | H/IMC | ∆Habs. ±δ, kJ/mol H2 | H/IMC | ∆Hdes. ±δ kJ/mol H2 |
|---|---|---|---|---|
| 250 | 0.3–2.0 | −81.7 ± 0.5 | 0.5–0.7 | 81.6 ± 0.5 |
| 250 | 2.1–3.0 | −75.6 ± 1.0 | 1.0–2.3 | 76.8 ± 0.8 |
| 200 | 0.8–1.6 | −80.2 ± 0.8 | 1.2–1.6 | ~81 |
| 200 | 1.8–2.6 | −74.9± 2.2 | 1.6–2.6 | 73.0 ± 1.1 |
| T, °C | H/IMC | ∆Habs. ±δ, kJ/mol H2 | H/IMC | ∆Hdes. ±δ, kJ/mol H2 |
|---|---|---|---|---|
| 50 | 0.3–0.9 | −69.3 ± 1.3 | ||
| 50 | 2.0–3.0 | −36.6 ± 1.6 | 2.0–3.0 | 38.7 ± 0.8 |
| 50 | 3.2–3.7 | −34.0 ± 1.3 | 3.2–3.7 | 33.5 ± 1.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anikina, E.; Verbetsky, V.; Savchenko, A. Study of Hydrogen Interactions with Nd2Fe17 and Nd2Fe14B by Means of Calorimetric Method. ChemEngineering 2018, 2, 15. https://doi.org/10.3390/chemengineering2020015
Anikina E, Verbetsky V, Savchenko A. Study of Hydrogen Interactions with Nd2Fe17 and Nd2Fe14B by Means of Calorimetric Method. ChemEngineering. 2018; 2(2):15. https://doi.org/10.3390/chemengineering2020015
Chicago/Turabian StyleAnikina, Elena, Victor Verbetsky, and Alexander Savchenko. 2018. "Study of Hydrogen Interactions with Nd2Fe17 and Nd2Fe14B by Means of Calorimetric Method" ChemEngineering 2, no. 2: 15. https://doi.org/10.3390/chemengineering2020015




