Traditional Chinese Medicine (TCM) and Herbal Hepatotoxicity: RUCAM and the Role of Novel Diagnostic Biomarkers Such as MicroRNAs
Abstract
:1. Introduction
2. Data Sources and Searches
2.1. Search Terms
2.2. Data Extraction
3. Herbal Traditional Chinese Medicine, Its History, Philosophy and Challenges
3.1. History
3.2. Philosophical Background
3.3. Herbal TCM Use and Current Issues
3.4. Integration of Herbal TCM into Western Medicine
3.5. New TCM Drugs and Approaches of Research and Development
3.6. Therapeutic Efficacy
3.7. Safety
3.8. Nomenclature
4. TCM Herbs with Published Claims of Hepatotoxic Potential
4.1. Compilation of TCM Herbal Products with Reported Claims of Liver Injury
4.2. Worldwide Reports
5. Narratives of TCM HILI Cases
5.1. An Shu Ling
5.2. Ba Jiao Lian
5.3. Bai Fang
5.4. Bai Xian Pi
5.5. Ban Tu Wan
5.6. Bo He
5.7. Bofu Tsu Sho San
5.8. Boh Gol Zhee
5.9. Bupleurum
5.10. Camellia Sinensis
5.11. Chai Hu
5.12. Chaso
5.13. Chi R Yun
5.14. Chinese Herbal Mixtures
5.15. Chinese Skullcap
5.16. Chuan Lian Zi
5.17. Ci Wu Jia
5.18. Da Chai Hu Tang
5.19. Da Huang
5.20. Dai Saiko To
5.21. Dan Zhi Xiao Yao San
5.22. Dictamnus Dasycarpus
5.23. Gan Cao
5.24. Ge Gen
5.25. Glycyrrhiza Uralensis
5.26. Ho Shou Wu
5.27. Huang Qin
5.28. Hwang Geun Cho
5.29. Ji Gu Cao
5.30. Ji Xue Cao
5.31. Jia Wei Xiao Yao San
5.32. Jiguja
5.33. Jin Bu Huan
5.34. Jing Tian San Qi
5.35. Ju San Qi
5.36. Jue Ming Zi
5.37. Kamishoyosan
5.38. Kudzu
5.39. Liquorice
5.40. Long Dan Xie Gan Tang
5.41. Long Dan Xie Gan Wan
5.42. Lu Cha
5.43. Ma Huang
5.44. Mao Guo Tian Jie Cai
5.45. Onshido
5.46. Phyllanthus Urinaria
5.47. Polygonum Multiflorum
5.48. Qibao Meiran Wan
5.49. Rhen Shen
5.50. Sairei To
5.51. Shan Chi
5.52. Shen Min
5.53. Shi Can
5.54. Shou Wu Pian
5.55. Syo Saiko To
5.56. TJ-8
5.57. TJ-9
5.58. TJ-24
5.59. White Flood
5.60. Xiao Chai Hu Tang
5.61. Yin Chen Hao
5.62. Zexie
5.63. Zhen Chu Cao
5.64. Zhi Gan Cao
6. Pathogenetic Aspects of Liver Injury from Herbal TCM
6.1. Abundance of Herbs Used as TCM
6.2. TCM Herbs and Their Molecules
6.3. Idiosyncratic and Intrinsic Liver Injury from Herbal TCM
6.3.1. Idiosyncratic Type of Liver Injury
6.3.2. Intrinsic Type of Liver Injury
6.3.3. Tentative Toxic Components or Intermediates
7. Key Clinical Features of Liver Injury Due to TCM Herbs
7.1. Duration of Herbal Use and Time to Onset
7.2. Symptoms and Clinical Features
7.3. Laboratory Results
7.4. Severity of Liver Disease and Hy’s Law
7.4.1. Liver Adaptation
7.4.2. Severe Liver Injury and Acute Liver Failure
7.4.3. Natural Course and Outcome
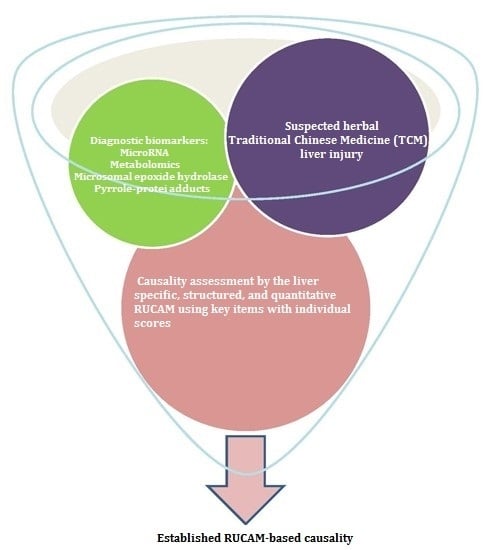
8. Biomarkers: Microsomal Epoxide Hydrolase, Pyrrole-Protein Adducts, HLA, MicroRNA, and Metabolomics
8.1. Diagnostic Biomarkers
8.1.1. Established Diagnostic Biomarkers
8.1.1.1. Microsomal Epoxide Hydrolase
8.1.1.2. Pyrrole-Protein Adducts
8.1.1.3. Metabolomics
8.2. Progress in Developing Valid Diagnostic Biomarkers
8.2.1. Human Leucocyte Antigen (HLA)
8.2.2. MicroRNA
9. Diagnostic Challenges
9.1. Minimum Quality Requirements of Herbal TCM Products
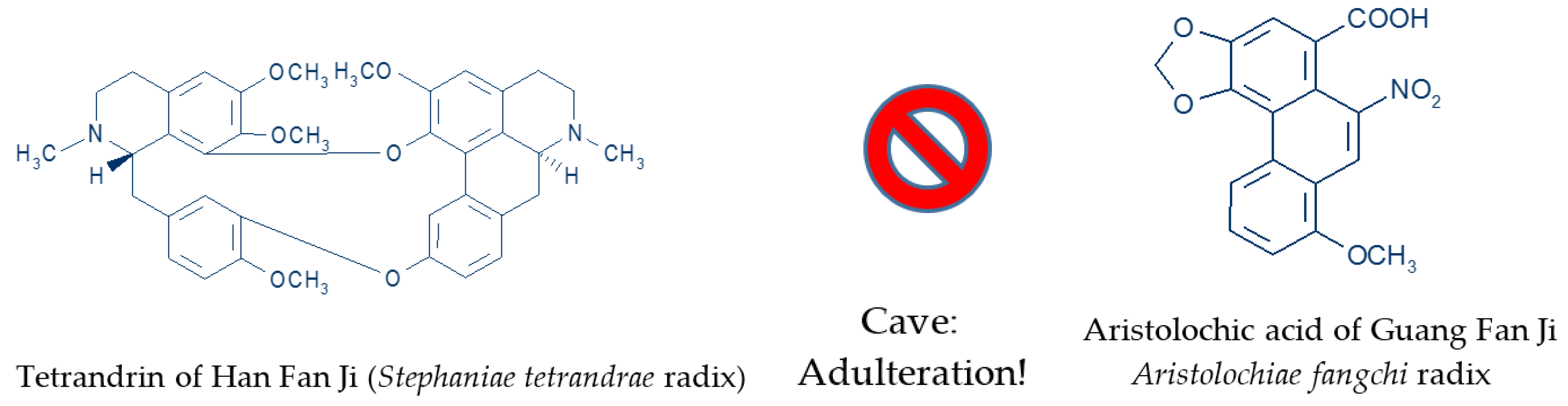
9.2. Variability of Herbal TCM Product Names, Quality, Misidentifications, Adulterants, and Impurities
9.3. Case Data Quality
10. Hepatotoxicity Criteria and Liver Injury Pattern
11. RUCAM as the Preferred Method to Assess Causality in Liver Injury Caused by TCM Herbs
12. Reexposure
13. TCM Herbs with Established, Questionable, or Lacking Liver Injury and Listing Compilation
14. Chronic Courses and Injury Risk from TCM Herbs for Preexisting Liver Disease
15. Comparison with Drug Induced Liver Injury
16. Outbreaks of Liver Injury from TCM Herbs
17. Conclusions
Conflicts of Interest
References
- Wu, W.Y.; Yang, W.Z.; Hou, J.J.; Guo, D.A. Current status and futures perspective in the globalization of Traditional Chinese Medicine. World J. Tradit. Chin. Med. 2015, 1, 1–4. [Google Scholar] [CrossRef]
- Leonti, M.; Casu, L. Traditional medicine and globalization: Current and future perspectives in ethnopharmacology. Front. Pharmacol. 2013, 4, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Pei, L.; Lu, J. Filling the gap between traditional Chinese medicine and modern medicine, are we heading to the right direction? Complement. Ther. Med. 2013, 21, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Wang, T. Development and expectation of modernization of herbal medicine. Zhongguo Zhong Xi Yi Jie He Za Zhi 2013, 33, 1018–1029. (In Chinese) [Google Scholar] [PubMed]
- NIH; National Center for Complementary and Alternative Medicine (NCCAM). Traditional Chinese Medicine: An Introduction. 2016. Available online: http://nccam.nih.gov/health/whatiscam/chinesemed.htm (accessed on 15 May 2016). [Google Scholar]
- National Institutes of Health (NIH) and LiverTox: Chinese and Other Asian Herbal Medicines. 2016. Available online: http://livertox.nih.gov/ChineseAndOtherAsianHerbalMedicines.htm (accessed on 15 May 2016).
- Liu, S.H.; Chuang, W.C.; Lam, W.; Jiang, Z.; Cheng, Y.C. Safety surveillance of Traditional Chinese Medicine: Current and future. Drug Saf. 2015, 38, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Manheimer, E.; Wieland, S.; Kimbrough, E.; Cheng, K.; Berman, B.M. Evidence from the Cochrane Collaboration for Traditional Chinese Medicine therapies. J. Altern. Complement. Med. 2009, 15, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Eickhoff, A.; Schulze, J. Herbal traditional Chinese medicine and its evidence base in gastrointestinal disorders. World J. Gastroenterol. 2015, 21, 4466–4490. [Google Scholar] [PubMed]
- Zhang, G.P.; Hou, H.P.; Ye, Z.G. Toxicity classification and detoxification strategies of Chinese Materia Medica. World J. Tradit. Chin. Med. 2015, 1, 33–37. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, J.; Liu, X.; Ye, Z.; Yang, X.; Meyboom, R.; Chan, K.; Shaw, D.; Duez, P. Pharmacovigilance practice and risk control of Traditional Chinese Medicine drugs in China: Current status and future perspective. J. Ethnopharmacol. 2012, 140, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Melchart, D.; Hager, S.; Dai, J.; Weidenhammer, W. Quality control and complication screening programme of Chinese medicinal drugs at the first German hospital of Traditional Chinese Medicine—A retrospective analysis. Forsch. Komplementmed. 2016, 23, 21–28. [Google Scholar] [PubMed]
- Melchart, D.; Linde, K.; Weidenhammer, W.; Hager, S.; Shaw, D.; Bauer, R. Liver enzyme elevations in patients treated with traditional Chinese medicine. 1999, 282, 28–29. [Google Scholar] [CrossRef]
- Efferth, T.; Kaina, B. Toxicities by herbal medicines with emphasis to Traditional Chinese medicine. Curr. Drug Metab. 2011, 12, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Zhang, A.H.; Wang, X.J. Metabolomics and its potential in drug discovery and development from TCM. World J. Tradit. Chin. Med. 2015, 1, 26–32. [Google Scholar] [CrossRef]
- Pelkonen, O.; Xu, O.; Fan, T.P. Why is research on herbal medicinal products important and how can we improve quality? J. Tradit. Complement. Med. 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Romero, M.R.; Wolf, D.G.; Stamminger, T.; Marin, J.J.G.; Marschall, M. The antiviral activities of Artemisinin and Artesunate. Clin. Infect. Dis. 2008, 47, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.A. Nobel Prize for Artemisinin inspires modern TCM Research. World J. Tradit. Chin. Med. 2015, 1, 1. [Google Scholar] [CrossRef]
- Efferth, T. Artemisinin-second career as anticancer drug? World J. Tradit. Chin. Med. 2015, 1, 2–25. [Google Scholar] [CrossRef]
- Tu, Y.; Ni, M.Y.; Zhong, Y.R.; Li, L.N.; Cui, S.L.; Zhang, M.Q.; Qang, W.Z.; Hi, Z.; Li, X.T. Studies on the constituents of Artemisia annua Part II. Planta Medica 1982, 44, 143–145. [Google Scholar]
- Tu, Y. The development of new antimalarial drugs: Qinghaosu and dihydro-qinghaosu. Chin. Med. J. 1999, 112, 976–977. [Google Scholar] [PubMed]
- World Health Organization. World Malaria Report 2014. World Health Organization, 2014. Available online: http://www.who.int/malaria/publications/world_malaria_report_2014/wmr-2014-no-profiles.pdf (accessed on 15 April 2016).
- Centers for Disease Control and Prevention (CDC). Anopheles mosquitoes. Available online: http://www.cdc.gov/malaria/about/biology/mosquitoes/ (accessed on 15 April 2016).
- Teschke, R.; Zhang, L. Chinese herbs and their molecules: Clinical and pathophysiological implications for the liver. J. Mol. Pathophysiol. 2015, 4, 85–92. [Google Scholar] [CrossRef]
- Xue, R.; Fang, Z.; Zhang, M.; Yi, Z.; Wen, C.; Shi, T. TCMID: Traditional medicine integrative database for herb molecular mechanism analysis. Nucleic Acids Res. 2013, 41. [Google Scholar] [CrossRef] [PubMed]
- Meshnick, S.R. Artemisinin: Mechanisms of action, resistance, and action. Int. J. Parasitol. 2002, 32, 1655–1660. [Google Scholar] [CrossRef]
- Bunchorntavakul, C.; Reddy, K.R. Review article: Herbal and dietary supplement hepatotoxicity. Aliment. Pharmacol. Ther. 2013, 37, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Calitz, C.; du Plessis, L.; Gouws, C.; Steyn, D.; Steenekamp, J.; Muller, C.; Hamman, S. Herbal hepatotoxicity: Current status, examples, and challenges. Expert Opin. Drug Metab. Toxicol. 2015, 11, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- Harper, D. Warring States Natural Philosophy and Occult Thought. In The Cambridge History of Ancient China: From the Origins of Civilization to 221 BC; Loewe, M., Shaughnessy, E., Eds.; Cambridge University Press: Cambridge, UK, 1999; pp. 813–884. [Google Scholar]
- Lu, Z.J. The Yellow Emperor’s Inernal Classic, an ancient medical canon of traditional Chinese medicine. J. Tradit. Chin. Med. 1985, 5, 153–154. [Google Scholar] [PubMed]
- Du, X. Study on the monographs about Jin Kui Yao Lue (synopsis of prescriptions of the Golden Chamber) in the past dynasties. J. Chin. Physician 1989, 19, 38–42. [Google Scholar]
- Beijing Declaration on International Traditional Chinese Medicine Cooperation in Science and Technology. Available online: http://www.most.gov.cn/eng/pressroom/200712/t20071206_57649.htm (accessed on 24 April 2016).
- Hager, S.; Dai, J.; Fischer, V.; Lüthke, F.; Staudinger, A. East Meets West: Synergy through Diversity. Forsch. Komplementmed. 2016, 23, 3–7. [Google Scholar] [PubMed]
- Zhang, G.G.; Lee, W.; Bausell, B.; Lao, L.; Handwerger, B.; Berman, B. Variability in the traditional Chinese medicine (TCM) diagnoses and herbal prescription provided by three TCM practitioners for 40 patients with rheumatoid arthritis. J. Altern. Complement. Med. 2005, 11, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Yifan, Y.; Al-Khafaji, M. The theory and concepts of Chinese herbal medicine. In Chinese Herbal Medicines, 2nd ed.; Churchill Livingstone: Edinburgh, Scotland, 2010; pp. 209–215. [Google Scholar]
- Yang, Y.; Al-Khafaji, M. The theory and concepts of Chinese herbal medicine. In Chinese Herbal Medicines, 2nd ed.; Churchill Livingstone: Edinburgh, Scotland, 2010; pp. 3–22. [Google Scholar]
- Sun, W.; Yang, L.; Qiu, Y.; Ren, J.; Huang, R.; Fu, J. Identify nature N-acylethanolamide-hydrolyzing acid amide (NAAA) inhibitor: Effect of angelicae pubescentis radix on anti-inflammation. China J. Chin. Mater. Med. 2011, 36, 3161–3166. [Google Scholar]
- Xu, L.L.; Wang, L.; Wang, Y.Q. Effects of radix salvia militiorrhia, radix aconiti lateralis preparata and rhizoma anemarrhena on nitric oxide systems of endotoxemia mice. Chin. J. Integr. Med. 2006, 26, 102–107. [Google Scholar]
- Lipsky, P.E.; Tao, X.L. A potential new treatment for rheumatoid arthritis: thunder god vine. Semin. Arthritis Rheum. 1997, 26, 13–723. [Google Scholar] [CrossRef]
- Ernst, E. Methodological aspects of Traditional Chinese Medicine (TCM). Ann. Acad. Med. Singap. 2006, 35, 773–774. [Google Scholar] [PubMed]
- Wang, G.; Mao, B.; Xiong, Z.Y.; Fan, T.; Chen, X.D.; Wang, L.; Liu, G.J.; Liu, J.; Guo, J.; Chang, J.; et al. The quality of reporting of randomized controlled trials of traditional Chinese medicine: A survey of 13 randomly selected journals from mainland China. Clin. Ther. 2007, 29, 1456–1467. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.L.; Zhan, S.Y.; Ernst, E. Review of randomised controlled trials of traditional Chinese medicine. BMJ 1999, 319, 160–161. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.L.; Liu, B.Y.; Ma, K.W. Traditional Chinese medicine. Lancet 2008, 372, 1938–1940. [Google Scholar] [CrossRef]
- Nortier, J.L.; Vanherweghem, J.L. Renal interstitial fibrosis and urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi). Toxicology 2002, 181–182, 577–580. [Google Scholar] [CrossRef]
- Nortier, J.L.; Martinez, M.C.; Schmeiser, H.H.; Arlt, V.M.; Bieler, C.A.; Petein, M.; Depierreux, M.F.; de Pauw, L.; Abramowicz, D.; Vereerstraeten, P.; et al. Urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi). N. Engl. J. Med. 2000, 342, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Vanherweghem, L.J. Misuse of herbal remedies: The case of an outbreak of terminal renal failure in Belgium (Chinese herbs nephropathy). J. Altern. Complement. Med. 1998, 4, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H. Introduction. In Evidence and Rational Based Research on Chinese Drugs; Wagner, H., Ulrich-Merzenich, G., Eds.; Springer: Vienna Austria, 2013; pp. 1–26. [Google Scholar]
- WHO (World Health Organization) Traditional Medicine Strategy 2014–2023. Available online: http://apps.who.int/iris/bitstream/10665/92455/1/9789241506090_eng.pdf?ua=1 (accessed on 26 April 2016).
- Frenzel, C.; Teschke, R. Herbal hepatotoxicity: Clinical characteristics and listing compilation. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Danan, G.; Teschke, R. RUCAM in drug and herb induced liver injury: The update. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Shaw, D.; Simmonds, M.S.J.; Leon, C.J.; Xu, Q.; Lu, A.; Sutherland, I.; Ignatova, S.; Zhu, Y.P.; Verpoorte, R.; et al. General practice in reviewing and publishing studies on herbal medicine, with special emphasis on traditional Chinese medicine and Chinese materia medica. J. Ethnopharmacol. 2012, 140, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Traditional Chinese Medicine induced liver injury. J. Clin. Transl. Hepatol. 2014, 2, 80–94. [Google Scholar] [CrossRef] [PubMed]
- CAMbrella Report. Available online: http://cordis.europa.eu/result/rcn/57185_en.html (accessed on 26 April 2016).
- Teschke, R.; Wolff, A.; Frenzel, C.; Schulze, J. Letter: Herbal hepatotoxicity—An update on traditional Chinese medicine preparations; authors’ reply. Aliment. Pharmacol. Ther. 2014, 40, 738–740. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.J.; Ruan, Y.X.; Lee, C.C.; Lee, M.S.; Chiou, W.Y.; Lin, H.Y.; Hsu, F.C.; Su, Y.C.; HSU, T.W.; Hung, S.K. Use of Chinese medicine among colorectal cancer patients: a nationwide population-based study. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 343–349. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4202644/ (accessed on 28 January 2014). [Google Scholar] [CrossRef] [PubMed]
- Efferth, T. Inhibition of ATP-binding cassette transporters by Chinese herbs and phytochemicals. In Evidence and Rational Based Research on Chinese Drugs; Wagner, H., Ulrich-Merzenich, G., Eds.; Springer: Wien, Austria, 2013; pp. 283–331. [Google Scholar]
- Liu, J.; Wang, S.; Zhang, Y.; Fan, H.T.; Lin, H.S. Traditional Chinese medicine and cancer: History, present situation, and development. Thoracic. Cancer 2015, 6, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Koh, H.L.; Gao, Y.; Gong, Z.Y.; Lee, E.J.D. Herbal bioactivation: The good, the bad, and the ugly. Life Sci. 2004, 74, 935–968. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A. Herbal hepatotoxicity in traditional and modern medicine: Actual key issues and new encouraging steps. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Avigan, M.I.; Mozersky, R.P.; Seeff, L.B. Scientific and regulatory perspectives in herbal and dietary supplement associated hepatotoxicity in the United States. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. Adulteration of Chinese herbal medicines with synthetic drugs: a systematic review. J. Intern. Med. 2002, 252, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. Toxic heavy metals and undeclared drugs in Asian herbal medicines. Trends Pharmacol. Sci. 2002, 23, 136–139. [Google Scholar] [CrossRef]
- Wu, M.L.; Deng, J.F.; Lin, K.P.; Tsai, W.J. Lead, mercury, and arsenic poisoning due to topical use of traditional Chinese medicines. Am. J. Med. 2013, 126, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Ting, A.; Chow, Y.; Tan, W. Microbial and heavy metal contamination in commonly consumed traditional Chinese herbal medicines. J. Tradit. Chin. Med. 2013, 33, 119–124. [Google Scholar] [CrossRef]
- Bauer, R.; Gasser, U.; Oettmeier, R.; Rausch, H. Quality of TCM drugs and TCM products—Current status in the European Union. Pharm. Read. Trib. 2013, 3, 1–10. [Google Scholar]
- Shaw, D. Toxicological risks of Chinese herbs. Planta Med. 2010, 76, 2012–2018. [Google Scholar] [CrossRef] [PubMed]
- Chan, K. Chinese medicinal materials and their interface with Western medical concepts. J. Ethnopharmacol. 2005, 96, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Pharmacopoeia of the People’s Republic of China; Peoples Medical Publishing House: Beijing, China, 2005; Volume 1.
- Haller, C.A.; Dyer, J.E.; Ko, R.; Olson, K.R. Making a diagnosis of herbal-related toxic hepatitis. West. J. Med. 2002, 176, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Kao, W.F.; Hung, D.Z.; Tsai, W.J.; Lin, K.P.; Deng, J.F. Podophyllotoxin intoxication: Toxic effect of Bajiaolian in herbal therapeutics. Hum. Exp. Toxicol. 1992, 11, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Chou, S.L.; Chou, M.Y.; Kao, W.F.; Yen, D.H.; Yen, L.Y.; Huang, C.I.; Lee, C.H. Bajiaolian poisoning—A poisoning with high misdiagnostic rate. Am. J. Emerg. Med. 2010, 28, 85–89. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Ba Jiao Lian (Dysosma pleianthum). Last updated 6 June 2016. Available online: http://livertox.nih.gov/BaJiaoLian.htm (accessed on 12 June 2016).
- Estes, J.D.; Stolpman, D.; Olyaei, A.; Corless, C.L.; Ham, J.M.; Schwartz, J.M.; Orloff, S. High prevalence of potentially hepatotoxic herbal supplement use in patients with fulminant hepatic failure. Arch. Surg. 2003, 138, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.S.; Seo, E.G.; Han, C.; Chae, H.B.; Kim, S.J.; Lee, J.D.; Wang, J.H. Four cases of toxic liver injury associated with Dictamnus dasycarpus. Korean J. Hepatol. 2008, 14, 206–212, (Abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Graham-Brown, R. Toxicity of Chinese herbal remedies. Lancet 1992, 340, 673–674. [Google Scholar] [CrossRef]
- Kane, J.A.; Kane, S.P.; Jain, S. Hepatitis induced by traditional Chinese herbs: Possible toxic components. Gut 1995, 36, 146–147. [Google Scholar] [CrossRef] [PubMed]
- Vautier, G.; Spiller, R.C. Safety of complementary medicines should be monitored. BMJ 1995, 311, 633. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Kim, J.I.; Jeong, K.H.; Ko, K.H.; Ko, P.G.; Hwang, S.W.; Kim, E.M.; Kim, S.H.; Lee, H.Y.; Lee, B.S. Korean J. Hepatol. 2008, 14, 483–492, (Abstract in English, article in Korean).
- Sohn, C.H.; Cha, M.I.; Oh, B.J.; Yeo, W.H.; Lee, J.H.; Kim, W.; Lim, K.S. Liver transplantation for acute toxic hepatitis due to herbal medicines and preparations. J. Korean Soc. Clin. Toxicol. 2008, 6, 110–116, (Abstract in English, article in Korean). [Google Scholar]
- Cortez, E.; Boulger, C.; Bernard, A. Ban Tu Wan hepatotoxicity. BMJ Case Rep. 2012. [Google Scholar] [CrossRef] [PubMed]
- Yuen, M.F.; Tam, S.; Fung, J.; Wong, D.K.H.; Wong, B.C.Y.; Lai, C.L. Traditional Chinese Medicine causing hepatotoxicity in patients with chronic hepatitis B infection: A 1-year prospective study. Aliment. Pharmacol. Ther. 2006, 24, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, H.; Enomoto, M.; Yasuda, T.; Fujii, H.; Kobayashi, S.; Iwai, S.; Morikawa, H.; Takeda, T.; Tamori, A.; Sakaguchi, H.; et al. Drug-induced liver injury caused by a herbal medicine, bofu-tsu-sho-san. Nihon Shokakibyo Gakkai Zasshi 2008, 105, 1234–1239, (Abstract in English, article in Japanese). [Google Scholar] [PubMed]
- Sakamoto, S.; Takeshita, S.; Sassa, S.; Suzuki, S.; Ishikawa, Y.; Kudo, H. Effects of colestimide and/or Bofu-tsusho-san on plasma and liver lipids in mice fed a high-fat diet. In Vivo 2005, 19, 1029–1034. [Google Scholar] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Ma Huang (Ephedra sinica). Last updated 10 March 2012. Available online: http://livertox.nih.gov/Ephedra.htm (accessed on 15 May 2016).
- Hwang, S.H.; Park, J.A.; Jang, Y.S.; Lee, K.M.; Lee, D.S.; Ahn, B.M.; Lee, E.H. Case of acute cholestatic hepatitis caused by the seeds of Psoralea-corylifolia. Korean J. Hepatol. 2001, 7, 341–344, (Abstract in English, article in Korean). [Google Scholar]
- Nam, S.W.; Baek, J.T.; Lee, D.S.; Kang, S.B.; Ahn, B.M.; Chung, K.W. A case of acute cholestatic hepatitis associated with the seeds of Psoralea corylifolia (Boh-Gol-Zhee). Clin. Toxicol. 2005, 43, 589–591. [Google Scholar] [CrossRef]
- Cheung, W.I.; Tse, M.L.; Ngan, T.; Lin, J.; Lee, W.K.; Poon, W.T.; Mak, T.W.; Leung, V.K.S.; Chau, T.N. Liver injury associated with the use of Fructus Psoraleae (Bol-gol-zhee or Bu-gu-zhi) and its related propriety medicine. Clin. Toxicol. 2009, 47, 683–685. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Bahre, R. Severe hepatotoxicity by Indian Ayurvedic herbal products: A structured causality assessment. Ann. Hepatol. 2009, 8, 258–266. [Google Scholar] [PubMed]
- Lee, C.H.; Wang, J.D.; Chen, P.C. Risk of liver injury associated with Chinese herbal products containing Radix bupleuri in 639,779 patients with hepatitis B virus infection. Plos One 2011, 6, e16064. [Google Scholar] [CrossRef] [PubMed]
- Kamiyama, T.; Nouchi, T.; Kojima, S.; Murata, N.; Ikeda, T.; Sato, C. Autoimmune hepatitis triggered by administration of an herbal medicine. Am. J. Gastroenterol. 1997, 92, 703–704. [Google Scholar] [PubMed]
- Inoue, H.; Yamazaki, S.; Shimizu, M.; Uozki, H.; Goto, T.; Ohnishi, S.; Koike, K. Liver injury induced by the Japanese herbal drug kamishoyosan. Gastroenterol. Hepatol. 2011, 7, 692–695. [Google Scholar]
- Adachi, M.; Saito, H.; Kobayashi, H.; Horie, Y.; Kato, S.; Yoshioka, M.; Ishii, H. Hepatic injury in 12 patients taking the herbal loss aids Chaso and Onshido. Ann. Intern. Med. 2003, 139, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Lai, V.; Smith, A.; Thorburn, D.; Raman, V.S. Severe hepatic injury and adulterated Chinese medicines. BMJ 2006, 332, 304–305. [Google Scholar] [CrossRef] [PubMed]
- Yuen, Y.P.; Lai, C.K.; Poon, W.T.; Ng, S.W.; Chan, A.Y.W.; Mak, T.W.L. Adulteration of over-the-counter slimming products with pharmaceutical analogues—An emerging threat. Hong Kong Med. J. 2007, 13, 216–220. [Google Scholar] [PubMed]
- Kanda, T.; Yokosuka, O.; Tada, M.; Kurihara, T.; Yoshida, S.; Suzuki, Y.; Nagao, K.; Saisho, H. N-nitroso-fenfluramine hepatotoxicity resembling chronic hepatitis. J. Gastroenterol. Hepatol. 2003, 18, 999–1000. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.J.; Tsai, M.S.; Chiou, N.M.; Deng, J.F.; Chiu, N.Y. Hepatotoxicity caused by Breynia officinalis. Vet. Hum. Toxicol. 2002, 44, 87–88. [Google Scholar] [PubMed]
- Lin, T.J.; Su, C.C.; Lan, C.K.; Jiang, D.D.; Tsai, J.L.; Tsai, M.S. Acute poisonings with Breynia officinalis—An outbreak of hepatotoxicity. J. Toxicol. Clin. Toxicol. 2003, 41, 591–594. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Chi R Yun (Breynia officinalis). Last updated 25 October 2013. Available online: http://livertox.nih.gov/ChiRYun.htm (accessed on 15 May 2016).
- Davies, E.G.; Pollock, I.; Steel, H.M. Chinese herbs for eczema. Lancet 1990, 336, 117. [Google Scholar] [CrossRef]
- Sanders, D.; Kennedy, N.; McKendrick, M.W. Monitoring the safety of herbal remedies: Herbal remedies have a heterogeneous nature. Br. Med. J. 1995, 311, 1569. [Google Scholar] [CrossRef]
- Yoshida, E.M.; McLean, C.A.; Cheng, E.S.; Blanc, P.D.; Somberg, K.A.; Ferrell, L.D.; Lake, J.R. Chinese herbal medicine, fulminant hepatitis, and liver transplantation. Am. J. Gastroenterol. 1996, 91, 2647–2648. [Google Scholar] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Sho-Saiko-To, Dai-Saiko-To, Xiao Chai Hu Tang (Herbal mixtures). Last updated 5 January 2014. Available online: http://livertox.nih.gov/ShoSaikoTo_DaiSaikoTo.htm (accessed on 15 May 2016).
- Kim, S.Y.; Yim, H.J.; Ahn, J.H.; Kim, J.H.; Kim, J.N.; Yoon, I.; Kim, D.I.; Lee, H.S.; Lee, S.W.; Choi, J.H. Two cases of toxic hepatitis caused by arrowroot juice. Korean J. Hepatol. 2009, 15, 504–509, (Abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Bae, S.H.; Kim, D.H.; Bae, Y.S.; Lee, K.J.; Kim, D.W.; Yoon, J.B.; Hong, J.H.; Kim, S.H. Toxic hepatitis associated with Polygoni multiflori. Korean J. Hepatol. 2010, 16, 182–186, (Abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Gono, Y.; Odaguchi, H.; Hayasaki, T.; Suzuki, K.; Oikawa, T.; Muranushi, A.; Akahoshi, T.; Hanawa, T. Clinical analysis of cases with drug-induced liver injury for Kampo medicine. Kampo Med. 2010, 61, 828–833, (Abstract in English, article in Japanese). [Google Scholar] [CrossRef]
- Linnebur, S.A.; Rapacchietta, O.C.; Vejar, M. Hepatotoxicity associated with Chinese skullcap contained in Move Free Advanced dietary supplement: Two case reports and review of the literature. Pharmacotherapy 2010, 750, 258e–262e. [Google Scholar] [CrossRef]
- Yang, L.; Aronsohn, A.; Hart, J.; Jensen, D. Herbal hepatotoxicity from Chinese skullcap: A case report. World J. Hepatol. 2012, 4, 231–233. [Google Scholar] [CrossRef] [PubMed]
- Dhanasekaran, R.; Owens, V.; Sanchez, W. Chinese skullcap in Move Free arthritis supplement causes drug induced liver injury and pulmonary infiltrates. Case Rep. Hepatol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Papafragkakis, C.; Ona, M.A.; Reddy, M.; Anand, S. Acute hepatitis after ingestion of a preparation of Chinese skullcap and black catechu for joint pain. Case Rep. Hepatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Choi, H.S.; Yun, T.J.; Lee, K.G.; Seo, Y.S.; Yeon, J.E.; Byun, K.S.; Um, S.H.; Kim, C.D.; Ryu, H.S. A case of acute cholestatic hepatitis induced by Corydalis speciosa Max. Korean J. Hepatol. 2009, 15, 517–523, (Abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Jorge, O.A.; Jorge, A.D. Hepatotoxicity associated with the ingestion of Centella asiatica. Rev. Esp. Enferm. Dig. 2005, 97, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Ryu, S.L.; Shim, J.W.; Kim, D.S.; Shim, J.Y.; Park, M.S.; Jung, H.L. A pediatric case of toxic hepatitis induced by Hovenia dulcis. Pediatr. Gastroenterol. Hepatol. Nutr. 2012, 15, 111–116. [Google Scholar] [CrossRef]
- Woolf, G.M.; Petrovic, L.M.; Rojter, S.E.; Wainwright, S.; Villamil, F.G.; Katkov, W.N.; Michieletti, P.; Wanless, I.R.; Stermitz, F.R.; Beck, J.J.; et al. Acute hepatitis associated with the Chinese herbal product Jin Bu Huan. Ann. Intern. Med. 1994, 121, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.S.; Feldhaus, K.; Dart, R.C.; Stermitz, F.R.; Beck, J.J. The clinical spectrum of Jin Bu Huan toxicity. Arch. Intern. Med. 1996, 156, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Divinsky, M. Case report: Jin Bu Huan—Not so benign herbal medicine. Can. Fam. Physician 2002, 48, 1640–1642. [Google Scholar] [PubMed]
- Picciotti, A.; Campo, N.; Brizzolara, R.; Giusto, R.; Guido, G.; Sinelli, N.; Lapertosa, G.; Celle, G. Chronic hepatitis induced by Jin Bu Huan. J. Hepatol. 1998, 28, 165–167. [Google Scholar] [CrossRef]
- National Institutes of Health (NIH) and LiverTox: Drug record. Jin Bu Huan (Lycopodium serratum). Last updated 12 May 2013. Available online: http://livertox.nih.gov/JinBuHuan.htm (accessed on 15 May 2016).
- Wu, G.L.; Yu, G.Y.; Chen, J. Clinical analysis of hepatic veno-occlusive disease induced by Sedum aizoon. Zhongguo Zhong Yao Za Zhi 2008, 33, 2402–2404, (Abstract in English, article in Chinese). [Google Scholar] [PubMed]
- Lin, G.; Wang, J.Y.; Li, N.; Li, M.; Gao, H.; Ji, Y.; Zhang, F.; Wang, H.; Zhou, Y.; Ye, Y.; et al. Hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. J. Hepatol. 2011, 54, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.S.; Xiao, S.S.; He, J.F. Analysis of alkaloids in Sedum aizoon and establishment of hepatic veno-occlusive model in mice. Chin. J. Integr. Trad. West. Med. Dig. 2006, 14, 311–313. [Google Scholar]
- Gao, H.; Li, N.; Wang, J.Y.; Zhang, S.C.; Lin, G. Definitive diagnosis of hepatic sinusoidal obstruction syndrome induced by pyrrolizidine alkaloids. J. Dig. Dis. 2012, 13, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Gao, H. Tusanqi and hepatic sinusoidal obstruction syndrome. Dig. Dis. 2014, 15, 105–107. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.F.; Gao, Y.; Yang, M.; Yu, C.H.; Gu, Z.Y.; Chen, W.X. Hepatic veno-occlusive disease induced by Gynura segetum: Report of two cases. Hepatobiliary Pancreat. Dis. Int. 2006, 5, 406–408. [Google Scholar] [PubMed]
- Kumana, C.R.; Ng, M.; Lin, H.J.; Ko, W.; Wu, P.C.; Todd, D. Herbal tea induced hepatic veno-occlusive disease: Quantification of toxic alkaloid exposure in adults. Gut 1985, 26, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Kumana, C.R.; Ng, M.; Lin, H.J.; Ko, W.; Wu, P.C.; Todd, D. Hepatic veno-occlusive disease due to toxic alkaloid in herbal tea. Lancet 1983, 2, 1360–1361. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Edgar, J.A.; Smith, L.W.; Kumana, C.R.; Lin, H.J. Heliotropium lasiocarpum Fisch and Mey identified as cause of veno-occlusive disease due to herbal tea. Lancet 1986, 1, 978. [Google Scholar] [CrossRef]
- Chen, M.Y.; Cai, J.T.; Du, Q. Hepatic veno-occlusive disease associated with the use of Gynura segetum. Eur. J. Intern. Med. 2007, 18, 609. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Liang, X.S.; Li, C.Z. Sinusoidal obstruction syndrome associated with the ingestion of gynura root. Clin. Toxicol. 2010, 48, 962–964. [Google Scholar] [CrossRef] [PubMed]
- Fenkel, J.M.; Navarro, V.J. Review: Herbal and dietary supplement-induced liver injury. Gastroenterol. Hepatol. 2011, 7, 695–696. [Google Scholar]
- Duenas Sadornil, C.; Fabregas Piugtio, S.; Durandez, R. Hepatotoxicity due to Camelia sinensis. Med. Clin. (Barc.) 2004, 122, 677–678. (In Spanish) [Google Scholar]
- Garcia-Moran, S.; Saez-Royuela, F.; Gento, E.; Lopez Morante, A.; Arias, L. Acute hepatitis associated with Camellia tea and Orthosiphon stamineus ingestion. Gastroenterol. Hepatol. 2004, 27, 559–560. [Google Scholar] [CrossRef]
- Peyrin-Biroulet, L.; Petitpain, N.; Kalt, P.; Ancel, D.; Petit-Laurent, F.; Trechot, P.; Barraud, H.; Bronowicki, J.P. Probable hepatotoxicity from epigallocatecol gallate used for phytotherapy. Gastroenterol. Clin. Biol. 2004, 28, 404–406. [Google Scholar] [CrossRef]
- Jimemez-Saenz, M.; del Martinez-Sanchez, M.C. Acute hepatitis associated with the use of green tea infusions. J. Hepatol. 2006, 44, 616–617. [Google Scholar] [CrossRef] [PubMed]
- Bonkovsky, H.L. Hepatotoxicity associated with supplements containing Chinese green tea (Camellia sinensis). Ann. Intern. Med. 2006, 144, 68–71, Erratum in: Ann. Intern. Med. 2006, 144, 380. [Google Scholar] [CrossRef]
- García-Cortés, M.; Borraz, Y.; Lucena, M.I.; Peláez, G.; Salmerón, J.; Diago, M.; Martínez-Sierra, M.C.; Navarro, J.M.; Planas, R.; Soria, M.J.; et al. Liver injury induced by “natural remedies”: An analysis of cases submitted to the Spanish Liver Toxicity Registry. Rev. Esp. Enferm. Dig. 2008, 100, 688–695. [Google Scholar] [PubMed]
- Sarma, D.N.; Barrett, M.L.; Chavez, M.L.; Gardiner, P.; Ko, R.; Mahady, G.B.; Marles, R.J.; Pellicore, L.S.; Giancaspro, G.I.; Low Dog, T. Safety of green tea extract: A systematic review by the US Pharmacopeia. Drug Saf. 2008, 31, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Mazzanti, G.; Menniti-Ippolito, F.; Moro, P.A.; Cassetti, F.; Raschetti, R.; Santuccio, C.; Mastrangelo, S. Hepatotoxicity from green tea: A review of the literature and two unpublished cases. Eur. J. Clin. Pharmacol. 2009, 65, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Navarro, V.J.; Bonkovsky, H.L.; Hwang, S.I.; Vega, M.; Barnhart, H.; Serrano, J. Catechins in dietary supplements and hepatotoxicity. Dig. Dis. Sci. 2013, 58, 2682–2690. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Zhang, L.; Melzer, L.; Schulze, J.; Eickhoff, A. Green tea extract and the risk of drug-induced liver injury. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1663–1676. [Google Scholar] [CrossRef] [PubMed]
- Mazzanti, G.; Di Sotto, A.; Vitalone, A. Hepatotoxicity of green tea: An update. Arch. Toxicol. 2015, 89, 1175–1191. [Google Scholar] [CrossRef] [PubMed]
- Huseini, A.B.; Bekjarovski, N. Hepatotoxicity after 51 days use of green tea. Sch. J. Med. Case Rep. 2016, 4, 80–82. [Google Scholar]
- Rossi, S.; Navarro, V.J. Herbs and liver injury: A clinical perspective. Clin. Gastroenterol. Hepatol. 2014, 12, 69–76. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Green tea (Camellia sinensis). Last updated 17 March 2014. Available online: http://livertox.nlm.nih.gov/GreenTea.htm (accessed on 15 May 2016).
- Nadir, A.; Agrawal, S.; King, P.D.; Marshall, J.B. Acute hepatitis associated with the use of a Chinese herbal product, ma-huang. Am. J. Gastroenterol. 1996, 91, 1436–1438. [Google Scholar] [PubMed]
- Reuben, A.; Koch, D.G.; Lee, W.M.; the Acute Liver Failure Study Group. Drug-induced acute liver failure: Results of a U.S. multicenter, prospective study. Hepatology 2010, 52, 2065–2076. [Google Scholar] [CrossRef] [PubMed]
- Borum, M.L. Fulminant exacerbation of autoimmune hepatitis after the use of Ma Huang. Am. J. Gastroenterol. 2001, 96, 1654–1655. [Google Scholar] [CrossRef] [PubMed]
- Skoulidis, F.; Alexander, G.J.; Davies, S.E. Ma huang associated acute liver failure requiring liver transplantation. Eur. J. Gastroenterol. Hepatol. 2005, 17, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Joshi, D.; Cross, T.J.S.; Wong, V.S. Acute drug induced hepatitis secondary to a weight loss product purchased over the internet. Nutr. J. 2007, 6. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Shou Wu Pian (Polygonum multiflorum). Last updated 24 March 2016. Available at: http://livertox.nih.gov/ShouWuPian.htm (accessed on 12 April 2016).
- Cárdenas, A.; Restrepo, J.C.; Sierra, F.; Correa, G. Acute hepatitis due to shen-min: A herbal product derived from Polygonum multiflorum. J. Clin. Gastroenterol. 2006, 40, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Panis, B.; Wong, D.R.; Hooymans, P.M.; de Smet, P.A.G.M.; Rosias, P.R. Recurrent toxic hepatitis in a Caucasian girl related to the use of Shou-Wu-Pian, a Chinese herbal preparation. J. Pediat. Gastroenterol. Nutr. 2005, 41, 256–258. [Google Scholar] [CrossRef]
- Jung, K.A.; Min, H.J.; Yoo, S.S.; Kim, H.J.; Choi, S.N.; Ha, C.Y.; Kim, H.J.; Kim, T.H.; Jung, W.T.; Lee, O.J.; et al. Drug-induced liver injury: Twenty five cases of acute hepatitis following ingestion of Polygonum multiflorum Thun. Gut Liver 2011, 5, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Valente, G.; Sanges, M.; Campione, S.; Bellevicine, C.; de Franchis, G.; Sollazzo, R.; Matera, D.; Cimino, L.; Vecchione, R.; D’Arienzo, A. Herbal hepatotoxicity: A case of difficult interpretation. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 865–870. [Google Scholar] [CrossRef]
- But, P.P.H.; Tomlinson, B.; Lee, K.L. Hepatitis related to the Chinese medicine Shou-wu-pian manufactured from Polygonum multiflorum. Vet. Hum. Toxicol. 1996, 38, 280–282. [Google Scholar] [PubMed]
- Park, G.J.H.; Mann, S.P.; Ngu, M.C. Acute hepatitis induced by Shou-Wu-Pian, a herbal product derived from Polygonum multiflorum. J. Gastroenterol. Hepatol. 2001, 16, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Mazzanti, G.; Battinelli, L.; Daniele, C.; Mastroianni, C.M.; Lichtner, M.; Coletta, S.; Costantini, S. New case of acute hepatitis following the consumption of Shou Wu Pian, a Chinese herbal product derived from Polygonum multiflorum. Ann. Intern. Med. 2004, 140, W30. [Google Scholar] [CrossRef] [PubMed]
- Laird, A.R.; Ramchandani, N.; deGoma, E.M.; Avula, B.; Khan, I.A.; Gesundheit, N. Acute hepatitis associated with the use of an herbal supplement (Polygonum multiflorum) mimicking iron-overload syndrome. Clin. Gastroenterol. 2008, 42, 861–862. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, M.; Kasajima, S.; Nakamura, Y.; Shouzushima, M.; Nagatani, N.; Takinishi, A.; Taguchi, A.; Fujita, M.; Niimi, A.; Misaka, R.; et al. Toxic hepatitis induced by Show-Wu-Pian, a Chinese herbal preparation. Intern. Med. 2010, 49, 1537–1540. [Google Scholar] [CrossRef] [PubMed]
- Banarova, A.; Koller, T.; Payer, J. Toxic hepatitis induced by Polygonum multiflorum. Vnitr. Lek. 2012, 58, 958–962, (Abstract in English, article in Slovak). [Google Scholar] [PubMed]
- Zhang, L.; Yang, X.; Sun, Z.; Qu, Y. Retrospective study of adverse events of Polygonum multiflorum and risks control. Zhongguo Zhong Yao Za Zhi 2009, 34, 1724–1729, (Abstract in English, article in Chinese). [Google Scholar] [PubMed]
- Dong, H.; Slain, D.; Cheng, J.; Ma, W.; Liang, W. Eighteen cases of liver injury following ingestion of Polygonum multiflorum. Complement. Ther. Med. 2014, 22, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Chen, J.; Ren, J.; Li, Y.; Zhai, J.; Mu, W.; Zhang, L.; Zheng, W.; Tian, G.; Shang, H. Liver damage associated with Polygonum multiflorum Thunb: A systematic review of case reports and case series. Evid. Based Complement. Altern. Med. 2015. [Google Scholar] [CrossRef]
- Wang, J.; Ma, Z.; Niu, M.; Zhu, Y.; Liang, Q.; Zhao, Y.; Song, J.; Bai, Z.; Zhang, Y.; Zhang, P.; et al. Evidence chain-based causality identification in herb-induced liver injury: Exemplification of a well-known liver-restorative herb Polygonum multiflorum. Front. Med. 2015, 9, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Qu, C.; He, Q.; Chen, W.; Zhang, X.; Liu, X.; Liu, Y.; Tang, Y. Acute hepatitis induced by a Chinese herbal product Qibao Meiran Wan: A case study. Int. J. Clin. Exp. Med. 2015, 8, 11624–11627. [Google Scholar] [PubMed]
- Aiba, T.; Takahashi, T.; Suzuki, K.; Okoshi, S.; Nomoto, M.; Uno, K.; Aoyagi, Y. Liver injury induced by a Japanese herbal medicine, sairei-to (TJ-114, Bupleurum and Hoelen combination, Chai-Ling-Tang). J. Gastroenterol. Hepatol. 2007, 22, 762–763. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Yashiro, S.; Gamo, Y.; Watanabe, K.; Hoshino, T.; Oikawa, T.; Hanawa, T. Discrepancy between clinical course and drug-induced lymphocyte stimulation tests in a case of saireito-induced liver injury accompanied by Sjögren syndrome. J. Altern. Complement. Med. 2010, 16, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D.; Vial, T.; Pauwels, A.; Castot, A.; Biour, M.; David, M.; Michel, H. Hepatitis after germander (Teucrium chamaedrys) administration: another instance of herbal medicine hepatotoxicity. Ann. Intern. Med. 1992, 117, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Castot, A.; Larrey, D. Hepatites observées au cours d’un traitement par un medicament ou d’une tisane contenant de la germander petit-chêne. Bilan des 26 cas rapportés aux centres régionaux de pharmacovigilance. Gastroenterol. Clin. Biol. 1992, 16, 916–922. (In French) [Google Scholar] [PubMed]
- Mostefa-Kara, N.; Pauwels, A.; Pines, E.; Biour, M.; Levy, V.G. Fatal hepatitis after herbal tea (letter). Lancet 1992, 340, 674. [Google Scholar] [CrossRef]
- Diaz, D.; Ferroudji, S.; Heran, B.; Barneon, G.; Larrey, D.; Michel, H. Hepatite aiguë à la germander petit-chêne. Gastroenterol. Clin. Biol. 1992, 16, 1006–1007. (In French) [Google Scholar] [PubMed]
- Ben Yahia, M.; Mavier, P.; Metreau, J.M.; Zafrani, E.S.; Fabre, M.; Gatineau-Saillant, G.; Dhumeaux, D.; Mallat, A. Hepatite chronic active et cirrhose induites par la germander petit-chêne. Gastroenterol. Clin. Biol. 1993, 17, 959–962. (In French) [Google Scholar] [PubMed]
- Dao, T.; Peytier, A.; Galateau, F.; Valla, A. Chronic hepatitis due to germander. Gastroenterol. Clin. Biol. 1993, 17, 614–615. [Google Scholar]
- LiverTox. Germander. Last update 24 March 2014. Available online: http://livertox.nih.gov/Germander.htm (accessed on 5 May 2016).
- Mattéi, A.; Rucay, P.; Samuel, D.; Feray, C.; Michel, R.; Bismuth, H. Liver transplantation for acute liver failure after herbal medicine (Teucrium polium) administration (letter). J. Hepatol. 1995, 22, 597. [Google Scholar] [CrossRef]
- Starakis, I.; Siagris, D.; Leonidou, L.; Mazakopakis, E.; Tsamandas, A.; Karatza, C. Hepatitis caused by the herbal remedyTeucrium polium L. Eur. J. Gastroenterol. Hepatol. 2006, 18, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.M.; Heywood, E.; Pillai, A.; Ahn, J. Hepatotoxicity associated with the use of White Flood, a nutritional supplement. Pract. Gastroenterol. 2012, 36, 45–48. [Google Scholar]
- Itoh, S.; Marutani, K.; Nishijima, T.; Matsuo, S.; Itabashi, M. Liver injuries induced by herbal medicine, Syo-saiko-to (xiao-chai-hu-tang). Dig. Dis. Sci. 1995, 40, 1845–1848. [Google Scholar] [CrossRef] [PubMed]
- Hsu, L.M.; Huang, Y.S.; Tsay, S.H.; Chang, F.Y.; Lee, S.D. Acute hepatitis induced by Chinese hepatoprotective herb xiao-chai-hu-tang. J. Chin. Med. Assoc. 2006, 69, 86–88. [Google Scholar] [CrossRef]
- Teschke, R.; Zhang, L.; Long, H.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Genthner, A.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Traditional Chinese Medicine and herbal hepatotoxicity: A tabular compilation of reported cases. Ann. Hepatol. 2015, 14, 7–19. [Google Scholar] [PubMed]
- Ma, X.; Peng, J.H.; Hu, Y.Y. Chinese herbal medicine-induced liver injury. J. Clin. Transl. Hepatol. 2014, 2, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Suk, K.T.; Kim, D.J.; Kim, C.H.; Park, S.H.; Yoon, J.H.; Kim, Y.S.; Baik, G.H.; Kim, J.B.; Kweon, Y.O.; Kim, B.I.; et al. A prospective nationwide study of drug-induced liver injury in Korea. Am. J. Gastroenterol. 2012, 107, 1380–1387. [Google Scholar] [CrossRef] [PubMed]
- Douros, A.; Bronder, E.; Andersohn, F.; Klimpel, A.; Kreutz, R.; Garbe, E.; Bolbrinker, J. Herb-induced liver injury in the Berlin Case-Control Surveillance Study. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, L.; Liao, Z.; He, X.; Zhou Y. Guo, H. Epidemiology of drug-induced liver injury in China: A systematic analysis of the Chinese literature including 21,789 patients. Eur. Gastroenterol. Hepatol. 2013, 25, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Schulze, J. Review article: Herbal hepatotoxicity—An update on Traditional Chinese Medicine preparations. Aliment. Pharmacol. Ther. 2014, 40, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Hao, K.; Yu, Y.; He, C.; Wang, M.; Wang, S.; Li, X. RUCAM scale-based diagnosis, clinical features and prognosis of 140 cases of drug-induced liver injury. Zhonghua Gan Zang Bing Za Zhi 2014, 22, 938–941, (Abstract in English, article in Chinese). [Google Scholar] [PubMed]
- Zhu, Y.; Liu, S.H.; Wang, J.B.; Song, H.B.; Li, Y.G.; He, T.T.; Ma, X.; Wang, Z.X.; Wang, L.P.; Zhou, K.; et al. Clinical analysis of drug-induced liver injury caused by Polygonum multiflorum and its preparations. Zhongguo Zhong Xi Yi Jie He Za Zhi 2015, 35, 1442–1447, (Abstract in English, article in Chinese). [Google Scholar] [PubMed]
- Zhu, Y.; Niu, M.; Chen, J.; Zou, Z.S.; Ma, Z.J.; Liu, S.H.; Wang, R.L.; He, T.T.; Song, H.B.; Wang, Z.X.; et al. Comparison between Chinese herbal medicine and Western medicine-induced liver injury of 1985 patients. J. Gastroenterol. Hepatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, Y.G.; Wang, J.B.; Liu, S.H.; Wang, L.F.; Zhao, Y.L.; Bai, Y.F.; Wang, Z.X.; Li, J.Y.; Xiao, X.H. Causes, features, and outcomes of drug-induced liver injury in 69 children from China. Gut Liver 2015, 9, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, Y.G.; Wang, Y.; Wang, L.P.; Wang, J.B.; Wang, R.L.; Wang, L.F.; Meng, Y.K.; Wang, Z.X.; Xiao, X.H. Analysis of clinical characteristics in 595 patients with herb-induced liver injury. Zhongguo Zhong Xi Yi Jie He Za Zhi CJITWM 2016, 36, 43–47. [Google Scholar]
- Patwardhan, B.; Warude, D.; Pushpangadan Bhatt, N. Ayurveda and traditional Chinese medicine: A comparative overview. Evid. Based Complement. Altern. Med. 2005, 2, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Sarges, P.; Steinberg, J.M.; Lewis, J.H. Drug-induced liver injury: Highlights from a review of the 2015 literature. Drug Saf. 2016, 39, 561–575. [Google Scholar] [CrossRef] [PubMed]
- Urban, T.J.; Daly, A.K.; Aithal, G.P. Genetic basis of drug-induced liver injury: Present and future. Semin. Liver Dis. 2014, 34, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Ai, C.Z.; Liu, Y.; Zhang, Y.Y.; Jiang, M.; Fan, X.R.; Lv, A.P.; Yang, L. Interactions between phytochemicals from Chinese Medicines and human cytochrome P450 enzymes. Curr. Drug Metab. 2012, 13, 599–614. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.; Yoon, I.S. Pharmacogenetic interactions of herbs with cytochrome P450 and p-glycoprotein. Evid. Based Complement. Altern. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.F.; Zhang, X.G.; Jia, H.Y. CYP1A2 polymorphism in Chinese patients with acute liver injury induced by Polygonum multiflorum. Genet. Mol. Res. 2014, 13, 5637–5643. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, H.J. Hepatotoxicity, 2nd ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 1999. [Google Scholar]
- Teschke, R.; Danan, G. Diagnosis and management of drug-induced liver injury (DILI) in patients with pre-existing liver disease. Drug Saf. 2016, 39, 729–744. [Google Scholar] [CrossRef] [PubMed]
- Loeper, J.; Descatoire, V.; Letteron, P.; Moulis, C.; Degott, C.; Dansette, P.; Fau, D.; Pessayre, D. Hepatotoxicity of germander in mice. Gastroenterology 1994, 106, 464–472. [Google Scholar] [CrossRef]
- Fau, D.; Lekehal, M.; Farrell, G.; Moreau, A.; Moulis, C.; Feldman, G.; Haouzi, D.; Pessayre, D. Diterpenoids from germander, an herbal medicine, induce apoptosis in isolated rat hepatocytes. Gastroenterology 1997, 117, 1334–1336. [Google Scholar] [CrossRef]
- Larrey, D.; Faure, S. Herbal medicine hepatotoxicity: A new step with development of specific biomarkers. J. Hepatol. 2011, 54, 599–601. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Borlak, J.; Tong, W. High lipophilicity and high daily dose of oral medications are associated with significant risk for drug-induced liver injury. Hepatology 2013, 58, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Geng, M.; Zhang, J.; Wang, B.; Ilic, K.; Tong, W. High daily dose and being a substrate of cytochrome P450 enzymes are two important predictors of drug induced liver injury. Drug Metab. Dis. 2014, 42, 744–750. [Google Scholar] [CrossRef] [PubMed]
- Lammert, C.; Einarsson, S.; Saha, C.; Niklasson, A.; Bjornsson, E.; Chalasani, N. Relationship between daily dose of oral medications and idiosyncratic drug-induced liver injury: Search for signals. Hepatology 2008, 47, 2003–2009. [Google Scholar] [CrossRef] [PubMed]
- Lammert, C.; Bjornsson, E.; Niklasson, A.; Chalasani, N. Oral medications with significant hepatic metabolism at higher risk for hepatic adverse effects. Hepatology 2010, 51, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Suzuki, A.; Borlak, J.; Andrade, R.J.; Lucena, M.I. Drug induced liver injury: Interactions between drug properties and host factors. J. Hepatol. 2015, 63, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Andrade, R.J. Drug-induced liver injury: Expanding our knowledge by enlarging population analysis and appropriate prospective causality assessment. Gastroenterology 2015, 148, 1271–1273. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Herbal hepatotoxicity: A tabular compilation of reported cases. Liver Int. 2012, 32, 1543–1556. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Eickhoff, A.; Frenzel, C.; Wolff, A.; Schulze, J. Clinical and causality assessment in herbal hepatotoxicity. Expert Opin. Drug Saf. 2013, 12, 330–366. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Hennermann, K.H. Kava hepatotoxicity: A clinical survey and critical analysis of 26 suspected cases. Eur. J. Gastroenterol. Hepatol. 2008, 20, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Pantano, F.; Tittarelli, R.; Mannochi, G.; Zaami, S.; Ricci, S.; Giorgetti, R.; Terranova, D.; Busardò, F.P.; Marinelli, E. Hepatotoxicity induced by “the 3Ks”: Kava, Kratom and Khat. In Int. J. Mol. Sci.; 2016; Volume 17, p. 580. [Google Scholar]
- Teschke, R. Kava hepatotoxicity—A clinical review. Ann. Hepatol. 2010, 9, 251–265. [Google Scholar] [PubMed]
- Teschke, R. Kava hepatotoxicity: Pathogenetic aspects and prospective considerations. Liver Int. 2010, 30, 1270–1279. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Glass, X.; Schulze, J. Herbal hepatotoxicity by Greater Celandine (Chelidonium majus): Causality assessment of 22 spontaneous reports. Regul. Toxicol. Pharmacol. 2011, 61, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Glass, X.; Schulze, J.; Eickhoff, A. Greater Celandine hepatotoxicity: A clinical review. Ann. Hepatol. 2012, 11, 838–848. [Google Scholar] [PubMed]
- Teschke, R.; Glass, X.; Schulze, J.; Eickhoff, A. Suspected Greater Celandine hepatotoxicity: Liver specific causality evaluation of published case reports from Europe. Eur. J. Gastroenterol. Hepatol. 2012, 24, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Z.; Liu, X.; Pan, R. Review on the toxic effects of radix Bupleuri. Curr. Opin. Complement. Altern. Med. 2014, 1, 3–7. [Google Scholar]
- Wu, X.; Chen, X.; Huang, Q.; Fang, D.; Li, G.; Zhang, G. Toxicity of raw and processed roots of Polygonum multiflorum. Fitoterapia 2012, 83, 469–475. [Google Scholar] [CrossRef] [PubMed]
- National Pharmacopoeia Committee. Chinese Pharmacopoeia; China Medical Science Press: Beijing, China, 2010. [Google Scholar]
- Frank, J.; George, T.W.; Lodge, J.K.; Rodriguez-Mateos, A.M.; Spencer, J.P.E.; Minihane, A.M.; Rimbach, G. Daily consumption of an aqueous green tea extract supplement does not impair liver function or alter cardiovascular disease risk biomarkers in healthy men. J. Nutr. 2009, 139, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Bun, S.S.; Bun, H.; Guédon, D.; Rosier, C.; Ollivier, E. Effect of green tea extracts on liver functions in Wistar rats. Food Chem. Toxicol. 2006, 44, 1108–1113. [Google Scholar] [CrossRef] [PubMed]
- Galati, G.; Lin, A.; Sultan, A.M.; O’Brien, P.J. Cellular and in vivo hepatotoxicity caused by green tea phenolic acids and catechins. Free Radic. Biol. Med. 2006, 15, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.C.; Ramot, Y.; Malarkey, D.E.; Blackshear, P.; Kissling, G.E.; Travlos, G.; Nyska, A. Fourteen-week toxicity study of green tea extract in rats and mice. Toxicol. Pathol. 2010, 38, 1070–1084. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.C. Tea. The Drink That Changed the World; Tuttle Publishing: North Clarendon, VT, USA, 2007. [Google Scholar]
- Chow, H.H.S.; Hakim, I.A.; Vining, D.R.; Crowell, J.A.; Cordova, C.A.; Chew, W.M.; Xu, M.J.; Hsu, C.H.; Ranger-Moore, J.; Alberts, D.S. Effects of repeated green tea catechins administration on human cytochrome P450 activity. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2473–2476. [Google Scholar] [CrossRef] [PubMed]
- Chacko, S.M.; Thambi, P.T.; Kuttan, R.; Nishigaki, I. Beneficial effects of green tea: A literature review. Chin. Med. 2010, 5, 13. Available online: http://www.cmjournal.org/content/5/1/13 (accessed on 22 May 2016). [Google Scholar] [CrossRef] [PubMed]
- Gavilan, J.C.; Bermudez, F.J.; Salgado, F.; Pena, D. Phytotherapy and hepatitis. Rev. Clin. Esp. 1999, 199, 693–694. [Google Scholar] [PubMed]
- Patel, S.S.; Beer, S.; Kearney, D.L.; Phillips, G.; Carter, B.A. Green tea extract: A potential cause of acute liver failure. World J. Gastroenterol. 2013, 19, 5174–5177. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.P.; Xia, Q.; Lin, G.; Chou, M.W. Pyrrolizidine alkaloids—Genotoxicity, metabolism enzymes, metabolic activation, and mechanisms. Drug Metab. Rev. 2004, 36, 1–55. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.P.; Yang, Y.C.; Xia, Q.; Chou, M.W.; Cui, M.M.; Lin, G. Pyrrolizidine alkaloids—Tumorigenic components in Chinese herbal medicines and dietary supplements. J. Food Drug Anal. 2002, 10, 198–211. [Google Scholar]
- Roulet, M.; Laurini, R.; Rivier, L.; Calame, A. Hepatic veno-occlusive disease in newborn infant of a woman drinking herbal tea. J. Pediatr. 1988, 11, 433–436. [Google Scholar] [CrossRef]
- Committee on Herbal Medicinal Products (HMPC). Public statement on the use of herbal medicinal products containing toxic, unsaturated pyrrolizidine alkaloids (PAs). 2014. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Public_statement/2014/12/WC50017955.pdf (accessed on 15 May 2016).
- Allgaier, C.; Franz, S. Risk assessment on the use of herbal medicinal products containing pyrrolizidine alkaloids. Regul. Toxicol. Pharmacol. 2015, 73, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Avula, B.; Wang, Y.H.; Wang, M.; Smillie, T.J.; Khan, I.A. Simultaneous determination of sesquiterpenes and pyrrolizidine alkaloids from the rhizomes of Petasites hybridus (L.) G.M. et Sch. and dietary supplements using UPLC-UV and HPLC-TOF-MS methods. J. Pharm. Biomed. Anal. 2012, 70, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Schenk, A.; Drewe, J.; Siewert, B. Determination of pyrrolizidine alkaloids in Petasites hybridus leaf native CO2-extract by using UHPLC-HRMS. Planta Med. 2014, 80. [Google Scholar] [CrossRef]
- Gao, H.; Ruan, J.Q.; Chen, J.; Li, N.; Ke, C.Q.; Ye, Y.; Lin, G.; Wang, J.Y. Blood pyrrole-protein adducts as a diagnostic and prognostic index in pyrrolizidine alkaloid-hepatic sinusoidal obstruction syndrome. Drug Des. Develop. Ther. 2015, 9, 4861–4868. [Google Scholar] [CrossRef] [PubMed]
- DeLeve, L.D.; Valla, D.C.; Garcia-Tsao, G. Vascular disorders of the liver. Hepatology 2009, 49, 1729–1764. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Kim, H.W.; Lee, H.Y.; Son, C.G. Systematic review of herb-induced liver injury in Korea. Food Chem. Toxicol. 2015, 84, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.P.; Hayashi, P.H.; Bonkovsky, H.L.; Navarro, V.J.; Lee, W.M.; Fontana, R.J. ACG Clinical guideline: The diagnosis and management of idiosyncratic drug-induced liver injury. Am. J. Gastroenterol. 2014, 109, 950–966. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, P.H.; Fontana, R.J.; Chalasani, N.P.; Stolz, A.A.; Talwalkar, J.A.; Navarro, V.J.; Lee, W.M.; Davern, T.J.; Kleiner, D.E.; Gu, J.; et al. For the US Drug-Induced Liver Injury Network Investigators. Under-reporting of poor adherence to monitoring guidelines for severe cases of Isoniazid hepatotoxicity. Clin. Gastroenterol. Hepatol. 2015, 13, 1676–1682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teschke, R.; Schulze, J.; Eickhoff, A.; Wolff, A.; Frenzel, C. Mysterious Hawaii liver disease case—Naproxen overdose as cause rather than OxyELITE Pro? J. Liver Clin. Res. 2015, 2, 1013. [Google Scholar]
- Teschke, R.; Schwarzenboeck, A.; Frenzel, C.; Schulze, J.; Eickhoff, A.; Wolff, A. The mystery of the Hawaii liver disease cluster in summer 2013: A pragmatic and clinical approach to solve the problem. Ann. Hepatol. 2016, 15, 91–119. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Ruan, J.; Fu, P.P.; Lin, G. Cytotoxicity of pyrrolizidine alkaloid in human hepatic parenchymal and sinusoidal endothelial cells: Firm evidence for the reactive metabolites mediated pyrrolizidine alkaloid-induced hepatotoxicity. Chem. Biol. Interact. 2016, 243, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Yang, M.; Fu, P.; Ye, Y.; Lin, G. Metabolic activation of pyrrolizidine alkaloids: Insights into the structural and enzymatic basis. Chem. Res. Toxicol. 2014, 27. [Google Scholar] [CrossRef] [PubMed]
- DeLeve, L.D.; Wang, X.; Kuhlenkamp, J.F.; Kaplowitz, N. Toxicity of azathioprine and monocrotoline in murine sinusoidal endothelial cells and hepatocytes: The role of glutathione and relevance to hepatic venoocclusive disease. Hepatology 1996, 23, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Chojkier, M. Hepatic sinusoidal-obstruction syndrome: Toxicity of pyrrolizidine alkaloids. J. Hepatol. 2003, 39, 437–446. [Google Scholar] [CrossRef]
- Braet, F.; Wisse, E. Structural and functional aspects of liver sinusoidal endothelial cell fenestrae: A review. Comp. Hepatol. 2002, 1, 1. Available online: http://comparative-hepatology.biomedcentral.com/articles/10.1186/1476-5926-1-1 (accessed on 12 June 2016). [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeLeve, L.D. Vascular liver disease and the liver sinusoidal endothelial cell. In Vascular Liver Disease: Mechanisms and Management; DeLeve, L.D., Garcia-Tsao, G., Eds.; Springer+Buisiness Media: Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Senior, J.R. New biomarker for drug induced liver injury: Are they really better? What do they diagnose? Liver Int. 2014, 34, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.H. The art and science of diagnosing and managing drug-induced liver injury in 2015 and beyond. Clin. Gastroenterol. Hepatol. 2015, 13. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Jaeschke, H. MicroRNAs as signaling mediators and biomarkers of drug- and chemical-induced liver injury. J. Clin. Med. 2015, 4, 1063–1078. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Ji, C.; Lu, X.; Tong, W.; Fan, X.; Gao, Y. Integrated expression profiles of mRNA and microRNA in the liver of Fructus Meliae Toosendan water extract injured mice. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Enache, L.S.; Enache, E.L.; Ramière, C.; Diaz, O.; Bancu, L.; Sin, A.; André, P. Circulating RNA molecules as biomarkers in liver disease. Int. J. Mol. Sci. 2014, 15, 17644–17666. [Google Scholar] [CrossRef] [PubMed]
- Li, L.M.; Wang, D.; Zen, K. MicroRNAs in drug-induced liver injury. Clin. Transl. Hepatol. 2014, 2, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Aithal, G.P. Pharmacogenetic testing in idiosyncratic drug induced liver injury: Current role in clinical practice. Liver Int. 2015, 35, 1801–1808. [Google Scholar] [CrossRef] [PubMed]
- Daly, A.K.; Donaldson, P.T.; Bhatnagar, P.; Shen, Y.; Pe’er, I.; Floratos, A.; Daly, M.J.; Goldstein, D.B.; John, S.; Nelson, M.R.; Graham, J.; et al. For the DILIGEN Study & International SAE Consortium. HLA-B*5701 genotype is a major determinant of drug-induced liver injury due to flucloxacillin. Nat. Genet. 2009, 41, 816–819. [Google Scholar]
- Lewis, P.J.S.; Dear, J.; Platt, V.; Simpson, K.J.; Craig, D.G.N.; Antoine, D.J.; French, N.S.; Dhaun, N.; Webb, D.J.; Costello, E.M.; et al. Circulating microRNAs as potential markers of human drug-induced liver injury. Hepatology 2011, 54, 1767–1776. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Salminen, W.F.; Schnackenberg, L.K. Current and emerging biomarkers of hepatotoxicity. Curr. Biomark. Find 2012, 20, 43–55. [Google Scholar]
- Thulin, P.; Nordahl, G.; Gry, M.; Yimer, G.; Aklillu, E.; Makonnen, E.; Aderaye, G.; Lindquist, L.; Mattsson, C.M.; Ekblom, B.; et al. Keratin-18 and microRNA-122 complement alanine aminotransferase as novel safety biomarkers for drug-induced liver injury in two human cohorts. Liver Int. 2014, 34, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.W.; Chen, X.; Jiang, Z.Z.; Wang, T.; Wang, C.; Zhang, Y.; Wen, J.; Xue, M.; Zhu, D.; Zhang, Y.; et al. A panel of serum microRNAs as specific biomarkers for diagnosis of compound- and herb-induced liver injury in rats. 2012, 7, e37395. [Google Scholar] [CrossRef] [PubMed]
- Watkins, P.B.; Merz, M.; Avigan, M.I.; Kaplowitz, N.; Regev, A.; Senior, J.R. The clinical liver safety assessment best practices workshop: Rationale, goals, accomplishments and the future. Drug Saf. 2014, 37 (Suppl. S1), S1–S7. [Google Scholar] [CrossRef] [PubMed]
- Senior, J.R. Evolution of the Food and Drug Administration approach to liver safety assessment for new drugs: Current status and challenges. Drug Saf. 2014, 37, S9–S17. [Google Scholar] [CrossRef] [PubMed]
- Merz, M.; Lee, K.R.; Kullak-Ublick, G.A.; Brueckner, A.; Watkins, P.B. Methodology to assess clinical liver safety data. Drug Saf. 2014, 37, S33–S45. [Google Scholar] [CrossRef] [PubMed]
- Regev, A.; Seeff, L.B.; Merz, M.; Ormarsdottir, S.; Aithal, G.P.; Gallivan, J.; Watkins, P.B. Causality assessment for suspected DILI during clinical phases of drug development. Drug Saf. 2014, 37, S47–S56. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Black cohosh and suspected hepatotoxicity—Inconsistencies, confounding variables, and prospective use of a diagnostic causality algorithm: A critical review. Menopause 2010, 17, 426–440. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J. Risk of kava hepatotoxicity and the FDA consumer advisory. J. Am. Med. Assoc. 2010, 304, 2174–2175. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Qiu, S.X.; Xuan, T.D.; Lebot, V. Kava and kava hepatotoxicity: Requirements for novel experimental, ethnobotanical, and clinical studies based on a review of the evidence. Phytother. Res. 2011, 25, 1262–1274. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Lebot, V. Proposal for a Kava Quality Standardization Code. Food Chem. Toxicol. 2011, 49, 2503–2516. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Sarris, J.; Lebot, V. Kava hepatotoxicity solution: A six point plan for new kava standardization. Phytomedicine 2011, 18, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Special report: Kava and the risk of liver toxicity: Past, current, and future. Am. Herb. Prod. Assoc. 2011, 26, 1–17. [Google Scholar]
- Teschke, R.; Sarris, J.; Lebot, V. Contaminant hepatotoxins as culprits for hepatotoxicity—Fact or fiction? Phytother. Res. 2011, 27, 472–474. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Sarris, J.; Schweitzer, I. Kava hepatotoxicity in traditional and modern use: The presumed Pacific kava paradox hypothesis revisited. Br. J. Clin. Pharmacol. 2012, 73, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D.; Pageaux, G. Hepatotoxicity of herbal remedies and mushrooms. Semin. Liver Dis. 1995, 15, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Genthner, A.; Frenzel, C.; Wolff, A.; Schulze, J. Clinical review: Herbal hepatotoxicity and the call for systematic data documentation of individual cases. J. Liver Clin. Res. 2015, 2, 1008. [Google Scholar]
- Danan, G.; Bénichou, C. Causality assessment of adverse reactions to drugs-I. A novel method based on the conclusions of international consensus meetings: Application to drug-induced liver injuries. J. Clin. Epidemiol. 1993, 46, 1323–1330. [Google Scholar] [CrossRef]
- Bénichou, C.; Danan, G.; Flahault, A. Causality assessment of adverse reactions to drugs-II. An original model for validation of drug causality assessment methods: Case reports with positive rechallenge. J. Clin. Epidemiol. 1993, 46, 1331–1336. [Google Scholar] [CrossRef]
- Yuan, D.; Yang, X.; Guo, J.C. A great honor and a hugh challenge for China: You-you TU getting the Nobel Prize in Physiology or Medicine. J. Zhejiang Univ.-Sci. B (Biomed. & Biotechnol.) 2016, 17, 405–406. [Google Scholar]
- Gloro, R.; Hourmand-Ollivier, I.; Mosquet, B.; Mosquet, L.; Rousselot, P.; Salamé, E.; Piquet, M.A.; Dao, T. Fulminant hepatitis during self-medication with hydroalcoholic extract of green tea. Eur. J. Gastroenterol. Hepatol. 2005, 17, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.N.; Kim, D.J.; Kim, Y.M.; Kim, B.H.; Sohn, K.M.; Choi, M.J.; Choi, Y.H. Aloe-induced toxic hepatitis. J. Korean Med. Sci. 2010, 25, 492–495. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.J.; Kim, H.Y.; Choi, E.S.; Cho, Y.; Kim, Y.; Lee, J.H.; Jang, E. Drug-induced liver injury: A 2-year retrospective study of 1169 hospitalized patients in a single medical center. Phytomedicine 2015, 13, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Heidemann, L.A.; Navarro, V.J.; Ahmad, J.; Hayashi, P.H.; Stolz, A.; Kleiner, D.E.; Fontana, R. Severe acute hepatocellular injury attributed to OxyELITE Pro: A case series. Dig. Dis. Sci. 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A. The Honolulu Liver disease cluster at the Medical Center: Its mysteries and challenges. Int. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Wai, CT. Presentation of drug-induced liver injury in Singapore. Singap. Med. J. 2006, 47, 116–120. [Google Scholar]
- Björnsson, E.; Olsson, R. Serious adverse liver reactions associated with herbal weight loss supplements. J. Hepatol. 2007, 47, 295–297. [Google Scholar] [CrossRef] [PubMed]
- García-Cortés, M.; Lucena, M.I.; Pachkoria, K.; Borraz, Y.; Hidalgo, R.; Andrade, R.J. Evaluation of Naranjo Adverse Drug Reactions Probability Scale in causality assessment of drug-induced liver injury. Aliment. Pharmacol. Ther. 2008, 27, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Chau, T.N.; Cheung, W.I.; Ngan, T.; Lin, J.; Lee, K.W.S.; Poon, W.T.; Leung, V.K.S.; Mak, T.; Tse, M.L.; The Hong Kong Herb-Induced Liver Injury Network (HK-HILIN). Causality assessment of herb-induced liver injury using multidisciplinary approach and the Roussel Uclaf Causality Assessment Method (RUCAM). Clin. Toxicol. 2011, 49, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Schulze, J.; Schwarzenboeck, A.; Eickhoff, A. Herbalife hepatotoxicity: Evaluation of cases with positive reexposure tests. World J. Hepatol. 2013, 5, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Zambrone, F.A.D.; Correa, C.; Sampaio do Amaral, L.M. A critical analysis of the hepatotoxicity cases described in the literature related to Herbalife® products. Braz. J. Pharm. Sci. 2015, 51, 785–796. [Google Scholar] [CrossRef]
- Mahady, G.B.; Low Dog, T.; Barrett, M.L.; Chavez, M.L.; Gardiner, P.; Ko, R.; Marles, R.J.; Pellicore, L.S.; Giancaspro, G.I.; Sarma, D.N. United States Pharmacopeia review of the black cohosh case reports of hepatotoxicity. Menopause 2008, 15, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J. Suspected herbal hepatotoxicity: Requirements for appropriate causality assessment by the US Pharmacopeia. Drug Saf. 2012, 35, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Liss, G.; Lewis, J.H. Drug-induced liver injury: What was new in 2008? Expert Opin. Drug Metab. Toxicol. 2009, 5, 843–860. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Genthner, A.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Herbal hepatotoxicity: Analysis of cases with initially reported positive reexposure tests. Dig. Liver Dis. 2014, 46, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Hillman, L.; Gottfried, M.; Whitsett, M.; Rakela, J.; Schilsky, M.; Lee, W.M.; Ganger, D. Clinical features and outcomes of complementary and alternative medicine induced acute liver failure and injury. 2016, 111, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Mohabbat, O.; Younos, M.S.; Merzad, A.A.; Srivastava, R.N.; Sediq, G.G.; Aram, G.N. An outbreak of hepatic veno-occlusive disease in north-western Afghanistan. Lancet 1976, 308, 269–271. [Google Scholar] [CrossRef]
- Kakar, F.; Akbarian, Z.; Leslie, T.; Mustafa, M.L.; Watson, J.; van Egmond, H.P.; Omar, M.F.; Mofleh, J. An outbreak of hepatic veno-occlusive disease in western Afghanistan associated with exposure to wheat flour contaminated with pyrrolizidine alkaloids. J. Toxicol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Tandon, R.K.; Tandon, B.N.; Tandon, H.D. Study of an epidemic of venoocclusive disease in India. Gut 1976, 17, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Tandon, B.N.; Tandon, H.D.; Tandon, R.K.; Narndranathan, M.; Joshi, Y.K. An epidemic of veno-occlusive disease of the liver in central India. Lancet 1976, 308, 272–272. [Google Scholar] [CrossRef]
- Abdualmjid, R.J.; Sergi, C. Hepatotoxic botanicals—An evidence-based systematic review. J. Pharm. Pharm. Sci. 2013, 16, 376–404. [Google Scholar] [CrossRef] [PubMed]


| Issues | Details of Issues and Proposed Solutions | First Author |
|---|---|---|
| Therapeutic efficacy is rarely provided for most TCM herbs. Randomized controlled trials (RCTs) are urgently needed, based on criteria of evidence based medicine (EBM). | Manheimer, 2009 [9] |
| Teschke, 2015 [10] | ||
| Ernst, 2006 [41] | ||
| Wang, 2007 [42] | ||
| Tang, 1999, 2008 [43,44] | ||
| Most TCM herbs are well tolerated by the majority of users, but few are not. RCTs should evaluate the individual benefit-risk profile for each herbal TCM product. It is important to define product quality specifications and adherence to the principles of cGAPs and cGMPs. | Ekor, 2014 [3] |
| Liu, 2015 [8] | ||
| Melchart, 2016 [13] | ||
| Northier, 2000, 2002 [45,46] | ||
| Vanherweghem, 1998 [47] | ||
| Wagner, 2013 [48] | ||
| WHO, 2016 [49] | ||
| Frenzel, 2016 [50] | ||
| Causality assessment is often missing, must be verified by RUCAM for each spontaneous and clinical trial report submitted to regulatory agencies and for each case report submitted to a scientific journal for consideration of publication. | Frenzel, 2016 [50] |
| Danan, 2016 [51] | ||
| Less strict regulatory surveillance is risky for consumers, calling for substantial improvement. Regulatory definitions of indications and use are mandatory. Herbal TCM products for medicinal use should be approved by regulatory agencies and registered as herbal drugs, provided efficacy is validly established and their benefit-risk balance is favorable. | Liu, 2015 [8] |
| Zhang, 2012 [12] | ||
| Frenzel, 2016 [50] | ||
| Variable nomenclature of herbal TCM products causes confusion, requiring CMM international name standards. Unlike Latin scientific names, CMM names, be they Chinese, pin yin, English, or according to pharmacopoeia, have not been standardized and their use, spelling and occasionally even the plant species to which they refer vary from publication to publication. | Chan, 2012 [52] |
| Reported Side Effects | Patients (n) | Patients (%) |
|---|---|---|
| Liver adaptation | 3 | 0.3 |
| Liver injury | 3 | 0.3 |
| Diarrhea | 46 | 4.6 |
| Nausea | 11 | 1.1 |
| Vomiting | 4 | 0.4 |
| Other gastrointestinal symptoms | 103 | 13.4 |
| Neuro-vegetative symptoms | 61 | 6.1 |
| Aggravation of pain | 36 | 3.6 |
| Chinese Name | Botanical Names, Main Ingredients |
|---|---|
| Artemisia argyi. |
| Lycopodium serratum or rarely, Corydalis species, Panax ginseng, Pseudo ginseng, or two species of Stephania. |
| Angelica sinensis, Cyperus rotundus, Ginseng, Ligusticumwallichii, Paeonia alba, Rehmannia glutinosa. |
| Atractylis, Carthamus tinctorius, Dalbergia odorifera, Dioscorea bulbifera, Glycyrrhiza, Lithospermum erythrorhizon, Paeonia suffruticosa, Polygonum multiflorum, Psoralea corylifolia, Salvia miltiorrhiza; Endoconcha sepiae, Ganoderma lucidum (mushroom). |
| Rhicinus communis, Chaenomeles, Codonopsis pilosula, Notopterygium, Polygonum multiflorum, Rehmannia, Schisandra. |
| Mentha haplocalyx. |
| Tinospora crispa. |
| Angelica, Atractylis, Cnidium, Gardenia, Ephedra, Forsythia, Glycyrrhhiza, Gypsum fibrosum, Ledebouriella, Mentha, Paeonia, Platycodon, Rheum, Schizonepeta, Scutellaria, Zingiber ; Kadinum (talcum powder), sodium sulfuricum. |
| Psoralea corylifolia. |
| Xanthium sibiricum. |
| Dichora febrifuga Lour. |
| Bupleurum falcatum. |
| Camellia sinensis, Cassia tora (syn. Senna), Crataegus, N-nitroso-fenfluramine. |
| Breynia officinalis. |
| Dictamnus dasycarpus, Gentiana scabra, Hedyotis diffusa, Paeonia suffructicosa, Paris polyphylla, Rehmannia glutinosa, Smilax glabra, Sophora subprostrata; Angelica sinensis, Bupleurum chinese, Dictamnus dasycarpus, Paeonia suffructiosa, Philodendron chinese, Saposhnikovia divaricata, Shisandra chinesis, Shizonepeta tenuifolia, Tribulus terrestris; Cocculus trilobus, Dictamnus dasycarpus, Eurysolen gracilis, Glycyrrhiza, Lophatherum, Paeonia, Potentilla, Rehmannia glutinosa; Alisma plantago aquatica, Artemisia capillaris, Gardenia jasminoidis, Gentiana scabra, Glycyrrhiza, Magnolia, Paeonia, Plantago asiatica, Saussurea Lappa. |
| Melia toosendan. |
| Acanthopanax senticosus. |
| Bupleurum falcatum, Ginseng, Glycyrrhiza glabra, Pinellia, Scutellaria, Zingiber officinale, Zizyphus jujuba. |
| Rheum palmatum. |
| Angelica archangelica. |
| Angelica dahurica, Isatis indigotica (Indigo naturalis), Massa medicata fermentata (yeast), Salvia milthiorrhiza, Smilax glabra. |
| Glycyrrhiza uralensis, syn. Liquorice. |
| Pueraria lobata, syn. Arrowroot. |
| Albizia julibrissin. |
| Polygonum multiflorum, syn. He Shou Wu. |
| Mentha pulegium, syn. Pennyroyal oil. |
| Polygonum cuspidatum. |
| Scutellaria baicalensis. |
| Dioscorea bulbifera. |
| Corydalis speciosa. |
| Abrus cantoniensis. |
| Chloranthus serratus. |
| Centella asiatica, syn. Gotu Kola. |
| Hovenia dulcis. |
| Lycopodium serratum or rarely, Corydalis species, Panax ginseng, Pseudo ginseng, or two species of Stephania. |
| Cassia obtusifolia, syn. Senna. |
| Angelica sinensis, Atractylodes racea, Bupleurum falcatum, Gardenia, Glycyrrhiza glabra, Mentha haplocalyx, Moutan, Paeonia alba, Sclerotium Poriae Cocos, Zingiber officinale. |
| Pueraria thunbergiana. |
| Melia azedarach. |
| Tripterygium wilfordii Hook. |
| Aloe vera. |
| Acebia, Alisma, Angelica sinensis, Bupleurum, Gardenia, Gentiana, Glycyrrhiza, Plantago, Rehmannia, Scutellaria. |
| Camellia sinensis, syn. Chinese green tea. |
| Ephedra sinica. |
| Heliotropium lasiocarpum. |
| Aloe, Camellia sinensis, Crataegus, Gynostemma pentaphyllum makino, Raphanus; N-nitroso-fenfluramine. |
| Senecio scandens. |
| Polygonum multiflorum, Angelica sinensis, fructus psoraleae (Yan Shuizhi), wolfberry fruit, Cuscuta species, poria cocos, achyranthes bidentata. |
| Panax ginseng. |
| Alisma, Atractylis, Bupleurum, Cinnamomum, Ginseng, Glycyrrhiza, Pinellia, Polyporus, Poria, Scutellaria, Zingiber, Zizyphus. |
| Gynura segetum. |
| Phytolacca acinosa. |
| Black cohosh, Burdock, Cayenne pepper, Ginkgo biloba, Horse chestnut, Piper nigrum, Polygonum multiflorum, uva ursi; biotin, collagen (hydrolyzed), niacin, pantothenic acid, silica (from plant sources), soy isoflavones, vitamin A, vitamin B6. |
| Teucrium chamaedrys, syn. Germander. |
| Pericarpium granati. |
| Achyranthes bidentata, Cuscuta chinensis, Eclipta prostrata, Ligustrum lucidum, Lonicera japonica, Morus alba, Polygonum multiflorum, Psoralea corylifolia, Rehmannia glutinosa, Rosa aevigat, Sesemum indicum, Siegesbeckia orientalis. |
| Trichosanthes kirilowii. |
| Qian Ceng Ta (Huperzia serrata), Wu Zhu Yu Evodia rutaecarpa); beet root, caffein, cocoa bean, vinpocetine (from Vinca plant); acesulfame potassium, calcium silicate, carnitine tartrate, Carno-Syn® beta-alanine, citrulline, cryptoxanthin, folic acid, gamma-aminobutyric acid (GABA), glucuronolactone, selenium, L-norvaline, L-tyrosine, lutein, malic acid, ornithine, potassium gluconate, sucralose, sugar cane, water melon flavor, zeaxanthin. |
| Galla chinensis. |
| Camptotheca acuminate. |
| Abrus Precatorius. |
| Bupleurum falcatum, Ginseng, Glycyrrhiza glabra, Pinellia tuber, Scutellaria baicalensis, Zingiber officinale, Zizyphus jujuba. |
| Artemisia capillaris. |
| Alisma orientalis. |
| Phyllanthus urinaria. |
| Country/Area | Cases (n) | References with First Author |
|---|---|---|
| 3 | Jorge, 2005 [112] |
| 1 | Park, 2001 [156] |
| 1 | Divinsky, 2002 [116] |
| 1 | Cárdenas, 2006 [151] |
| 7 | Larrey, 1992 [168] |
| 26 | Castot, 1992 [169] | |
| 1 | Mostefa-Kara, 1992 [170] | |
| 1 | Diaz, 1992 [171] | |
| 1 | Ben Yahia, 1993 [172] | |
| 1 | Dao, 1993 [173] | |
| 1 | Mattéi, 1999 [175] | |
| 1 | Peyrin-Biroulet, 2004 [133] | |
| 1 | Starakis, 2006 [176] |
| 2 | Dai, 2006 [124] |
| 1 | Chen, 2007 [128] | |
| 41 | Wu, 2008 [119] | |
| 1 | Li, 2010 [129] | |
| 116 | Gao, 2012 [122] | |
| 146 | Zhou, 2013 [184] | |
| 1 | Li, 2015 [165] | |
| 40 | Wang, 2015 [164] | |
| 15 | Zhu, 2015 [189] | |
| 563 | Zhu, 2016 [188] | |
| 4 | Kumana, 1983, 1985 [125,126] |
| 1 | But, 1996 [155] | |
| 7 | Yuen, 2006 [82] | |
| 3 | Cheung, 2009 [88] | |
| 52 | Lin, 2011 [120] | |
| 1 | Picciotti, 1998 [117] |
| 1 | Mazzanti, 2004 [157] | |
| 36 | Mazzanti, 2009 [138] | |
| 1 | Valente, 2010 [154] | |
| 19 | Mazzanti, 2015 [141] | |
| 4 | Itoh, 1995 [178] |
| 1 | Kamiyama, 1997 [91] | |
| 12 | Adachi, 2003 [93] | |
| 1 | Aiba, 2007 [166] | |
| 1 | Motoyama, 2008 [83] | |
| 21 | Gono, 2010 [106] | |
| 1 | Furukawa, 2010 [159] | |
| 1 | Tsuda, 2010 [167] | |
| 1 | Inoue, 2011 [92] | |
| 1 | Hwang, 2001 [86] |
| 1 | Nam, 2005 [87] | |
| 4 | Jang, 2008 [75] | |
| 120 | Kang, 2008 [79] | |
| 24 | Sohn, 2008 [80] | |
| 1 | Kang, 2009 [111] | |
| 2 | Kim, 2009 [104] | |
| 1 | Bae, 2010 [105] | |
| 25 | Jung, 2011 [153] | |
| 1 | Yang, 2012 [108] | |
| 3 | Kim, 2012 [113] | |
| 1 | Huseini, 2016 [142] |
| 1 | Panis, 2005 [152] |
| 1 | Banarova, 2012 [160] |
| 1 | Garcia-Moran, 2004 [132] |
| 1 | Jimenez-Saenz, 2006 [134] | |
| 8 | Garcia-Cortés, 2008 [136] | |
| 61 | Lee, 2011 [90] |
| 2 | Lin, 2002 [97] | |
| 19 | Lin, 2003 [98] | |
| 1 | Hsu, 2006 [179] | |
| 1 | Davies, 1990 [100] |
| 1 | Graham-Brown, 1992 [76] | |
| 1 | Sanders, 1995 [101] | |
| 3 | Kane, 1995 [77] | |
| 1 | Vautier, 1995 [78] | |
| 1 | Skoulidis, 2005 [148] | |
| 7 | Woolf, 1994 [114] |
| 1 | Nadir, 1996 [145] | |
| 6 | Horowitz, 1996 [115] | |
| 1 | Borum, 2001 [147] | |
| 2 | Haller, 2002 [70] | |
| 7 | Estes, 2003 [74] | |
| 1 | Bonkovsky, 2006 [135] | |
| 1 | Laird, 2008 [158] | |
| 34 | Sarma, 2008 [137] | |
| 1 | Linnebur, 2010, [107] | |
| 1 | Cohen, 2012 [177] | |
| 1 | Cortez, 2012 [81] | |
| 1 | Yang, 2012 [108] | |
| 1 | Dhanasekaran, 2013 [109] | |
| 47 | Navarro, 2013 [139] | |
| 1 | Papafragkakis, 2016 [110] |
| Chinese Name | Scientific Name | Tentative Hepatotoxic Components |
|---|---|---|
| Ai Ye | Artemisia argyi | Volatile oil |
| Bi Ma Zi | Rhicinus communis | Ricin, toxic proteins |
| Cang Shan | Xanthium | Glycosides (kaurene), diterpenoids |
| Chang Shan | Dichor febrifuga Lour | Alkaloids (dichroine) |
| He Huan Pi | Albizia julibrissin | Glycosides (saponine) |
| He Shou Wu | Polygonum multiflorum | Anthraquinones |
| Huang Yao Zi | Discorea bulbifera L | Glycosides (steroids, diosgenin), diterpenoids-lactones |
| Ku Lian Zi | Melia azedarach | Glycosides (tetranortriterpenoids) |
| Lei Gong Teng | Tripterygium wilfordii hook F | Glycosides (tripterygium), diterpenoid-lactones |
| Qian Li Guang | Senecio scandens | Pyrrolizidine alkaloids |
| Shan Lu | Phytolacca acinosa Roxb. | Alkaloids (phytolaccine) |
| Xiang Si Zi | Abrus Precatorius | Abrin |
| Conditions | Results |
|---|---|
| n = 116 |
| Males 57 |
| Females 56 | |
| (NA 3) | |
| 17–76 years |
| 115/116 cases |
| 104/113 cases |
| 95/113 cases |
| 47/60 cases |
| (NA 56 cases) | |
| 50/58 cases |
| (NA 58 cases) | |
| Recovery 75 cases |
| Chronicity 27 cases | |
| Death 11 cases | |
| (NA 3 cases) |
| TCM Product/TCM Herb | Fatality (n) | LTX (n) | Selected References with First Author |
|---|---|---|---|
| 2 | Sohn, 2008 [80] | |
| 2 | Sohn, 2008 [80] | |
| 1 | Sohn, 2008 [80] | |
| 4 | Mazzanti, 2015 [141] | |
| 2 | Estes, 2003 [74] | |
| 1 | Skoulidis, 2005 [148] | ||
| 3 | Sohn, 2008 [80] | |
| 1 | Jung, 2011 [153] | ||
| 1 | 1 | Wang, 2015 [164] | |
| 1 | Gao, 2012 [122] | |
| 11 | 1 | Sohn, 2008 [80] |
| 1 | Sohn, 2008 [80] | |
| 3 | 3 | Lee, 2015 [238] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Differential Diagnosis | Diagnostic Parameters | Diagnostic Exclusion Done for Patient’s Causality Assessment | ||
|---|---|---|---|---|
| Yes | No | Partial | ||
| Anti-HAV-IgM | □ | □ | □ |
| HBV-DNA, anti-HBc-IgM | □ | □ | □ |
| HCV-RNA, anti-HCV | □ | □ | □ |
| HEV-RNA , titer change for anti-HEV-IgM/anti-HEV-IgG | □ | □ | □ |
| CMV-PCR, titer change for anti-CMV-IgM/anti-CMV-IgG | □ | □ | □ |
| EBV-PCR, titer change for anti-EBV-IgM/anti-EBV-IgG | □ | □ | □ |
| HSV-PCR, titer change for anti-HSV-IgM/anti-HSV-IgG | □ | □ | □ |
| VZV-PCR, titer change for anti-VZV-IgM/anti-VZV-IgG | □ | □ | □ |
| Specific serology of Adenovirus, Coxsackie-B-Virus, Echovirus, Measles virus, Rubella virus, Flavivirus, Arenavirus, Filovirus, Parvovirus, HIV, and others | □ | □ | □ |
| Specific assessment of bacteria, fungi, parasites, worms, and others | □ | □ | □ |
| Gamma globulins, ANA, SMA, AAA, SLA/LP, Anti-LSP, Anti-ASGPR | □ | □ | □ |
| Gamma globulins, Anti-LKM-1 (CYP 2D6), Anti-LKM-2 (CYP 2C9), Anti-LKM-3 | □ | □ | □ |
| AMA, Anti PDH-E2 | □ | □ | □ |
| p-ANCA, MRC | □ | □ | □ |
| ANA, SMA | □ | □ | □ |
| See AIH, PBC, PSC, and AIC | □ | □ | □ |
| BMI, insulin resistance, hepatomegaly, echogenicity of the liver | □ | □ | □ |
| Patient’s history, clinical and laboratory assessment, other alcoholic disease(s) | □ | □ | □ |
| Patient’s history, clinical and laboratory assessment, sonography, use of the updated RUCAM | □ | □ | □ |
| Toxin screening | □ | □ | □ |
| Toxin screening for household and occupational toxins | □ | □ | □ |
| Serum ferritin, total iron-binding capacity, genotyping for C2824 and H63D mutation, hepatic iron content | □ | □ | □ |
| Copper excretion (24 h urine), ceruloplasmin in serum, free copper in serum, Coombs-negative hemolytic anemia, hepatic copper content, Kayser-Fleischer-ring, neurologic-psychiatric work-up, genotyping | □ | □ | □ |
| Porphobilinogen in urine, total porphyrines in urine | □ | □ | □ |
| α1—Antitrypsin in serum | □ | □ | □ |
| Clinical and laboratory assessment, hepatobiliary sonography, MRC | □ | □ | □ |
| Clinical and laboratory assessment, sonography, CT, MRT | □ | □ | □ |
| TTG antibodies, endomysium antibodies, duodenal biopsy | □ | □ | □ |
| Clinical context | □ | □ | □ |
| Clinical context | □ | □ | □ |
| Cardiopulmonary assessment of congestive heart disease, myocardial infarction, cardiomyopathy, cardiac valvular dysfunction, pulmonary embolism, pericardial diseases, arrhythmia, hemorrhagic shock, and various other conditions | □ | □ | □ |
| Plasma cortisol | □ | □ | □ |
| TSH basal, T4, T3 | □ | □ | □ |
| Clinical context of epileptic seizure (duration > 30 min) | □ | □ | □ |
| Shock, hyperthermia | □ | □ | □ |
| Shock, liver injury | □ | □ | □ |
| Specific assessment of sarcoidosis, amyloidosis, metastatic tumor, sepsis, and others | □ | □ | □ |
| Clinical context | □ | □ | □ |
| Items | RUCAM | MV | DILIN | Naranjo | WHO | AD HOC |
|---|---|---|---|---|---|---|
| + | + | 0 | 0 | 0 | 0 |
| + | + | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | 0 | 0 | + | 0 | 0 |
| + | 0 | 0 | 0 | 0 | |
| + | + | 0 | 0 | 0 | 0 |
| + | + | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | + | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | + | 0 | + | 0 | 0 |
| + | + | 0 | + | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| + | + | + | 0 | 0 | 0 |
| + | + | + | 0 | 0 | 0 |
| + | + | + | 0 | 0 | 0 |
| + | + | 0 | 0 | 0 | 0 |
| + | + | 0 | 0 | 0 | 0 |
| + | 0 | 0 | 0 | 0 | 0 |
| Cases | Products | Country/Region | Year | First Author |
|---|---|---|---|---|
| HILI | Ji Xue Cao | Argentina/South America | 2005 | Jorge [112] |
| HILI | Lu Cha (Green tea , Camellia sinensis) | France/Europe | 2005 | Gloro [278] |
| HILI | Bo He, Chuan Lian Zi, and various other herbal TCM | Korea/Asia | 2006 | Yuen [82] |
| HILI | Lu Cha | Spain/Europe | 2006 | Jimenez-Saenz [134] |
| HILI | Polygonum multiflorum | Columbia/South America | 2006 | Cárdenas [151] |
| HILI | Bai Xian Pi, Kudzu, Lu Cha, Yin Chen Hao | Korea/Asia | 2008 | Kang [111] |
| HILI | Bai Xian Pi, Ci Wu Jia, Shou Wu Pian, Yin Chen Hao | Korea/Asia | 2008 | Sohn [80] |
| HIL | Polygonum multiflorum | China/Asia | 2009 | Zhang [161] |
| HILI | Lu Cha | Italy/Europe | 2009 | Mazzanti [138] |
| HILI | Corydalis speciosa | Korea/Asia | 2009 | Kang [111] |
| HILI | Ge Gen | Korea/Asia | 2009 | Kim [104] |
| HILI | Ho Shou Wu | Korea/Asia | 2010 | Bae [105] |
| HILI | Aloe | Korea/Asia | 2010 | Yang [279] |
| HIL | Polygonum multiflorum | Korea/Asia | 2011 | Jung [153] |
| HILI | Gynura segetum | Hong Kong/Asia | 2011 | Lin [120] |
| HILI | Juguju | Korea/Asia | 2012 | Kim [113] |
| HILI | Gynura segetum | Hong Kong/Asia | 2012 | Gao [122] |
| HILI DILI | Multiple herbs synthetic drugs and | Korea/Asia | 2012 | Suk [182] |
| DILI HILI | Multiple TCM herbs and synthetic drugs | China/Asia | 2014 | Hao [186] |
| HILI | Polygonum multiflorum | China/Asia | 2014 | Dong [162] |
| HILI | Lu Cha | Italy/Europe | 2015 | Mazzanti [221] |
| HILI | Polygonum multiflorum | China/Asia | 2015 | Lei [163] |
| HILI | Polygonum multiflorum | China/Asia | 2015 | Wang [164] |
| HILI | Polygonum multiforme | China/Asia | 2015 | Zhu [187] |
| HILI | Single TCM herbs | Germany/Europe | 2016 | Douros [183] |
| HILI DILI | Multiple TCM herbs, synthetic drugs | Korea/Asia | 2016 | Woo [280] |
| HILI | Chinese skullcap, black catechu | USA | 2016 | Papafragkakis [110] |
| HILI | Various TCM herbs | Germany/Europe | 2016 | Melchart [14] |
| Cases | Suspected Products | Country/Region | Year | First Author of the Group/Agency |
|---|---|---|---|---|
| HILI DILI | Various herbal TCM, synthetic drugs | Singapore/Asia | 2006 | Wai [283], National University of Singapore |
| HILI | Lu Cha | Sweden/Europe | 2007 | Björnsson [284], Swedish Adverse Drug Reactions Advisory Committee |
| HILI | Lu Cha, other TCM herbs | Spain/Europe | 2008 | García-Cortés [136], Spanish Liver Toxicity Registry |
| HILI DILI | Few TCM herbs, multiple drugs | Spain/Europe | 2008 | García-Cortés [285], Spanish Group for the Study of Drug-induced Liver Disease |
| HILI | Various TCM herbs | Hong Kong | 2011 | Chau [286], Hong Kong Herb-Induced Liver Injury Network (HK-HILIN), Hong Kong |
| HILI | Polygonum multiflorum | Korea/Asia | 2011 | Jung [153], School of Medicine, Changwon Gyeongsang National University School of Medicine, Jinju/Sungkyunkwan University |
| Reexposure Test Result | Hepatocellular Injury | Cholestatic or Mixed Liver Injury | ||
|---|---|---|---|---|
| ALTb | ALTr | ALPb | ALPr | |
| <5 N | ≥2 ALTb | <2 N | ≥2 ALPb |
| <5 N | <2 ALTb | <2 N | <2 ALPb |
| ≥5 N | ≥2 ALTb | ≥2 N | ≥2 ALPb |
| ≥5 N | <2 ALTb | ≥2 N | <2 ALPb |
| <5 N | n.a. | <2 N | n.a. |
| n.a. | ≥2 ALTb | n.a. | ≥2 ALPb |
| n.a. | n.a. | n.a. | n.a. |
| Herbal TCM | RUCAM Based Causality | Reexposure Based Causality |
|---|---|---|
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| + | − |
| − | − |
| − | + |
| − | + |
| − | − |
| + | + |
| + | − |
| + | − |
| − | − |
| + | − |
| + | + |
| − | + |
| + | + |
| + | − |
| + | − |
| + | − |
| + | + |
| + | − |
| + | − |
| + | + |
| − | + |
| + | − |
| + | − |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teschke, R.; Larrey, D.; Melchart, D.; Danan, G. Traditional Chinese Medicine (TCM) and Herbal Hepatotoxicity: RUCAM and the Role of Novel Diagnostic Biomarkers Such as MicroRNAs. Medicines 2016, 3, 18. https://doi.org/10.3390/medicines3030018
Teschke R, Larrey D, Melchart D, Danan G. Traditional Chinese Medicine (TCM) and Herbal Hepatotoxicity: RUCAM and the Role of Novel Diagnostic Biomarkers Such as MicroRNAs. Medicines. 2016; 3(3):18. https://doi.org/10.3390/medicines3030018
Chicago/Turabian StyleTeschke, Rolf, Dominique Larrey, Dieter Melchart, and Gaby Danan. 2016. "Traditional Chinese Medicine (TCM) and Herbal Hepatotoxicity: RUCAM and the Role of Novel Diagnostic Biomarkers Such as MicroRNAs" Medicines 3, no. 3: 18. https://doi.org/10.3390/medicines3030018