Trace Element Concentrations in Drinking Water and Urine among Saharawi Women and Young Children
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Water and Urine Samples
- Water source 1: El Aiun and Auserd. Water is supplied from groundwater wells and reverse osmosis plant for both of the camps. The plant does not have capacity to serve treated water continuously, hence each camp receives treated water for 21 days and raw water for 21 days in different turns. A chlorination step is required for all distributed water.
- Water source 2: Bojadour/Smara. Water is supplied from ground water wells and the reverse osmosis plant. A chlorination step is required for all distributed water.
- Water source 3: Dakhla. Water is supplied from groundwater wells followed by chlorination.
2.3. Chemical Analyses
2.4. Thyroid Hormones and Background Characteristics
2.5. Ethical Considerations
2.6. Statistics and Data Management
3. Results
4. Discussion
4.1. Trace Element Concentration in Distributed Public Drinking Water
4.2. Associations between Zones and Urinary Excretion of Elements
4.3. Elements in Urine
4.4. Elements in Urine Attributable to Their Occurrence in Drinking Water
4.5. Elements in Urine Elements Attributable to Other Sources
4.6. Trace Elements in Urine Relevant for Thyroid Metabolism
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cornelis, R.; Heinzow, B.; Herber, R.; Christensen, J.M.; Poulsen, O.; Sabbioni, E.; Templeton, D.; Thomassen, Y.; Vahter, M.; Vesterberg, O. Sample collection guidelines for trace elements in blood and urine. J. Trace Elem. Med. Biol. 1996, 10, 103–127. [Google Scholar] [CrossRef]
- United Nations. Progress towards the Sustainable Development Goals; United Nations: New York, NY, USA, 2017. [Google Scholar]
- United Nations General Assembly. 64/292 the Human Right to Water and Sanitation. United Nations General Assembly, Ed. 2010; Vol. 64/292. Available online: http://www.un.org/es/comun/docs/?symbol=A/RES/64/292&lang=E (accessed on 12 May 2017).
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Ebdon, L.; Pitts, L.; Cornelis, R.; Crews, H.; Donard, O.F.X.; Quevauviller, P. Trace Element Speciation for Environment, Food and Health; Royal Society of Chemistry: Cornwall, UK, 2007. [Google Scholar]
- Bevan, R.; Jones, K.; Cocker, J.; Assem, F.; Levy, L. Reference ranges for key biomarkers of chemical exposure within the UK population. Int. J. Hyg. Environ. Health 2013, 216, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Herber, R.F.; Duffus, J.H.; Christensen, J.M.; Olsen, E.; Park, M.V. Risk assessment for occupational exposure to chemicals. A review of current methodology (iupac technical report). Pure Appl. Chem. 2001, 73, 993–1031. [Google Scholar] [CrossRef]
- Angerer, J.; Ewers, U.; Wilhelm, M. Human biomonitoring: State of the art. Int. J. Hyg. Environ. Health 2007, 210, 201–228. [Google Scholar] [CrossRef] [PubMed]
- Hoet, P.; Jacquerye, C.; Deumer, G.; Lison, D.; Haufroid, V. Reference values and upper reference limits for 26 trace elements in the urine of adults living in Belgium. Clin. Chem. Lab. Med. 2013, 51, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Díaz-CadÓrniga, F.J.; Delgado, E.; Tartón, T.; Valdés, M.M.; Méndez, A.; Fernández, M.T.; Rojo, C. Bocio endémico por exceso de yodo en la población escolar de los campos de refugiados de la rasd (república árabe saharaui democrática). Endocrinología y Nutrición 2003, 50, 357–362. [Google Scholar] [CrossRef]
- Barikmo, I.; Henjum, S.; Dahl, L.; Oshaug, A.; Torheim, L.E. Environmental implication of iodine in water, milk and other foods used in saharawi refugees camps in Tindouf, Algeria. J. Food Comp. Anal. 2011, 24, 637–641. [Google Scholar] [CrossRef] [Green Version]
- Aakre, I.; Bjøro, T.; Norheim, I.; Strand, T.A.; Barikmo, I.; Henjum, S. Excessive iodine intake and thyroid dysfunction among lactating saharawi women. J. Trace Elem. Med. Biol. 2015, 31, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Docampo, E. Estudio de Las Aguas Subterráneas y los Abastecimientos en Los Campamentos de Refugiados Saharauis en Tindouf. Master’s Thesis, Universidad de Santiago de Compostela, Santiago, Spain, 2006. [Google Scholar]
- Leung, A.M. The effects of iodine excess. In Iodine Deficiency Disorders and Their Elimination; Pearce, E.N., Ed.; Springer International Publishing: New York, NY, USA, 2017; pp. 75–89. [Google Scholar]
- Meenakshi; Maheshwari, R.C. Fluoride in drinking water and its removal. J. Hazard. Mater. 2006, 137, 456–463. [Google Scholar]
- Lv, S.; Wang, Y.; Xu, D.; Rutherford, S.; Chong, Z.; Du, Y.; Jia, L.; Zhao, J. Drinking water contributes to excessive iodine intake among children in Hebei, China. Eur. J. Clin. Nutr. 2013, 67, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Lv, S.; Xu, D.; Wang, Y.; Chong, Z.; Du, Y.; Jia, L.; Zhao, J.; Ma, J. Goitre prevalence and epidemiological features in children living in areas with mildly excessive iodine in drinking-water. Br. J. Nutr. 2014, 111, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.L.; Peng, L.; Su, X.H.; Liu, S.J. Surveys in areas of high risk of iodine deficiency and iodine excess in China, 2012–2014: Current status and examination of the relationship between urinary iodine concentration and goiter prevalence in children aged 8–10 years. Biomed. Environ. Sci. 2017, 30, 88–96. [Google Scholar] [PubMed]
- Vivar, M.; Pichel, N.; Fuentes, M.; Martínez, F. An insight into the drinking-water access in the health institutions at the saharawi refugee camps in Tindouf (Algeria) after 40 years of conflict. Sci. Total Environ. 2016, 550, 534–546. [Google Scholar] [CrossRef] [PubMed]
- Henjum, S.; Barikmo, I.; Gjerlaug, A.K.; Mohamed-Lehabib, A.; Oshaug, A.; Strand, T.A.; Torheim, L.E. Endemic goitre and excessive iodine in urine and drinking water among saharawi refugee childrren. Public Health Nutr. 2010, 13, 1472–1477. [Google Scholar] [CrossRef] [PubMed]
- Aakre, I.; Strand, T.A.; Bjøro, T.; Norheim, I.; Barikmo, I.; Ares, S.; Alcorta, M.D.; Henjum, S. Thyroid function among breastfed children with chronically excessive iodine intakes. Nutrients 2016, 8, 398. [Google Scholar] [CrossRef] [PubMed]
- UNHCR. Map Portal. Available online: http://maps.unhcr.org/en/view?id=289 (accessed on 17 August 2008).
- Aakre, I.; Bjøro, T.; Norheim, I.; Strand, T.A.; Barikmo, I.; Henjum, S. Development of thyroid dysfunction among women with excessive iodine intake—A 3-year follow-up. J. Trace Elem. Med. Biol. 2015, 31, 61–66. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Database on Body Mass Index. Bmi Classification. Available online: http://apps.who.int/bmi/index.jsp?introPage=intro_3.html (accessed on 29 May 2004).
- WHO. Who Child Growth Standards. Length/Height-for Age, Weight-for -Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age. Methods and Development; World Health Organization: Geneva, Switzerland, 2006; Available online: http://www.who.int/childgrowth/standards/Technical_report.pdf?ua=1 (accessed on 20 July 2018).
- WHO. Who Anthro (Version 3.2.2, January 2011) and Macros. Available online: http://www.who.int/childgrowth/software/en/ (accessed on 10 September 2011).
- Helsel, D.R. Fabricating data: How substituting values for nondetects can ruin results, and what can be done about it. Chemosphere 2006, 65, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Drinking Water Standards and Health Advisories Table; United States Environmental Protection Agency: Washington, DC, USA, 2007; p. 30.
- Morton, J.; Tan, E.; Leese, E.; Cocker, J. Determination of 61 elements in urine samples collected from a non-occupationally exposed UK adult population. Toxicol. Lett. 2014, 231, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Heitland, P.; Köster, H.D. Biomonitoring of 30 trace elements in urine of children and adults by ICP-MS. Clin. Chim. Acta 2006, 365, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Aakre, I.; Lilleengen, A.M.; Aarsand, M.L.; Strand, T.A.; Barikmo, I.; Henjum, S. Infant feeding practices in the saharawi refugee camps algeria, a cross-sectional study among children from birth to six months of age. Int. Breastfeed J. 2017, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Opinion of the scientific panel on dietetic products, nutrition and allergies on a request from the commission related to the tolerable upper intake level of vanadium. EFSA J. 2004, 33, 1–22. [Google Scholar]
- Crebelli, R.; Leopardi, P. Long-term risks of metal contaminants in drinking water: A critical appraisal of guideline values for arsenic and vanadium. Ann. Ist. Super. Sanita 2012, 48, 354–361. [Google Scholar] [CrossRef] [PubMed]
- IPCS. International Programme on Chemical Safety; World Health Organization: Geneva, Switzerland, 1998; p. 170. [Google Scholar]
- Edmunds, W.M.; Guendouz, A.H.; Mamou, A.; Moulla, A.; Shand, P.; Zouari, K. Groundwater evolution in the continental intercalaire aquifer of southern Algeria and Tunisia: Trace element and isotopic indicators. Appl. Geochem. 2003, 18, 805–822. [Google Scholar] [CrossRef]
- Asante, K.A.; Agusa, T.; Subramanian, A.; Ansa-Asare, O.D.; Biney, C.A.; Tanabe, S. Contamination status of arsenic and other trace elements in drinking water and residents from tarkwa, a historic mining township in Ghana. Chemosphere 2007, 66, 1513–1522. [Google Scholar] [CrossRef] [PubMed]
- WFP; UNHCR. Joint Assessment Mission-Assistance to Refugees from Western Sahara; United Nations High Comissioner for Refugees, World Food Programme: Rome, Italy, 2007. [Google Scholar]
- Barceloux, D.G.; Barceloux, D. Vanadium. J. Toxicol. Clin. Toxicol. 1999, 37, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Aakre, I.; Strand, T.A.; Moubarek, K.; Barikmo, I.; Henjum, S. Associations between thyroid dysfunction and developmental status in children with excessive iodine status. PLoS ONE 2017, 12, e0187241. [Google Scholar] [CrossRef] [PubMed]
- Ting, B.G.; Paschal, D.C.; Jarrett, J.M.; Pirkle, J.L.; Jackson, R.J.; Sampson, E.J.; Miller, D.T.; Caudill, S.P. Uranium and thorium in urine of united states residents: Reference range concentrations. Environ. Res. 1999, 81, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Orloff, K.G.; Mistry, K.; Charp, P.; Metcalf, S.; Marino, R.; Shelly, T.; Melaro, E.; Donohoe, A.M.; Jones, R.L. Human exposure to uranium in groundwater. Environ. Res. 2004, 94, 319–326. [Google Scholar] [CrossRef]
- Robberecht, H.J.; Deelstra, H.A. Selenium in human urine: Concentration levels and medical implications. Clin. Chim. Acta 1984, 136, 107–120. [Google Scholar] [CrossRef]
- Alexander, J.S. Handbook on the Toxicology of Metals; Nordberg, G.F., Fowler, B.A., Nordberg, M., Eds.; Academic Press: London, UK, 2015; Volume 40, pp. 1175–1208. [Google Scholar]
- UNHCR; WFP; INRAN. Nutrition Survey Saharawi Refugee Camps Tindouf-Algeria; United Nations High Commissioner for Refugees, World Food Programme, National Institute for Research on Food and Nutrition: Rome, Italy, 2005. [Google Scholar]
- Molin, M.; Ulven, S.M.; Meltzer, H.M.; Alexander, J. Arsenic in the human food chain, biotransformation and toxicology—Review focusing on seafood arsenic. J. Trace Elem. Med. Biol. 2015, 31, 249–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- WHO. Preventing Disease through Healthy Environments. Action Is Needed on Chemicals of Major Public Health Concern; World Health Organization: Geneva, Switzerland, 2010; Available online: http://www.who.int/ipcs/features/10chemicals_en.pdf (accessed on 15 May 2018).
- Fowler, B.A.; Selene, C.H.; Chou, R.J.; Jones, D.L.; Sullivan, W., Jr.; Chen, C.J. Chapter 28—Arsenic. In Handbook on the Toxicology of Metals, 4th ed.; Academic Press: San Diego, CA, USA, 2015; pp. 581–624. [Google Scholar]
- White, M.A.; Sabbioni, E. Trace element reference values in tissues from inhabitants of the european union. X. A study of 13 elements in blood and urine of a United Kingdom population. Sci. Total Environ. 1998, 216, 253–270. [Google Scholar] [CrossRef]
- Sanborn, M.D.; Abelsohn, A.; Campbell, M.; Weir, E. Identifying and managing adverse environmental health effects: 3. Lead exposure. Can. Med. Assoc. J. 2002, 166, 1287–1292. [Google Scholar]
- WHO. Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination. A Guide for Programme Managers; World Health Organisation, International Council for Control of Iodine Deficiency Disorders, United Nations Children’s Fund: Geneva, Switzerland, 2007; Available online: http://apps.who.int/iris/bitstream/handle/10665/43781/9789241595827_eng.pdf;jsessionid=764E64BD38CAF3D74A2EE604E550966A?sequence=1 (accessed on 17 June 2018).
| Characteristics Women | Zone 1 (n = 34) | Zone 2 (n = 43) | Total (n = 77) |
|---|---|---|---|
| Age, years b | 33.0 (25.3–36.8) | 38.0 (30.8–40.0) | 34.0 (30.0–39.0) |
| Height, cm | 158.1 ± 5.6 | 156.0 ± 6.1 | 156.9 ± 6.9 |
| Weight, kg | 70.0 ± 11.3 | 70.4 ± 14.0 | 70.2 ± 12.8 |
| BMI, kg/m2 | 27.1 ± 5.2 | 27.9 ± 5.3 | 28.5 ± 5.1 |
| <18.5 | 0 | 1 [2.3] | 1 [1.3] |
| 18.5–24.9 | 9 [26.5] | 11 [25.6] | 20 [26.0] |
| ≥ 25 | 25 [73.6] | 31 [72.2] | 56 [72.7] |
| Household size, number | 5.5 ±1.8 | 5.3 ± 1.7 | 5.4 ± 1.7 |
| Children < 5 years | 1.5 ± 0.7 | 1.4 ± 0.5 | 1.4 ± 0.6 |
| Characteristics Children | Zone 1 (n = 192) | Zone 2 (n = 104) | Total (n = 296) |
| Age, months | 30.3 (25.1–34.8) | 32.7 (24.9–35.4) | 31.4 (25.2–35.2) |
| Male | 83 [43.2] | 58 [55.8] | 141 [47.6] |
| Female | 109 [56.8] | 46 [44.2] | 155 [52.4] |
| Still breastfed, yes | 23 [12.0] | 18 [17.3] | 41 [13.9] |
| Weight-for-age, z-score c | −1.0 ± 0.9 | −1.0 ± 0.9 | −1.0 ± 0.9 |
| <−2 (underweight) | 20 [10.4] | 14 [13.5] | 34 [11.5] |
| Length/height-for-age, z-score c | −1.6 ± 1.1 | −1.6 ± 1.0 | −1.6 ± 1.0 |
| <−2 (stunted) | 61 [31.9] | 37 [35.6] | 98 [33.2] |
| Weight-for-length/height, z-score c | −0.1 ± 1.0 | −0.2 ± 0.9 | −0.2 ± 1.0 |
| <−2 (wasted) | 7 [3.7] | 4 [3.8] | 11 [3.7] |
| Element | Water Samples (n = 24) | Urine Samples | ||||||
|---|---|---|---|---|---|---|---|---|
| Women (n = 80) | Children (n = 296) | |||||||
| LOQ (µg/L) | <LOQ (%) | <LOD (%) | LOQ (µg/L) | <LOQ (%) | <LOD (%) | <LOQ (%) | <LOD (%) | |
| a, c Chlorine (Cl) | 38 | 0 | 0 | 2.8 × 103 | 0 | 0 | 0 | 0 |
| a Potassium (K) | 19 | 4 | 0 | nd | ||||
| a Sulphur (S) | nd | 2.0 × 103 | 0 | 0 | 0 | 0 | ||
| a Aluminum (Al) | 5.7 | 8 | 0 | nd | ||||
| a Arsenic (As) | 28 × 10−3 | 0 | 0 | 6.3 | 1 | 3 | 0 | |
| a Barium (Ba) | 5.2 | 13 | 4 | nd | ||||
| a Boron (B) | 16 | 0 | 0 | nd | ||||
| a Bromine (Br) | 92 × 10−3 | 0 | 0 | 42 | 0 | 0 | 0 | 0 |
| a Cadmium (Cd) | 18 × 10−3 | 92 | 67 | nd | ||||
| a Calcium (Ca) | 28 | 0 | 0 | nd | ||||
| a Cerium (Ce) | 7.4 × 10−3 | 46 | 8 | nd | ||||
| a Cesium (Cs) | 38 × 10−3 | 75 | 54 | nd | ||||
| a Chromium (Cr) | 0.54 | 88 | 38 | nd | ||||
| a Copper (Cu) | 0.56 | 50 | 13 | nd | ||||
| a Iodine (I) | 0.18 | 0 | 0 | 6.3 | 0 | 0 | 0 | 0 |
| a Iron (Fe) | 2.3 | 33 | 0 | 15.0 | 9 | 6 | 0.3 | |
| a Lead (Pb) | 0.12 | 71 | 38 | 5.6 | 49 | 34 | 13 | 7 |
| a Lithium (Li) | 2.7 | 13 | 8 | nd | ||||
| a Magnesium (Mg) | 8.1 | 0 | 0 | nd | ||||
| a Manganese (Mn) | 0.25 | 29 | 8 | nd | ||||
| a Molybdenum (Mo) | 23 × 10−3 | 0 | 0 | nd | ||||
| a Nickel (Ni) | 0.84 | 92 | 38 | nd | ||||
| a Rubidium (Rb) | 0.19 | 13 | 8 | nd | ||||
| a Selenium (Se) | 0.27 | 17 | 8 | 0.79 | 0 | 0 | 0 | 0 |
| a Sodium (Na) | 13 × 101 | 0 | 0 | nd | ||||
| a Silicon (Si) | 6.8 | 0 | 0 | nd | ||||
| a Strontium (Sr) | 0.19 | 0 | 0 | nd | ||||
| a Thallium (Tl) | 6.2 × 10−3 | 58 | 29 | nd | ||||
| a Vanadium (V) | 25 ×10−3 | 0 | 0 | 80 × 10−3 | 0 | 0 | 0 | 0 |
| a Zinc (Zn) | 3.2 | 29 | 13 | nd | ||||
| a Uranium (U) | 4.5 × 10−3 | 0 | 0 | 0.13 d | 25 | 0 | 23 | 0 |
| b Fluoride (F−) | 0.14 × 103 | 25 | 21 | |||||
| b Chloride (Cl−) | 0.49 × 103 | 0 | 0 | |||||
| b Nitrate (NO3−) | 0.35 × 103 | 0 | 0 | |||||
| b Nitrite (NO2−) | 0.27 × 103 | 0 | 0 | |||||
| b Sulfate (SO42−) | 0.34 × 103 | 0 | 0 | |||||
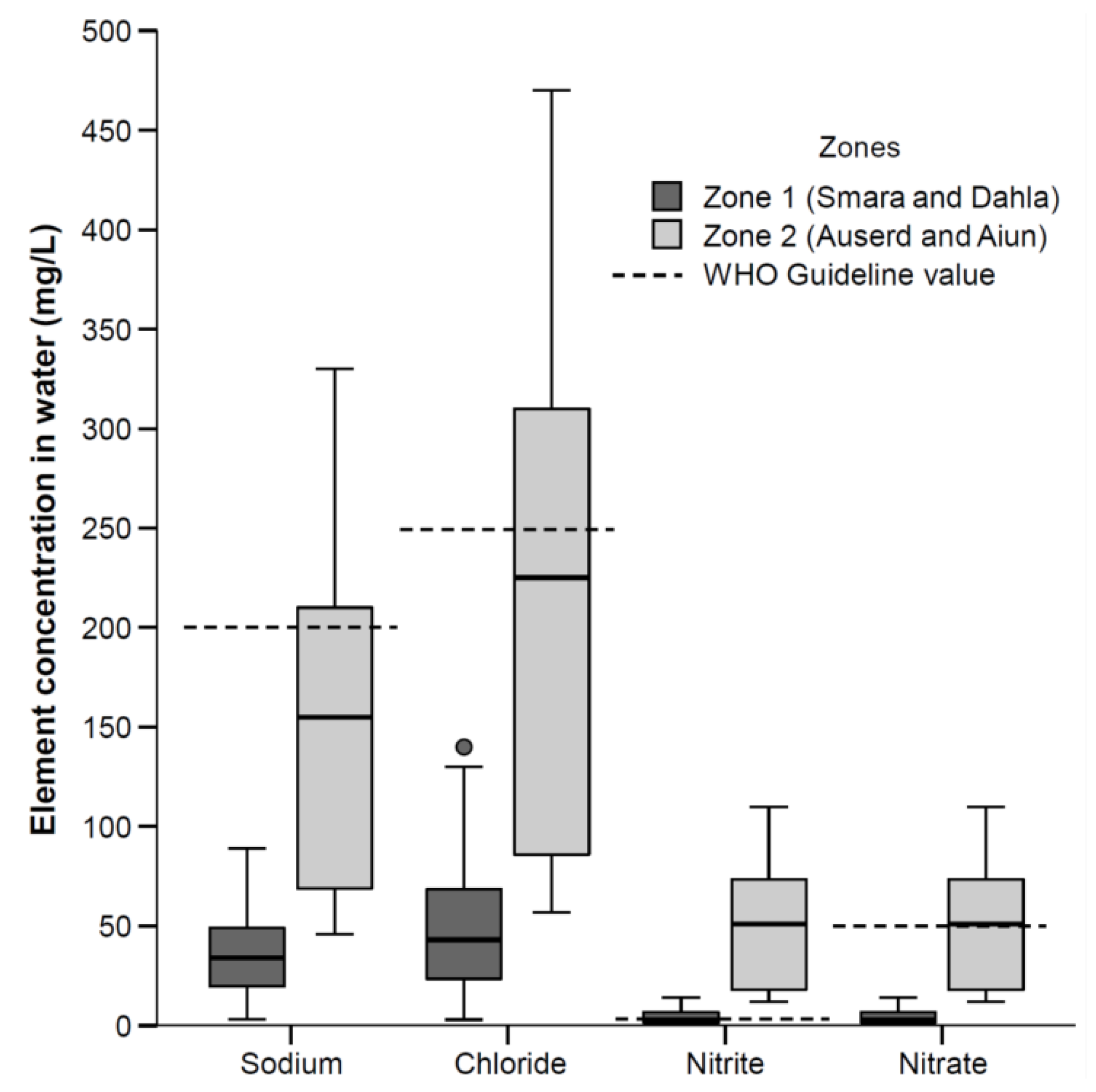
| Elements and Anions | Zone 1 (n = 12) | Zone 2 (n = 12) | p | Total (n = 24) | WHO/US Guideline Values [4,28] | |||
|---|---|---|---|---|---|---|---|---|
| Median | P25–P75 | Median | P25–P75 | Median | P25–P75 | |||
| V, µg/L | 0.93 | 0.28–1.9 | 55 | 47–62 | <0.001 | 20 | 0.85–73 | - |
| Fe, µg/L | 3.9 | 2.8–5.5 | 1.1 | 1.1–4.2 | 0.039 | 3.0 | 1.1–5.1 | 2000 |
| As, µg/L | 0.12 | 0.086–0.21 | 2.8 | 1.9–4.5 | <0.001 | 0.83 | 0.12–2.9 | 10 |
| Se, µg/L | 0.50 | 0.14–1.3 | 5.0 | 2.0–7.0 | <0.001 a | 1.6 | 0.42–5.3 | 40 |
| I, µg/L | 80 | 73–92 | 250 | 230–270 | <0.001 | 100 | 80–260 | - |
| Pb, µg/L | <0.12 | <0.12–0.20 | <0.12 | <0.12 ≤ 0.12 | 0.689 | <0.12 | <0.12–0.13 | 10 |
| U, µg/L | 0.7 | 0.04–2.1 | 5.1 | 3.9–7.7 | <0.001 | 2.9 | 0.44–5.2 | 30 |
| Li, µg/L | 11 | 2.5–27 | 59 | 22–80 | <0.001 a | 23 | 9.5–60 | - |
| B, µg/L | 99 | 73–270 | 330 | 230–430 | 0.002 | 230 | 93–400 | 2400 |
| Na, mg/L | 34 | 15–53 | 155 | 68–210 | <0.001 | 62 | 34–220 | 200 |
| Mg, mg/L | 3.0 | 1.2–5.3 | 57 | 22–78 | <0.001 | 11 | 2.9–58 | - |
| Al, µg/L | 8.3 | 6.9–11 | 8.7 | 7.7–13 | 0.583 a | 8.6 | 7.6–11 | 200 b |
| Si, mg/L | 4.2 | 0.78–7.7 | 21 | 20–21 | <0.001 | 13 | 2.7–21 | - |
| K, µg/L | 1300 | 580–2200 | 6200 | 2600–8900 | 0.002 a | 2500 | 1300–6400 | - |
| Ca, mg/L | 45 | 5.0–95 | 64 | 55–66 | 0.977 | 64 | 21–76 | - |
| Cr, µg/L | <0.54 | <0.54 ≤ 0.54 | <0.54 | <0.54–0.6 | 0.999 a | <0.54 | <0.54 ≤ 0.54 | 50 |
| Mn, µg/L | 0.6 | 0.5–1.3 | <0.25 | <0.25–0.7 | 0.097 a | 0.5 | <0.25–1.0 | 400 |
| Ni, µg/L | <0.84 | <0.84 ≤ 0.84 | <0.84 | <0.84 ≤ 0.84 | 0.999 a | <0.84 | <0.84 ≤ 0.84 | 70 |
| Cu, µg/L | <0.56 | <0.56–0.89 | <0.56 | <0.56–0.95 | 0.999 a | <0.56 | <0.56–0.89 | 2000 |
| Zn, µg/L | 6.0 | <3.2–590 | 5.9 | <3.2–35 | 0.225 a | 5.9 | <3.2–160 | 3000 |
| Rb, µg/L | 1.0 | 0.16–1.8 | 3.1 | 1.4–4.3 | <0.001 a | 1.5 | 0.6–3.1 | - |
| Sr, µg/L | 550 | 62–1200 | 2100 | 1900–2600 | <0.001 | 1600 | 390–2200 | 17,000 b |
| Mo, µg/L | 0.37 | 0.08–0.78 | 2.8 | 1.4–3.9 | <0.001 | 1.1 | 0.30–2.9 | 70 |
| Cd, µg/L | <0.02 | <0.02 ≤ 0.02 | 0.02 | <0.02 ≤ 0.02 | 0.941 a | <0.02 | <0.02 ≤ 0.02 | 3 |
| Cs, µg/L | 0.09 | <0.038–0.4 | <0.038 | <0.038 ≤ 0.038 | 0.999 a | <0.038 | <0.038–0.14 | - |
| Ba, µg/L | 20 | 3.7–25 | 11 | 9.3–14 | 0.478 a | 12 | 8.0–20 | 700 |
| Ce, µg/L | 0.01 | <0.007–0.02 | <0.007 | <0.007–0.02 | <0.001 a | 0.008 | <0.007–0.02 | - |
| Tl, µg/L | 0.02 | <0.006–0.05 | <0.006 | <0.006–0.006 | <0.001 a | <0.006 | <0.006–0.02 | 2 b |
| F−, mg/L | 0.17 | 0.02–0.32 | 1.1 | 0.8–1.2 | <0.001 a | 0.56 | 0.12–1.1 | 1.5 |
| Cl−, mg/L | 43 | 18–73 | 230 | 84–310 | <0.001 | 80 | 43–240 | 250 |
| NO2−, mg/L | 2.2 | 1.1–2.8 | 5.4 | 2.8–5.9 | <0.001 | 2.8 | 2.1–5.5 | 3 |
| NO3−, mg/L | 2.7 | 1.4–8.3 | 51 | 18–74 | <0.001 | 13 | 2.6–54 | 50 |
| SO42−, mg/L | 88 | 7.0–200 | 270 | 170–360 | 0.004 | 200 | 50–290 | 500 |
| Elements and Anions | Unit | Zone 1 (n = 34) | Zone 2 (n = 43) | Total (n = 77) | Reference Data | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hoet et al., 2013 [9] | Morton et al., 2014 [29] | ||||||||||
| Median | P25–P75 | Median | P25–P75 | Median | P25–P75 | Median | URL a | Median | P95 | ||
| Sulphur | µg/L | 620 | 330–910 | 1000 | 540–1600 | 820 | 485–1200 | - | - | - | - |
| Chlorine | mg/L | 2800 | 1200–4800 | 3600 | 2000–5400 | 3100 | 1800–5050 | - | - | - | - |
| Vanadium | µg/L | 0.19 | 0.11–0.37 | 2.6 | 1.3–5.8 | 0.83 | 0.22–3.1 | 0.25 | 1.5 | 1.6 | 3.8 |
| Iron b | µg/L | 22 | 9.0–36 | 22 | 12–32 | 22 | 11–33 | - | - | - | - |
| Arsenic | µg/L | 28 | 15–48 | 31 | 18–53. | 31 | 16–51 | 14.1 | 300 | 10.5 | 152.4 |
| Bromine | mg/L | 5.0 | 3.6–7.5 | 6.9 | 5.2–9.5 | 6.1 | 4.8–8.6 | - | - | 2.3 | 5.4 |
| Selenium | µg/L | 36 | 25–54 | 58 | 31–88 | 46 | 28–64 | 25.1 | 80 | 13.4 | 33.4 |
| Iodine | µg/L | 150 | 92–230 | 800 | 630–1500 | 500 | 170–900 | - | - | - | - |
| Lead | µg/L | 2.6 | 1.2–3.9 | 0.05 | 0.05–1.1 | 1.1 | 0.05–2.9 | 0.87 | 4 | 0.5 | 7.6 |
| Uranium | µg/L | <0.13 | <0.13 ≤ 0.13 | 0.22 | 0.15–0.32 | 0.15 | <0.13–0.26 | <0.007 | 0.05 | - | - |
| Elements and Anions | Unit | Zone 1 (n = 192) | Zone 2 (n = 104) | Total (n = 296) | Reference Data | ||||
|---|---|---|---|---|---|---|---|---|---|
| Heitland et al., 2005 [30] | |||||||||
| Median | P25–P75 | Median | P25–P75 | Median | P25–P75 | Median | Min–Max | ||
| Sulphur | µg/L | 830 | 480–1200 | 1300 | 620–1900 | 920 | 520–1400 | - | - |
| Chlorine | mg/L | 2700 | 1600–4800 | 4500 | 1850–6200 | 3300 | 1700–5300 | - | - |
| Vanadium | µg/L | 0.44 | 0.26–0.77 | 4.4 | 1.9–7.8 | 0.73 | 0.33–3.0 | <0.056 a | <0.056 a–0.16 |
| Iron | µg/L | 28 | 18–52 | 27 | 16–42 | 28 | 17–46 | ||
| Arsenic | µg/L | 27 | 12–68 | 36 | 22–68 | 32 | 16–68 | 25 | 1-260 |
| Bromine | mg/L | 6.1 | 4.5–8.7 | 7.8 | 5.0–11 | 6.5 | 4.6–9.4 | - | - |
| Selenium | µg/L | 43 | 25–65 | 72 | 32–100 | 50 | 27–76 | 17 | 4-39 |
| Iodine | µg/L | 320 | 180–480 | 1400 | 680–2500 | 430 | 240–1000 | - | - |
| Lead | µg/L | 4.1 | 2.6–6.8 | 2.8 | 1.1–6.3 | 3.7 | 2.1–6.7 | 1.3 | 0.1-4.6 |
| Uranium | µg/L | 0.14 | <0.13–0.20 | 0.27 | 0.18–0.47 | 0.18 | <0.13–0.27 | 0.004 | <0.004 a–0.003 |
| Elements in Urine Women | Adjusted Coefficient (95% CI) a | p | Stand Beta | R2 |
|---|---|---|---|---|
| Sulphur (n = 74) | 0.8 (0.4, 1.3) | <0.001 | 0.225 | 0.20 |
| Chlorine (n = 74) | 0.5 (0.04, 1.0) | 0.033 | 0.244 | 0.12 |
| Vanadium (n = 74) | 4.0 (3.2, 4.8) | <0.001 | 0.768 | 0.60 |
| Iron (n = 74) | 0.2 (−0.5, 0.8) | 0.644 | 0.055 | 0.03 |
| Arsenic (n = 74) | 0.3 (−0.3, 0.9) | 0.333 | 0.116 | 0.02 |
| Bromine (n = 74) | 0.6 (0.2, 0.9) | <0.001 | 0.397 | 0.19 |
| Selenium (n = 74) | 0.7 (0.3, 1.1) | 0.001 | 0.370 | 0.20 |
| Iodine (n = 66) | 3.0 (2.6, 3.3) | <0.001 | 0.867 | 0.83 |
| Lead (n = 74) | −3.6 (−4.5, −2.6) | <0.001 | −642 | 0.42 |
| Uranium (n = 72) | 1.2 (0.8, 1.6) | <0.001 | 0.581 | 0.35 |
| Elements in Urine Children | ||||
| Sulphur (n = 293) | 0.4 (6.3, 8.4) | 0.001 | 0.194 | 0.15 |
| Chlorine (n = 287) | 0.5 (0.2, 0.7) | <0.001 | 0.220 | 0.15 |
| Vanadium (n = 294) | 3.0 (2.6, 3.4) | <0.001 | 0.183 | 0.49 |
| Iron (n = 288) | −0.1 (−0.4, 0.2 | 0.449 | 0.161 | 0.07 |
| Arsenic (n = 292) | 0.2 (−0.2, 0.6) | 0.314 | 0.059 | 0.06 |
| Bromine (n = 293) | 0.3 (0.1, 0.4) | 0.002 | 0.078 | 0.08 |
| Selenium (n = 292) | 0.5 (0.3, 0.8) | <0.001 | 0.221 | 0.16 |
| Iodine (n = 294) | 2.1 (1.8, 2.4) | <0.001 | 0.617 | 0.43 |
| Lead (n = 294) | −1.4 (−1.9, −1.0) | <0.001 | −0.337 | 0.13 |
| Uranium (n = 290) | 1.1 (0.9. 1.3) | <0.001 | 0.495 | 0.25 |
| Thyroid Disturbances Women b | Thyroid Disturbances Children c | |||||
|---|---|---|---|---|---|---|
| Trace Elements | Yes (n = 20) | No (n = 44) | p | Yes (n = 40) | No (n = 246) | p |
| Iodine, µg/L | 690 (283–998) | 390 (173–798) | 0.202 | 365 (173–685) | 445 (258–1100) | 0.151 |
| Vanadium, µg/L | 1.5 (0.3–6.0) | 0.8 (0.3–2.5) | 0.171 | 0.5 (0.3–2.4) | 0.8 (0.4–3.2) | 0.123 |
| Selenium, µg/L | 49 (29–69) | 41 (27–65) | 0.805 | 44 (21–77) | 52 (28–76) | 0.301 |
| Arsenic, µg/L | 29 (17–34) | 32 (15–49) | 0.385 | 25 (12–57) | 33 (17–74) | 0.118 |
| Bromine, mg/L | 6.1 (5.0–9.2) | 6.2 (4.8–8.1) | 0.778 | 6.0 (4.7–9.2) | 6.6 (4.6–9.5) | 0.667 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aakre, I.; Henjum, S.; Folven Gjengedal, E.L.; Risa Haugstad, C.; Vollset, M.; Moubarak, K.; Saleh Ahmed, T.; Alexander, J.; Kjellevold, M.; Molin, M. Trace Element Concentrations in Drinking Water and Urine among Saharawi Women and Young Children. Toxics 2018, 6, 40. https://doi.org/10.3390/toxics6030040
Aakre I, Henjum S, Folven Gjengedal EL, Risa Haugstad C, Vollset M, Moubarak K, Saleh Ahmed T, Alexander J, Kjellevold M, Molin M. Trace Element Concentrations in Drinking Water and Urine among Saharawi Women and Young Children. Toxics. 2018; 6(3):40. https://doi.org/10.3390/toxics6030040
Chicago/Turabian StyleAakre, Inger, Sigrun Henjum, Elin Lovise Folven Gjengedal, Camilla Risa Haugstad, Marie Vollset, Khalil Moubarak, Tecber Saleh Ahmed, Jan Alexander, Marian Kjellevold, and Marianne Molin. 2018. "Trace Element Concentrations in Drinking Water and Urine among Saharawi Women and Young Children" Toxics 6, no. 3: 40. https://doi.org/10.3390/toxics6030040






